Abstract
Marrow stromal cells (MSCs) are capable of differentiating into various cell types including endothelial cells. Microenvironment is important in cell fate determination. Tumor necrosis factor-α converting enzyme (TACE), a well-characterized “sheddase,” participates in the differentiation process of multiple lineages by the proteolytic release of membrane-bound proteins such as tumor necrosis factor-α (TNF-α). We investigated the endothelial differentiation of MSCs under two coculture conditions: 1) direct MSCs-rat brain microvascular endothelial cells (rBMECs) contact coculture; and 2) indirect coculture of MSCs and rBMECs. Also, we examined the role of TACE/TNF-α signaling in the process of differentiation under direct coculture condition. We found that endothelial differentiation of MSCs was substantially enhanced in MSCs-rBMECs direct contact coculture, but not in indirect transwell coculture condition. Transcript levels of TACE and TNF-α as well as TACE protein expression were significantly upregulated in direct, but not in indirect, coculture condition. Addition of human recombinant TACE promoted gene expression of endothelial specific markers including vWF, CD31, VE-cadherin, Flk-1, and Flt-1 in the differentiating MSCs. Furthermore, inhibition of TACE with TAPI-2 or inhibition of TNF-α with Etanercept attenuated endothelial differentiation of MSCs in the direct coculture condition. We demonstrated for the first time that direct MSCs-rBMECs interaction stimulated the endothelial differentiation of MSCs via TACE/TNFα signaling.
Keywords
Introduction
Marrow stromal cells (MSCs), also referred to as mesenchymal stem cells, are capable of differentiating in vitro and in vivo into various cell types including endothelial cells (12). Because MSCs are relatively easy to acquire, and can be transplanted into the recipients without inducing adverse immune reactions, these cells are an ideal source of exogenous endothelial cells, which can be used for the treatment of endothelial injury-related cardio- and cerebrovascular diseases. However, the low differentiation rate of the MSC greatly limits the potential clinical application for endothelial cell therapy. Therefore, in order to improve the differentiation rate, it is important to optimize the differentiation conditions and elucidate the regulatory mechanisms underlying the differentiation process.
The mechanisms underlying the endothelial differentiation of stem cells are complex, and not fully understood. Previous work demonstrated that various growth factors such as vascular endothelial growth factor (VEGF) are required for endothelial differentiation of MSCs (15, 30, 40). Recently, we observed that simvastatin enhanced bone marrow stromal cell differentiation into endothelial cells through the Notch signaling pathway (39). Some studies indicated that the microenvironment was also important in promoting the differentiation of stem cells into multiple lineages, either via paracrine factors from the neighboring cells or through direct cell–cell interactions both in vitro and in vivo (6, 18, 35). However, it is not clear if the local factors in the microenvironment are also involved in the endothelial differentiation of MSCs.
Tumor necrosis factor-α converting enzyme [TACE; also known as disintegrin and metalloproteinases 17 (ADAM17)] is the first and best-characterized “sheddase” that belongs to the metalloprotease-disintegrin family, and plays important roles in cell–cell interactions and in the processing of protein ectodomain cleavage (4, 42). TACE is critically involved in the proteolytic release of diverse membrane-bound proteins including tumor necrosis factor-α (TNF-α), pro-transforming growth factor-α (pro TGF-α), and L-selectin (27), which are all important factors during mammalian development (24). Recently, it has been reported that TACE gene expression is essential for normal lung branching morphogenesis and cytodifferentiation (42). Mice lacking functional TACE have multiple defects during their development (27, 43), implicating a critical role of TACE in the process of cell differentiation and organ development. As one of the major substrates of TACE, TNF-α is a multifunctional cytokine. In addition to mediating inflammatory responses, TNF-α is involved in the differentiation of a variety of cell types including promotion of osteoclast differentiation from human peripheral blood mononuclear cells (20). TNF-α expression is significantly increased in the myocytes in the early phase of myogenesis (21).
In the present study, we investigated the effect of microenvironment on the endothelial differentiation of MSCs by performing two coculture experiments. We found that endothelial differentiation was significantly enhanced in MSCs-rBMECs (rat brain microvascular endothelial cells) direct contact coculture condition, but not in the indirect coculture system where MSCs and rBMECs were cultured in separate inserts. TACE and TNF-α gene expression were significantly upregulated in direct coculture, but only slightly increased in indirect coculture. In parallel to the change in transcriptional expression, similar change in TACE protein expression was observed in both conditions with significant upregulation in direct coculture system. Addition of human recombinant TACE into the culture promoted the gene expression of endothelial-specific markers in the differentiating MSCs. On the other hand, inhibition of TACE with TAPI-2 or inhibition of TNF-α with Etanercept attenuated endothelial differentiation of MSCs in the direct coculture condition. These results demonstrated for the first time that direct cell–cell interaction stimulated the endothelial differentiation of MSCs via TACE and TNF-α.
