Abstract
Liver transplantation is the only existing modality for treating decompensated liver cirrhosis. Several factors, such as nonavailability of donors, combined with operative risks, complications associated with rejection, usage of immunosuppressive agents, and cost intensiveness, make this strategy available to only a few people. With a tremendous upsurge in the mortality rate of patients with liver disorders worldwide, there is a need to search for an alternative therapeutic tool that can combat the above limitations and serve as a supportive therapy in the management of liver diseases. Cell therapy using human fetal liver-derived stem cells can provide great potential to conservatively manage end-stage liver diseases. Therefore, the present investigation aimed to study and prove the safety and efficacy of human fetal liver-derived stem cell transplantation in patients with end-stage liver cirrhosis. Twenty-five patients with liver cirrhosis of different etiologies were infused with human fetal liver-derived stem cells (EpCAM+ve) labeled with Tc-HMPAO through hepatic artery. Our high throughput analysis using flow cytometry, RT-PCR, and cellular characterization exemplifies fetal liver cells with their high proliferation rate could be the best source for rejuvenating the diseased liver. Further, no episodes related to hepatic encephalopathy recurred in any of the subjects following hepatic stem cell transplantation. There was marked clinical improvement observed in terms of all clinical and biochemical parameters. Further, there was decrease in mean MELD score (p < 0.01) observed in 6 months follow-up in all patients. Therapy using human fetal liver stem/progenitor cells offers a potentially supportive modality to organ transplantation in the management of liver diseases.
Introduction
Late-stage progressive hepatic fibrosis characterized by distortion of hepatic architecture, necrosis of hepatocytes, and formation of regenerative nodules leads to cirrhosis. Orthotopic liver transplantation (OLT), which includes cadaveric LT, split LT, and living donor LT (LDLT), brings hope to patients with these diseases. Globally, 1.4 million deaths occur annually as a result of chronic liver diseases and approximately 150,000 patients succumb to liver cirrhosis in India alone. The reasons for this high death toll include unavailability of healthy liver donor and highly expensive liver transplantation treatment. Furthermore, some other factors such as operative risks and posttransplant rejection are major limitations of OLT. Hepatocyte transplantation is emerging as an alternative bridge support until a healthy donor is arranged (14, 25), but mature hepatocytes have short-term survival and low proliferation both in vitro and in vivo. Recently, a study demonstrated the effect of bone marrow cell (BMC) transplantation in a chronic liver diseased murine model but failed to show reversal of cirrhosis (17).
Hepatic progenitor cells derived from murine embryonic stem cells showed less engraftment with limited capacity of liver tissue formation in recipient livers (22). Recent studies on isolation of hepatic progenitors have shown promising results to overcome these limitations (6, 33, 34). They actually give rise to cells that are functionally equal to mature hepatocytes found in the adult liver and suggest that these can be useful cell source for developing therapeutic strategies to treat various liver disorders (27).
In humans, the ductal plates of fetal liver and Canals of Herring to the adult liver are the richest source of human fetal liver-derived hepatic stem cells (hHSCs) (2, 19, 20, 29, 31, 35). These hHSCs express several specific markers, namely epithelial cell adhesion molecule (EpCAM), neural cell adhesion molecule (N-CAM), and cytokeratin CK-19 (1, 7, 12, 20, 30). The ability to purify hepatic stem cells has been made possible by using immunoselection for a defined EpCAM antigen (4). Adult liver and neonatal liver consist of low percentages (1.7–2.5%) of hHSCs (EpCAM+ve) (19), whereas human fetal liver serves as the richest repository of these cells, accounting to about 12%. These cells possess high proliferative capacity, are less immunogenic, and more resistant to cryopreservation and ischemic injury, properties that could enhance their engraftment within the recipient liver. The self-renewal capacity of hHSCs is indicated by phenotypic stability after expansion for >150 population doublings in a serum-free, defined medium and with a doubling time of ~36 h. In addition, immunophenotypic studies on EpCAM+ve cells revealed that these cells do not express HLA-DR marker but express liver-specific markers that envisage progenic potential that can be implemented for therapeutic intervention (11, 18).
