Abstract
Since their derivation 11 years ago, human embryonic stem (hES) cells have become a powerful tool in both basic biomedical research and developmental biology. Their capacity for self-renewal and differentiation into any tissue type has also brought interest from fields such as cell therapy and drug screening. We conducted an extensive analysis of 750 papers (51% of the total published about hES cells between 1998 and 2008) to present a spectrum of hES cell research including culture protocols developed worldwide. This review may stimulate discussions about the importance of having unvarying methods to culture hES cells, in order to facilitate comparisons among data obtained by research groups elsewhere, especially concerning preclinical studies. Moreover, the description of the most widely used cell lines, reagents, and procedures adopted internationally will help newcomers on deciding the best strategies for starting their own studies. Finally, the results will contribute with the efforts of stem cell researchers on comparing the performance of different aspects related to hES cell culture methods.
Introduction
Human embryonic stem (hES) cells are isolated from the inner cell mass of the blastocyst. They are pluripotent and, while kept in ideal conditions, can differentiate into specific cell types within the three germ layers and some extraembryonic lineages (21, 30, 49, 53, 63). These properties make hES cells a promising tool for cell-based therapies and regenerative medicine (33, 45). The majority of the studies about hES cells focus on their self-renewing capabilities, their pluripotent state, and differentiation.
The attention towards embryonic stem cells began over 25 years ago, after the establishment of pluripotent stem cells from mouse embryos (17). While the first human embryonic stem cells lines were derived only in 1998 (57), several groups have derived original hES cell lines since then (9, 24, 49, 59, 60).
Gradually, countries have established their own policies regarding the use of hES cells, which differs worldwide (18). In some countries, laws were created by governmental funding availability, while in others, legal conditions were developed to encourage or discourage governmental and private funding (11). For instance, until 2008 hES cell research in the US was allowed but not eligible for federal government funding, unless the used lines had been derived before August 9, 2001. By now the situation is changing with the US government bringing in new legislation to modify how federal funding of hES cell research occurs (27).
In Brazil, research with hES cells was approved in March 2005, for cell lines derived from embryos frozen at least 3 years. However, just after the bill was passed, a petition challenged the law. In 2008, Brazil's Supreme Court rejected the petition, ruling that embryonic stem cell research should be allowed (32, 55, 58). Countries like the UK, Sweden, Singapore, Israel, and Australia are among the most liberal. On the other hand, Switzerland, Spain, and France allow stem cell research with some limitations. Austria and Germany are restrictive; German scientists, for example, are not allowed to work with cell lines produced after January 1, 2002 (52).
Despite differences in the policies surrounding the field, it is clear that the interest in hES cells increases around the world. In this way, many groups have developed methods for derivation and maintenance of such cells (35, 38).
In the beginning, hES cell culture techniques were based on procedures designed originally for mouse embryonic stem cells (23), including the use of mitotically inactivated mouse embryonic fibroblasts (MEFs) as feeder layers (49, 57). hES cells were cultured in medium containing fetal bovine serum (FBS) and leukemia inhibitory factor (LIF). Later, researchers discovered that hES cells are not dependent on LIF (10, 23, 28) and, instead, fibroblast growth factor-2 (FGF-2) was the main factor that sustains pluripotency (62).
Over the years, hES cell culture procedures have undergone numerous modifications in order to improve handling and cell propagation, and at this time several protocols are available (39). This inevitable scenario of multiple ways to culture hES cells led to a new challenge: the comparison among findings obtained using multiple hES cell lines, propagation methods, media, supplements, feeder cells, or substrates may contribute to generate skewed conclusions.
In order to identify trends regarding hES cell culture methods, we analyzed 750 articles published by researchers from different regions, highlighting the differences and similarities within the adopted procedures. Our evaluation focused primarily on the fundamental variations for culturing hES cells, such as cell lines, media, substrates, reagents, and propagation methods.