Materials and Methods
Culture of MSCs
Rat MSCs (R-048, P4; Cognate BioServices, Inc., Baltimore, MD) were incubated in HyQ MEM Alpha Modification media (HyClone, Logan, UT) with 20% fetal bovine serum (Gibco, Grand Island, NY), and 1% antibiotin (penicillin/streptomycin; Gibco). The cells were maintained at 37°C in 5% CO2/95% ambient mixed air, and split every 2–3 days. All the cells were used within passage 8.
Primary rBMEC Culture
All visible surface blood vessels, midbrain, cerebellum, and the white matter were removed immediately after the brain was obtained surgically from normal rats. The remaining brain tissue was washed twice with wash buffer containing RMPI-1640 (Invitrogen, Carlsbad, CA), 2% FBS, and 1% Pen/Strep to remove the blood. The tissue was cut into small pieces, and briefly homogenized in 2 ml of wash buffer using three strokes of a Wheaton “200” Dounce tissue grinder. The resulting suspension was mixed with a 30% dextran solution, and centrifuged at 10,000 × g for 15 min. The pellet was resuspended in digestion buffer (RPMI-1640 + 2% FBS) containing collagenase/dispace (Roche Science, Indianapolis, IN), and shaken once hourly at 37°C until all visible clumps were digested. The resulting suspension was centrifuged at 1000 × g for 5 min, mixed with Percoll (Sigma, St. Louis, MO), and then centrifuged in a fixed angle centrifuge at 20,000 × g for 20 min at 10°C. The uppermost band was then carefully removed, washed twice with PBS, and centrifuged each time at 2000 × g for 5 min. The final cell pellet was resuspended in endothelial cell growth media [80% MCDB 131 (Invitrogen), 0.15% NaHCO3 (7.5% stock), 10% FBS, 1% endothelial cell growth supplement (15 mg/ml, BD Bioscience, San Jose, CA), 1.5% heparin, 1% penicillin/streptomycin, 5% L-glutamine (200 nM), 0.02% L-ascorbic acid (25 mg/ml), 1% insulin-transferrin-selenium (Sigma), and 0.1% 2-mecaptomethanol (1000×)] (1), and allowed to grow at 37°C in 5% CO2/95% ambient mixed air for 24 h before changing the media to remove all nonadherent cells and debris. The cells used for experiments were from passage 2 to 6.
Endothelial Differentiation of MSCs
MSCs within passage 8 were prelabeled with DiI (1,1′-dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine perchlorate) as per manufacturer's recommendations. Briefly, cells were incubated with DiI (5 μl/ml cell suspension) for 15 min at 37°C. After washing three times with PBS, MSCs were plated in a 75-cm2 flask alone or with endothelial cells. The cells were allowed to grow to 90–100% confluence in full media, and then switched to the differentiation media containing 2% FBS (Gibco) and 1% antibiotic/antimyotic. The cells in different groups were incubated for 7 days in the differentiation media. Experimental groups were described as follows.
Control Group: MSCs Alone. MSCs prelabeled with DiI were plated in a 75-cm2 flask at a density of 6,700 cells/cm2. Cells were allowed to grow in HyQ MEM Alpha Modification media with 20% FBS to 90–95% confluence before being switched to low serum differentiation medium containing 2% FBS. Media was changed every 48 h. The cells were collected in TRIzol Reagent for isolation of RNA and protein after 7 days of culture.
Direct Coculture. MSCs were cocultured with rBMECs in direct contact. After being prelabeled with DiI, MSCs were plated in a 75-cm2 flask at a density of 5,000 cells/cm2. rBMECs within passage 6 were mixed with MSCs at a ratio of 1:1. The cells were allowed to grow in an equal mixture of HyQ MEM Alpha Modification and Dulbecco's modified Eagle's media (DMEM) with 20% FBS and 1% antibiotic/antimyotic to 90–95% confluence. Confluent cells were then switched to low serum (2% FBS) media for 7 days with a media change every 48 h. At the end of the experiment, cells were trypsinized and washed with PBS. FACS (fluorescent activated cell sorting) was performed to separate the DiI-positive and -negative cells. DiI-positive cells were lysed with TRIzol Reagent for the isolation of RNA and protein for real-time PCR and Western blot analysis.
