Abstract
The choroid plexus (CP), located at the blood–brain interface, is partially responsible for maintaining the composition of cerebrospinal fluid. Epithelial cell clusters isolated from the CP secrete numerous biologically active molecules, and are neuroprotective when transplanted in animal models of Huntington's disease and stroke. The transcriptomic and proteomic profiles of CP may extend beyond CNS applications due to an abundance of trophic and regenerative factors, including vascular endothelial growth factor, transforming growth factor-β, and others. We used microarray to investigate the transcriptome of porcine CP epithelium, and then assessed the in vitro and in vivo regenerative capability of secreted CP products in cell monolayers and full-thickness cutaneous wounds. In vitro, CP reduced the void area of fibroblast and keratinocyte scratch cultures by 70% and 33%, respectively, compared to empty capsule controls, which reduced the area by only 35% and 6%, respectively. In vivo, after 10 days of topical application, CP conditioned medium lyophilate dispersed in antibiotic ointment produced a twofold improvement in incision tensile strength compared to ointment containing lyophilized control medium, and an increase in the regeneration of epidermal appendages from roughly 50–150 features per wound. Together, these data identify the CP as a source of secreted regenerative molecules to accelerate and improve the healing of superficial wounds and potentially other similar indications.
Introduction
The choroid plexus (CP) is an ependymal structure composed of loosely connected networks of capillaries interspersed within epithelial fronds that extend into the brain's ventricular system. The interface is characterized by ciliated cuboidal epithelium rich in tight junctions that produces cerebrospinal fluid (CSF), making up the blood–CSF barrier. The CP also has a substantial role in detoxifying and regulating the ionic composition of CSF, as well as secreting an assortment of factors, some of which are trophic or regenerative in nature (6–8,10,26,28). Because of its copious production of these factors, the CP has recently been examined for its potential role as a cell-based therapeutic. Rescue of embryonic neurons by CP culture supernatant has been observed in vitro, while transplants of CP promoted regeneration of damaged spinal cord (14) and reduced the pathologic sequelae associated with stroke in animal models (2,3). Additionally, intrastriatal implantation of encapsulated CP has protected against excitotoxic lesions in rodent (5) and primate models (9) of Huntington's disease.
The complex profile of bioactive substances secreted by the CP and the mechanism by which they exert their beneficial effects in these models remains unclear and is a fertile area of investigation, but certain proteins have been identified in the CP secretome that may contribute to its pervasive effect. Vascular endothelial growth factor (VEGF), a potent molecule associated with angiogenesis and neurotrophic activity, is secreted continuously from isolated CP epithelium when cultured as a monolayer, as free-floating cell clusters, and when encapsulated in microcapsules (27). A number of other trophic factors have been identified in the CP, including fibroblast growth factor 2 (FGF2), netrin 1, pentraxin 3, pigment epithelium-derived factor (PEDF), cardiotrophin-1, Wnt-7B, Wnt-10A, nerve growth factor (NGF), neurotrophin-4 (NT-4), transforming growth factor-β (TGF-β), growth hormone (GH), and others (6,12,13,28). Some of these factors have been implicated in regenerative processes outside of the central nervous system, including wound healing. Certain subsets of the Wnt family, for example, participate in the regeneration of hair follicles in healing full-thickness wounds (16). VEGF, FGF2, and TGF-β are known mediators of the wound healing response that have chemotaxic and mitogenic properties (1). Interleukin-1 (IL-1), IL-6, connective tissue growth factor (CTGF), and others expressed by the CP (see Results section) mediate other repair processes such as inflammation, reepithelialization, granulation tissue formation, and matrix remodeling.
The current work explored the potential of CP-secreted products to aid in cutaneous wound healing. Based on the abundance and significance of its components relevant to the wound healing process, we hypothesized that the simultaneous application of the entire CP cocktail would provide relevant molecules “as needed,” avoiding an attempt to orchestrate delivery with the precisely staged action of cells, proteins, and matrix elements that occurs in the natural healing cascade. In vitro, hydrogel-encapsulated porcine CP was cocultured with scratched monolayers of fibroblast or keratinocytes to assess migration and proliferation triggered by CP molecules. In vivo, we used CP conditioned media lyophilate to treat full-thickness open wounds and sutured skin incisions. Additionally, we collected mRNA from CP used to generate conditioned media to identify genes associated with potential regenerative factors expressed in this specific culture system.
Materials and Methods
Choroid Plexus Isolation
Three separate groups of four neonatal Yorkshire pigs aged 7–10 days were induced with ketamine (500 mg/kg) and xylazine (0.15 mg/kg), and then maintained on 3% isoflurane during controlled exsanguination. Following craniotomy, the brain was removed and hemisected along the midline to expose the CP in the lateral ventricles. The CP was extricated and placed in Hanks balanced salt solution (HBSS, 0–4°C) supplemented with ciprofloxacin (3 μg/ml, Sigma, St. Louis, MO), 2% human serum albumin, and digested as previously described (4,9). Briefly, following mechanical disruption with scissors, CP epithelial clusters were released after light enzymatic digestion over 20 min (Liberase HI, Roche, NJ). Epithelial clusters were washed and maintained in RPMI-1640 (Sigma) with 2% neonatal porcine serum and 3 μg/ml ciprofloxacin (RPMI-CPN) and placed in nonadherent flasks at 37°C with 5% CO2. Half of the medium was removed and replaced with fresh medium after 24 h, 48 h, and every other day thereafter. After 0, 2, and 8 days in RPMI, clusters were counted and separated into individual flasks. After 10 days, the medium was replaced with serum-free endothelial cell culture medium (ENDO-SFM, Gibco, USA), which was used throughout the experiment for generation of conditioned media over intervals of 24, 72, or 96 h as previously described (27).