Methodology
The articles were accessed directly through the “PubMed Central” (www.ncbi.nlm.nih.gov/pubmed) and the search criteria were based on the keyword “human embryonic stem” for articles published between 1998 and 2008. This search revealed a total of 1,473 articles. In this review, the scientific articles with free access were used, including the journals that most published in this research field (24). Our survey resulted in a total of 750 articles from journals listed in Table 1, which represents ~51% of the total articles found in PubMed Central. Only articles that had used hES cells as the subject of the study were considered. For all analyzed parameters, a cut-off point of at least five publications for a given culture method was considered in this review and the items with five or less total occurrences were put together as “others” in the figures.
Journals and the Total Number of Analyzed Articles in the Survey

The journals are listed in descending order of number of publications.
Results and Discussion
Since the primary study conducted by Thomson et al. in 1998 (57), the number of hES cell publications has seen an extraordinary increase, mainly concentrated in the last 5 years (Fig. 1). This trend in the number of papers published between 1998 and 2008 was probably due to the period requested by various groups to learn and adapt their technique to culture hES cells. Indeed, it is probable that specific grants to fund stem cells research released in this period and permissive laws in some countries have contributed to the increase as well. Besides, the distribution and derivation of cell lines worldwide and the increasing number of groups working with hES cells may also have had an effect in the number of publications (1, 11, 38, 43).

Distribution of hES cell publications worldwide. Number of publications between 1998 and 2008 achieved by the cited search criteria are expressed as the total amount of papers per year, and separated by all continents: North America, Europe, Asia, Oceania, Latin America, and Africa.
From Thomson's work (57) to now, the US leading position is evident. Compared with the other continents, North America, with the US as its primary contributor, can easily be considered the world leader in the number of publications, with Europe and Asia coming in second (Fig. 1). Furthermore, even though the continents of Europe and Asia have both gained significant ground in the number of hES cell publications within the same time frame during the last 5 years, they display very different publication profiles. Up to 2007, Europe was clearly led by the UK, which has been responsible for ~40% of the total number of publications from this continent. By 2008, other countries also started to publish significantly and the UK contribution dropped to 30% in relation to whole Europe. Sweden came in second in the number of publications from this continent (Fig. 2A). On the other hand, in Asia there is not a clear leading country. The first position is split among different countries, with a great contribution of Israel, Singapore, Korea, China, and, more recently, Japan and India (Fig. 2B). The observed discrepancies in publication numbers worldwide may be a consequence of differences in available funding opportunities, accessibility of techniques for manipulating hES cells, or legal issues among countries (11, 35). These trends provide a clear indication of the state of the art of hES cell research in the world. However, the current trend is more likely expected to evolve during the next few years, especially considering the fact that new research groups are interested in working with hES cells around the world.

Leading countries in Europe and Asia. (A) Total number of European hES cell publications per year. (B) Total number of Asian hES cell publications per year.
Our data indicate that the only continents that had not published articles with hES cells until 2008 were Africa and Latin America (Fig. 1). Brazil has changed this scenario, with its first article published in 2009 (19).
The nontrivial differences in developmental potential among hES cell lines pointed to the importance of screening and deriving new cell lines (4, 46). One interesting feature of working with hES cells is that each cell line has a different genetic background, and this characteristic may reflect on their behavior. The increasing amount of cell lines used per year (Fig. 3A), especially after 2003, could be indicative of researchers' efforts to increase the number of hES cell lines, for three main reasons: facilitating research with hES cells, facilitating the comparison between cell lines, studying the capacity of a variety of lines to differentiate into a specific cell type. In this review we observed that a total of 244 cell lines were used in articles published between 1998 and 2008, which represents ~35% of the total registered cell lines (56). The discrepancy between the number of cell lines used and registered ones may indicate that the majority of the cell lines have not been adopted by academic laboratories.
