Abstract
The therapeutic mechanism of mesenchymal stromal/stem cells (MSC) for the treatment of acute myocardial infarction is not well understood. Our goal was to get insights into this mechanism by analyzing the survival kinetics of allogeneic and syngeneic cell transplants under different tissue conditions. Two MSC cell banks, stably and equally expressing the luciferase reporter construct, were developed for these studies and injected directly to the myocardium of Lewis rat recipients under syngeneic or allogeneic transplantation conditions. Cell survival was monitored by real-time fashion for up to 2 weeks, using optical imaging device (IVIS, Xenogen Corp.). We found that both syngeneic and allogeneic grafts reduced significantly in size during the first week of transplantation, either in the normal or in the late infarcted heart (5 days after MI) and allotransplants became always smaller than syngeneic grafts during this period. Low dose of cyclosporine A treatment had a benefit on both allo- and syngeneic graft sizes, suggesting that multiple mechanisms play a role in early graft reduction. The MSC characteristic factors IL-6, IL-8, MCP-1, and VEGF were well above the control level in the heart tissue at 4 days after cell injection, suggesting that the peak therapeutic effect of MSC can be expected during the first week of the administration. Although allogeneic cells induced immunoglobulin production, their biological effects (cell survival, factor productions) are very similar to the syngeneic transplants and therefore they could deliver the same therapeutic effect as the syngeneic cells. Finally, freshly infarcted tissue (30 min) supported better the survival of MSC than late postischemic tissue (5 days) but only “off the shelf” allogeneic cell transplants fits with this treatment strategy.
Introduction
Multipotent mesenchymal stem/stromal cells (MSC) with potential capacity to differentiate into cardiomyocytes and/or other cell lines have been applied in numerous preclinical studies (4,27,30,36) and a recent clinical trial is testing their benefit for patients with acute MI (12). In most investigations, transplantation of MSC resulted in improved cardiac function, but despite these encouraging data the mechanism of how the injected MSC lead to functional improvement of the infarcted heart is not well understood. It is reasonable to assume that a positive correlation exists between the improved myocardial function and the residence time of the injected cells in the myocardium. Consequently, the transplantation conditions should have a strong impact on injected cell survival and may determine their life span in the injured myocardium. Understanding the factors that affect tissue residence time may contribute to the development of more effective therapeutic strategies.
Several laboratories have reported follow-up studies of transplanted MSC in the heart (1,9,16). These investigations, however, characterized the transplanted cells under a single condition and little information is available for a comparative analysis of graft survival under different circumstances. This occurs, at least partially, because the techniques used to monitor the fate of the injected cells have significant limitations. For example, histological evaluations of cell engraftment using cells labeled with BrdU, GFP (8,19,31,34,44) offer only an end of life data point for cell engraftment studies and therefore cannot be employed for kinetics assessment of graft size in the same animal. Moreover, these methods also involve a tedious quantitative analysis. Application of radioisotopes for in vivo imaging (2,6) also has limitations because of the short biological and/or physical half-life of different radioligands and the nonspecific labeling by secondary uptake of radioactive compounds.
To circumvent these difficulties, in the current study we used in vivo biophotonic imaging technology (IVIS, Xenogen Corp.) that allows monitoring cell survival in vivo within the living animal in a real-time fashion (7,33). IVIS has already been applied for the spatial and temporal detection of cancer cells during drug and gene therapy (13,14,24,42,48) to follow the survival and biodistribution of cell transplants (20,21,40,47) or expression of viral genes (35). We further improved this technique by combining it with retroviral overexpression of the firefly luciferase in the MSC to be injected. The retrovirus system offers stable integration of the target gene into the genome and therefore provides longer follow-up periods than the adenoviruses. Additionally, this virus system allows the development of cell banks, providing uniform and highly comparable transplants for different studies. The bioluminescent signal released from the luciferin substrate after enzymatic cleavage by the reporter enzyme is also preferred to a potentially applicable fluorescent signal, because of its higher signal to background ratio (41).
