Abstract
Donors after cardiac death (DCD) have recently become an important source of renal transplants to alleviate the shortage of renal grafts in kidney transplantation (KTx), although DCD kidneys often have complications associated with a delayed graft function (DGF). A microarray-based approach using renal biopsy samples obtained at 1 h after KTx from DCD identified the tissue inhibitor of metalloproteinases 1 (TIMP-1) gene as a potential predictive marker for DGF. The current study measured serum TIMP-1 in patients undergoing KTx and analyzed the time course after KTx. The average serum TIMP-1 level before KTx was 240 ± 10 ng/ml (n = 34). In patients undergoing KTx from a living donor (n = 23), the serum TIMP-1 levels showed no increase after KTx (POD1: 226 ± 12, POD2: 211 ± 12, and POD3: 195 ± 10 ng/ml), but in one case, the only patient who required post-KTx HD due to DGF, the level on POD1 was the highest among subjects (361 ng/ml). In contrast, patients undergoing KTx from DCDs (n = 11), the serum TIMP-1 levels increased rapidly after a KTx (POD1: 418 ± 32, POD2: 385 ± 42, and POD3: 278 ± 25 ng/ml). However, two patients who avoided post-KTx HD due to the immediate function of the graft did not show increased levels (<370 ng/ml) on either POD1 or POD2. The peak serum TIMP-1 values appeared to correlate to the post-KTx dialysis period. Furthermore, the increment of serum TIMP-1 on the early POD was found to be predictive of immediate or delayed function of the grafts. These data suggest that monitoring of serum TIMP-1 levels allow the prediction of graft recovery and the need for HD after a KTx from a DCD.
Keywords
Introduction
Because of a worldwide shortage of renal grafts, transplantation from donors after cardiac death (DCD) is becoming an alternative method to that from brain-dead donors (22, 32, 33). KTx of DCD grafts are still associated with a high incidence of delayed graft function (DGF) and/or primary nonfunction (PNF) during the early posttransplantation period. The incidence of PNF is as small as 5%, but the majority of grafts undergo some degree of DGF (9, 13, 14). DGF predisposes the graft to both acute and chronic rejection, thus indicating that DGF is a crucial risk factor for the short- and long-term survival of the graft and prognosis of the recipients (5, 10, 11, 20, 25, 27, 28, 34). These situations prompted the attempt to understand the etiology of DGF and to establish highly sensitive prognostic markers for DGF in KTx from DCD.
Gene expression profiling was conducted using renal biopsy samples obtained at 1 h after KTx from DCD. Several genes significantly upregulated in DCD kidneys were identified and they might reflect the performance of the graft and be potential noninvasive biomarkers (19). One of the good candidate genes that are potentially useful as a serum marker is the gene encoding tissue inhibitor of metalloproteinases 1 (TIMP-1). The matrix metalloproteinases (MMPs) and TIMPs system is responsible for the degradation of extracellular matrix (ECM) components involved in normal physiology and tissue remodeling (31). There is mounting evidence that the expression of TIMP-1 in the kidney is upregulated in various condition related to degeneration of renal tissue, such as lupus nephritis (29), diabetic nephropathy (28, 30), drug nephrotoxicity (26), and acute pyelonephritis (6). The serum TIMP-1 levels have also been shown to be elevated in patients with glomerulonephritis, nephrosclerosis, and diabetic nephropathy, and its potential usefulness as a biomarker has been discussed (2, 8, 12).
TIMP-1 is upregulated in the kidneys after ischemia/reperfusion injury, cyclosporine-induced interstitial fibrosis, and chronic renal allograft rejection in animal KTx models (1, 3, 15). In a clinical study, TIMP-1 was upregulated in the transplanted kidneys treated by cyclosporine 1 week after Tx (4). TIMP-1 is also associated with chronic renal transplant fibrosis (15, 21, 23). TIMP-1 is upregulated in the transplanted kidneys 1 week after Tx from DCD (16). However, no report has yet analyzed the serum TIMP-1 levels in KTx. This study determined the serum TIMP-1 level in patients undergoing KTx and analyzed the time course of the TIMP-1 expression after KTx to evaluate the usefulness of serum TIMP-1 as a potential marker for predicting the early functional recovery of transplanted DCD kidneys.
Materials and Methods
Study Design
This investigation was approved by the Institutional Review Boards. Written informed consent was obtained from each patient or legal guardian before enrollment. Patients undergoing either a living-related (LD; n = 23) or DCD KTx (n = 11) were prospectively enrolled. The immunosuppressive regimen of basiliximab, tacrolimus, or cyclosporine with prednisone and mycophenolate mofetil was similar in all patients.
