Abstract
Intramyocardial transplantation of bone marrow-derived stem cells is a potential therapeutic option after myocardial infarction (MI). Intramyocardial administration is invasive but allows efficient and targeted stem cell delivery. Aims of this study were validation of minimal-invasive, echo-guided closed-chest cell transplantation (CTx) of mononuclear (MNC) or mesenchymal stem cells (MSC) and quantification of systolic left ventricular function and assessment of contractile reserve with high-resolution reconstructive 3D-echocardiography (r3D-echo) 3 weeks after CTx. Female Fischer344 rats received syngeneic male MNC, MSC, or medium after myocardial ischemia and reperfusion via echo-guided percutaneous injection (open-chest for control). Left ventricular systolic function was measured and dysfunctional myocardium was quantified with r3D-echo. For investigation of contractile reserve and myocardial viability r3D-echo was additionally conducted during low-dose dobutamine 3 weeks after CTx. Cell persistence after echo-guided CTx was quantified via real-time PCR; scar size was measured histologically. Echo-guided percutaneous CTx was feasible in all animals (n = 30) without periprocedural complications. After 3 weeks, 1.4 ± 1.1% of transplanted MNC and 1.9 ± 1.2% of MSC were detected. These numbers were comparable to those after open-chest intramyocardial injection of MNC (0.8 ± 1.1%; n = 8, p = 0.3). In r3D-echo no functional benefit was associated with CTx after MI and reperfusion. All groups (MNC, MSC, and controls) revealed a significant decrease of dysfunctional myocardium and similar contractile reserve during inotropic stimulation. In conclusion, percutaneous echo-guided closed-chest CTx promises to be an effective and safe approach for CTx in small-animal research. However, intramyocardial CTx of MNC or MSC had no influence on systolic function and contractile reserve after reperfused MI.
Keywords
Introduction
Intramyocardial cell transplantation (CTx) of bone marrow cells (BMC) has been under investigation for the treatment of acute myocardial infarction for several years. Therefore, subpopulations, such as mononuclear stem cells (MNC) and mesenchymal stem cells (MSC), have been investigated to assess their functional impact. The largest randomized, placebo-controlled study to date demonstrated beneficial effects of intracoronary cell therapy on left ventricular ejection fraction in patients after myocardial infarction (20), despite the low persistence of injected cells (8,24). On the other hand, other well-performed studies revealed less favorable results (12). While intracoronary CTx can be performed via coronary catheter, which is less invasive than myocardial injection, the intravascular mode of delivery is less effective (24) and does not allow targeting of cell delivery into distinct dysfunctional myocardial segments. Therefore, novel strategies of targeted CTx that are less invasive than open-chest surgery are of major interest.
Despite the advance of CTx with BMC to the clinical level, further animal studies are needed to estimate its therapeutic effect, as well as to identify the most suitable cell type. Therapeutic goals should not only aim at global and regional left ventricular function, but also on the amount of dysfunctional myocardium and contractile reserve demonstrating myocardial viability. Furthermore, because first-line therapy of acute myocardial ischemia is revascularization with consecutive reperfusion, animal models should implement this primary treatment strategy in the experimental protocol for evaluation of impact of CTx on myocardial dysfunction. In the present study we performed sonographically guided minimal-invasive myocardial closed-chest BMC-CTx. The impact of two distinct BMC populations (MNC or MSC) on global and regional left ventricular systolic function was assessed with reconstructive 3D-echocardiography (r3D-echo). Further, the impact of BMC-CTx on the quantity of dysfunctional myocardium and contractile reserve was assessed in rats after acute myocardial infarction with reperfusion.
Materials and Methods
Experimental protocol was approved by the regional government's Animal Care and Use Committee (50.203.2-K 47, 34/03), and conformed to the Guide for the Care and Use of Laboratory Animals (NIH publication No. 85-23, National Academy Press, Washington DC, revised 1996) (2).