Our previous report on intrahepatic transplantation of hepatic progenitors in Crigler-Najjar syndrome patients successfully demonstrated a decline in total bilirubin and elevation in conjugated bilirubin (10). Although considerable progress has been made in hepatic stem/progenitor cell transplantation in preclinical studies, there is lack of clinical safety data to treat end-stage liver cirrhosis. In a recent study, Kang et al. reported an effective noninvasive imaging procedure to localize transplanted cells in the liver of mice using 99mTc-Ceretec® labeling (9). Therefore, the present study was designed to evaluate the long-term safety and efficacy of human fetal liver-derived hepatic stem cell transplantation to the patients with end-stage liver cirrhosis via the hepatic artery. In addition, tracking of cells through noninvasive imaging was also performed in order to localize the transplanted cells in the liver.
Materials and Methods
Patient Selection
Twenty-five male subjects (50.6 ± 8.53 years; range 38–60 years) with established cirrhosis and having an average disease length of 5.8 ±1.0 years were included in the study. Written informed consent of the patients was obtained after briefing them about the objectives of the study. The study protocol was approved by the Institutional Ethics Committee, Deccan College of Medical Sciences, Hyderabad, India.
Patients were categorized into four classes on the basis of clinical conditions and Mayo's Model for End Stage liver Disease (MELD) score, which was calculated by the formula [0.957 × ln(serum Cr) + 0.378 × ln(serum bilirubin) + 1.120 × ln(INR) + 0.643] x 10 [Cr = creatinine, INR = International Normalized Ratio (prothrombin)]. MELD Class I: 16–19 (n = 5); Class II: 20–24 (n = 7); Class III: 25–28 (n = 6); Class IV: 29–34 (n = 7).
Investigation of the underlying etiology revealed chronic alcohol intoxication in 23 patients, one each of hepatitis B (MELD score 28) and hepatitis C (MELD score 25) virus-related cirrhosis, respectively. Eighteen of the 25 patients (72%) included in the study presented with ascites. Two patients in group I and three patients in group II showed mild to moderate ascites (grade 1 and 2), respectively, whereas 13 patients [group III (n = 6) and group IV (n = 7)] showed gross ascites (grade 3) with distended abdomen. Ultrasound examination of the abdomen showed splenomegaly in all groups with shrunken liver and portal hypertension in all the patients of groups III and IV. Encephalopathy was observed in two subjects in group III and four subjects in group IV; however, recurrent episodes of encephalopathy were seen in one patient in group II.
Cell Harvest and Characterization
Human fetal livers were obtained from spontaneous abortions from 16 to 20 weeks of gestation. Liver cells from a single fetal liver were used solitarily for the transplantation. All donors of the fetus were thoroughly screened serologically for syphilis, toxoplasmosis, rubella, hepatitis B and C, human immunodeficiency virus 1, cytomegalovirus, parvovirus, and herpes simplex types 1 and 2. Liver cells were isolated using two-step collagenase perfusion method as described elsewhere (8). Briefly, resected livers from the aborted fetuses were collected under sterile conditions within 2 h of the termination of pregnancy and then cut into thin slices (1–2 mm thickness). Thereafter, each fetal liver was digested with 0.02% collagenase type IV (Sigma Aldrich Inc., St. Louis, MO, USA) containing penicillin G (10 U/ml), streptomycin (10 U/ml), amphoteracin B (0.025 μg/ml) at 37°C for 30 min. Single cell suspension of liver was collected by centrifugation at 250 × g and then washed with sterile phosphate-buffered saline (PBS, pH 7.0) (Sigma Aldrich). Cells (8 × 109) were taken using hemocytometry (Marienfeld, Germany) and then 80 μl of FcR (Miltenyi Biotec, Germany) blocking reagent and 80 μl of anti-CD326 (EpCAM) microbeads (Miltenyi Biotec, Bergisch Gladbach, Germany) were added and finally incubated at 4°C for 30 min. The CD326+ve cells were sorted by using the magnetic associated cell sorter (MACS) according to the manufacturer's instructions (Miltenyi Biotec; http//www.miltenyibiotec.com). The enriched CD326+ve cells were then washed with Dulbecco's modified Eagle's medium (DMEM, Sigma Aldrich Inc.). Subsequently, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was done to assess the viability and proliferation of cells by taking an aliquot of 1 × 106 cells. Immunocytochemical analysis was carried out to determine the albumin, CK18, CK-19, and AFP+ve cells in sorted cells using FITC conjugated anti-human albumin, CK18, CK-19, and AFP under fluorescence microscope (Axio Plan upright; Carl Zeiss Vision Co., Ltd., Hallbergmoos, Germany). Immunohistochemical staining of fetal liver tissue with anti-albumin-FITC was also performed in order to localize and quantify hepatic stem cell proportion in tissue.