Isolation of RNA and Gene Array
Selected Genes Expressed by CP as Detected in Porcine Microarray
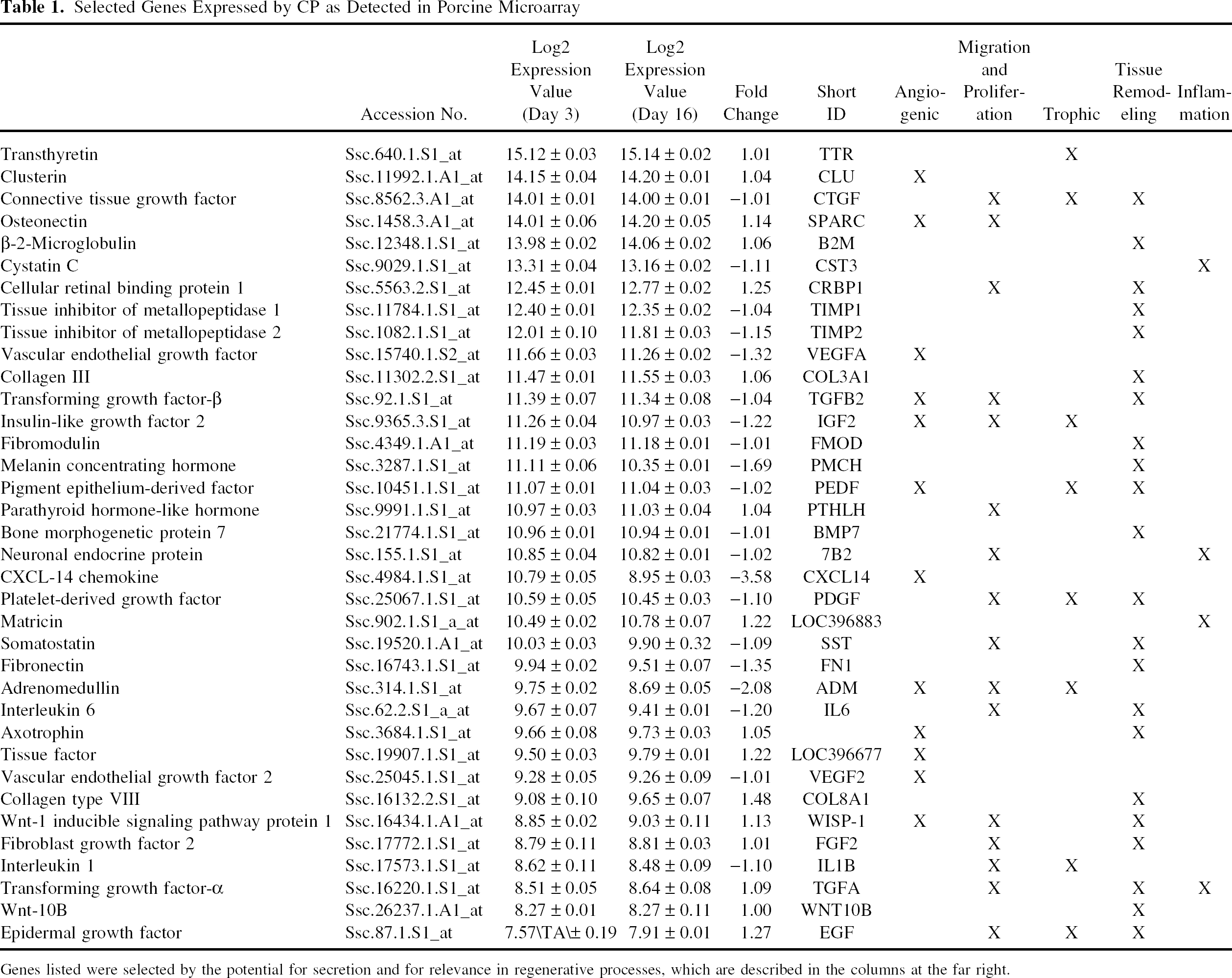
Genes listed were selected by the potential for secretion and for relevance in regenerative processes, which are described in the columns at the far right.
Selected CP Genes That Change Between Day 3 and Day 16 in ENDO-SFM

Genes were selected based on amplitude of change (>1.8-fold) and statistical significance.
