Abstract
Duchenne muscular dystrophy (DMD) is the most frequent muscular dystrophy. Currently, there is no cure for the disease. The transplantation of muscle precursor cells (MPCs) is one of the possible treatments, because it can restore the expression of dystrophin in DMD muscles. In this study, we investigated the effects of myoblasts injected with cardiotoxin on the contractile properties and resistance to eccentric contractions of transplanted and nontransplanted muscles. We used the extensor digitorum longus (EDL) as a model for our study. We conclude that the sole presence of dystrophin in a high percentage of muscle fibers is not sufficient by itself to increase the absolute or the specific force in the EDL of transplanted mdx muscle. This lack of strength increase may be due to the extensive damage that was produced by the cardiotoxin, which was coinjected with the myoblasts. However, the dystrophin presence is sufficient to protect muscle from eccentric damage as indicated by the force drop results.
Introduction
Duchenne muscular dystrophy (DMD) is the most frequent inherited lethal muscle-wasting disease as it affects 1 of 3,500 newborn boys worldwide. Patients are usually diagnosed between 3 and 5 years of age. Early symptoms of delayed walking and unsteady gait rapidly progress to general muscle wasting, first affecting the muscles of the hips, pelvic area, thighs, and shoulders (9, 12, 30). By age 12, 95% of patients are confined to a wheelchair and most of them develop severe scoliosis. In the natural course of the disease, survival is rare beyond the early 20s, most likely because of respiratory and heart failure (20). The current treatment options for DMD patients focus primarily to control some of the consequences of the disease, slow down the muscle weakness for a few years, and prolong life expectation (5).
DMD is caused by a mutation in the dystrophin gene located on the X chromosome (17). Dystrophin is a 427-kDa protein, localized under the sarcolemma of the myofibers and cardiomyocytes, and it is an essential component of the dystrophin glycoprotein complex (6, 13, 14, 25). The dystrophin complex normally links the extracellular matrix to the actin cytoskeleton. The lack of dystrophin results in the absence of the entire complex. Its physiological role would be to provide mechanical stability and maintain membrane integrity during normal contraction. Consequently, lack of dystrophin in muscles of DMD patients is responsible for an increased fragility of myofibers and repeated cycles of degeneration and regeneration, which induce muscle atrophy and subsequently muscle replacement by fat and fibrosis (11, 15, 26).
Mdx mice have been extensively used as a model of DMD. A consequence of the lack of dystrophin in mdx muscles is an increased frequency of damaged myofibers compared to normal mice, and this phenomenon is accentuated following eccentric contractions or experimental lengthening contractions (8, 11, 15, 22, 23, 31).
Transplantation of normal muscle precursor cells (MPCs) is one of the possible treatments of muscular dystrophies (27, 29). Incorporation of normal MPCs in DMD myofibers leads to the expression of dystrophin. In previous mouse studies, many research teams have shown that MPC transplantation led to expression of dystrophin or of a reporter gene (present in the donor MPCs) in the fibers of injected muscles (16, 18, 21, 28, 30, 32).
Even if MPC transplantation could be a potential therapy for DMD, several important questions remain to be answered regarding the functional consequences of MPC transplantation. Previous studies have tried to answer these questions. Brussee et al. reported that MPC transplantation in biceps brachii of mdx mice, resulting in at least 40% dystrophin-positive fibers, significantly reduced the damage induced by exercise in the grafted muscle (7). Wernig et al. studied the function of irradiated soleus after MPC transplantation. They observed that transplanted muscles produced considerable higher twitch and tetanic forces compared with sham-injected muscles (34). However, it is still unknown whether normal MPC transplantation and the consequent dystrophin expression could improve the strength of mdx muscles and/or protect them from contraction-induced injury. To address these issues, we transplanted normal and mdx MPCs in the extensor digitorum longus (EDL) muscle of mdx mice and performed functional assays on muscle contractility.