Flow Cytometry and Gene Expression Analysis
The enriched CD326+ve cell suspension of 5 × 105 was taken in test tubes for flow cytometry analysis. Cells were stained with FITC-conjugated human anti-CD326 (1:100, Chemicon, USA), anti-CK18 (1:100, BD Biosciences, San Jose, CA, USA), anti-CK19 (1:100, BD Biosciences), anti-albumin (1:100, BD Biosciences) anti-α-fetoprotein (1:100, BD Biosciences) antibodies and were analyzed on a tricolor flow cytometer (FACS Calibur E2390, BD Biosciences) using Cell Quest (Version 7.5.3) software. EpCAM+ve cells were also subjected to RT-PCR using standard procedure to assess the expression levels of all hepatic stem cell markers (18). Amplification was performed using cDNA amount representing 100 ng of total RNA and 1 U of Taq DNA polymerase (Invitrogen, reaction mixture containing 10 mmol/L Tris-HCl, 200 mmol/L dNTPs, and 10 pmol of both forward and reverse primers). The list of oligonucleotide primers used in the study is the same as reported by us previously (18).
Labeling of Stem Cells with 99mTc-Hexamethylpropyleneamine Oxime (99mTc-HM-PAO)
99mTc-HM-PAO mixture was prepared by mixing 99mTc pertechnetate with biocompatible HM-PAO (Ceretec, GE Healthcare, Chalfont St. Giles, UK). EpCAM+ ve-enriched cells (80 × 106) in 5 ml volume were taken and added to 20 mCi 99mTc-HM-PAO and incubated at 37°C for 10 min. Unlabeled 99mTc-HM-PAO was removed by washing with sterile dextrose normal saline (DNS). Labeled cells were resuspended in 5 ml of DNS and transplanted to the patients. The labeling efficiency and viability of cells were analyzed in vitro and biodistribution of cells in the liver was analyzed in vivo after transplantation in patients via hepatic artery.
Transplantation of Fetal Liver Stem Cells
Iohexol (350 mg/ml) (GE Healthcare, Cork, Ireland) was infused through the catheter to locate the hepatic artery. After localization, 99mTc-HM-PAO-labeled fetal hepatic stem cells (EpCAM+ve) of 80 × 106 were infused through the hepatic artery to the liver using femoral artery catheterization. In brief, a mini-guide wire (0.035 in.) was inserted into the femoral artery through puncture needle. Using guide wire support, selective catheterization of hepatic artery was done. Cells were infused through the catheter (5 flinch polyurethane) at the rate of 1 ml/min as a single dose in a sterile condition. During transplantation, hepatic arteriogram was recorded using Recor (Version 4.3) software under Coroskop (Siemens AG, Erlangen, Germany) in a sterile catheterization lab. After the transplantation, appropriate safety measures were taken to stop bleeding from the insertion site. To assess the condition of the hepatic artery, hepatic angiogram was performed after 3 and 6 months.