In Vitro Bioactivity: Scratch Assay
CP clusters were encapsulated in alginate-polyornithine-alginate (APA) microcapsules after 10 days in culture as previously described at 50,000 clusters/ml using a coaxial air jet system (9). Capsules (600 μm diameter) were produced and were incubated in RPMI-CPN for 5 days prior to transfer to fibroblast or keratinocyte cultures. A sample was pulsed in fresh RPMI-CPN for 24 h to quantify secreted VEGF. Normal human dermal fibroblasts (NHDF, Cambrex, USA) or normal human epidermal keratinocytes (NHEK, Cambrex, USA) were expanded in T75 flasks in supplemented cell culture medium. NHEK cells were grown in keratinocyte growth medium (KGM, Cambrex, USA) supplemented with epidermal growth factor, insulin, and hydrocortisone. NHDF cells were grown in fibroblast growth medium (FGM, Cambrex, USA) supplemented hFGF-B and insulin. After several passages, cells were seeded into 24-well plates at 5,000 cells/well (N = 8 per treatment group) and grown to 95% confluency. To initiate the scratch assay, the culture medium was removed, the cells were washed with HBSS three times, and a defect (approximately 9 mm2) was created with the tip of a 20-μl pipette. The HBSS was aspirated several times to loosen the cells surrounding the defect, and then replaced with basal culture medium. The defects were then photographed at 4x magnification. Transwell inserts with a pore size of 10 μm were placed on top of the wells, and encapsulated CP were added for an equivalent dose of 500, 1,500, 2,500, or 5,000 CP clusters per well. Empty capsules served as controls and were also incubated in RPMI-CPN for 5 days, and then added to wells at the peak dose range of the encapsulated cell group, corresponding to a 100-μl volume of capsules. Cultures were allowed to incubate for 7 h (NHEK) or 14 h (NHDF), the capsules and inserts were removed, and the defects were photographed again at 4x magnification. Calibrated image analysis software (Motic, China) was used to measure the scratched area prior to and following exposure to encapsulated CP.
Collection and Preparation of CP Proteins for Topical Application
CP conditioned medium (CM) was collected at 96-h intervals in ENDO-SFM from flasks containing roughly 100,000 CP clusters in 28 ml medium. Following the initial culture period in RPMI-CPN and subsequent transition to ENDO-SFM, CM was collected repeatedly for a period of 2 months. Briefly, cell clusters were allowed to sediment by gravity, and then conditioned media was aspirated and replaced with fresh ENDO-SFM warmed to 37°C. The CM was collected in 50-ml tubes and immediately flash-frozen in liquid nitrogen. In some cases, the CM was dialyzed against distilled water in a 1000 Da MWCO membrane to remove salts prior to freezing. An aliquot was also taken prior to processing for measuring VEGF content. Frozen CM was lyophilized for 96 h (Virtis, USA) and stored at −80°C prior to use. Fresh medium was processed in parallel for use in control animals.
Lyophilized CM (LCM) or dialyzed lyophilized CM (DLCM) was prepared for topical administration immediately prior to application by mixing the lyophilate in bacitracin ointment (Johnson & Johnson, USA) at roughly 50 mg LCM/350 mg ointment for the high dose, and roughly 17 mg LCM/350 mg for the low dose. The actual amounts were calculated based on the amount of VEGF measured during the media collection period, with the final doses normalized to a VEGF dose of 6 ng at the low dose, and 20 ng at the high dose. Fresh ENDO-SFM, not exposed to choroid plexus, does not contain VEGF, and is not associated with improved healing in tissue culture models or cutaneous autografts (data not shown).
Detection of VEGF Using ELISA
VEGF was measured in culture supernatants as a surrogate marker of potency as previously described (27). After 96 h of incubation (37°C, 5% CO2), the supernatant was removed and frozen for subsequent analysis. Thawed samples were analyzed in triplicate using a human VEGF ELISA (Quantikine®, R&D Systems, USA). Sequence homology between porcine and human VEGF is greater than 90% (24). Samples of conditioned media (200 μl) were combined with assay diluent (50 μl) and the optical density was measured at 450 nm on a Beckman Coulter DTX-880 spectrophotometer after development. The value obtained was compared to a standard curve of recombinant human VEGF using a logarithmic curve fit. Values were expressed as picograms per milliliter.
In Vivo Evaluation of CPCM Formulations
CPCM was evaluated in full-thickness wounds in male Long-Evans rats approximately 3 months old and weighing 250–350 g housed in pairs in a temperature-controlled environment with a 12:12 hour light/dark cycle. All procedures met or exceeded NIH guidelines and were approved in advance by the Brown University IACUC governing body.
Immediately prior to surgery, rats were anesthetized with isoflurane (2–3%) and placed in a nose cone in a prone position. Prior to the creation of the skin defect, animals were clipped and the skin around the area was disinfected with betadine. Following the surgery, animals were treated with an analgesic (buprenorphine, 0.02 mg/kg subcutaneous) and observed until fully recovered from anesthesia. Animals were euthanized using carbon dioxide gas, and explanted tissue was removed as indicated for histological processing. Briefly, after fixation in isotonic 4% paraformaldehyde for 2–3 days, tissue segments were transversely embedded in paraffin for cutting 4-μm-thick sections, which were subsequently stained with either hematoxylin and eosin (open wound tissue), or Masson's trichrome stain (sutured linear wounds). Morphometry on healing wounds was carried out using image analysis software (Motic, China) to measure the healing area within the margin of hair growth, and statistics were calculated with Prism software (GraphPad, USA), using a one-way ANOVA with a 95% confidence interval and post hoc Dunnett's test for comparison against the control group.