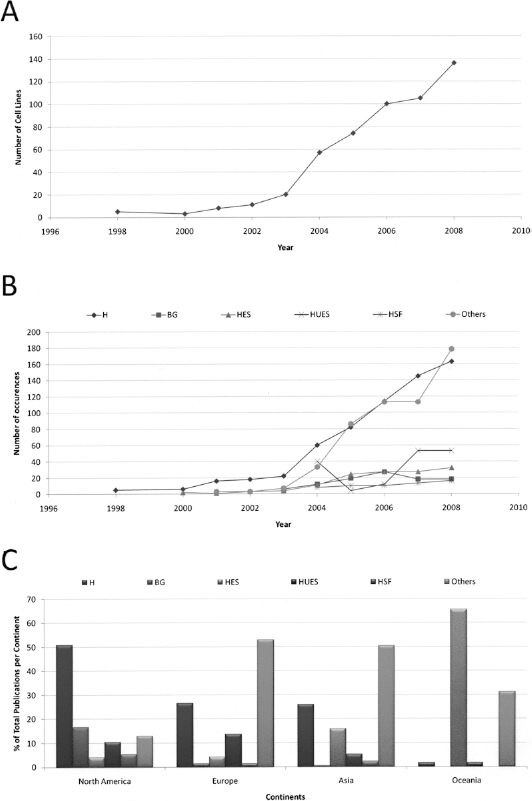
Description in peer-reviewed journals of new derived and most used human embryonic stem cell lines. (A) Total cell lines described in the world over the years. (B) The most published groups of hES cell lines: H group, BG group, HES group, HUES group, HSF group, and other groups. (C) Percentage of the groups of cell lines used in different continents: H group, BG group, HES group, HUES group, HSF group, and other groups. Numbers are expressed as the total amount of articles containing cell line information.
Although each cell line has its own characteristics, historically multiple lines have been derived with similar protocols and by the same research group; therefore, they were grouped together in our analysis. Among the different groups of cell lines derived, certain ones should be highlighted (Fig. 3B): H group (57) (WiCell Research Institute), HES group (49) (ES Cell International), BG group (40) (BresaGen), HUES group (9) (HUES Cell Facility), and HSF group (2) (University of California at San Francisco). All the published groups of hES cell lines other than these were placed as “others” in this analysis. Overall, the H group (i.e., H1, H9) leads, followed by HES (i.e., HES1, HES2), BG (i.e., BG01, BG02), HUES (i.e., HUES1, HUES3), and HSF (i.e., HSF1, HSF6).
We also analyzed the frequency in the use of individual cell lines. We found that the H1 and H9 (WiCell) cell lines were notably the most used ones (data not shown). These lines were described in the first article of human embryonic stem cells (57) and precursors of oligodendrocytes derived from H1, for the first time worldwide, will be used in a clinical trial for human spinal cord injury by Geron (5, 22).
When the distribution of cell lines per continent is taken into consideration, we observe regional preferences for their uses (Fig. 3C). In North America, the H group is more frequently used, and this profile might be explained by its availability and by the past policy of NIH funding. In Europe and Asia the use of other cell lines, generally locally derived, was more prominent. Oceania, represented by Australia, uses the HES group more than other cell lines (1). This trend could be explained by the high costs in order to import cell lines from WiCell, which were distributed mainly in the US (38) and authorized for research by NIH.
With regard to the culture methods, adherent substrate and feeder layers were narrowed down to four major categories: mouse embryonic fibroblasts (MEFs), Matrigel (a matrix derived from mouse sarcoma, composed mostly by laminin, collagen IV, and heparan sulfate proteoglycan), human feeders (represented by human fibroblasts or human mesenchymal cells), and STO (a cell line derived from mouse embryonic fibroblasts). Despite the attempt to cultivate hES cells in animal-free or defined conditions (6, 15, 36, 44, 50, 61), MEFs remain the most widely used substrate, which is probably due to their efficacy and lower costs for keeping pluripotent hES cells, when compared to other methods (Fig. 4A). Several strains of mice have been used to prepare MEFs and CF1 is the most frequently used one (data not shown). Regardless of the extensive use of MEFs, the uses of Matrigel and human feeders have increased in the last years (Fig. 4A). These may reflect an intent to culture hES cells for clinical purposes or an attempt to reduce the expression of unwanted immunogenic molecules (37), although these unwanted molecules could only be vanished by complete from the culture with the removal of nonhuman products (12).