Optical imaging of the heart in living animals by the biophotonic imaging method has been reported by Wu et al. (46) after injecting an adenovirus firefly lucerase reporter construct directly into the heart. The same method was also utilized by Min and Bom for engraftment study of human MSC in the rat heart (25) after being transduced by a luciferase adenovirus construct. Our studies investigated MSC survival in a comprehensive manner under different transplantation conditions and found short survival time for both allogeneic and syngeneic transplants. Furthermore, quantitative differences between syngeneic and allogeneic transplant sizes were consistent during these studies but the tissue levels of MCP-1, IL-6, IL-8, and VEGF were very comparable after cell injection, suggesting a similar paracrine function of the two graft types in situ.
Materials and Methods
All experimental protocols were approved by the Institutional Animal Care and Use Committee of Genzyme Corporation and conformed to the US National Institute of Health Guideline for the Care and Use of Laboratory Animals.
Isolation and Cultivation of Mesenchymal Stromal Cells
Bone marrow samples of male Norway Brown and Lewis rats were harvested from the femurs and tibias of multiple animals. Mononuclear fractions were isolated, plated, and cultured according to a standard MSC cultivation protocol (11).
Vector Construction, Virus Preparation
The coding sequence of the firefly luciferase (Luc) originating from pGL3 vector (Promega) was inserted into the BamH1 site of the replication incompetent recombinant retrovirus vector pDON-AI (Takara Bio Inc). The construct was further modified by replacing the 5′ LTR sequences with the EF1a promoter to increase luciferase expression (50). pDON-EF1aLuc.Neo vector (Fig. 1) was propagated in HEK293 cells, and the optimal dilution of the unpurified culture supernatants was titrated in pilot experiments on MSC.

Vector structure of pDON-EF1aLuc.Neo. The viral construct was generated to constitutively express the firefly luciferase gene driven by a CMV promoter. pDON-A1 was modified by replacing the 5′ LTR cassette by EF1a sequences for long-term expression of the reporter gene.
Preparation of Luc+/MSCL and Luc+/MCSNR Banks
Subconfluent Norway Brown and Lewis MSC were infected with the Luc+/retrovirus construct overnight. Positive cells were selected with 0.2 mg/ml neomycin and propagated up to passage 10. The expanded neomycin-resistant cells were then screened for luciferase activity and MSC surface marker expression before being cryopreserved in small aliquots for further in vivo work. Luc+ MSC from Lewis rats were called MSCL, and Luc+ MSCs from Norway Brown rats were called MSCNB.
Preparation of Cells for Injection
Cryopreserved aliquots of MSCL and MSCNB banks were propagated in culture for 3–4 days. On the day of injection, cells were harvested by Accutase (Millipore), washed, counted, and aliquoted into 4 × 106 cells/80 μl injection buffer for cell transplantation. The injection buffer consisted of DMEM and 0.2% human serum albumin. The average viability of the different preps was >95%, as determined by propidium iodide staining.
Surgical Procedures
All in vivo analyses were performed in adult, female Lewis rats (200–250 g). Rats received 44 mg/kg ketamine, 5 mg/kg xylazine, and 1.5 mg/kg diazepin intraperitoneally for anesthesia and they were mechanically ventilated during the surgery. The myocardial infarction was created by permanent ligation of the left anterior descendent coronary artery via thoracotomy. Cell suspension or buffer was injected into four symmetrical locations of the left ventricle, corresponding to the infarct border zone.
Assessment of Long-Term Transplant Survival by CCD Camera/IVIS
Animals received 300 mg/kg luciferin substrate by IP injection. Image acquisition was performed on lightly anesthetized animals receiving inhaled isoflurane. Bioluminescence signals were collected by the IVIS 200 Imaging System (Xenogen Corp., Alameda, CA) equipped with the standard CCD camera and the Living Image™ software, using bioluminescence mode of operation. Bioluminescence light intensity was measured at the region of interest and expressed in relative light units (RLU/min), as shown on one of the representative images (Fig. 2). Serial images were collected from the heart by the ventral/lateral oblique view for 40 min after luciferin injection in 5-min intervals. Due to some differences in the luciferin absorption, the highest RLU/min value of each animal was selected as the representative data point for image analysis. We observed large fluctuation in signal intensity at the very early postinjection period; therefore, data acquisition for quantitative analysis started 2–3 days after cell injection. Misdelivery of cells into the LV cavity was verified by total body screening shortly after injection. Bright signals in the brain and the kidney area were observed in these animals, according to the arterial distribution. These animals were removed from the follow-up studies. Typically each study group was represented by six animals.