Indirect Coculture. MSCs were cocultured with rBMECs that were separated in transwell inserts. MSCs labeled with DiI were plated in the lower chambers of a six-well plate at a density of 6,700 cells/cm2. Rat BMECs within passage 6 were added into the upper chambers of a Falcon 0.4 mm cell-culture inserts at the same density. After growing to confluence in an equal mixture of HyQ MEM Alpha Modification and DMEM with 20% FBS and 1% antibiotic/antimyotic, cells were switched to 2% FBS media, and maintained in the differentiation media for 7 days. RNA and protein from the cells in the lower chambers were obtained and prepared for real-time PCR and Western blot assays.
Quantitative Real-Time PCR
Total RNA was extracted from rat MSCs with TRIzol (Invitrogen) following a standard protocol (22). Contaminating DNA was eliminated by two sequential steps of DNase (Invitrogen) treatment. The mRNA was reverse transcribed, and the cDNA underwent 40 rounds of amplification (ABI PRISM 7000, Applied Biosystems, Foster City, CA) with the following reaction conditions: 40 cycles of a two-step polymerase chain reaction (95°C for 15 s and 60°C for 60 s) after initial denaturation (50°C for 2 min and 95°C for 10 min) with 1 μl of DNA solution in 2× SYBR green PCR master mix reaction buffer (22). Each sample was tested in triplicate. The relative gene expressions were analyzed using the 2–ΔΔCT method. The authenticity and size of PCR products were confirmed by melting curve analysis (using software provided by Perkin-Elmer). The mRNA levels were normalized by using GAPDH as a housekeeping gene, and compared with the control group. The primers used for amplication were listed in Table 1.
Primers Used for Amplification

Western Blotting Analysis
Protein was isolated from the cells with TRIzol (Invitrogen) following a standard protocol. Protein concentrations were determined by a BCA protein assay kit (Pierce, Rockford, IL). Cell lysates (30 μg protein/lane) were loaded on a 10% sodium dodecyl sulfate-polyacrylamide gel (Bio-Rad), and subsequently transferred to nitrocellulose membranes after electrophoresis. Membranes were treated with blocking buffer (5% skimmed milk in TBS-T: 25 mm Tris-HCl, pH 8.0, 125 mm NaCl, and 0.1% Tween 20) for 1 h at room temperature, followed by incubation with primary antibodies at 4°C for 16 h. The primary antibodies included polyclonal rabbit anti-human vWF antibody (Daco), mouse anti-rat Pecam-1 (CD31) antibody (BD) at a dilution of 1:1000, and rabbit anti-human monoclonal TACE antibody (Santa Cruz Biotechnology, Inc.) at a dilution of 1:500. The membranes were washed three times with TBS-T, and then incubated with horseradish peroxidase-conjugated secondary antibodies (1:10,000) in blocking buffer for 1 h. Signals were developed with Western blotting luminal reagent (Santa Cruz Biotechnology, Inc). Normalization of results was ensured by running parallel Western blots with mouse anti-rat monoclonal β-actin antibody (Abcam) at dilution of 1:10,000. The optical density was quantified using an image processing and analysis program (Scion image, Ederick, MA).
FACS
After 7 days of incubation, cells from each group were trypsinized, and washed with PBS three times. DiI-positive cells from the direct cell–cell contact coculture group were isolated with a BD FACS Calibur cell sorter (BD Biosciences).
Statistical Analysis
Data were expressed as mean ± SE for all experiments. Statistical analysis was performed using one-way ANOVA and least significant difference (LSD) analysis (SlideWrite 5.0 32-Bit Edition, Advanced Graphics Software, Inc., Encinitas, CA). A value of p < 0.05 was considered statistically significant.