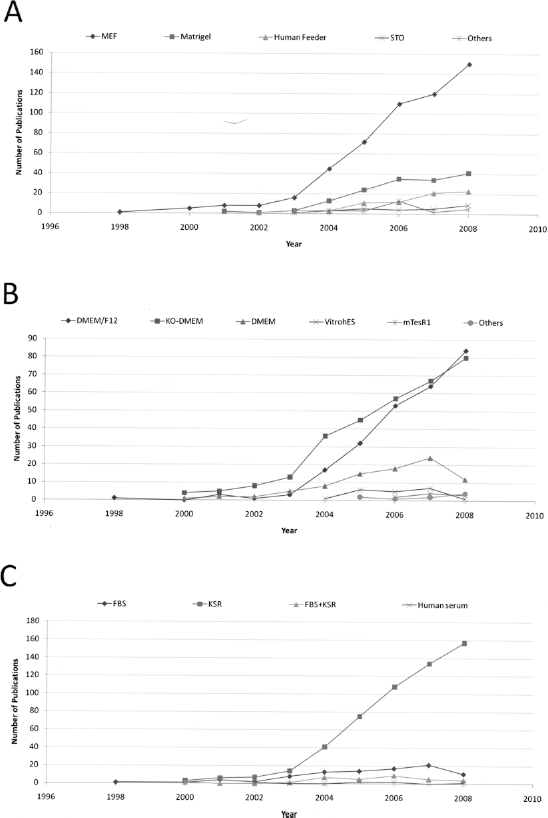
Fundamental components for growing hES cells. (A) Different substrates of adhesion: MEF, Matrigel, human feeders, STO, and others. (B) Media used in the hES cell culture: DMEM/F12, KO-DMEM, DMEM, VitrohES, mTeSR1, and others. (C) Serum and/or serum replacement used as supplement: FBS, KSR, FBS + KSR, and human serum. Numbers are expressed as the total amount of articles with enough information per year.
When the basal media are taken into consideration, we found that the most commonly used are knockout-DMEM (KO-DMEM) and DMEM/F12 (a mixture of DMEM and Ham's F12) (Fig. 4B). The use of these media could be explained by the following reasons. Knockout-DMEM is a basal medium with reduced osmolality, which reproduce the embryonic microenvironment and may better maintain hES cells in a undifferentiated state; DMEM/F12, richer in nutrients, is also the medium recommended by WiCell, which distribute the most used hES cells. According to our data, the defined medium most frequently used for culturing hES cells is mTeSR1. Two others defined media (the X-VIVO system and the chemically defined medium CDM) had also appeared in the survey, but less than five times between 1998 and 2008 and were included as “others” in Figure 4B. In contrast to “DMEM-based” media, which in general are supplemented with serum or serum replacement, these defined systems use recombinant growth factors and other components such as insulin, transferrin, and albumin, without the use of serum/serum replacement.
In addition, the use of knockout serum replacement (KSR) in hES cell cultures has been prevalent since the earliest years (Fig. 4C). Other sources, like fetal bovine serum (FBS), are still in use, but at a lower frequency. This is one factor that has dramatically changed in the hES cell culture, as the first articles with hES cells used FBS (49, 57).
This issue can be explained by the fact that KSR represents a way to achieve reproducible culture conditions. FBS, on the other hand, has unknown components, which can contribute to variations in the culture conditions. Furthermore, studies showed that FBS contains factors that may lead to hES cell differentiation (29, 31). In an attempt to achieve a xeno-free culture, some groups have been studying alternatives, including human serum or recombinant human factors to replace the main components of serum (48). In this way, KSR represents the first step for the transition to a culture medium free of animal components (7).