In vivo imaging of Luc+/MSC transplants. Representative image of two Luc+/MSC-injected animals, 2 weeks after direct cardiac delivery. Luminescence light intensity was measured at the region of interest (ROI).
Preparation of Heart Tissue Extracts and Cytokine Quantitation by Linco Beadlyte
Animals were euthanized by pentobarbital overdose. The hearts were removed quickly and washed with ice-cold saline solution. The buffer or cell-injected area of the left ventricle was quickly dissected out, transferred into protease inhibitors containing PBS, prepared according to the manufacturer (Protease Inhibitor Cocktail Tablet, Roche). Tissue pieces were then mechanically homogenized on ice by OMNI TH tissue homogenizer. Cell debris were separated by high speed centrifugation (13,000 x g for 20 min at 4°C), the clear tissue supernatants were transferred into clean tubes and stored at −80°C until further testing. Cytokines/chemokines were assayed by the premade rat 24 plex Linco beadlyte kit, according to the manufacturer and quantified by Luminex 200. Protein concentrations of the same samples were also measured by Bradford assay for normalization purposes.
Quantitative PCR
Genomic DNA was isolated from heart samples by proteinase K digestion. The Y chromosome was quantified by sry gene-specific primers and probe as described previously (26). In simultaneous experiments, the single copy gene osteopontin was also analyzed for normalization.
Statistical Analysis
The data are expressed as means ± SE. Statistical analysis was performed using Student's t-test and analysis of covariance (ANCOVA). Multiple comparisons were corrected for by using the Student-Newman-Keuls test. A value of p < 0.05 was considered statistically significant.
Results
Characterization of Allo- and Syngeneic Luciferase-Positive MSC Banks for In Vivo Studies
Reporter gene expression of the Luc+/MSCL and Luc+/MSCNB cell banks was compared in vitro by serial dilution of Luc+/MSCL and Luc+/MSCNB cells, measuring the luminescent light emission after substrate addition. In this analysis there was a linear relationship between the signal intensity (RLU/min) and the cell number in both MSC preparations and their dose–response curves almost completely overlapped with each other (Fig. 3). Next, we investigated the effect of the reporter gene expression on the character of cells by measuring the expression of MSC surface markers such as CD14, CD11b, CD29, CD31, CD45, CD73, CD90, MHC1 (RT1A), and MHCII (RT1) through FACS analysis (not shown). The level of these markers did not differ between Luc+ and Luc- cells.

Dose–response curves of Norway Brown and Lewis MSC, expressing the luciferase reporter gene. After multiple selections for neomycin resistance, cell banks were prepared from passage 10 cells. Expression levels of the reporter gene were compared by measuring the emitted light (y-axis) at different cell concentrations (x-axis) (open symbol: Lewis MSC; filled symbols: Norway Brown MSC).
To confirm the correlation between the in vivo signal intensity and cell number, heart samples from gender-mismatched male MSC transplants were assayed for Y chromosome copy number by quantitative PCR. We observed that high signal intensity associated with high Y copy number while low signal intensity with low copy number of sry gene (Fig. 4), indicating that light emission is proportional to the presence of the injected cells. In addition, when Luc+ cells were killed by freeze and thaw cycles, bioluminescent signal was not detectable from these preparations either in vitro or in vivo after injecting the dead cells into animals (not shown), demonstrating that only viable cells can emit bioluminescence light.

Correlation between chemiluminescence light intensity and Y chromosome number. One week after receiving Luc+/MSC animals were subjected to IVIS analysis, then heart samples were collected for genomic DNA extraction. Quantitative analysis of Y chromosome was performed by qPCR and normalized to osteopontin genes. Higher flux values (y-axis) associated with higher SRY/OSTP copy numbers (x-axis) and vice versa.