Open Wound
An 8-mm Keyes dermal punch was used to completely remove patches of skin to create full-thickness skin defects to the depth of subcutaneous tissue between the shoulder blades. Topical CM ointment was applied for 10 days following the procedure, and animals were sacrificed at day 14 for tissue harvesting. A control group (lyophilized ENDO-SFM in bacitracin ointment, N = 5) and two treated groups (high-dose LCM in bacitracin ointment, N = 7, and high-dose DLCM in bacitracin ointment, N = 7) were observed. At the conclusion of the study, following euthanasia with CO2 gas, large sections of skin containing the wounded area were removed and centered in devices consisting of two plastic rings held under tension by a series of peripherally located screws. The devices were created to overcome histological artifact associated with skin deformation during the fixation process (Fig. 1A, B). The skin was kept relaxed during insertion into the devices, preventing additional artifact caused by tension in the wound. Devices with skin samples were immersion fixated in isotonic 4% paraformaldehyde overnight and were subsequently processed for histology and morphometry, which included measurement of cross-sectional wound area, nuclear counts within the central 2-mm2 area, and counts of epidermal appendages within the entire wound area.
Tissue handling devices: (A) histology fixture containing control tissue; (B) histology fixture containing LCM-treated tissue; (C) explanted skin around incision placed in sampling fixture for tensiometry sample preparation. Clamping mechanism ensured centering of incision in jig and even tissue sampling. An aluminum block was placed on top of the razor blades, and then a press was used to cut through the tissue.
Incision
A 6-cm midline full thickness incision was made between the shoulder blades with an unused number 15 scalpel through a plastic template designed for uniformity of straightness and length. The incision was immediately closed with eight interrupted stitches using 6-0 silk equipped with a PC-3 cutting needle. Based on prior experience within our group, silk was chosen to minimize interference in the healing of the wounds caused by irritancy of other bristle-like materials. For the next 10 days, wound sites were treated topically once daily in the following groups: a control group (lyophilized ENDO-SFM in bacitracin ointment, N = 5) and two treated groups (LCM in bacitracin ointment, N = 5, and DLCM in bacitracin ointment, N = 5). After 14 days, the entire 6-cm wound strip was removed following euthanasia with carbon dioxide gas. After shaving the area and removing the sutures, an area of 7 × 3 cm was excised around the wound and processed for histology and tensiometry. Because pilot studies demonstrated intersample variability related to skin harvesting, a specialized instrument was engineered to create uniform 1-cm width strips of skin centered horizontally at the incision with fresh razor blades (Fig. 1C). A total of five strips of tissue were created per wound site by excluding the outer 0.5 cm on each side. The area of skin was placed onto slabs of dental wax that were visually centered onto the jig, and six fresh razorblades were inserted into the positions shown in the picture. A press was used to exert force on an aluminum block resting on top of the blades, creating an evenly distributed force completely perpendicular to the plane of tissue, and resulting in precise 1-cm-wide tissue segments for histological and physical analysis. For each animal, the central 1-cm segment was processed for histology, while the outer two on each side were analyzed for tensile strength at break using an Instron tensiometer (Instron, USA). Skin samples were immediately transported on ice on their wax backing material to the instrument. Samples were removed from the wax, any residual panniculus carnosus was moved from the underside of the wound, and the tissue was placed in the serrated jaws of the instrument under maximum clamping pressure. With a 50 N load cell in place, the tissue was pulled at a rate of 8 mm/min until breakage occurred. Statistics were calculated using Prism software (GraphPad, USA), using a one-way ANOVA with a 95% confidence interval and post hoc Dunnet's comparison.
Results
CP Isolation and Culture
CP was successfully isolated from neonatal pigs and digested into CP clusters ranging in size from approximately 50 to 250 μm. After culturing the clusters in nonadherent flasks in RPMI-CPN for 8 days, manual counting revealed that the yield per piglet decreased from 128,854 ± 16,159 to 50,962 ± 5,690 CP clusters. This is consistent with the temporal loss of nonepithelial clusters of cells due to attachment or death, in addition to gradual coalescence of small aggregates into larger clusters (Fig. 2). After the transition to serum-free medium after 10 days, CM was collected in parallel with fresh medium held under the same conditions. VEGF levels were measured in aliquots taken from CM generated throughout the experiment (Fig. 3) as well as control medium. Ten days after isolation, daily levels of VEGF secreted per CP increased from approximately 41 ng to around 70 ng at 2 weeks, and remained relatively stable thereafter. Following processing of the conditioned medium to generate a pooled purified lyophilate, VEGF levels were verified to ensure that the molecule was conserved throughout the process.