Fibroblast growth factor-2 (FGF-2) is one of the most important factors for keeping pluripotency of hES cells. In order to optimize hES cell culture conditions and maintain the pluripotent state, FGF-2 needs to bind its receptor and activate the ERK1/2 pathway (14). In our survey we found that the concentration of 4 ng/ml is the most used one (Fig. 5A) and has been accepted as sufficient for maintaining the cells on MEFs. However, it was evident by the data analyzed here that if hES cells were grown in feeder-free conditions without MEF conditioned medium, higher concentrations of FGF-2 are necessary (Fig. 5B) (6, 34, 61). These differences observed in FGF-2 concentration did not seem to have a correlation with the cell line used but with the use of MEFs or MEF conditioned medium. Matrigel and others matrices, despite of acting as an adherent substrate, do not produce/stabilize the factors in the way that MEFs and other feeders do (8, 14).

FGF-2 concentrations and other factors used in the hES cell culture. (A) The most used FGF-2 concentrations to culture hES cells: 4 ng/ml, 5 ng/ml, 8 ng/ml, 10 ng/ml, 12 ng/ml, 20 ng/ml, 40 ng/ml, and others. (B) Analysis of FGF concentrations used to culture hES cells in feeder-free conditions: with MEF conditioned medium, fresh medium, without conditioning by MEFs. (C) Other factors added in the hES cell culture: LIF, ITS, TGF-β, activin A, heparin, and noggin. Numbers are expressed as the total amount of articles with enough information per year.
In addition to FGF-2, some research groups have used other factors as supplements in culture. As briefly mentioned, LIF was erroneously thought to be the key factor in maintaining hES cells in a pluripotent state (10, 23, 28). In spite of this, LIF is the factor with major incidence in the protocols analyzed, followed by the combination of insulin, transferrin, and selenium (ITS), even though the use of this mix have fallen in the last 2 years (Fig. 5C). Activin A, TGF-β, heparin, and noggin have also appeared in our survey as complementary factor to maintain pluripotency (Fig. 5C). Other factors, such as IGF, IL-6, heregulin, and inhibitors of signaling pathways were observed in a reduced number and were not included in the analysis.
An important issue regarding the hES cell culture is the maintenance of their genetic stability even after long periods in culture. A few years ago, aneuploidy, a phenomenon characterized by loss or gain of chromosomes, was described in hES cells. Different groups showed aneuploidy such as trissomies of chromosomes 12 and 17 (13), an extra chromosome X (41), or gain of chromosome 20 (51) in hES. It has already been demonstrated that the propagation of hES by enzymatic means, with trypsin for example, can contribute to aneuploidy (25, 26, 40, 42). In our research, we have noticed a broad range of propagation methods. The most commonly employed techniques include the use of collagenase or mechanical dissociation (Fig. 6). While mechanical dissociation is a way to propagate hES cells minimizing microenvironmental disturbance (42), the preference for collagenase over other methods may have been influenced by the following: 1) simplifying routine for expanding hES cells when compared to mechanical dissociation, which is very laborious and time consuming depending on the amount of samples to be propagated (42); 2) the adverse effects of collagenase on hES cells are less prominent than trypsin (4), which may actually contribute to a higher occurrence of aneuploidy (7, 25); and 3) collagenase specifically cleaves collagen chains, while trypsin, with its wider spectrum of cleavage sites, may affect cell membrane proteins. In 2007, a new enzymatic mean, using a recombinant enzyme replacement for cell dissociation (TrypLE™) was found in our survey (Fig. 6). Some articles have suggested that this method does not cause karyotype abnormalities, increases colony formation (16), and easily detaches cells from adherent surfaces, thus bringing to the field a new alternative for passing hES cells (47).

Propagation methods for culturing hES cells. Numbers are expressed as the total amount of papers containing each technique per year: mechanical, collagenase, trypsin, dispase, cell dissociation buffer, TrypLE™, and others.
With so many different possibilities to culture hES cells, we decided to compare in detail protocols for culturing H1 and H9 cell lines, as they are the most published hES cell lines in the world (data not shown). Among the publications using H1 and/or H9 cell lines, 250 articles contained enough data for gathering information on protocols (Table 2). Protocols used exclusively for H1 or H9 or as a combination of both cell lines were analyzed. We found 61 different protocols for H1, 95 for H9, and 94 protocols for H1 + H9. Despite the great variability found within the H1 and H9 culture methods, it was possible to highlight 12 protocols with more than four occurrences among the cultivation of H1 and H9 alone or in combination (Table 2). The use of KSR as supplement, MEFs as substrate, collagenase as passage method, and 4 ng/ml of FGF-2 are the most commonly found similarities. The use of DMEM/F12 or KO-DMEM as culture media is the most significant difference. DMEM/F12 is mostly used in H1 cultures while KO-DMEM is mostly used in H9 and H1 + H9 cultures.