These data together indicate that MSCL and MSCNB were suitable for in vivo studies and that the bioluminescent signal intensity (RLU/min) represented the number of transplanted cells (graft size).
Survival of Allogeneic and Syngeneic Transplants in the Normal Heart
First, we investigated whether autologous (syngeneic) cell therapy has any benefit over allotransplantation by comparing cell survival. To this end, the same number of Luc+/MSCNB or Luc+/MSCL were injected into the normal hearts of Lewis recipients. We found that 3 days after delivery, the bioluminescence light intensity of the allogeneic and syngeneic groups was similar (9.12 × 107 and 1.16 × 108 RLU/min, respectively) as shown by the first data points of the solid lines in Figure 5. This was followed by a parallel decrease of both transplant types up to 9 days. At this time point the signal intensity became significantly higher in the syngeneic group (p < 0.01) and the transplant size stabilized for the rest of the study. In contrast, the allogeneic graft was barely detectable after 9 days and became undetectable after 14 days.
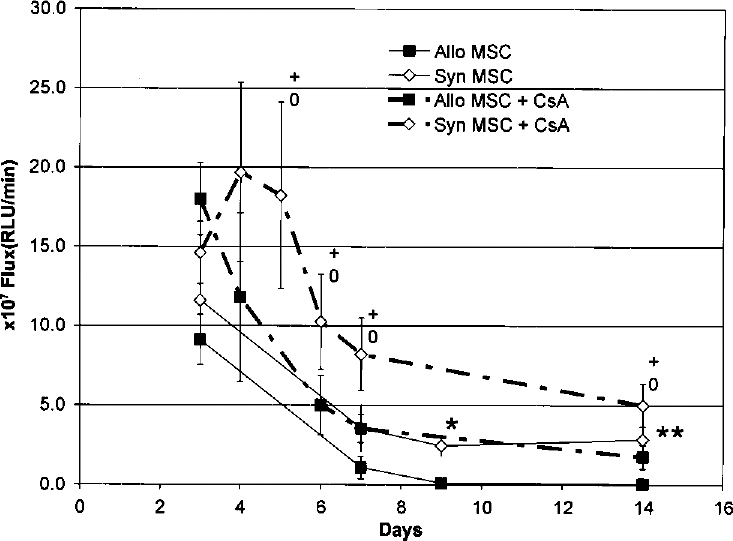
Allogeneic and syngeneic graft survival and the effect of immunosuppressive therapy. MSCL (open symbol, continuous line) or MSCNB (filled symbol, continuous line) were injected into the myocardium of the healthy, noninfarcted animals and graft survival was monitored for up to 2 weeks (six animals in each group). Differences between allo- and syngeneic transplants became statistically significant by day 9 (∗p < 0.01) and stayed significant by the end of the study (∗∗p < 0.05). The 10 mg/kg cyclosporine A treatment started 1 day before cell injection and continued for 4 more consecutive days (dashed lines, six animals in each group). Syngeneic grafts sizes became significantly larger under cyclosporine treatment for the whole duration of the study compared to non-cyclosporine-treated syngeneic controls (+p < 0.05) or to the cyclosporine-treated animals, receiving allotransplantation (0p < 0.05). Cyclosporine administration also improved allotransplants survival. No differences were observed between allo- and syngeneic (without cyclosporine) graft sizes when animals received cyclosporine treatment.
Mild Immunosuppression Enhanced Transplant Survival
Next we evaluated the effect of mild immunosuppression on the survival of newly transplanted syngeneic and allogeneic cells. Animals were treated with 10 mg/kg cyclosporine A for 5 consecutive days, starting 1 day before cell injection. This treatment approximately doubled the size of both the allogeneic and syngeneic grafts, compared with controls that did not receive cyclosporine A. The improvement in graft size was detectable as early as 3 days and stayed similar throughout the 14-day duration of the experiment (Fig. 5, dashed lines). Cyclosporine A treatment resulted superior survival of syngeneic transplants at every time point of the study, compared to any other treatment groups. Allotransplant sizes were also significantly larger (p < 0.02) under cyclosporine A administration than without. With this dosing regime, the survival curves of the cyclosporine-treated allogeneic and the untreated syngeneic groups became similar to each other even at the late posttransplantation time points (9 and 14 days). Cyclosporine A effect on both transplant types suggests that the protective effect of this drug is mediated not just by immunosuppression.