Materials and Methods
Fetal bovine serum (FBS), Dulbecco's modified Eagle medium (DMEM), Hank's balanced salt solution (HBSS), and penicillin–streptomycin were obtained from Invitrogen (Burlington, Ontario, Canada). Collagenase was purchased from Sigma Aldrich (St. Louis, MO) and dispase II from Boehringer Mannheim (Ontario, Canada). Anti-rabbit antibody coupled to ALEXA 546 was purchased from Molecular Probes (Eugene, OR), and polyclonal rabbit anti-dystrophin was produced in our laboratory. FK506 was a gift from Fuji-sawa Pharmaceutical Co. Ltd. (Osaka, Japan).
Animals
The experiments were conducted using C57BL/10J and C57BL10J mdx/mdx mice originally obtained from Jackson Laboratories and reproduced in our animal facilities. All the experiments performed on these animals were approved by the Animal Protection Committee of CRCHUL.
Mouse MPC Culture
Primary MPC cultures were prepared from normal C57BL/10J and C57BL10J mdx/mdx mice (2–3 days old). Briefly, mice were first anesthetized by hypothermia and killed by decapitation. Muscles were dissected from arms and legs, and were then cut into small fragments that were digested with collagenase (2 mg/ml) and dispase II (2 mg/ml) at 37°C for 1 h. The cell suspension was plated in DMEM supplemented with 10% FBS and antibiotics [penicillin G (10,000 IU/ml) and streptomycin (10 mg/ml)]. The cells were cultured at 37°C in a humidified atmosphere with 5% CO2. A pre-plating of 2 h was performed to discard most of the fibroblasts. After 48 h in culture, cells were frozen in nitrogen until transplantation. Desmin labeling was performed to investigate the percentage of MPCs (approximately 50% of desmin-positive cells).
MPC Transplantation
The day of transplantation, cells were thawed and washed in DMEM containing 10% FBS and resuspended in 10 μl of cardiotoxin solution (100 ng/ml) in HBSS. The left and right EDL of 12-month-old female mice were surgically exposed and 1.5 million cells were injected in several sites throughout each muscle. There were four groups of animals: 1) control mdx mice without treatment, 2) mdx mice transplanted with 10J MPCs (mdxT10J), 3) mdx mice transplanted with mdx MPCs (mdxTmdx), and 4) control mdx mice injected with cardiotoxin solution alone (cardiotoxin damaged).
Immunosuppression
The host mice were immunosuppressed with 2.5 mg/kg/day, IP, of FK506 until the end of the experiment. The immunosuppression was used to avoid allogenic MPC rejection in the mdxT10J group, but was also used in the other two groups (syngenic transplantation) to exclude an effect of FK506 in the force measurements.
Muscle Contractile Properties
Muscle Preparation
Mice were anesthetized IP with buprenorphine (0.1 mg/kg) and pentobarbital (30 mg/kg). The EDL was prepared as described previously (19, 33). Briefly, the EDL was carefully dissected and placed in an organ bath maintained at 30°C. The muscle was incubated with oxygenated Kreps-Ringer's solution (137 mM NaCl, 5 mM KCl, 2 mM CaCl2, 24.7 mM NaHCO3, 2 mM MgSO4, 1.75 mM NaH2PO4, and 2 g/L dextrose, pH 7.4).
The proximal tendon was fixed in a stationary clamp with a 3-O suture (Harvard Apparatus, St. Laurent, Canada). The distal tendon was connected to a dual-mode level arm system 305B-LR (Aurora Scientific, Inc., Aurora, ON, Canada) that provided control of force and positioning of the motor arm so that both dynamic and isometric muscle contractions could be elicited. The stimulations were delivered by a pair of platinum electrodes in either side of the muscle. The force generated by the muscle was measured by the LabView-based DMC program (Dynamic Muscle Control and Data Acquisition; Aurora Scientific, Inc.). Length and force data were analyzed by the LabView-based DMA program (Dynamic Muscle Data Analysis; Aurora Scientific, Inc.).
