Abstract
Despite recent advances and promising results in children, liver cell transplantation (LCT) should still be regarded as an experimental therapy. Several substantial complications are known from animal studies and individual patients. However, safety data on liver cell infusion in children are scarce. We used LCT in four children of different ages (3 weeks to 11 years, 3–40 kg) and underlying diseases [acute liver failure (n = 1), urea cycle disorders (n = 2), and Crigler-Najjar syndrome (n = 1)]. Vital parameters, portal vein flow (PVF), portal vein pressure (PVP), and liver enzymes were measured every 5 min during cell application and hourly thereafter between applications. An application protocol with discontinuation rules depending on changes in PVF and PVP was developed and successfully applied. Application was feasible in all children despite the catastrophic overall condition of the patient with acute liver failure. No application-related changes in vital parameters were found, and none of the children experienced clinical signs of portal vein thrombosis, pulmonary embolism, or anaphylactic reactions. Time courses for changes in PVF, PVP, and liver enzymes were obtained. Thorough monitoring of portal vein pressure and duplex sonography according to a defined protocol is likely to increase safety of cell application in pediatric LCT.
Keywords
Introduction
Liver cell transplantation (LCT) is an innovative therapy for the treatment of various acute and chronic liver diseases. It can be used in patients that otherwise would not be considered for a traditional liver transplantation. Furthermore, it is especially tempting to use the technique in pediatric patients because it is much less invasive than whole-organ transplantation. Since its first use in a boy with a urea cycle disorder in 1997, about 30 children underwent LCT (6). This very first pediatric patient died from complications of a routine liver biopsy 42 days after LCT. Although it is very unlikely that the fatal course was directly related to the procedure of cell application, it enforces the question what safety issues have to be dealt with to use this still experimental technique in children.
Three main complications of LCT have been identified in preclinical animal studies: portal vein thrombosis, pulmonary embolism, and elevation of liver enzymes. Microembolization of portal vein branches is expected and even desired during hepatocyte infusion, because local ischemia has been shown to improve cell engraftment (12). However, thrombosis of larger branches or even the portal vein stem has to be regarded as a serious complication. As it is thought that formation of portal venous thrombosis would be paralleled or favored, respectively, by a decrease in local blood flow, portal vein flow (PVF) and/or portal vein pressure (PVP) have been studied in several animal models of LCT (8, 13, 18, 24, 33). Portal vein flow velocity, measured by Doppler ultrasound, has been shown to be inversely related to PVP (13, 18). In all these animal studies, transient increases of PVP and/or decreases of PVF were observed. Moreover, it could be shown that the increase of PVP was directly related to the amount of cells given (18). Shunting of infused hepatocytes into the systemic circulation (i.e., pulmonary embolism) is only expected when infusing very large amounts of hepatocytes in the presence of already substantial occlusion of the portal vein bed (23, 26). Elevation of liver enzymes after cell application was found in some animal studies (8, 13, 18, 26). Similar to the changes in PVP, the levels returned to baseline levels a few days after LCT.
In contrast to these findings, safety data from human LCT are scarce. For a safe use of human LCT, especially in children, it is of utmost importance to thoroughly monitor the patients for signs of significant adverse effects. In this article, we present a comprehensive set of safety parameters that was measured in four children with different ages and different underlying diseases.
Materials and Methods
Patients
Since 2006, four children underwent LCT in our institution. All investigations were done in accordance with the ethical guidelines given in the 6th version of the Declaration of Helsinki. Patient characteristics are given in Table 1. Three children had liver-based inborn errors of metabolism [urea cycle disorders in two patients (15), defective bilirubin conjugation in one child], and one child was treated for acute liver failure from neonatal herpes infection. The overall condition was normal in the three metabolic patients before elective LCT. In contrast, the infant girl with acute liver failure was in catastrophic clinical state (dependent on mechanical ventilation, inotropes, hemofiltration, and plasma substitution), and LCT was performed under emergency conditions as a desperate therapeutic attempt.
Patient and Cell Characteristics

Liver Cell Transplantation
Cryopreserved human liver cells were provided by Cytonet GmbH&Co KG (Weinheim, Germany). In all four children, a portal vein catheter was introduced surgically into the middle colic vein as previously described (16). We used single-lumen Broviac catheters (Lifecath Broviac Expert, Vygon, Aachen, Germany) with diameters of 4.2F (patients 1–3) and 5.0F (patient 4), respectively. All cell applications were done at our pediatric intensive care unit. Vital parameters were recorded continuously every 5 min during application and every 60 min thereafter. Patient 1 had an arterial line for recording of arterial blood pressure; in the other patients it was recorded noninvasively.