Cell Isolation
The femurs, humeral, and tibial bones (male Fischer344 rats, 2 months old, Charles River Laboratories, Sulzfeld, Germany) were dissected as described previously (13). Bone marrow was eluted with a 19-gauge needle and DMEM, high glucose, 10% FCS, 1% penicillin/streptomycin. The cell suspension was filtered through 70-μm filters, centrifuged, and resuspended in PBS. A ficoll gradient centrifugation (NycoPrep, Progen GmbH, Heidelberg, Germany) was performed and the MNC were collected and washed three times with PBS. For isolation of MSC 50 × 106 of the remaining cells were plated on 10-cm dishes, and nonadherent cells were removed after 72 h. MSC were expanded to passage 3 and then detached with trypsin/EDTA. The cells were centrifuged at 800 × g for 1 min and resuspended in serum-free DMEM medium at a concentration of 106 cells/60 μl. Cell viability was 90.0 ± 2.1% as assessed by trypan blue staining before cell injection.
Rat Model of Acute Myocardial Infarction
For the induction of myocardial infarction, female Fischer 344 rats (125–150 g) were anesthetized with 1.0–1.5% isoflurane, intubated, and ventilated as previously described (6). The thorax was opened via the third inter-costal space and a 7-0 polypropylene suture was placed around the left anterior descending artery (LAD). Both ends of the suture were led through a short plastic tube, forming a loose snare around the LAD. Each end of the suture was externalized laterally through each side of the thorax and placed under the skin. Then the chest was closed layer by layer. Tramadol and carprofen were given subcutaneously (40 mg/kg/24 h and 5 mg/kg/24 h, respectively) as postoperative analgetics.
To avoid stress-induced immunological response to the thoracotomy, acute myocardial ischemia was induced 7 days after the initial operation by occlusion of the LAD (17). The ends of the suture were externalized from the skin pouches, carefully taped to heavy metal picks, and pulled apart carefully for 60 min. Acute myocardial ischemia was controlled by means of electro- and echocardiography, resulting in elevation of ST segment and regional dysfunction of anterolateral wall segments, respectively.
Transplantation of BMC
Minimal-invasive BMC-CTx was performed immediately after myocardial ischemia and reperfusion. After 1 h the coronary occlusion was released and blood flow was reestablished. Subsequently, rats were randomized to receive myocardial injections of either MNC, MSC, or medium. During continuous echocardiographic guidance, 60 μl of cell suspension was injected percutaneously into three separate sites of the anterior, lateral, and posterior myocardial peri-infarction border zone. This procedure was performed using a 100-μl Hamilton syringe attached to a 30-gauge needle. Efficacy of intramyocardial cell delivery was monitored sonographically after the intervention (Fig. 1). Open-chest CTx of MNC was conducted similarly into peri-infarction border zone myocardium as described recently (13).
Minimal-invasive, echo-guided closed-chest cell transplantation (CTx). (A) Parasternal short-axis view observed with high-resolution echocardiography during minimal-invasive, echo-guided closed-chest cell transplantation. Acute myocardial ischemia was induced in anterolateral segments and is characterized by wall thinning and dysfunctional wall motion (akinetic myocardium is marked with open arrowheads). The tip (arrow) of a 30-gauge needle (arrowheads) is placed percutaneously, echo-guided from a left thoracal (open arrows) access into the posterior border zone of the ischemic myocardium. (B, C) Ultimately, this method allows qualitative control of intramyocardial stem cell delivery (closed arrowheads). Scale bars: 5 mm.