Posttransplantation Follow-up
Posttransplantation patients were kept under close surveillance in the Intensive Liver Care Unit (ILCU) for 5 days and were subsequently discharged. All the patients were then followed up consecutively for 2 weeks and then every fortnight until 6 months. All relevant biochemical, pathological, and radiological examinations were performed at each visit. Ultrasound was performed to analyze the portal vein caliber and presence of ascitis.
Statistical Analysis
Computed data are presented as mean ± SD. Means of variables at baseline and after follow-up were compared using Student's t-test (paired, two-tailed) and at different intervals by ANOVA. We performed the post hoc Turkey homogeneously significant difference test to determine where the significance lies after ANOVA. Computational analysis was performed using SPSS (Version 16). A value of p < 0.05 was considered to be statistically significant.
Results
Cell Enrichment Recovery and Characterization
Enrichment of EpCAM+ve cells from 16–20-week gestation resulted in the recovery of 10–12% of EpCAM+ve cells in total isolated liver cell suspension. The isolated cells showed 95 ± 2.5% of viability. Sorting of EpCAM+ve cells from 16–20-week gestation fetus resulted in a population predominantly consisting of hepatoblasts and hepatic progenitors. By immunocytochemical analysis, the presence of liver specific markers in these gestational fetal liver was shown. The expression of both albumin (hepatic) and CK-19 (biliary) reflects the bipotential nature of hepatic stem cell towards both hepatic and biliary lineages. Hepatic stem cells (EpCAM+ve) were more intensely stained for ALB and CK19 (Fig. 1), where positive stained cells possessed large nuclei surrounded by a thin rim of granulated cytoplasm. Immunohistochemical staining of liver tissue identified cells with oval-shaped, small-sized and large nuclei to cytoplasmic ratio proved the characteristic feature of the hepatic stem/progenitor cells (Fig. 2). Viability of cells after radiolabeling with 99mTc-Ceretec® remained unchanged.

Liver-specific gene expression analysis by immunocytochemistry of sorted cells on cytospin. The same was counterstained with 4,6-diamidino-2-phenylindole (blue) and observed under a fluorescent microscope. (a) CK-19, (b) CK-18, (c) albumin, and (D) α-fetoprotein. All the positive cells showed large nucleus encircled by thin cytoplasmic rim. Original magnification: 100×.
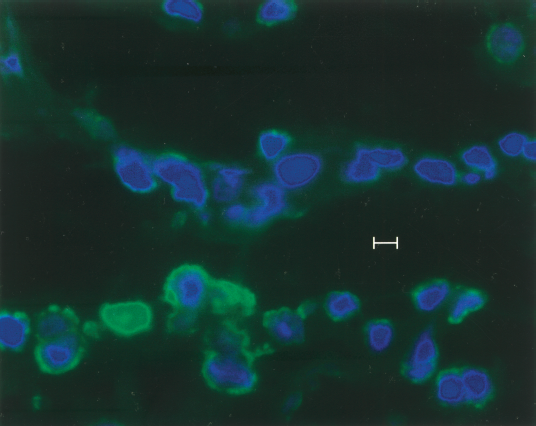
Immunohistochemistry of human fetal liver with FITC conjugated anti-alubmin showing location of hepatic stem cells in ductal plates in the tissue. The same was counterstained with 4,6-diamidino-2-phenylindole (blue) and observed under a fluorescent microscope Original magnification: 40×.
RT-PCR and Flow Cytometric Analysis
Isolated positive fraction of EpCAM cells showed elevated expressions of EpCAM, CK-19 markers with low levels of albumin expression, and intense AFP expression by RT-PCR (Fig. 3). We also analyzed the expression of hepatic progenitor cell markers such as CD29, CD49f, and CD90, and hepatic-specific markers such as albumin and AFP and HLA class I and II in EpCAM+ve cells by flow cytometry. HLA class I and II expression was seen in approximately 1.8% and 0.6%, respectively. HLA class I and II expression levels were not significantly different with respect to their gestational age groups (p < 0.05). We observed strong levels of albumin expression accounting to ~19.2% and 21.3% and AFP showed strong expression levels of approximately 7.2–7.3%, but their expression did not differ significantly in gestational ages of human fetus.