Isometric Contraction Protocol
Once dissected and placed in the bath, the muscle was initially set at a resting tension of 1 g for 10 min. Lo was determined as the muscle length at which the maximal twitch force was elicited. The muscle was subjected to a force-frequency measurement protocol. Briefly, the muscle was stimulated for 500 ms at 80, 120, and 150 Hz, respectively, with 1 min rest between each stimulation. The optimal fiber length (Lf) was determined by multiplying Lo by an Lf/Lo ratio of 0.44. The maximal force response at each stimulation frequency was normalized to muscle cross-sectional area {CSA (N/cm2) = (muscle mass, in g)/[(optimal fiber length, in cm) x (muscle density, in g/cm3)]}.
Eccentric Contraction Protocol
Before beginning the eccentric contraction protocol, the muscle was rested 10 min. Briefly, the muscle was stimulated at 150 Hz for 700 ms. After 500-ms stimulation, the muscle was lengthened to 110% Lo at 0.5 Lo/s for 200 ms. When stimulation ended, the muscle length was reset to Lo at the same rate. This stimulation-stretch cycle was repeated every 2 min for a total of seven cycles. To determine the percentage of the force drop after each contraction, the maximal isometric tetanic force developed during the first 500 ms of the first stimulation was considered as 100%. The formula (F1 - Fn)/F1 was used to calculate the percentage of tetanic force loss at each cycle; F1 was the tetanic force developed during the first cycle and Fn represented the tetanic force obtained during the nth cycle.
Immunohistochemistry
After the contraction protocol, the muscles were frozen in liquid nitrogen and then stored at −80°C. Transverse serial cryostat sections throughout the muscles were collected successively on slides. The presence of dystrophin in the muscle sections was detected with a rabbit polyclonal antibody directed against the dystrophin C-terminal region. The secondary antibody was an anti-rabbit antibody coupled to red ALEXA 546.
Statistical Analysis
All statistical analysis was performed using repeated measures ANOVA. Following a significant effect of any sources of variation, multiple comparisons were done in order to evaluate which factor's level differs from the others. The false discovery rate technique was used to maintain the global type I error rate at the desired level. The normality assumption was verified using the Shapiro-Wilk's statistic and the homogeneity of variances was verified graphically with the residuals plots. The interpretation of the results was done at the 0.05 level of significance and all ANOVA analysis was done using SAS software (SAS Inc., NC).
Results
Presence of Dystrophin-Positive Fibers After Transplantation of Normal C57BL/10J MPCs in mdx EDL
In the present series of experiments, myoblasts were coinjected with cardiotoxin to induce muscle fiber damage and thus increase the fusion of the transplanted cells with the existing muscle fibers. Two months after the myoblast transplantation, the EDL muscles were prepared to evaluate the contractile properties, as describe in Materials and Methods. Subsequently, muscles were frozen and 10-μm transverse sections were collected on slides for dystrophin immunohistochemistry. Very few dystrophin-positive fibers (approximately six per muscle) were observed in the EDL of mdx, mdxTmdx, or cardiotoxin-damaged groups, these being likely “revert-ant fibers” (11). In the mdxT10J group a range of 38–59% of muscle fibers were dystrophin positive, with a mean of 44.6 ± 10%. These hybrid dystrophin-positive fibers were formed by the fusion of the donor MPCs with the host muscle fibers (Fig. 1).
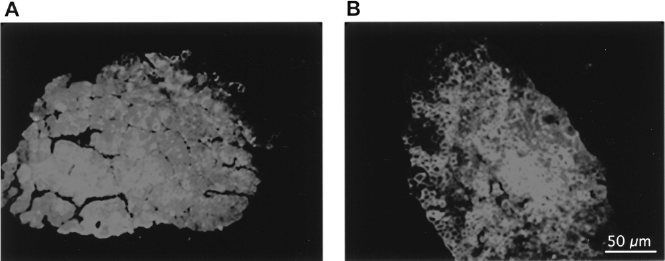
Immunofluorescence showing dystrophin-positive myofibers. (A) Representative cross sections of mdx, mdxTmdx, or cardiotoxin-damaged EDL. Only one dystrophin staining can be observed. (B) A high percentage of dystrophin-positive fibers was observed in mdx EDL transplanted with 10J myoblasts. The average percentage of positive fibers was 44 ± 10%. Scale bar (A, B): 50 μm.