Cell doses, viability after thawing, donors, mean duration of the applications and volume of the cell suspensions are given in Table 1. The mean concentration of total/viable cells/ml was 7.8–15.7 × 106/5.7–9.5 × 106, which is consistent with previously published protocols (2.4–22.0 × 106 total cells/ml) (1, 2, 7, 17, 21, 22, 27, 30, 31). After thawing, the cells were manually infused over 10–40 min. The maximum application rate was 2.0 ml/min in the patients with 4.2F catheters, and 6.1 ml/min in the patient with the 5.0F catheter. These application rates were based on in vitro data about mechanical damage to the infused hepatocytes by shear forces (14). To prevent intravasal clotting of the cells, heparin was added to the cell suspension at a concentration of 2 U/ml.
Tacrolimus (Prograf™, Astellas, Tokio) and methyl-prednisolone (Urbason™, Sanofi-Aventis, Vienna) were used for immunosuppression in all patients according to common protocols in pediatric liver transplantation. In patients 3 and 4, basiliximab (Simulect™, Novartis, Basel) was given additionally at days 1 and 4 of cell applications.
Portal Vein Pressure
Portal vein pressure could not be measured in patient 3 due to technical problems. In the other three patients, it was recorded every 5 min during application and every 60 min thereafter. We used a standard pressure transducer connected via a three-way valve to the patient monitor. Between applications, the portal vein catheter was continuously rinsed with a solution containing heparin to prevent clotting.
Portal Vein Flow Velocity
Doppler ultrasound was performed by an experienced pediatric radiologist. Because of easier measurement, portal hemodynamic was analyzed by max flow velocity (cm/s) instead of portal flow rate (ml/min). Patient 3 occasionally needed light sedation to facilitate ultrasound examinations, as described before in pediatric LCT patients (5). Maximum flow velocities were measured in the portal vein stem 3–5 cm before branching and its main left and right branches directly after branching every 5 min during application. Doppler ultrasound was performed with a high-end ultrasound system (Antares, Acuson Siemens) with a 2.5–6.67 MHz curved array transducer.
Application Protocol
A protocol was used during all cell infusions that defined tolerable changes in PVP and PVF. An increase in PVP and/or a decrease of PVF velocity of more than 50% was used as the cut-off for temporary discontinuation of the application procedure. If these changes returned to 25% compared to the initial values, the infusion could be continued.
Statistics
All statistical analyses were done with the SPSS 11 software package (SPSS Inc., Chicago, IL). Changes of vital parameters during cell applications were examined with Student's t-test for paired samples. Flow velocities in the portal vein stem and its main branches were compared with the t-test for unpaired samples. The two-tailed Pearson test was used to calculate correlations between PVP and Vmax. For all tests, statistical significance was assumed at a level of p < 0.05.
Results
Vital Signs
Mean values of heart rate, blood pressures, oxygen saturation, and body temperature before and 5 min after the end of the cell applications are given in Table 2. Systolic, diastolic, and mean blood pressure significantly decreased in patient 1 during cell applications. However, the changes were only small (5–8%) and thus are not regarded clinically relevant. No changes in blood pressure were noted in the other three patients. Heart rate, oxygen saturation, and body temperature were unaffected by cell application in all four patients. The time interval between two applications was 8–12 h in patient 1, 12–24 h in patient 2, and 24 h in patients 3 and 4. No significant changes in any parameter were found in these observation periods (data not shown).
Vital Signs

Values are expressed as mean and SD of all cell applications.
p < 0.05.
p < 0.01.
Portal Vein Flow
The maximum flow velocities (Vmax) during cell application are given in Figure 1. Vmax before cell application was similar in the right and left portal vein branches. In the portal vein stem, flow velocity was significantly higher in the patients with metabolic diseases (patient 2 and 4: p < 0.01; patient 3: p < 0.05). Interestingly, no differences could be detected in the girl with acute liver failure (Fig. 1, patient 1).

Maximum flow velocities (Vmax) in the portal vein stem (A), and its right (B) and left (C) main branches during cell applications. Values are expressed as mean and SEM. Open circles: patient 1 (acute liver failure); filled circles: patient 2 (CPS1 deficiency); open triangles: patient 3 (citrullinemia); filled triangles: patient 4 (Crigler-Najjar syndrome).