Reconstructive 3D-Echocardiography
For high-resolution r3D-echo a HDI-5000 ultrasound system (HDI-5000, Philips Medical Systems, The Netherlands) was used, equipped with a 15 MHz linear array transducer allowing frame rates of up to 230 Hz making use of harmonic imaging (4,23). The transducer was positioned perpendicular to the long axis of the left ventricle in consecutive short-axis sections starting at the aortic root. ECG and respiratory gated imaging was performed in 12–18 consecutive short-axis views of 0.5mm increment (Fig. 2). Cineloops of 2-s duration were stored digitally for each short-axis view and analyzed offline. Additionally, the imaging protocol was repeated during intravenous low-dose dobutamine infusion for assessment of myocardial viability (10 μg/kg/min) (6). r3D-echo was performed during myocardial ischemia and 3 weeks after reperfusion by one investigator blinded to treatment groups. For echocardiography, rats were anesthetized with 1.0–1.2% isoflurane in O2, minimizing cardiodepressant effects (22).
Reconstructive 3D-echocardiography. (A) Representative sequential imaging planes marked as white lines in a parasternal long-axis view. (B) Volumetric model of the full-volume dataset as assessed with reconstructive 3D-echocardiography. (C) Representative high-resolution short-axis section at the position of the dark blue line in (A) and dark blue imaging plane in (B). Endocardial (green) and epicardial (red) borders of the left ventricle as well as akinetic (yellow) and hypokinetic (light blue) myocardium were traced manually after visual wall motion analysis. Scale bar: 5 mm.
All imaging planes from basal to apical were visually assessed for: 1) left ventricular wall motion, 2) wall thickness and systolic wall thickening, 3) echocardiographic criteria of scar formation (increased echodensity and wall thinning), and 4) contractile reserve. In addition, quantitative assessment was performed. Therefore, based on visual judgment, circumferential sections were placed to define: a) epicardial circumference, b) endocardial circumference, c) akinetic circumference, d) septal hypokinetic circumference, and e) lateral hypokinetic circumference in each plane at end-diastole (Fig. 2). Additionally, epi- and endocardial regions of interest (ROIs) were traced in end-systole and end-diastole, allowing calculation of left ventricular end-diastolic (LVEDV), end-systolic (LVESV), and stroke volumes (SV), as well as left ventricular mass (LVM) and left ventricular ejection fraction (LVEF). To facilitate visual analysis, intermittently loops were replayed at slow speed (25% of initial speed). For assessment of myocardial surfaces, circumferential sections were multiplied with the intersection interval (0.5 mm) and summarized along the long axis, as validated previously (6).
Myocardial compartments were differentiated by visual assessment following established echocardiographic criteria: a) akinetic/dyskinetic wall motion (collagenous scar): thinned, echodense wall, no systolic wall thickening, no inotropic response to dobutamine; b) hypokinetic wall motion (peri-infarction border zone myocardium): normal myocardial thickness, markedly reduced systolic wall thickening during baseline, reduced inotropic response to dobutamine; c) normokinetic wall motion (remote myocardium): normal myocardial thickness, good systolic wall thickening, good inotropic response to dobutamine.
DNA Preparation and Quantitative Analysis of Cell Survival
Genomic DNA preparation and quantitative real-time PCR analysis of cell survival in the hearts were performed with primers against Sry gene and osteopontin as internal standard (to normalize the amount of DNA) as described previously (14). Samples with known dilutions of male with female DNA were included to verify consistency and to derive a calibration curve for the calculation of male/total ratio. DNA (1 μg) was used for the analysis, and we were able to detect 1 male cell in 100,000 cells.
Histology
Animals were euthanized after the last echocardiographic evaluation; the hearts were excised and placed in 4% paraformaldehyde for 24 h and washed in PBS. Then they were cryoprotected in 18% sucrose and finally embedded with Tissue Tec and stored at −80°C. To investigate scar size and collagen deposition cryosections (5 μm) were stained with Masson's trichrome in intervals of 250 μm from base to apex. The sections were scanned and the infarction areas planimetrically analyzed using Sigmascan Pro 5.0 (Software SigmaScan Pro, Image Analysis, Version 5.0.0, SPSS Science, Germany). The volumes of infarcted and noninfarcted myocardium were calculated from planimetric analysis using modified Simpson's method.