Both Allogeneic and Syngeneic Grafts Survived Better in Normal Hearts Compared to Infarcted Hearts
Infarcted tissue with the associated inflammatory reactions (mononuclear, neutrophil invasion, cytokine release, etc.) and the hypoxic conditions most likely presents a hostile environment for the fresh cell transplant. Therefore, we asked whether the MSC grafts are sensitive to these tissue conditions and whether the different transplant types are similarly affected by this environment. For this purpose, recipient animals received allogeneic or syngeneic cells 5 days after infarction. The 5-day postligation time point was selected because many cell therapy treatments are more feasible at a later time point of the postischemic episode. Graft viability was monitored only for 1 week following the treatment because the major effect of infarct on graft size was expected during the first postinjection week. Similar to noninfarct conditions, we observed parallel decreases of allogeneic and syngeneic graft sizes by time (Fig. 6), but the survival in the infarcted tissue was smaller for both graft types, comparing day 7 flux values between the infracted (allo = 6.35 × 106 RLU/min, syn = 2.52 × 107 RLU/min) and normal hearts (allo = 1.09 × 107 RLU/min, syn = 3.55 × 107 RLU/min) (Fig. 5). Moreover, infarct environment better reduced allograft sizes than the syngeneic ones, because the difference between allo-and syngeneic transplants became significant by day 7 (p < 0.01), while in the normal heart only by day 9 (see Fig. 5).

Comparison of allogeneic and syngeneic cell survival in the infarcted myocardium. Same doses of MSCL (open symbol) and MSCNB (filled symbol) were delivered into the peri-infarct zone of animals underwent permanent coronary ligation 5 days earlier (nsyn = 6, nallo = 6). Cell survival during the first week was monitored by scanning the animals at multiple time points. Only the last time point data indicated significant differences between the two graft types (∗p < 0.01).
Shorter Postinfarction Time Promoted the Survival of Transplanted Cells
To further investigate the infarction effect on transplant survival, syngeneic MSC were also injected into the heart at the onset of the hypoxia (30 min after coronary ligation) and transplant survival at two treatment points 5 days vs. 30 min were compared.
When cells were injected soon after coronary occlusion (30 min), into the hypoxic tissue, graft sizes were significantly larger at most post-injection time points of the study, when compared to grafts injected 5 days after infarction (Fig. 7). For example, 1 week after cell injection, the 30-min grafts were still three times larger than the grafts transplanted 5 days after the myocardial insult (compare 1.4 × 108 vs. 4.13 × 107 RLU/min, p < 0.02). This difference, however, disappeared by the end of the second week when the emitted light of the two treatment groups were markedly decreased and did not differ significantly from each other.

Survival of syngeneic MSC in the infarcted myocardium delivered at different postinjury time points. Luc+/MSCL were injected into the peri-ischemic zone 30 min (continuous line, n = 5) or into the peri-infarct zone 5 days (dashed line, n = 6) after permanent coronary ligation. Cell survival (y-axis) was monitored at multiple time points by measuring luciferase activity (x-axis). Injection of the cells soon after coronary ligation (30 min post-MI time) transiently increased graft sizes, resulting in approximately threefold differences by day 7 (∗p < 0.05) between the two groups.
These data suggest that graft sizes vary by the postischemic treatment time and the conditions in the acute phase of ischemia (30 min) are more amenable for cell retention and/or survival than later postinfarction time point (5 days). Moreover, this effect was only temporary and did not result in long-term cell survival.