Looking at the mean values of all applications, no significant changes in Vmax could be observed at any 5-min time point (Fig. 1). In individual applications, increases (maximum 113% of baseline values) as well as decreases (maximum 50% of baseline values) of Vmax were observed at the end of the infusions. The maximum decrease between two 5-min values was 54%. A discontinuation of the infusion for 5 min as foreseen in the application protocol was necessary only three times in 29 cell infusions.
Portal Vein Pressure
The changes in PVP during cell applications are shown in Figure 2. Measurement of PVP was not possible in patient 3 due to the position of the portal vein catheter. In the remaining three patients, a significant increase of PVP of 24–36% was observed during the infusions. This increase lasted for 6 h after cell application before it returned to baseline values (Fig. 2).
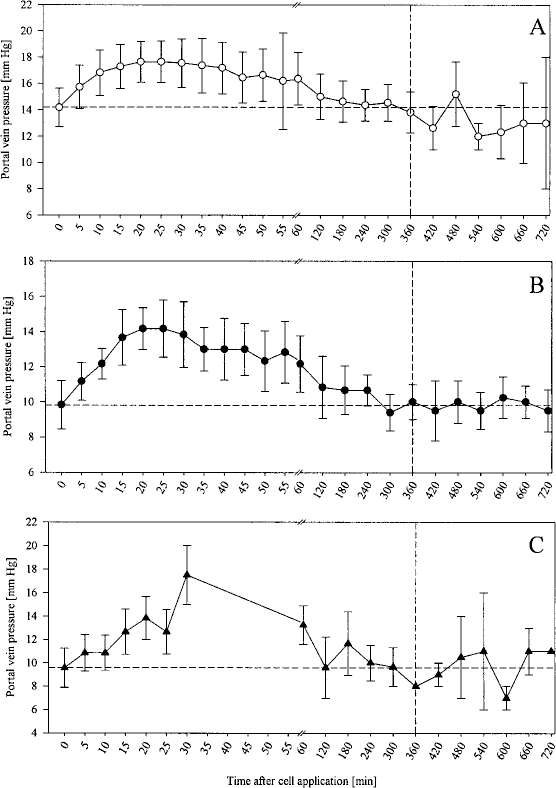
Portal vein pressure (PVP) during cell applications. Values are expressed as mean and SEM. (A) Patient 1 (acute liver failure), (B) patient 2 (CPS1 deficiency), (C) patient 4 (Crigler-Najjar syndrome).
During the individual sessions, increases as well as decreases in portal vein pressure were noted. Maximum decrease of PVP was 27% (compared to baseline value) and 46% (compared to the immediately preceding value), respectively. The increases were more distinct, up to 200% (baseline value) and 100% (preceding value). Pauses in the infusions according to the application protocol were sporadic, but more frequent than those caused by decreases of Vmax. Their typical duration was 1–2 min with a maximum of 5 min before the application could be continued.
Correlation Between PVP and PVF
Values of PVP and Vmax were correlated for each application time point. Three individual significant correlations (one positive, two negative) were found in different patients and different sections of the portal vein and thus are not thought to be relevant. These findings correspond well to the differences in Vmax and PVP before and after cell application. Whereas PVP values uniformly increased during application sessions and needed 6 h to return to baseline levels, no significant changes in Vmax were obeserved. Thus, a positive or negative correlation between PVP and Vmax could not be expected.
Liver Enzymes
We determined levels of lactate dehydrogenase (LDH), aspartate aminotransferase (AST), and alanine transaminase (ALT) over a period of 24 h after cell application. The results are shown in Figure 3. In patient 1 (acute liver failure; ALF), liver enzymes were grossly elevated due to the underlying disease, and were therefore not included in the analysis. In the remaining three patients, similar patterns were found. LDH and AST values initially increased and returned to baseline values after 8 h. ALT levels decreased more slowly, and did not reach baseline values at the time of the next application, thus the baseline increased. However, ALT values normalized within 7 days after the last cell application in all patients (data not shown).

Liver enzymes after cell applications. Figures are expressed as mean and SEM of all applications. Filled circles: patient 2 (CPS1 deficiency); open triangles: patient 3 (citrullinemia); filled triangles: patient 4 (Crigler-Najjar syndrome).
Clinical Outcomes
In the patient with acute liver failure, beneficial effects of the transplanted cells (reduction of hyperammonaemia, decrease of plasma demand) were only transient, and the patient died after 11 days from a complicating hemophagocytic histiolymphocytosis. Some aspects of the clinical courses of patients 2 and 3 have been published (15). In both children, metabolic crises ceased after LCT. While the older girl is still on conventional therapy 30 months after LCT, the younger boy received a liver graft 15 months later. The Crigler-Najjar patient experienced a 20% reduction of his bilirubin levels and is listed for liver transplantation. No signs of portal vein thrombosis have been observed in frequent ultrasound investigations during the follow-up of the patients. In the explanted liver of patient 3, no thromboses or infarctions were detected. Autopsy was rejected by the parents of the ALF girl; a small postmortem liver biopsy did not show any vessel obstructions.