Statistical Analysis
All analyses were performed blinded to randomization. Data were presented as mean ± SD. GraphPad InStat® was used for computerized statistical analysis applying one-way ANOVA and post-hoc Tukey test between the groups. Differences were considered significant if p < 0.05.
Results
A total of 55 animals underwent primary surgery; eight died shortly after surgery, five during infarct induction due to extensive ischemic area, and in four cases ischemia could not be induced, so that 38 animals were subject to analysis (n = 10 for each MNC, MSC, and medium; n = 8 for open-chest control).
Safety and Efficacy of Minimal-Invasive, Echo-Guided Closed-Chest CTx
During the intramyocardial injection the development of an echo-dense intramyocardial deposit was recognized in all injection sites of all animals (3 sites × 30 animal/3 sites × 30 animals). Three weeks after the injection of 106 MNC, 1.4 ± 1.1% of initially injected cells were detected (corresponding to 14,100 cells). Similar engraftment efficacy was found for 106 MSC, namely 1.9 ± 1.2% (i.e. 18,800 cells) and for open-chest CTx (0.8 ± 1.1%, i.e. 8,400 cells; p = 0.3).
r3D-Echo After Percutaneous Bone Marrow Cell Therapy
Left ventricular end-diastolic and end-systolic volumes revealed no significant differences between the groups (Table 1). Global left ventricular ejection fraction (LVEF) after 1 h of ischemia was reduced to similar levels in all groups (Fig. 3). Three weeks after CTx no difference in left ventricular volumes between the groups evolved (Table 1). Similarly, the global systolic function was not different in animals treated with MNC (LVEF 53 ± 3%), MNC open-chest (52 ± 5%) as in animals treated with MSC (50 ± 5%) and in control animals (60 ± 4%). If anything, there was trend towards higher LVEF in control animals (Fig. 3). Stroke volume (SV) revealed no significant differences between the groups with a trend to increased SV in the control animals (Table 2). Stress echocardiography revealed a preserved dobutamine-induced increment in ejection fraction, reflecting a contractile reserve and preserved myocardial viability, in the treated groups after CTx, which however was not higher than that of the medium group (Table 2). Interestingly, the individual amount and absolute number of surviving bone marrow cells found after 3 weeks was neither associated with global and regional left ventricular function nor with the amount of dysfunctional myocardium.