Initial tissue culture of digested choroid plexus clusters. (Top) Clusters were counted over the first 8 days in separate samples each containing 2 CP equivalents. Values shown are mean ± SEM. (Bottom) Light microscopy of CP clusters in RPMI-CPN tissue culture medium at 2 and 8 days postisolation. Scale bar: 250 μm. VEGF output as measured by ELISA in separate collection flasks normalized per CP equivalent, as aliquoted at the initiation of the experiment. Values represent mean ± SEM.

CP Gene Array
CP cohorts from the same animal group were cultured, after 10 days in RPMI-CPN, in ENDO-SFM for an additional 3 or 16 days specifically for microarray analysis. Whole genome porcine arrays were performed to characterize the CP expression pattern as well as to evaluate the plasticity of the CP transcriptome over the first 16 days following media transition. The microarray data were selected and sorted based on the potential of mRNAs to transcribe for secreted molecules, relevance in tissue regeneration, expression levels, and statistical significance. Shown in Table 1 is a list of those factors that could play a role in wound healing, along with a descriptive matrix of their potential roles. As anticipated (27,28), transthyretin was expressed at extremely high levels in tissue culture, and a number of other factors were expressed at similarly high levels. In general, the regenerative genes listed encode for growth factors, angiogenic factors, extracellular matrix remodeling components, and other factors with regenerative properties. The regenerative factor compliment expressed by the CP clusters in ENDO-SFM was significant, and included several relevant factors (see Table 1 for abbreviations): CTGF (14.01), VEGF (11.66), TGF-β (11.39), IGF-2 (11.26), PEDF (11.07), BMP-7 (10.96), PDGF (10.59), somatostatin (10.03), Il-6 (9.67), tissue factor (9.50), WISP-1 (8.85), FGF-2 (8.79), TGF-α (8.51), Wnt-10B (8.27), EGF (7.57), and others. The difference in expression levels of most of these regenerative components between day 3 and 16 was nearly indistinguishable, with most factors remaining within a very narrow range, except for CXCL-14 chemokine and adrenomedullin, which decreased by 3.58- and 2.08-fold, respectively. The entire array database was subsequently sorted to compare statistically significant changes greater than 1.8-fold in all expression levels between day 3 and day 16, the output of which is shown in Table 2. These genes that exhibit the highest variability in temporal expression could be involved in the acclimation of CP to tissue culture conditions, and do not include major regenerative candidates.
Monolayer Scratch Assay