Results
MSCs Differentiated Into Endothelial Cells in Direct Cell–Cell Contact, but not in Indirect Coculture Condition
Microenvironment plays important roles in stem cell differentiation either through direct communication between cell membranes or through soluble factors secreted from the neighboring cells (11, 12). To evaluate the effect of microenvironment on cell fate determination, we introduced two coculture systems into our study, and compared the potential of MSCs to differentiate into endothelial cells under these conditions. We found that after 7 days of culture, the transcription levels of endothelial-specific markers including vWF, CD31, VE-cadherin, Flk-1, and Flt-1 were significantly increased by 3.1 ± 0.1-, 3.4 ± 0.7-, 12.6 ± 0.9-, 35.0 ± 1.34-, and 4.0 ± 0.6-fold over control, respectively, in the cells from the direct coculture group as determined by quantitative PCR, indicating an acquisition of endothelial phenotype by the MSCs (Fig. 1A). In parallel, Western blot analysis showed a 2.1 ± 0.15- and 3.8 ± 0.20-fold increase in protein expression of vWF and CD31 within these cells, respectively, compared with the control (Fig. 1B, C). Immunofluorescence staining was performed to further confirm the endothelial phenotype. To rule out the possibility of cross-staining on DiI-positive cells, we prelabeled the endothelial cells instead of MSCs with DiI, and stained the cocultured cells using FITC-conjugated CD31 after 7 days of culture. We observed that 39.3 ± 5.1% of DiI-negative cells (MSCs) from the direct cell–cell contact coculture group were positive for CD31, while only 7.0 ± 1.5% of the cells were positive for CD31 in the control group (Fig. 2), suggesting that direct cell–cell contact between MSCs and rBMECs promote endothelial differentiation of MSCs.
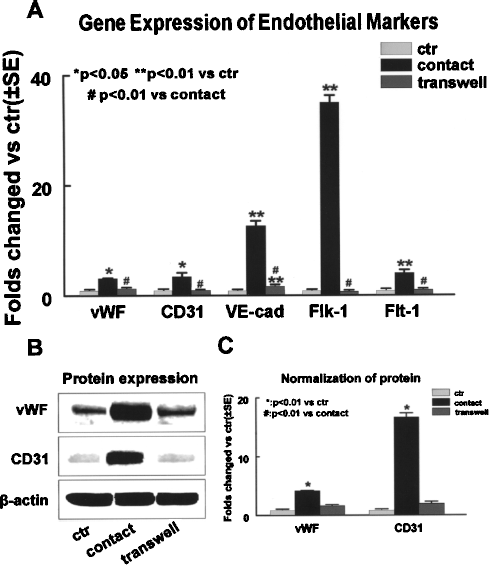
Differentiation of rat MSCs into endothelial cells by day 7 in coculture conditions. (A) Transcript levels of endothelial specific genes detected by Q-RT-PCR. Gene expression of endothelial specific markers including vWF, CD31, VE-cadherin, Flk-1, and Flt-1 was significantly increased in cell–cell contact coculture (direct), but not in transwell coculture (indirect) group. (B) Western blot analysis shows that protein expression of endothelial specific markers (vWF and CD31) was upregulated in direct cell–cell contact culture, but not in transwell coculture system. (C) Normalization of protein expression by β-actin. Data are presented as mean ± SE from at least three independent experiments. Western blot results are representative of three independent experiments. VE-cad: VE-cadherin; ctr: control group; contact: direct cell–cell contact coculture; transwell: idirect transwell coculture.
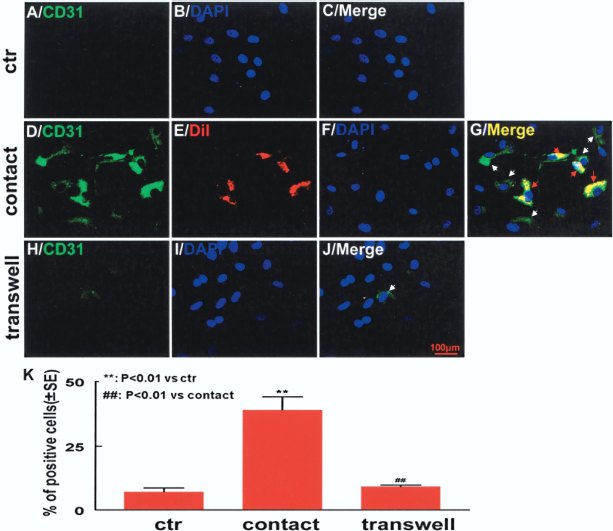
Immunofluorescence staining of CD31. (A–C) Control; (D–G) direct cell–cell contact coculture; (H–J) indirect transwell coculture. Green fluorescence: CD31-positive cells; red fluorescence: DiI prelabeled rBMECs; blue fluorescence: DAPI. CD31-positive cells were significantly increased in direct cell–cell contact, but not in transwell coculture group. White arrows indicate CD31-positive cells; red arrows show CD31-positive rBMECs. (K) Percent CD31-positive cells after 7 days of coculture. Scale bar: 100 μm. Results were from at least three independent experiments.