Transplantation of Normal C57BL/10J MPCs in mdx EDL Is Not Sufficient to Increase the Total or the Specific Force
To test whether the coinjection of MPCs and of cardiotoxin has an effect on the contractile properties of the mdx EDL, we injected both EDL of mdx mice. The right muscle was grafted with normal C57BL/10J MPCs (mdxT10J) and the left muscle with mdx MPCs (mdxTmdx) (i.e., cells that did not contain the normal dystrophin gene). Mice were sacrificed 2 months after the transplantation to permit good muscle regeneration after the cardiotoxin damage. Contractile properties of untreated EDL of mdx mice (mdx) and of EDL injected only with the cardiotoxin solution (cardiotoxin damaged) were also measured. This last group was included to verify whether the damage induced by cardiotoxin could influence the contractile properties in EDL of mdx mice. Muscles were stimulated at three different frequencies (80, 120, and 150 Hz). All treated groups were compared to the untreated mdx group. As observed in Figure 2A, no significant force difference was observed between the different groups, even in those injected with cardiotoxin alone. We observed the same phenomena for all three frequencies tested (80, 120, and 150 Hz).

Dystrophin expression did not increase the muscle absolute force (g) or the muscle specific force (N/cm2). The force–frequency curves of mdx, mdxTmdx, mdxT10J, and mdx cardiotoxin-damaged groups were compared (A). No significant difference was observed on absolute force between these groups. (B) The specific force was calculated by dividing the absolute force by the muscle cross-sectional area. There was also no significant difference of the specific force between the different groups. The slight changes observed between mdxTmdx, mdxT10J, or cardiotoxin-damaged EDL were not significant. However, the mdxT10J group differed significantly from the mdx group.
The force was normalized to the cross-sectional area (CSA) to verify whether significant differences could be observed between the different groups. No significant increase of the normalized force was observed. Thus, in these experimental conditions, coinjection of normal MPCs with cardiotoxin did not induce a force increase in mdx muscles. This means that dystrophin expression in mdx muscle is not sufficient by itself to induce a force increase. Moreover, as observed in Figure 2A and 2B, a force decrease can be observed for all treated groups, compared to the untreated mdx group. This trend was observed at all stimulation frequencies but was not statistically significant except for the mdxT10J group. However, the strength of the mdxT10J was not significantly lower than the strength of the muscles injected only with cardiotoxin. This may be due to the extensive muscle fiber damage produced by cardiotoxin.
Transplantation of Normal C57BL/10J MPCs in mdx EDL Increases Muscle Resistance to Eccentric Contractions
Based on previous results, showing no force increase, we verified whether the presence of dystrophin increased the muscle resistance to eccentric damage. To verify this hypothesis, the EDL muscles from myoblast-transplanted and nontransplanted mdx mice were submitted to an eccentric contraction protocol. The EDL was stimulated at 150 Hz for 700 ms; during the last 200 ms of stimulation, the EDL muscle was stretched from its optimal length (Lo) to 110% of Lo (Fig. 3A). A total of seven stimulation/stretching cycles were applied to each EDL. Eccentric contraction occurs when a muscle is contracting and lengthened simultaneously. Such forced lengthening damages the myofibers and reduces subsequent tetanic force development.

Dystrophin expression resulted in a better protection of EDL against eccentric contractions in mdx mice. (A) Schematic representation of the eccentric contraction protocol. Before the contraction, muscle length was adjusted at Lo. The stimulation of the muscle began at 200 ms; the muscle was stimulated at 150 Hz for 700 ms. After 500-ms stimulation, muscle was stretched at the speed of 50% Lo/s, to 110% of Lo. At the end of the stimulation, muscle length was returned to Lo at the same speed. For each EDL, a total of seven eccentric contractions were performed. The change of tetanic force between each contraction reflects the degree of muscle damage. (B) EDL eccentric contraction protocol was performed on each different group and compared to mdx EDL, without treatment. The tetanic tension developed during the first cycle was designated as 100%. ***The curve obtained with myoblasts obtained from C57BL/10J mice (i.e., normal myoblasts containing the normal dystrophin gene, mdxT10J) is statistically different from the EDL of mdx (not transplanted and not treated with cardiotoxin), from the EDL of mdx mice damaged with cardiotoxin and transplanted with mdx myoblasts (mdxTmdx) (p < 0.0023, p < 0.0177, and p < 0.0477, respectively). Differences observed between mdx and mdxTmdx or cardiotoxin-dam-aged mdx were not statistically different.