The clinical characteristics of the patients undergoing KTx from DCD are shown in Table 1. All donors after cardiac death from this hospital were classified as type IV in this study. The cause of donor death was cerebrovascular disease in all cases. Although most of those cases required hemodialysis (HD: 0–17 days) after KTx because of DGF, the function of all of transplanted kidneys eventually recovered. There were no cases of either rejection or calcineurin inhibitor nephrotoxicity, which were confirmed by a biopsy during DGF.
Patient Characteristics: Donation After Cardiac Death (DCD)
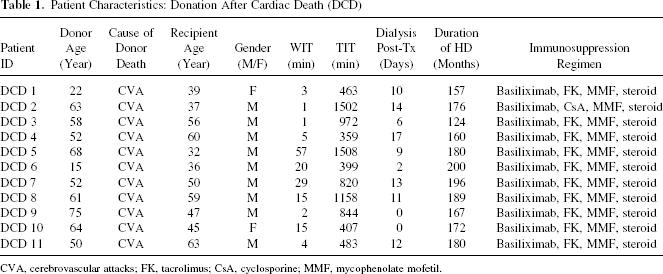
CVA, cerebrovascular attacks; FK, tacrolimus; CsA, cyclosporine; MMF, mycophenolate mofetil.
Serum samples were collected before and following KTx and stored in aliquots at −80°C. The primary outcome variable was the development of DGF, defined as the need for HD within the first few weeks after transplantation. The decision to initiate HD was taken by the primary transplant nephrologists and transplant surgeons, without any involvement from the study investigators. Other variables included age, gender, original kidney disease, warm and total ischemic time, urine output, and the serial serum creatinine level.
ELISA for TIMP-1 Quantification
The ELISA for serum TIMP-1 was performed as previously described. Briefly, microtiter plates were precoated with a mouse monoclonal antibody raised against human TIMP-1 (DTM100, R&D Systems, Minnesota, USA) and blocked with buffer containing 1% BSA. The wells were then coated with 50 μl of serum samples or standards (TIMP-1 concentrations ranging from 0.08 to 10 ng/ml), and incubated with a polyclonal antibody against TIMP-1-conjugated HRP. TMB substrate was added for color development, which was read within 30 min at 450 nm with a microplate reader (Benchmark Plus, BioRad, Hercules, CA, USA). All measurements were made in triplicate. The inter- and intra-assay coefficient variations were 5–10% for batched samples analyzed on the same day. The laboratory investigators were blinded to the sample sources and clinical outcomes until the end of the study.
Statistical Analyses
The results are expressed as the mean ± SEM. Statistical comparisons between groups were performed by Student's t-test, and differences were considered to be significant at p < 0.05. Correlations were evaluated with a linear straight-line regression. In significant difference tests, p-values were calculated with the z conversion of Fisher's r. Values of p < 0.05 were also considered to be statistically significant.
Results
The mean serum TIMP-1 level pre-KTx (n = 34) was 240 ± 10 ng/ml. The values were similar to the reported normal levels (190 ng/ml) and there was little variation among the samples. Next, samples obtained at post-KTx were examined. Patients who required HD in the post-KTx period due to DGF (LD: n = 1, DCD: n = 9) had serum TIMP-1 levels on POD1 (430 ± 33 ng/ml) that were significantly higher than those who did not (LD: n = 22, DCD: n = 2; 229 ± 12, ng/ml; p < 0.001) (Fig. 1).

Comparison of serum TIMP-1 concentrations at POD 1 after KTx between patients who required HD during the post-KTx period due to DGF (LD: n = 1, DCD: n = 9) and those who did not (LD: n = 22, DCD: n = 2; ∗p < 0.001).
The time course of serum TIMP-1 levels were analyzed before and after the KTx. Serum TIMP-1 levels showed no increase after KTx (POD1: 226 ±12, POD2: 211 ± 12, and POD3: 195 ± 10 ng/ml) in patients undergoing a KTx from a LD. However, in one case, the serum TIMP-1 level was higher than 360 ng/ml at POD1. This patient was the only LD-KTx case who required HD due to DGF (Fig. 2). In contrast, 9 of the 11 patients undergoing KTx from DCD required HD due to DGF patients. In these nine cases, serum TIMP-1 levels increased rapidly after the KTx (POD1: 418 ± 32, POD2: 385 ± 42, and POD3: 278 ± 25 ng/ml). However, the serum TIMP-1 levels were under 370 ng/ml on both POD1 and POD2 in the two patients who avoided post-KTx HD due to the immediate function of the graft (Fig. 3).

The serum TIMP-1 concentrations following a KTx from a living donor (LD; n = 23). The serum TIMP-1 level showed no increase after KTx from a LD. However, one patient had serum TIMP-1 levels over 360 ng/ml on POD1, and HD was therefore required due to DGF (indicated by open circles with bold line).