Cultured monolayers of neonatal human keratinocytes and fibroblasts were disrupted and incubated with different doses of encapsulated CP in basal culture medium. In both cell types, the wounded area decreased during the incubation period and was significantly smaller in groups treated with > 1,500 encapsulated CP. The data, shown in Figure 4, demonstrate that the chemotaxic effect was dose dependent and the peak effect was associated with a 35.2% area reduction over control in NHDF cultures, and a 27.9% area reduction over control in the NHEK cultures. In NHEK cultures, 2,500 encapsulated CP was associated with the smallest wound area, while the NHDF cells showed an increased response up to the maximum dose of 5,000 encapsulated CP. To gauge the levels of VEGF secreted by the encapsulated CP, samples of 2,500 (N = 9) were assessed as previously described (27). At the dose of 2,500 encapsulated CP/well, roughly 2.4 ng VEGF was secreted over 24 h.
Percent reduction of areas in the in vitro scratch assay containing NHEK cells (top) and NHDF cells (bottom). Encapsulated CP was separated by Transwell membranes from cultured monolayers at the doses indicated on the x-axis, which represent the number of encapsulated CP clusters per well per milliliter basal culture medium. Values represent mean percent area reduction [(starting area – final area)/(starting area)] ± SEM.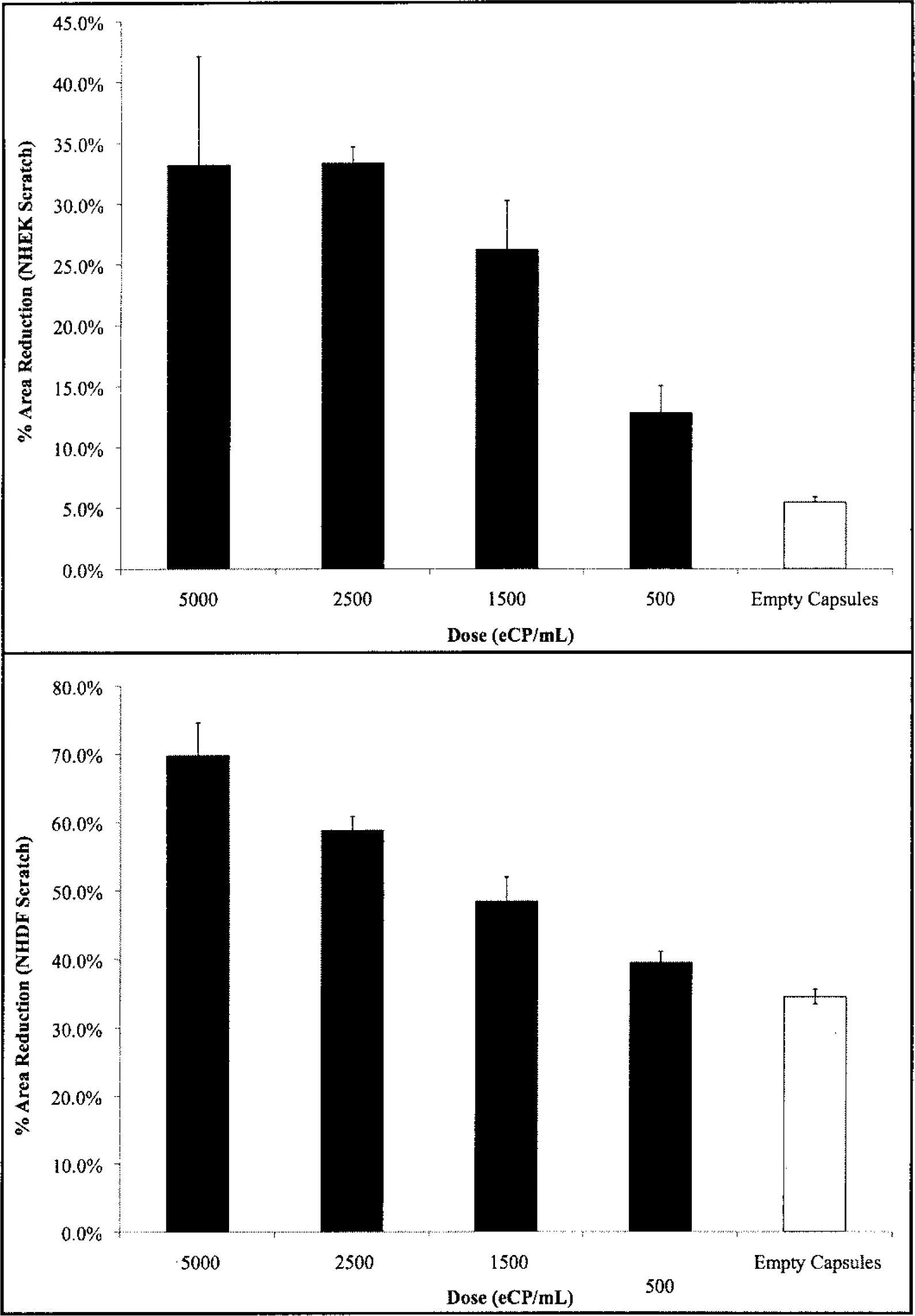
Full-Thickness Open Wounds
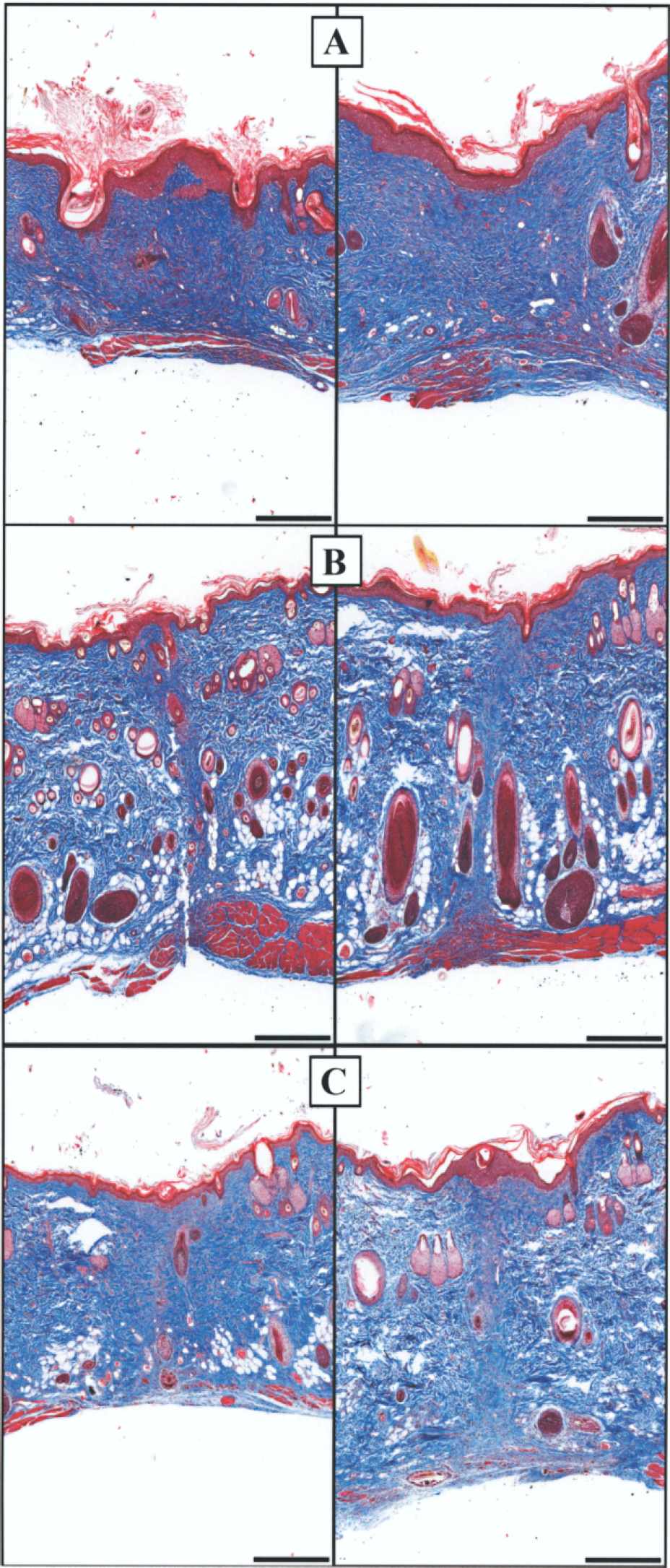
Uncovered open wounds were treated daily for 10 days with no sign of infection. By day 3, scabs formed on all wounds with no obvious difference between groups; after 8 days, the scabs began to detach as the underlying wounds contracted. After 2 weeks, wound surfaces appeared epithelialized and smooth with little evidence of scab remnants. Superficially, all wounds looked well healed by this time point, with some variability in the presence of hair follicles evident in the central wounded area. Figure 1 shows explanted skin containing the wounded area, with a control treatment (Fig. 1A) and LCM treatment (Fig. 1B), which contains more follicles within the wounded area. This observation is more apparent histologically, as shown in Figure 5. Here, a series of representative sections are shown located one third into the width of the wound. In Figure 5A, normal skin is shown, which contains epidermal appendages including hair follicles associated with sebaceous glands, dense collagen, and distinctly formed epidermal rete pegs. By comparison, a wound treated with lyophilized control medium in bacitracin, shown in Figure 5B, contains densely packed fibroblasts within the dermis, replete with newly formed vessels, and an epidermis of a thin, flat layer of keratinocytes. Wounds treated with LCM (Fig. 5C) contain tissue with a higher level of organization, with forming epidermal appendages, a lower cell density, and a more ridged profile suggestive of epidermal rete pegs. Similarly, DLCM-treated wounds (Fig. 5D) were rich in epidermal appendages, including sebaceous glands and hair follicles at various stages of development. These wounds also resembled the morphology of normal tissue in the density and ratio of cells to collagen. As shown in Figure 6, wounds treated with CP products contained a significantly higher density of epidermal appendages (5- and 5.8-fold higher for DLCM and LCM, respectively) and an overall smaller cross-sectional area. However, central cell density was morphometrically indistinguishable between groups.