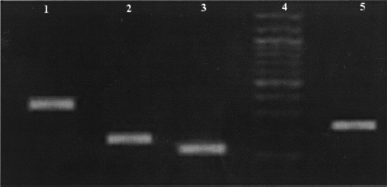
Liver-specific gene expression in sorted cells recovered from liver. RT-PCR products were electrophoresed on 1.5% agarose gel. Lane 1: CK-19, lane 2: albumin, lane 3: CK-18, lane 4: 100-bp marker, and lane 5: α-fetoprotein.
Patients' Performance and Clinical Results
After cell transplantation, all 25 patients demonstrated cumulative decrease in MELD score with increasing time interval (Table 1 and Fig. 4). Means of MELD score, serum bilirubin, SGOT/AST (serum glutamic-oxaloacetic transaminase), and alkaline phosphatase (p < 0.01) decreased after 6 months of follow-up except serum albumin levels, which were found elevated even after 6 months. In MELD score, statistically significant difference was observed between baseline and 4-month and 6-month follow-up (p < 0.05). Difference was also seen between 2 and 6 months in all etiologies. MELD score in hepatitis B and C virus-infected liver cirrhotic subjects was reduced to 19 and 17, respectively (Table 2).

Comparative analysis of biochemical parameters related to MELD score before and after transplantation. Patients were categorized into four groups according to Mayo Model for End Stage liver Disease (MELD) score. Class I: 16–18, Class II: 19–24, Class III: 25–29, Class IV: >30. Reduction in bilirubin and creatinine levels in four groups with respect to inset MELD score (mean) and International Normalized Ratio (mean) levels.
Descriptive Statistics of Clinical and Biochemical (Serum) Variables

MELD, Mayo's model for end stage liver disease; SGOT, serum glutamic-oxaloacetic transaminase; ALP, alkaline phosphatase.
p < 0.01 between baseline and follow-up and difference at different intervals.
Improvement in the MELD Score Among Patients in Various Groups

S, serum; INR, International Normalized Ratio.
Significant difference was observed between baseline and 2, 4, and 6 months and also between 2, 4, and 6 months in the case of SGOT (p < 0.05). In bilirubin levels, significant difference was observed between baseline and 2, 4, and 6 months, and also between 2, 4, and 6 months (p < 0.05). Statistically significant difference was observed between baseline and 2, 4, and 6 months and also between 2 and 6 and 4 and 6 months (p < 0.05) in serum albumin levels. In the case of serum ALP, significant difference was observed between baseline and 2, 4, and 6 months and also between 2 and 4, and between 4 and 6 months (p < 0.05).
No complications related to the procedure were observed during or after hepatic stem cell transplantation. Abdominal ascites in group I and II subjects was absent after 30 days of cell transplantation, whereas in group III and IV patients gross ascites with PHT reduced to moderate levels after 30 days with complete absence at 6 months follow-up was evident by ultrasonographic findings. None of the patients showed symptoms of recurrent hepatic encephalopathy after transplantation.
Hepatic angiogram showed no sign of thrombosis/narrowing/ischemia in the hepatic artery when analyzed after successive time intervals (Fig. 5). Hepatic scintiography showed that transplanted fetal hepatic stem cells homed in the total lobes of liver with a high rate of engraftment, thereby demonstrating the effectiveness of using hepatic artery in the delivery of the cells (Fig. 6). No other clinical complications were observed during and after 6 months of follow-up.

Comparative analysis of hepatic angiograms of the patients completed more than 6 months after hepatic stem cell transplantation. There is no sign observed related to thrombosis/ischemia/narrowing in the hepatic artery.