Intramyocardial MSC Injection Increased the Levels of MSC-Specific Cytokines in the Myocardium
To search for evidence of in situ MSC activity, we prepared tissue extracts of heart samples after MSC injection and assayed them for the presence of MCP-1, IL-2, IL-6, and VEGF. As observed with human MSC (22), these cytokines were elevated in the culture supernatant from rat MSC. In this study, luciferase-negative syngeneic or allogeneic cells were delivered into the normal, noninfarcted myocardium, while control hearts were treated only with injection buffer.
Based on the survival curves (Fig. 5), heart samples were harvested 1, 4, or 7 days after injection. Then the anterior heart wall corresponding to the injected area was isolated and processed for protein extraction and analysis. The relative cytokine levels after normalization to buffer control are shown on Table 1. No significant differences from the controls were observed on the first day after injection in either cell types. At 4 days there was a general increase in the levels of all four cytokines with both allogeneic and syngeneic transplants. Some of them had statistically higher levels than the buffer control (bold and italic in Table 1), but no significant differences were observed between heart samples treated by different cell types. By day 7, however, the levels of most of these cytokines returned close to the basal level.
Local Cytokine Levels After MSC Treatment

Normal hearts were injected with syngeneic (MSCL) or allogeneic (MSCNB) cells or buffer (control). Hearts were harvested 1, 4, or 7 days later for the assay of local cytokines. The table summarizes the relative level of MCP-1, IL-2, IL-6, and VEGF, having buffer controls as the baseline value (1.00). Bold italic font indicates statistically significant values (p < 0.05) compared to the control (buffer). Each time point group represents five animals.
These data suggest that during the first few days of transplantation both the syngeneic and allogeneic cells are functionally active and produce their characteristic cytokines, although the secretion may initially be compromised by the delivery procedures.
Serum IgG Concentration Was Significantly Higher in the Allogeneic Than in the Syngeneic Transplant Group
To investigate whether immune mechanisms played a role in reduced allograft survival, blood samples were collected from the same animals before cell injection and 2 weeks later. Two weeks following cell injections, animals that received allogeneic cells had IgG level (3143 ± 759 ng/ml) significantly higher (p < 0.02) than the animals treated with syngeneic cells (1710 ± 491 ng/ml) or their preimmune controls (1175 ± 231 ng/ml). Moreover, cyclosporine A treatment of the allogeneic transplant group reduced the serum IgG level to 1760 ± 86 ng/ml. Smaller reduction in the syngeneic treatment group (1601 ± 561 ng/ml) was also noticed. No difference was detected in the serum IgM levels when assayed simultaneously (not shown).
These observations suggest that allogeneic MSC provoke an immune response in the recipient animals and that mild immunosuppression can blunt this response.
Discussion
Utilizing bioluminescence imaging technique (IVIS), our study compares for the first time the in vivo survival of syngeneic and allogeneic cell transplants in normal and infarcted myocardium in a real-time fashion. The first optical imaging of the heart was reported by injection of an adenovirus-expressing firefly luciferase into the myocardium (46). Here we report the construction and application of a retrovirus-based reporter vector, which allows long-term propagation of infected cells without losing their biomarker expression and therefore offers opportunities for more comprehensive studies. Typically we followed the fate of the transplants between 2 days and 2 weeks after their direct injection into the heart tissue. Earlier than 2 days the signal intensity showed some fluctuations, probably because the high initial flux was above the linear range of the detection. From independent experiments utilizing neutron activation technique, we have reported that 85–90% of the europium prelabeled cells leave the heart in 5 min after injection (45). Therefore, the IVIS analysis from 2 days after injection describes the fate of this residual fraction, which is equal or less than 10–15% of the total delivered cells.
In general, we found very similar survival curves both for allogeneic and syngeneic transplants during the investigated time period. The kinetics can be characterized by rapid reduction in cell number during the first week and slower reduction during the second week of transplantation. Similar reduction has been reported for MSC, injecting them into the myocardium under xenografting condition using the same surveillance screening test (25). On the other hand, early graft reduction could be independent from cell type (51), and this loss could rather be related to common reasons like anoikis, inflammation that makes the transplanted cells more vulnerable during this early postdelivery period. The benefit of short course cyclosporine A treatment for both transplant types further underlines the existence of such a mechanism. Thus, the anti-inflammatory effect of cyclosporine A could mitigate the nonspecific local inflammation, associated with the delivery procedure, sparing the transplant. Moreover, cyclosporine A has an antiapoptotic effect, too, by blocking mitochondrial permeability (37), which is a critical mechanism of detachment-associated cell death (38). Clinical application of cyclosporine A during acute myocardial infarction is not suggested, but protection of the early transplant by targeting anchorage-dependent cell death (32) or improving the delivery technique (10,29) may enhance the performance of the transplant.