Selected images of open wounds processed for H&E histology. (A) Normal rat skin that was never injured, (B) open wound treated with lyophilized ENDO-SFM in bacitracin (control), (C) open wound treated with LCM in bacitracin, and (D) open wound treated with DLCM in bacitracin. Wounds were treated for 10 days and sampled 4 days later. Scale bar: 200 μm. Central morphometry of open wounds. Values show mean ± SEM. (A) Epidermal appendages were counted across the entire central cross sections. (B) Nuclear counts within the central portion midway through the wound, and (C) area of the wound midway through the tissue block.

Sutured Incisions
Incisions (6 cm) were treated topically with ointments for 10 days prior to skin harvest 4 days later, at day 14. All wounds healed completely during the study, with complete closure occurring after 8 days (Fig. 7). Incisions treated with CP proteins showed a marked improvement in the cosmesis of healing with LCM (Fig. 7B) and DLCM (Fig. 7C) appearing more symmetric and having narrower margins than the control group (Fig. 7A). This finding was consistent with the histology, which showed much thinner bands of fibrosis in the center of the wound, along with a greater density of epidermal appendages at the margins (Fig. 8). The control group (Fig. 8A) had a large area of fibrosis in the healing incision, while both LCM (Fig. 8B) and DLCM (Fig. 8C) reduced this area dramatically. Interestingly, LCM demonstrated the strongest effects, with the width of the central scar reduced roughly twofold over the DLCM treatment, and roughly fivefold over the control group. As demonstrated with end-to-end tensiometry, this improved cosmetic performance carried over to the strength of the tissue. As shown in Figure 9, both groups treated with CP proteins were associated with a statistically significant improvement in peak break strength over the control. LCM increased strength over the control group from 3.7 ± 0.5 to 7.7 ± 0.8 N, while DLCM was associated with a further increase in force to 9.5 ± 1.6 N.
Treatment of linear incisions with lyophilized media in bacitracin ointment. Representative macro photos are shown after 8 days of treatment. (A) Control ENDO-SFM, (B) LCM, and (C) DLCM. Treatment of linear incisions with lyophilized media in bacitracin ointment. Representative images following 10 days of treatment and an additional 4 days of healing. (A) Control ENDO-SFM, (B) LCM, and (C) DLCM. Scale bar: 100 μm. Treatment of linear incisions with lyophilized control or conditioned media in bacitracin ointment. Peak break strength in 1-cm-wide sections of skin, centered at the incision, as measured by Instron tensiometry (**p < 0.01).