Serum TIMP-1 concentrations following a KTx from DCDs (n = 11). Nine of 11 patients that underwent a KTx from a DCD required HD due to DGF (indicated by open circles with bold lines). The patients that required HD due to DGF showed a rapid increase in the serum TIMP-1 levels after a KTx (POD1: 435 ± 32, POD2: 412 ± 42, and POD3: 293 ± 26 ng/ml). However, two patients had serum TIMP-1 levels under 370 ng/ml on both POD1 and POD2, and showed immediate function (indicated by closed circles with narrow lines).
To validate the usefulness of serum TIMP-1 as a predictor of DGF, the peak serum TIMP-1 levels (POD 1 or POD 2) were analyzed and the length of the post-DCD KTx dialysis periods were compared. The peak TIMP-1 level was found to correlate well with the length of dialysis (p < 0.05) (Fig. 4A). However, it is possible that the peak serum TIMP-1 levels are affected by the primary diseases or pre-KTx levels, although the samples did not show a large variation. Therefore, the correlation with the increment value of TIMP-1 was determined. The increment values of TIMP-1 in DGF cases also appear to correlate with the post-KTx HD period (Fig. 4B). The cut off value of the increment level of 150 ng/ml was predictive for the requirement of post-KTx HD.

(A) Correlation between the peak serum TIMP-1 levels (POD 1 or POD 2) and the length of dialysis post DCD KTx. The regression line is shown with the correlation coefficients (R2) and p-values. (B) Correlation between the increment value (the peak TIMP-1 value minus the value of pre-KTx value) of serum TIMP-1 levels DGF and the length of dialysis post-DCD KTx. The regression line is shown with the correlation coefficients (R2) and p-values. The cut-off value of 150 ng/ml clearly distinguished patients who required HD due to DGF (open circles) and those who could avoid HD due to an immediate function (closed circles).
Discussion
This study evaluated the efficacy of serum TIMP-1 as a predictor of graft recovery after Ktx, particularly a KTx from DCD. The results demonstrated that serum TIMP-1 was elevated within a couple of days post-KTx and the levels were higher in DGF cases whether the origin of the graft was a living donor or DCD. These results suggest that the preexisting damage in the kidney grafts may induce expression of proinflammatory cytokines that subsequently induce expression of MMPs/TIMPs. Alternatively, the inflammation may accelerate ECM turnover after reperfusion leading to de novo production of MMPs/TIMPs. However, the precise mechanistic relationship between TIMP-1 and DGF following a KTx from a DCD is still unclear.
The correlation of the post-KTx HD period with the peak serum TIMP-1 levels as well as the increment of serum TIMP-1 levels in early POD indicates that the serum TIMP-1 levels may reflect the severity of tissue damage in the transplanted kidneys. This implicates the potential efficacy of the serum TIMP-1 levels as a biomarker for prediction of DGF in DCD-KTx. In fact, the cut-off value of the increment level of 150 ng/ml was found to be predictive for requirement of post-KTx HD, although the number of subjects in the current study was small. The increment values of the serum TIMP-1 levels might thus be a better marker than the peak values, because it is possible that the serum concentration itself may vary according to the nature and the severity of the underlying disease. The current results show that the pre-KTx serum TIMP-1 levels were almost similar to those of normal samples and not highly variable. However, another study has indicated higher serum TIMP-1 levels in patients with chronic renal diseases (24).
In addition to the critical shortage of renal allografts, legal and ethical problems preclude brain-dead organ donation in Japan. Therefore, most of the cadaveric kidneys used over the past 3 decades in Japan have been derived from DCDs. Since April 1979 through the end of 2008, 495 kidneys were retrieved from DCDs at this center alone, thus representing approximately 10% of all kidneys procured in Japan. These kidneys are often transplanted under suboptimal conditions. Approximately 40% of the kidney grafts were derived from expanded criteria donors (13, 17). The DCD from this hospital was classified mainly as type IV category donors, but most were from the recently categorized “uncontrolled” DCD (7, 9). Furthermore, these patients had been waiting for a KTx for many years. All recipients who received a KTx from a DCD in this study had required HD for at least 10 years. Under these conditions for both donors and recipients, there was a high incidence of DGF and a prolonged period of ATN. Although the in situ regional cooling technique allows excellent renal function and good long-term graft survival, the mean duration of posttransplant HD is still about 2 weeks (13, 17). This situation, particular to Japan, prompted the need for highly sensitive prognostic markers for DGF in KTx from DCDs.
In summary, although the number of patients in this study was small, the previous and present findings suggest that monitoring of serum NGAL (18) and TIMP-1 levels might be useful for the control of DGF during the post-KTx period, and it should lead to the better outcome in KTx from a DCD.