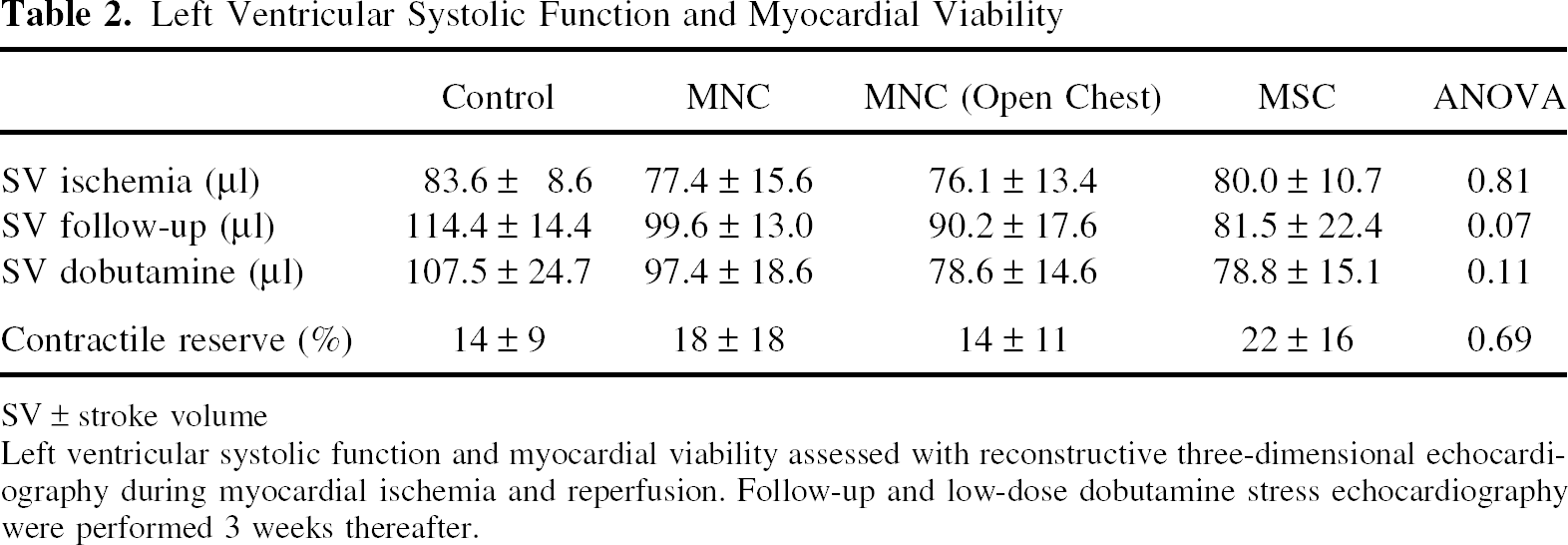
Left ventricular ejection fraction. Left ventricular ejection fraction during myocardial ischemia and reperfusion revealed no significant differences in global systolic function after percutaneous transplantation of mononuclear (MNC) or mesenchymal stem cells (MSC) and open-chest transplantation of MNC (MNC o.c.). Follow-up and low-dose dobutamine stress echocardiography was performed 3 weeks thereafter and demonstrated no significant differences between the therapeutic groups. Left Ventricular Dimensions Left ventricle dimensions were assessed by means of reconstructive three-dimensional echocardiography during myocardial ischemia and reperfusion. Follow-up and low-dose dobutamine stress echocardiography were performed 3 weeks thereafter. LVESV, left ventricular end-systolic volume; LVEDV, left ventricular end-diastolic volume; LVM, left ventricular mass. Left Ventricular Systolic Function and Myocardial Viability SV ± stroke volume Left ventricular systolic function and myocardial viability assessed with reconstructive three-dimensional echocardiography during myocardial ischemia and reperfusion. Follow-up and low-dose dobutamine stress echocardiography were performed 3 weeks thereafter.

To further evaluate the effect of cell therapy we examined the absolute and relative myocardial surface of dysfunctional, hypokinetic, and akinetic myocardium. Further regional analysis revealed no significant differences between the four groups. The surface of akinetic and hypokinetic myocardium at resting conditions as well as after inotropic stimulation decreased in all groups to similar levels, suggesting persistent dysfunction of left ventricular myocardium, which is not influenced by cell therapy with MNC or MSC. Consistently, the recruitment of dysfunctional, but viable myocardium that is reflected in the dobutamine-related reduction of dysfunctional myocardium did not reveal differences between groups (Fig. 4).