Discussion
The studies presented here demonstrate the ability of factors secreted by the CP to intervene in the wound healing process. In vitro, this activity was manifested by encapsulated CP cocultured with scratched monolayer cultures of human fibroblasts and keratinocytes as an accelerated wound area reduction due to chemotasix or cell proliferation. In these models, there was an increase in the amount of healing associated with the dose of VEGF in the conditioned medium. In vivo, open wounds treated with CPCM in ointment showed a five- to sixfold increase in replenishment of epidermal appendages after 14 days compared to the control, along with a significant reduction in cross-sectional wound area. And surgically closed incisions treated with CPCM were associated with a greater than twofold increase in the tensile strength of the healed tissue, and an improved cosmetic appearance with reduced central scarring and better preservation and/or repopulation of epidermal appendages surrounding the wound margins.
The healing mechanism associated with CP proteins remains unclear, but is likely related to the synergistic activity of a combination of expressed factors. The transcriptomic profile of CP contains a number of genes encoding for mediators of the natural wound healing response. While it is unknown if the full complement of these factors is secreted from CP in the tissue culture system described here, proteomic analysis of mouse CP supports an abundance of secreted proteins (28). VEGF, chosen as a surrogate marker of potency in these studies, was delivered at biologically efficacious levels (20), and is capable of upregulating intrinsic factors such as PDGF (11), bFGF (11), TGF-β1 (18), and others that are involved in wound healing. Still, it is likely that the overall effect of the CP cocktail extends beyond VEGF alone, as there are a number of other highly expressed factors contained within the CP transcriptome that act at similarly high levels.
CTGF, for example, is expressed by CP at levels nearly as high as transthyretin, the factor with the highest expression level in gene array (27) and proteomic analysis (28). CTGF promotes chemotaxis and proliferation of fibroblasts, the formation of granulation tissue, reepithelialization, and matrix remodeling (15). While the primary role of CTGF in wound healing is to promote tissue reconstruction through fibrosis, it has also been shown to regulate VEGF-mediated angiogenesis through negative feedback (17), and is angiogenic on its own in models of corneal neovascularization (25). TGF-β, also highly expressed by the CP, is one of the most well known mediators of cutaneous wound healing, and is involved from the inflammatory phase through tissue remodeling and reepithelialization (1). It has demonstrated therapeutic potential in topical application to venous stasis ulcers (21), and has been used to increase the strength of incisional wounds (29). IGF-2, BMP-7, PDGF, FGF-2, EGF, and other factors expressed by CP and listed in Table 1 also have potential wound healing effects as cited in the literature and as indicated in the table.
One of the most intriguing observations in these studies is the increased density of hair follicles within the wound margin following CP factor administration. While it is unclear if the follicles were generated de novo in both models, it is likely that follicles present in the healed open wounds were a result of neogenesis. These follicles, varying in their maturity but appearing in healing tissue prior to 2 weeks, may have developed as a result of Wnt-dependent signaling involving Wnt10b, which is expressed at moderate levels by CP. In vivo, Wnt10b is important in the differentiation of skin epithelium and formation of hair follicles (16), and recent work using recombinant Wnt10b has demonstrated positive effects on hair shaft growth and follicle formation in vitro (19). Perhaps as a component of the CP cocktail, Wnt10b works in concert with factors that are involved in the earlier stages of wound healing (e.g., PDGF, VEGF, CTGF, TGF-β, and others) to guide epithelial differentiation concurrent with angiogenesis and migration of fibroblasts and keratinocytes, effectively providing developmental cues as in embryogenesis.
The complexity of the porcine CP cocktail leaves open questions about the exact mechanism of action in these models, and further investigation with selective blocking of candidate factors with antagonists or antibody traps could be strategies for elucidation. Further investigation into the shared ectodermal origin of CP and skin may also provide additional insight into the healing mechanism. Still, the effects observed in these studies help to justify the continued analysis of therapeutic CP products outside of the central nervous system. They are a ready source of various factors that, when applied as individual molecules, are associated with improvements in wound healing. Recombinant PDGF, for example, has been studied extensively on its own to treat diabetic ulcers (22), and is approved by the FDA for topical use. TGF-β2 has also been tested clinically (23), and remains a fertile area of product development. Perhaps the application of these and other factors simultaneously, to be used as required by the healing tissue, would overcome issues related to targeting just one aspect of the wound healing cascade, and decrease the impact of delivery kinetics and the regulation of factors via feedback in the wound. Such a combination of factors could also prove useful in more severe wounds, such as deep ischemic ulcers, that would require a more coordinated rebuilding of tissue beginning with a restoration of blood supply and ending with superficial reconstruction.
Factors from the CP, delivered in a biologically determined ratio and composition as shown here, could be used to improve the healing of cutaneous wounds. Because of the potentially widespread effect imparted by the CP cocktail, ranging from angiogenesis to tissue rebuilding, there may be other indications that are also suitable for investigation. A therapeutic benefit has already been described in animal models of neurodegenerative disease, and other potential indications could include diseases where staged intervention is required to treat tissues suffering from ischemia and necrosis, such as diseases of the cardiovascular system, muscle wasting and repair, bone and joint healing, and other similar areas. The data presented here may serve as a starting point for the use of CP factors in regenerative medicine.