Discussion
Fortunately, clinically significant side effects of LCT have only very rarely been observed since the first use of the technique in humans in 1993. Besides the boy already mentioned who died from complications connected to a scheduled liver biopsy (31), deaths only occurred in patients with acute liver failure. However, these patients died from progress of their underlying disease, not from application-related complications. Another adult patient developed a complete thrombosis of the portal vein. Of note, LCT in this case was done for graft failure after OLT, and a preexisting stenosis of the portal vein anastomosis had to be dilated prior to application. Not surprisingly, the thrombosis developed at this predamaged location (3). Thrombosis of a mesenteric vein in another adult subject is mentioned in the review by Fisher and Strom (6), but no details are given. However, in the explanted livers of three children that received a whole liver graft subsequently to LCT, hepatocyte-containing thromboses were found in several small portal vein branches (11, 20, 22). We therefore think that at least formation of portal vein thrombosis remains a significant risk in pediatric LCT, and thus special attention should be paid to avoid it.
According to Virchow's triad, there are three factors contributing to thrombosis: venous stasis, endothelial injury, and hypercoagulability. Hypercoagulability of liver cell suspensions can be suspected from in vivo data on tissue factor-derived activation of coagulation by isolated hepatocytes (30). Besides these findings, stasis is the most significant risk factor for portal vein thrombosis in human LCT. PVF (by means of duplex sonography measurement of flow velocity) and PVP have been determined in several animal studies. The first observations are reported by Grossman et al. in three baboons treated with genetically modified hepatocytes. They found an increase in PVP up to 3.9-fold after LCT compared to baseline values, which normalized after 3–13 days (8). Rajvanshi et al. measured PVP in rats after repeated cell infusions and did not find significant differences from the control group. However, these were long-term observations 12 weeks after LCT (24). More detailed observations are given by Kocken et al. in dogs (13), Muraca et al. in pigs (18), and Yu in a rat model (33). In all three studies, PVP increased up to fivefold shortly after the start of the cell applications. An increase of the PVP baseline was observed in the pig model when cell infusions were repeated within 90 min (18). In two studies on dogs and pigs, respectively, it was shown that PVF was inversely proportional to PVP (13, 18) (i.e., PVF decreased during hepatocyte infusion and increased again to preapplication levels soon after the end of the infusions). In a rabbit model, Schneider et al. compared changes of the PVF during infusion with saline, hepatocytes, or macroalbumine aggregates. In the saline group, PVF increased steadily during the infusion interval, while it decreased in the groups treated with hepatocyte and macroalbumine aggregates, respectively. They speculate that the adaptive increase of PVF that was observed during saline infusion was suppressed by changes of the portal vasculature caused by the corpuscular suspensions (26).
Although changes in PVF during cell application can be easily determined by duplex sonography, the method is not widely used in human LCT. Some authors investigated the anatomy and the PVF prior to cell transplantation (1, 7, 21, 28), the Brussels group additionally confirmed the position of the portal vein catheter by using microbubbles (19). Absolute values for Vmax are only reported in two cases. Sokal et al. found a Vmax of 25–30 cm/s, which did not significantly change during cell application in a 4-year-old girl with Refsum's disease (27), while Muraca et al. observed a transient decrease of Vmax from 14 to 6 cm/s and a normalization within 24 h in their adult patient (17). In two other pediatric patients, Vmax did not change during cell application, but the authors do not give exact figures for Vmax (28, 29). The normal range for Vmax in the portal vein as determined in 61 healthy children is 40 ± 14 cm/s independently of age (9). In our three metabolic patients, Vmax was within this normal range. However, it was significantly lower in the patient with acute liver failure. Interestingly, the differences between portal vein stem and its main branches that were found in the other children were absent in this patient. Most probably, this reflects structural changes of the severely damaged liver. In all our patients, no significant decrease was observed in the mean Vmax values (Fig. 1), which is consistent with the literature data. However, individual applications had to be paused due to reductions in Vmax of more than 50%.