In Figure 3B, all treated groups (mdxT10J, mdxTmdx, and cardiotoxin damaged) were compared to the untreated mdx group. As observed in Figure 3B, only the curve representing the mdxT10J was significantly (p < 0.0023) different from the mdx curve. At each of the seven contractions, the percentage of force drop was always lower for the mdxT10J group than for the mdx group. At the seventh contraction, the force developed by the mdxT10J group was almost twice that developed by the untreated mdx group. Moreover, the mdxT10J curve was also significantly different from mdxTmdx and cardiotoxin-damaged groups (p < 0.0177 and p < 0.0477, respectively). The force drops for the mdxTmdx and cardiotoxin-damaged groups were not significantly different from those observed in the untreated mdx mice. Thus, although MPC transplantation did not induce a force increase, dystrophin expression protected the muscle fibers from damage induced by eccentric contractions.
Discussion
The most accepted physiological role attributed to dystrophin is to provide mechanical reinforcement of the sarcolemma during myofiber contraction. Lack of dystrophin in DMD patients may induce myofiber fragility, leading to a progressive degenerative process. Mdx mice are used as a model of DMD and knowledge of the contractile properties of dystrophin-deficient muscles is largely derived from experimentation on the mdx EDL muscle (23). Many groups have studied different potential therapies in mdx mice. Despite the different therapeutic approaches investigated, or the protocols to study muscle force or muscle damage, the same result seems to be obtained (i.e., dystrophin restoration in mdx muscles protects them from damage induced by stress) (7, 19, 33). However, few studies have focused on the effect of MPC transplantation on the contraction properties of muscles.
In this study, we analyzed the contractile properties of EDL muscles of mdx mice after the transplantation of MPCs combined with a cardiotoxin-induced damage. We thus compared three groups of treated muscles to a group of mdx untreated muscles (mdx); 1) mdx mice transplanted with normal C57BL/10J MPCs (mdxT10J), 2) mdx mice transplanted with mdx MPCs (mdxTmdx), and 3) mdx mice injected with only the cardiotoxin solution. First, we studied the effect of transplantation on the contractile properties of mdx EDL by verifying the absolute and the specific force. A high percentage of dystrophin-positive fibers was observed in EDL grafted with normal 10J MPCs. This high transplantation success was obtained because of the coinjection of the myoblasts with cardiotoxin, which damaged the host muscle fibers and thus favored the fusion of the donor myoblasts with these fibers. However, the consequent expression of dystrophin was not sufficient to increase the force of the mdx muscle. This may be due in part to the extensive damage induced by cardiotoxin.
This result is in agreement with those obtained by Ambrosio et al. (1, 2) following transplantation of normal muscle-derived stem cells (MDSC) in EDL to introduce the dystrophin gene in the mdx fibers, they also evaluated the effect of this transplantation on the muscle contractile properties. No statistical force difference was observed in the specific titanic force between the mdx muscles transplanted with normal MDSC and the mdx muscles injected with PBS. Thus, by itself the presence of dystrophin in the muscle fibers did not increase the strength. However, when their mice were submitted to an overload protocol, consisting in the ablation of the antagonist muscle, in their case the tibialis anterior, the muscles transplanted with MDSC produced a significantly greater specific force than the overloaded muscles simply injected with saline. Thus, in their protocol the presence of dystrophin protected the muscles against the loss of force induced by the repeated muscle fiber damages induced by the overload.