In contrast, no significant increase in the gene or protein expression of endothelial markers was observed in the cells from the indirect transwell coculture group when compared with control. The changes in transcription levels for vWF, CD31, VE-cadherin, Flk-1, and Flt-1 in these cells were measured using quantitative PCR, and were found to be 1.4 ± 0.05-, 1.0 ± 0.01-, 1.9 ± 0.06-, 0.8 ± 0.08-, and 1.1 ± 0.07-fold over control (p > 0.05), respectively (Fig. 1A). Similarly, Western blot analysis showed no significant changes in protein expression for vWF and CD31 (p > 0.05 vs. control) in these cells (Fig. 1B, C). Immunofluorescence staining revealed that percent CD31-positive cells (9.0 ± 0.6%) was the same in the indirect coculture group as that in the control group (7.0 ± 1.5%; p > 0.05) (Fig. 2), indicating that indirect coculture with rBMECs is not sufficient to initiate endothelial differentiation process for MSCs. These data also suggest that direct cell–cell connections, not paracrine factors from the neighboring endothelial cells, are crucial to endothelial differentiation of MSCs.
TACE and TNF-α Expressions Were Upregulated in Direct Cell–Cell Contact, but not in Indirect Coculture Condition
TACE is believed to function at the junctions on cell membrane (9). TACE and TACE-mediated protein secretion such as TNF-α may participate in diverse cell behavior, including proliferation, differentiation, migration, and survival (37). Therefore, in the present study, we examined if TACE/TNF-α signaling was involved in the endothelial differentiation of MSCs under the coculture conditions. After 7 days of culture, we detected a 7.3 ± 0.4- and 46.2 ± 1.8-fold increase in the transcription levels for TACE and TNF-α over control (p < 0.05), respectively, in the differentiating MSCs from the direct cell–cell contact coculture group as determined by quantitative PCR. However, the changes in transcriptional expression were minimal for TACE (0.7 ± 0.05-fold over control; p > 0.05), or moderate for TNF-α (4.4 ± 0.23-fold over control; p < 0.05) in the cells from the indirect coculture group (Fig. 3). Although the increase in TNF-α gene expression in the cells from transwell coculture was significant compared to the control, the extent of the upregulation was less than one tenth of that in the direct coculture group. Similarly, TACE protein expression was upregulated by 4.7 ± 0.2-fold over control (p < 0.01) in the direct coculture group, while no significant change (p > 0.05) was observed in the indirect coculture group when compared with control (Fig. 3B, C) after 7 days of culture. These data suggest that TACE/TNF-α signaling is activated in direct coculture condition, but not in the setting of indirect coculture.

Activation of TACE/TNF-α in coculture conditions. (A) Transcript levels of TACE and TNF-α. Q-RT-PCR results show that the gene expression of TACE and TNF-α was significantly increased in direct cell–cell contact, but not in indirect transwell coculture group after 7 days of culture. (B) Protein expression of TACE. Western blot analysis show a significant amount of TACE protein expression in the direct coculture condition, but not in the indirect coculture condition. (C) Normalization of the protein levels. Data are presented as mean ± SE from at least three independent experiments. ctr: control.
Exogenous Recombinant TACE Enhanced Endothelial Specification of MSCs
It has been reported by several groups that TACE from rodent and human origins shares similar biological and biochemical properties (7, 27, 31, 33). Recombinant human TACE has been shown to function on different rodent targets including keratinocyte (34) and mouse platelets (29). To confirm the stimulatory effect of TACE on the endothelial differentiation of MSCs, we incubated MSCs with exogenous human recombinant TACE at a final concentration of 100 ng/ml during differentiation. It was found that the transcription levels of endothelial-specific markers vWF, CD31, VE-cadherin, and Flk-1 were significantly increased by 2.2 ± 0.18-, 2.7 ± 0.25-, 4.9 ± 0.22-, and 1.7 ± 0.08-fold, respectively, in TACE-treated cells when compared with control after 3 days of incubation (Fig. 4), suggesting that TACE promote endothelial differentiation of MSCs.