Although MSCL and MSCNB graft sizes changed in parallel with time, allografts were always significantly smaller than syngeneic grafts, suggesting that immune mechanisms also play a role in cell survival. This observation was further supported by elevated serum IgG levels of animals receiving allogeneic cells. MSC have been reported as an immunoprivileged cell type (17,18), allowing their therapeutic application across the allogeneic barrier. While we did not observe major adverse effect, our data strongly suggest that allogeneic MSC directly injected into the myocardium are not neutral for the host immune system. Similar conclusions can be drawn from a rat spinal cord injury model, where the long-term immune suppression promoted the survival of the allogeneic MSC (39,49).
The tissue environment at the late postischemic time point (5 days post-MI) resulted in reduced MSCL and MSCNB graft sizes. In contrast, in the early postischemic environment (30 min after ligation) comparably larger graft sizes were observed, although this favorable effect was transient and did not promote long term (>2 weeks) survival. We suggest that local tissue edema, associated with the ligation procedure in the early infarct could passively increase the acute retention of the cells and may extend the effective period of cell therapy. In addition, the minimal level of tissue inflammation from the early injured tissue (30 min) could also be a contributing factor to improved transplant survival. Our tissue analysis on characteristic MSC factors such as IL-2, IL-6, and MCP-1 suggests that the injected cells are functioning during this period, either by directly expressing these factors (23) or by stimulating the receiving tissue through paracrine mechanism or both. According to the paracrine hypothesis, MSC therapy exerts its effect through the modulation of the inflammatory environment (43), and/or by promoting angiogenesis through elevated VEGF synthesis (3,5,15). Although the mechanism of MSC action is not completely understood, the short duration of significant levels of expression of these pararcrine factors indicate a narrow effective therapeutic time window for MSC.
The longer term survival of MSC was followed for 2 weeks after cell injection when small syngeneic transplants were still detectable but the allotransplants were not. In the same experimental conditions, a subgroup of animals was followed up to 8 weeks, when weak but still detectable signal was measured in the syngeneic transplants (not shown). We have little knowledge about these surviving cells. They may differentiate into endothelial, smooth, or cardiac muscle cells (28,30), but independently on their differentiation, these cells represent only a small fraction (<0.1%, <4,000 cells) of the total injectate. Therefore, their contribution to global heart function does not appear to be significant. It would also be interesting to know whether this long-term survival population represents a different subpopulation of the transplant with better survival property.
Taken together, our data indicate that for cell-mediated effect of MSC injected into the myocardium the most valuable time is the first week of the treatment, and during this time period both allo- and syngeneic cells may provide similar therapeutic effect, as measured by tissue factors and survival kinetics. Data from tissue extracts also suggest that the therapeutic effect of these cells is not prompt, but rather takes a few days to obtain elevated cytokine/growth factor levels. Therefore, it is important to enhance the benefit of these cells through increasing their number in the injected tissue. Cell administration in the early postischemic period, when the local environment appears to be more receptive, could be a simple approach to prolong the residence time of the delivered cells. Although syngeneic cells showed superior survival over allogenic ones, the early treatment strategy would only be feasible with an “off the shelf” allogeneic product.
Footnotes
Acknowledgments
We acknowledge Geoffrey Parson, Genzyme Molecular Biology, for constructing the retrovirus luciferase vector for this program, and Gary Jacques and Rob Fogle, Genzyme Preclinical Biology, for their technical support in in vivo images. Gitta Seleznik was sponsored by the Hungarian American Scholarship Fund, Graduate Scholarship Program.