Quantitative regional wall motion analysis. Percutaneous transplantation of mononuclear (MNC) and mesenchymal stem cells (MSC), as well as open chest transplantation of MNC (o.c.) were tested as cell therapeutic approaches. Regional wall motion analysis after cell transplantation revealed no significant impact on normalized amount of hypokinetic (left) and akinetic (right) myocardium during myocardial ischemia and reperfusion. Moreover, 3 weeks thereafter neither follow-up nor low-dose dobutamine stress echocardiography demonstrated additive functional effects.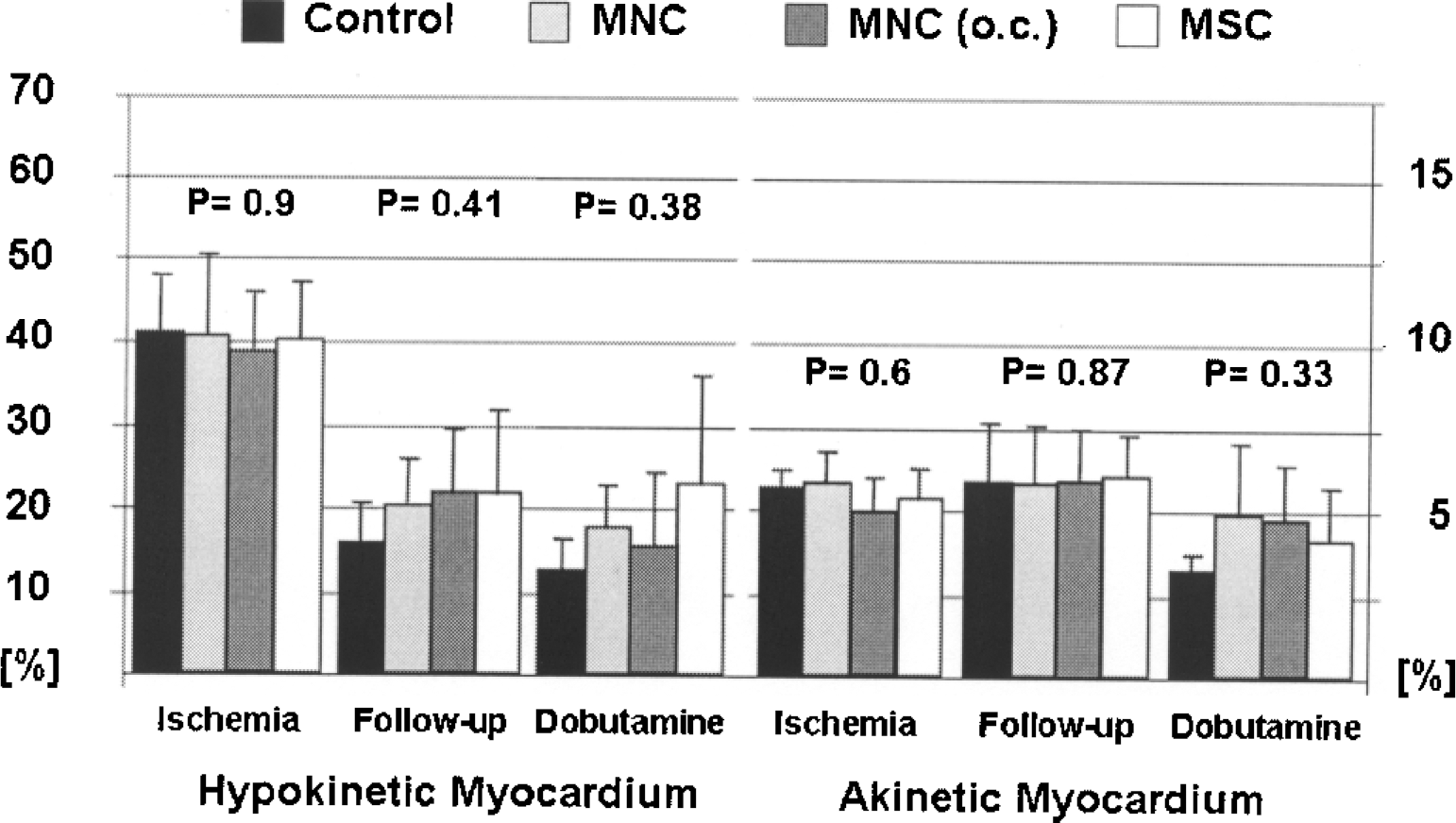
Histology
The size of the collagenous scar as quantified by histomorphometry was not significantly changed 3 weeks after CTx (Fig. 5).