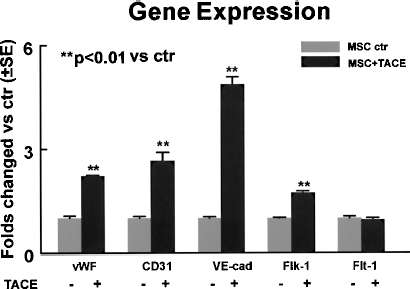
Exogenous human recombinant TACE increased endothelial-specific gene expression in the differentiating MSCs after 3 days of treatment. Transcript levels of endothelial specific markers vWF, CD31, VE-cadherin, and Flk-1 were significantly increased by TACE. VE-cad: VE-cadherin. Data are presented as mean ± SE from at least three independent experiments.
Inhibition of TACE or TNF-α Expression Attenuated Endothelial Differentiation of MSCs in Direct Coculture Condition
Because both TACE and TNF-α gene expression was upregulated in parallel to enhanced gene and protein expression of endothelial markers in the differentiating MSCs in the direct coculture condition, we further investigated the role of TACE and TNF-α in regulating the endothelial differentiation of MSCs. The metalloproteinase inhibitor TAPI2 is a specific TACE inhibitor, and prevents numerous membrane ectodomain shedding including TNF-α (3). Therefore, MSCs were cocultured with direct contact to rBMECs in the presence of 20 μM TAPI-2 for 7 days. We found that the gene expression of vWF, CD31, VE-cadherin, Flk-1, and Flt-1 in the differentiating MSCs was significantly decreased by 43.4 ± 1.1%, 44.3 ± 2.3%, 27.3 ± 4.0%, 27.4 ± 4.0%, and 31.9 ± 6.1% over control (without TAPI-2) (n = 3, p < 0.05), respectively (Fig. 5). These results indicate that TACE is important in the endothelial differentiation of MSCs associated with direct cell–cell communication.
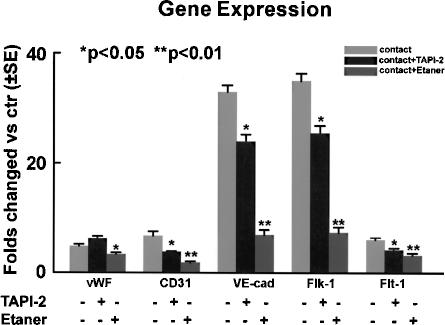
Inhibition of endogenous TACE or TNF-α attenuated endothelial differentiation of MSCs in direct cell–cell contact coculture condition. Q-RT-PCR analysis showed that the transcript levels of endothelia-specific markers in the differentiating MSCs were significantly down-regulated in the presence of TAPI-2 or Etanercept after 7 days of coculture in the cell–cell contact coculture condition. Etaner: Etanercept. Data are presented as mean ± SE from at least three independent experiments.
TNF-α is “shedded” off the cell membrane by the “sheddase” TACE, and is involved in multiple cell functions including differentiation (4, 37). To further examine the role of TACE/TNF-α signaling in endothelial differentiation of MSCs, we used Etanercept (Enbrel, Immunex, Seattle) as a soluble TNF-α receptor to neutralize the biological action of TNF-α during differentiation. MSCs were cocultured directly with rBMECs in the presence of Etanercept at the final concentration of 5 μg/ml for 7 days. Gene expression of vWF, CD31, VE-cadherin, Flk-1, and Flt-1 was significantly attenuated by Etanercept in the cells by 31.0 ± 6.5%, 74.0 ± 6.2%, 79.2 ± 2.9%, 79.0 ± 2.8%, and 48.7 ± 7.6% (n = 3, p < 0.05), respectively (Fig. 5), suggesting that TACE/TNF-α signaling mediates the endothelial differentiation of MSCs in direct coculture condition.
Discussion
In the present study we found that endothelial differentiation was significantly enhanced in MSCs-rBMECs direct cell–cell contact coculture condition, but not in indirect transwell coculture system. TACE and TNF-α gene expression was also upregulated in the direct coculture condition. Attenuation of TACE activity by the specific TACE inhibitor TAPI-2, or neutralization of the soluble TNF-α with Etanercept, resulted in a significant inhibition of endothelial differentiation of MSCs in the direct coculture condition. Moreover, addition of exogenous recombinant TACE increased the gene expression of the endothelial-specific markers in the differentiating MSCs. The results from this study demonstrated for the first time that direct cell–cell contact between MSCs and rBMECs promoted endothelial differentiation of MSCs via TACE/TNF-α signaling.