Surprisingly, we observed a slight decrease in force after our different treatments (mdxT10J, mdxTmdx, cardiotoxin-damaged). The trend appears to be independent of cell transplantation, because we observed the same phenomenon in EDL injected with only the cardiotoxin solution. When compared to the mdx group, the mdxTmdx and the cardiotoxin-damaged groups were not significant. However, a significant difference was observed for the mdxT10J group compared with the mdx group (see Fig. 2). The force diminution seems to be due to the limited muscle regeneration of mdx mice. This trend in the force diminution is in accordance with other studies. Indeed, Irintchev et al. studied the functional and structural recovery of mdx soleus after damage and observed that the recovery of dystrophic muscles was less efficient than that of normal mice, suggesting an exhaustion of the regenerative capacity in mdx muscles caused by previous cycles of degeneration and regeneration (10, 15). Moreover, in our study, we used 12-month-old mice and recently Chan et al. (10) have demonstrated that fiber branching may play a role in the pathogenesis of muscular dystrophy in mdx mice. In fact, at 7 months, roughly 90% of muscle fibers are branched, resulting in a higher force deficit than in the littermate controls. The decrease of total and specific forces in mdx mice in our study can be explained by an inefficient regeneration after the extensive damage caused by cardiotoxin and the numerous MPC injections. Those damages may have accelerated the development of branched fibers, inducing a small force reduction compared to the untreated mdx group.
Other authors demonstrated that dystrophin-deficient mdx muscle fibers were preferentially vulnerable to experimental damage and that the restoration of the dystrophin in the mdx muscle can increase its resistance to exercise (7, 19, 33). Our results confirm these findings, because we observed that the restoration of dystrophin expression in roughly 45% of the myofibers of mdx EDL gave it a protection from contraction-induced injury (Fig. 3). The comparison of the mdx group and the mdxT10J group clearly showed that the consequence of normal 10J MPC transplantation is a better protection from damage induced by eccentric contractions. Because no statistical difference could be observed between the mdxTmdx or cardiotoxin-damaged groups with the mdx group, we can easily attribute the higher resistance to eccentric contraction of mdxT10J group to the restoration of dystrophin expression. Supporting this observation, the mdxT10J group was also significantly different from the mdxTmdx and cardiotoxin-damaged groups.
Taken together, these observations indicate that dystrophin restoration is sufficient to protect muscle fibers from damage induced by an eccentric contraction protocol. However, the dystrophin restoration is not sufficient by itself to increase the strength of the muscle in mdx mice, perhaps because this animal model does not have an important muscle weakness. Myoblast transplantation should be eventually combined with another therapeutic strategy to increase muscle strength. One of them is to block the myostatin signal. Our group, for example, has induced muscle hypertrophy and improved the success of myoblast transplantation by blocking myostatin with follistatin (3, 4). Qiao et al. (24) have produced an AAV vector containing a mutated myostatin (MSTN) propeptide resistant to proteolytic cleavage. Thus, the propeptide inhibits the MSTN signal by blocking the MSTN interaction with its receptor. By treating mice with the AAV vector containing the propeptide, Qiao et al. (24) were thus able to induce muscle hypertrophy, increasing the strength observed in a grip test. Absolute and specific forces were also higher in the AAV-treated group. Therefore, for an eventual treatment of DMD patients, the sole restoration of dystrophin may not be sufficient to increase the strength of a muscle, but this treatment may increase the muscle resistance to damage. This will, over time, probably result in stabilization of the muscle strength and perhaps in a strength increase. In a clinical setting, it may also be possible to combine the protective effect induced by the restoration of dystrophin expression with an induction of muscle fiber hypertrophy to further improve the muscle strength.
Footnotes
Acknowledgments
This research project was supported by grants from the Association Française contre les Myopathies (AFM) and from the Jessee's Journey. Dr. Tremblay is the president of CellGene Inc., a biotechnology company involved in the development of cell and gene therapies for dystrophies. This work was supported by a fellowship (F.M.) and grants from the Association Française contre les Myopathies, and grants from the Muscular Dystrophy Canada, and the Canadian Institute for Health Research.