Published data on PVP in human LCT are available for 18 patients so far. PVP temporarily increased during cell application in 5 of 6 adults (17, 25) and 11 of 12 children (2, 5, 7, 11, 20–22, 27, 31). The extent of the PVP increase was between 50% and 500%. In all patients, PVP almost or completely returned to baseline values, but unfortunately the exact time spans are not given. However, this time span is of special importance, because actual LCT protocols foresee fractionated application of cells over several days to minimize adverse reactions. It was shown in preclinical studies that elevated PVP needs several hours to normalize after cell infusion, otherwise an up-shift of the PVP baseline will occur (13, 18). We therefore monitored PVP very thoroughly during and after each cell application and found that preapplication levels of PVP were reached only after 6 h. This should be the minimal time span between two applications in children. Keeping to our application protocol, mean PVP increase was only 24–36%.
Surprisingly, the inversely proportional relationship between PVP and PVF known from animal studies does not seem to exist in human LCT. Whenever measured, the expected transient increase in PVP was observed, but significant changes in Vmax were absent. Our detailed measurements confirm these observations. Whereas the increase of PVP reflects that something is happening inside the liver, the unaffected PVF seems to indicate that LCT is safe with regards to portal vein thrombosis.
Shunting of liver cells through the sinusoids into the systemic circulation seems to be a rare phenomenon even in animal models of LCT. It rats and rabbits, it could be demonstrated that hepatocytes only enter the pulmonary circulation at very high doses of cells and macroalbumine aggregate particles, respectively (23, 26). However, in a pig model of acute liver failure, hepatocytes were identified in the lungs of all seven animals, even at moderate cell doses. Pulmonary artery pressure transiently increased by up to 62%, but no clinical symptoms were observed (18). In another study, a monkey died with signs of respiratory distress 2 h after cell infusion, but thrombosis of lung vessels was not found at autopsy (32). In human LCT, pulmonary shunting of hepatocytes was suspected in two adult patients with acute liver failure and proven in one of them at autopsy (4). However, both subjects received huge doses of liver cells in these pioneer days of human LCT, about 50–100 times higher than foreseen in actual pediatric protocols. No more pulmonary complications have been reported since. However, published data on pulmonary monitoring are scarce. Normal oxygen saturation (2) and normal pulmonary artery pressures (7), respectively, have been reported in two children. In our patients, oxygen saturation was monitored thoroughly, and significant changes were not noted during any cell infusion. We therefore believe that more invasive techniques such as pulmonary artery catheters or PICCO are not necessary for routine use of LCT in children.
In some preclinical studies, transient elevations of liver enzymes have been observed (8, 13, 18, 26). Levels of AST, ALT, and LDH increased up to 20-fold shortly after cell infusion, and normalized after 1–28 days. Cho-line esterase, bilirubin, and bile duct enzymes remained unchanged. Only three authors reported changes in liver enzymes in human LCT. Fox et al. observed an eightfold increase of AST that lasted for 38 h, and a threefold increase of ALT that normalized after 5 days (7). Sokal et al. measured AST levels of 1733 U/L and ALT levels of 2260 U/L after cell infusion, but unfortunately they do not give preapplication levels. On day 10 after LCT, normal values were measured again (27). The same group reported threefold increases of AST and twofold increases of ALT in another pediatric patient. However, these are mean values of different applications over different periods of time (29). The changes observed in our patients were within these reported ranges, although much more detailed (Fig. 3). Our data shows that LDH normalizes within 12 h and AST after 24 h. ALT levels do not reach preapplication values after 24 h; thus, an up-shift of the baseline will result from multiple applications, even if only one application per day is performed. In all of our patients (as in the reported cases), enzyme levels normalized after a few days, and no clinical signs of liver dysfunction were noted. From animal studies, it is known that only about 30% of the transplanted cells engraft permanently while the rest is subject to rapid lysis (10). Therefore, it can be speculated that the transient rise of liver enzymes is partly caused by cell lysis, and partly reflects subclinical liver damage caused by the microembolization that is necessary for cell engraftment.
Although our patient series is small, we conclude from the consistent findings that LCT is a safe technique for the use in small and even critically ill children and infants. However, substantial complications have been observed in animal studies as well as in individual patients. Therefore, thorough monitoring of the application procedure is required. PVP seems to be the most significant parameter, although online visualization of the portal vein patency by means of Doppler ultrasound is a very useful additional technique. Our application protocol for PVF and PVP was feasible with individual variations in the application procedure, which in our belief increased patient safety. This protocol is being further tested in a prospective study on LCT in neonatal urea cycle disorders.
Footnotes
Acknowledgments
J.M. and G.F.H. are scientific consultants for the Cytonet GmbH that provided the cryopreserved human liver cells.