Description of the Most Used Protocols for H1 and H9 Cell Lines

Beyond the presented data from 1998 to 2008, we also analyzed articles published from January to August 2009. In spite of the numerous protocols, we noted that during these first 8 months of 2009 the tendencies were kept. We observed that MEFs still are the most used substrate to culture hES cells, and the media and supplements more frequently used are DMEM/F12 or KO-DMEM supplemented with KSR and 4 ng/mL of FGF-2, with collagenase as enzyme for propagation of the cells. To better understand and compare the data from January to August 2009 with the whole year of 2008, Table 3 presents the values of the items analyzed in this review as percentages.
Percentage of Publication of the Different Parameters Analyzed in 2008 and January to August 2009

To compare the data obtained in the first 8 months of 2009 with the whole year of 2008, the values were expressed in percentage of publication or occurrence.
In our own laboratory, reagents that best suit the requirements for the H9 cell line culture are: inactivated MEFs (C57/B16 strain) as the feeder layer in DMEM/F12 supplemented with 8 ng/ml of FGF-2 and 20% KSR, using TrypLE™ to dissociate the cells during passage.
Conclusion
Culturing hES cells with such a variety of protocols indicates the capacity of these cells to adapt into different conditions. As refereed by the number of protocols used for culturing H1 or H9 (Table 2), the capacity of hES cells to adapt to different substrates (as Matrigel or human feeders) and to defined media and supplements (as mTeSR and KSR) (20, 36, 54, 61). However, given all these available types of culture conditions, it is challenging to compare the results obtained by different research groups around the world. Therefore, the data presented in this review may contribute to the International Stem Cell Initiative (ISCI) (http://www.stemcellforum.org/isci_project.cfm), whose aim is to establish common standards and procedures between laboratories, upon comparing the performance of different media and other aspects related to the culture of hES cells.
The ISCI has published an article focusing on the characterization of 59 hES cell lines maintained under different culture conditions (3). Several similarities in gene and antigen expression were observed in these cell lines, as well as significant differences in this expression, that may be specific to some lines, but may also be caused by different culturing conditions (3). Beside this, differences in hES cells microenvironment can cause variations in their molecular signature, as observed by the differential expression of 1,417 genes in cells grown in serum-containing medium compared with those cultured in serum replacement medium (54). Indeed, standardization of protocols used for culturing hES cells may help with the reproducibility issue and also when comparing results obtained by different research groups.
The study of hES cells brings great perspectives not only to regenerative medicine but also for drug discovery and screening as well as studies of human development. The specific needs in growing these cells for use in basic or applied research are different, stimulating the development of protocols that best suit specific goals. With the aim of using hES cells for cell replacement therapies, a great effort has been made to eliminate animal components from culture. On the other hand, scientists focusing on basic research seek to reduce culture costs and to achieve effective culture conditions (an example is the number of publications using MEFs as a substrate even nowadays, representing an almost fourfold increase in relation to Matrigel culture publications) (Fig. 4A). In this regard, it will be imperative that researchers make sure that cells cultured in media containing animal-derived components will have the same characteristics when adapted to a xeno-free equivalent condition.
Herein, by describing trends for culturing hES cells, we hope to provide valuable information regarding the most adopted procedures to culture hES cells. Scrutinizing 750 published articles revealed important parameters in hES cell culture methods. This review could be used as a tool to help researchers already working in the field, and specially also newcomers who want to start their research.
Footnotes
Acknowledgments
This work was supported by grants from Faperj—Fundaç ão Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro; CNPq—Conselho Nacional para o Desenvolvimento Científico e Tecnológico (S.R.), and the Brazilian Ministry of Health/DECIT (S.R.).