MSCs are multipotent cells primarily found in the bone marrow. These cells have also been identified in a number of other tissues including peripheral blood, adipose tissue, and skin (10, 14, 23). MSCs can be induced to differentiate into various cell types including endothelium, bone, fat, cartilage, muscle, epithelium, and neural cells both in vitro and in vivo (12, 15, 25, 26, 40). Endothelial differentiation can be achieved from marrow stem cells in the presence of specific soluble factors such as VEGF (30). Local microenvironment has also been shown to be important in the differentiation of MSCs (12). The local niche leads implanted myoblasts to undergo a milieu-induced differentiation into cardiac-like cells in vivo (6). Cortical stem cells, which normally give rise to neural cells under high-density culture condition, differentiate exclusively into smooth muscle cells at low density, indicating the importance of cell–cell interaction signals in cell fate determination (35). Recently, it is reported that when cocultured with cardiomyocytes in a cell–cell contact condition, endothelial progenitor cells (EPC) can thereafter differentiate into functional cardiomyocytes (18). It is also shown that implanted mouse bone marrow-derived cells are able to reconstruct layered smooth muscle structures in injured urinary bladders (13). These studies indicate that the microenvironment plays a critical role in the cell fate determination of stem cells.
We used two coculture systems for MSCs and rBMECs to examine the effect of the microenvironment on endothelial lineage determination of MSCs. Our data demonstrated that endothelial differentiation, as indicated by endothelial-specific gene and protein expressions, was only triggered in direct cell–cell contact culture, but not in indirect coculture system, which allows the communication between two types of cells via paracrine factors without physical contact. These findings support the conclusion that cell junction signaling is the key determinant for the fate of MSCs, while the factors secreted by endothelial cells seem to be insufficient to trigger this process. A similar pattern is observed for EPCs. When directly cocultured with cardiomyocytes, EPCs are found to be differentiated into functional cardiomyocytes. This is not likely to be the result of cell fusion because the same outcome of differentiation is obtained using paraformaldehyde-fixed cardiomyocytes (19).
TACE belongs to a large family of disintegrin-metalloproteinases (ADAMs), a group of transmembrane proteases with preserved domain structures that were first discovered on sperm membranes (8, 28). TACE is usually activated at the cell–cell junctions, and facilitates cell–cell interactions (32, 36, 38). TACE is crucial to mammalian development as it mediates the epithelial–mesenchymal crosstalk in lung development (16). Mice lacking functional TACE have defects in epithelial maturation of multiple organs, in the formation of lung vasculature, airway branching, and the spongiotrophoblast layer of the placenta (27, 43). TNF-α was first isolated in 1975 as an agent capable of inducing necrosis of tumors, and has been used to treat malignancy (5). However, as one of the major substrates of TACE, TNF-α is also a multifunctional cytokine that mediates inflammation and other cellular functions such as differentiation. It has been reported that TNF-α induces osteoclast differentiation and bone resorption by stimulating osteoclast precursors (2, 17). In addition, TACE triggered release of TNF-α mediates myogenesis of C2C12 cell line or mouse primary myoblasts (41). In the present study, we showed that TACE, as well as TNF-α, was significantly upregulated in the differentiating MSCs in the direct cell–cell contact coculture condition. Inhibiting the function of TACE by TAPI-2, a specific inhibitor of TACE, or TNF-α by Etanercept, attenuated endothelial differentiation of MSCs. It will be very interesting and important to determine if a similar mechanism of TACE/TNF-α signaling exists for endothelial differentiation of human MSCs in future studies.
Taken together, the present study demonstrated that endothelial differentiation of MSCs was enhanced in MSCs-rBMECs direct cell–cell contact coculture condition, but not in indirect coculture condition. TACE and TNF-α gene expression were upregulated in the direct coculture condition. Suppression of TACE/TNF-α signaling inhibited endothelial differentiation of MSCs. Addition of exogenous recombinant TACE increased the expression of endothelial-specific markers in the differentiating MSCs. These data suggest that direct cell–cell interactions between MSCs and rBMECs promote endothelial differentiation of MSCs via activation of TACE and TNF-α.
Footnotes
Acknowledgments
This work was supported by NINDS RO1 NS047682 (J.C), AHA grant 0750048Z (J.C), NIHK 08HL0755410 (Z.L.), and NINDS grant PO1 NS23393 (M.C).