Hepatic scintiography of the liver. (Left) Anterior lobe after 4 h of hepatic progenitor cell transplantation and (right) anterior lobe after 24 h. Figure shows equal distribution (homing) of 99mTc-HM-PAO-labeled cells in the total liver lobes.
Discussion
Liver cirrhosis is one of the major causes of morbidity and mortality, accounting for eighth largest cause of deaths worldwide. In view of major limitations of orthotopic liver transplantation (36), cell-based therapy is emerging as one supportive therapy in the management of decompensated liver cirrhosis (24, 26). Clinical studies on transplantations of mature hepatocytes in end-stage liver diseases have shown some positive inclination; however, due to the inability of adult hepatocytes to proliferate and the requirement of large cell numbers, several investigators have identified and enriched hepatic stem/progenitors from various liver sources using different phenotypic markers (3, 5, 6, 12–15, 18, 28, 29, 32, 35). Recently, several groups have demonstrated the ability of rapid proliferation and regeneration of enriched liver cells derived from aborted human fetal liver in comparison to the adult liver (6, 12, 16, 18). Earlier pioneering work reported by our group had demonstrated the feasibility of using human fetal hepatocytes in the treatment of acute liver failure (8).
Our earlier investigation reported that the expression of HLA molecules in the total liver cells may be because of hematopoietic cell contamination in early gestation (18). Purification of hepatic stem cells using EpCAM transmembrane glycoprotein immunoselection has been made easier and is a compatible method for the isolation of pluripotent hepatic stem cells.
The gene expression profile and immunocytochemical patterns of EpCAM+ve cells from 16–20 weeks gestation fetal liver revealed the phenotypes of both adult hepatic stem cell and hepatoblasts. Size, morphology, and gene expression profiles of isolated EpCAM+ve cells from 16–20 weeks gestation showed similarity to hepatoblasts, which give rise to hepatocytes and cholangiocytes (19, 23). Presence of this population found in ductal plates of fetal liver is similar to those reported earlier (2, 19, 21, 30, 32, 36).
In the present study, we transplanted human fetal hepatic stem cells in patients with end-stage liver cirrhosis with MELD score >16 (patients with high mortality and for whom liver transplantation is the only proven treatment) and the results obtained are highly encouraging. The maximum amount of cells transplanted was 0.2–0.5% of normal liver mass, which was sufficient to replace the lost function in this disease condition without immunosuppression. We also found that EpCAM+ve cells expressed intermediate levels of HLA class I but not HLA class II. Earlier studies by others and that of ours have demonstrated that EpCAM+ve cells are less immunogenic as they failed to stimulate a significant amount of T-cell proliferation in in vitro mixed lymphocyte reaction (MLR) assay (18–21). Therefore, EpCAM+ ve cells might be ideal candidate donor cells for hepatic cell therapy in liver disorders.
The results obtained in the present study demonstrated that after hepatic stem cell transplantation, patients showed marked clinical recovery and significant decline in the MELD score with time. There was no recurrence of hepatic encephalopathy observed, which suggests marked improvement in the functioning of liver cell.
To our knowledge, this is the first major study to demonstrate the long-term safety and clinical efficacy of hepatic stem cells in the cirrhotic patients of different (both chemical and biological) etiologies. The results of the present study suggests that human fetal hepatic stem cell therapy may serve as effective therapeutic strategy to improve the overall quality of life of patients with end-stage liver diseases without the need for immunosuppression.
In conclusion, the results of the present study successfully demonstrated that transplantation of fetal hepatic stem cells has the potential to overcome the limitations of orthotopic liver transplantation and can serve as a supportive therapy in the management of end-stage liver diseases. Further, the study provides major impetus for successfully using liver stem cells in treating large patient populations that succumb to this disease.
Footnotes
Acknowledgments
This study was funded in part by grant from the ICMR, New Delhi, India and Vesta Therapeutics, Durham, NC, USA to C.M.H. The authors would also like to thank Dr. S. A. A. Lateef, Research Associate, Department of Biochemistry, University of Hyderabad, India for helping with statistical analysis.