Quantification of left ventricular scar size with histomorphometry. Three weeks after myocardial ischemia/reperfusion injury the extent of left ventricular collagenous scar was not significantly influenced after transplantation of mononuclear (MNC) or mesenchymal stem cells (MSC).
Discussion
In the present study, the validation of minimal invasive, percutaneous echo-guided closed-chest cell transplantation (CTx) of mononuclear (MNC) or mesenchymal stem cells (MSC) and the functional impact of such therapy were studied in a rat model of myocardial infarction and reperfusion. The main findings were that the new mode of cell delivery was comparably safe, feasible, and effective as the invasive approach. However, neither left ventricular dimensions were decreased nor global LVEF was increased 3 weeks after CTx compared to controls. Quantity of dysfunctional myocardium (as assessed with r3D-echo) and size of infarct scar (as assessed by histology) as well as assessment of contractile reserve after inotropic stimulation remained unaffected by bone marrow cell therapy in this model of infarction and reperfusion.
In contrast, previous results from animal experiments showed a wide discrepancy in heart function improvement from significant increase of LVEF to marginal level of recovery after BMC-CTx. Orlic et al. were the first to describe functional benefit after myocardial infarction and the injection of specifically selected bone marrow cells (18). The functional benefit of this procedure was confirmed by many studies using different kinds of bone marrow cells, although the mode of action of bone marrow cell therapy remains a matter of debate (7,15,19). Other studies found a transient benefit that was no longer detectable at later times (3). Meanwhile, there is a handful of well-performed randomized double-blind clinical studies that have assessed the functional impact of mononuclear cells after acute myocardial infarction. The results are controversial. While the largest study detected clear functional benefit for patients treated with bone marrow cells compared to controls (20), two other studies found no influence of bone marrow cell therapy on global myocardial function (9,12). Therefore, in spite of the many studies that were performed to date on bone marrow cell therapy after myocardial infarction, there is need for further information and additional investigations, which might be able to shed light onto this controversial issue.
In the present study no difference between groups (MNC, MSC, and control) was found for either the global systolic function measured as LVEF or the regional function quantified as dysfunctional myocardium. One obvious reason for our sobering results may be that the specific experimental setup with infarction/reperfusion and cell injection immediately after reperfusion is just not suitable to demonstrate the therapeutic effect of bone marrow cells. Thus, in the clinical setting there is some evidence that timing of cell injection should be a few days after infarction and reperfusion (20). Moreover, in most preclinical small-animal studies permanent coronary artery ligation was used, which may be the preferred condition for bone marrow cells to exert their beneficial therapeutic effect (3). The notion that ischemia/reperfusion, albeit clinically relevant, may not be ideal to demonstrate the beneficial effect of bone marrow cell therapy is supported by the partly negative results of the clinical studies. Along that line, we observed that cell persistence was lower in rats and pigs with reperfused myocardial infarcts than in animals with permanent vessel occlusion, which may serve as one reason for lack of effectiveness in ischemia/reperfusion (11). In the present study, however, cell persistence was measured and resulted in 1–2% of initially injected cells at 3 weeks, which is a similar number as previously observed in permanently ligated rat hearts after open-chest CTx (13). Because we do not know the optimal settings, optimal timings, and optimal cell types for cell therapy, it will not be possible to rule out that the experimental setup chosen for this study was suboptimal to demonstrate the efficacy of bone marrow cell therapy for myocardial infarction.
Nevertheless, the current study has several important strengths in the methodologies chosen, which may render some special impact to the data that were obtained. First, to obtain comparable systolic impairment for reproducible determination of therapeutic impact, left ventricular systolic function and extent of dysfunctional myocardium was measured before CTx. Only those animals were included in the study that showed similar impairment of global left ventricular systolic function and wall motion abnormalities. This selection lead to a rather low variability of infarct sizes while still allowing us to use a technique that mimics the pathophysiological conditions of myocardial infarction and reperfusion in the clinical setting (as opposed to cryoinfarction). Secondly, in contrast to almost all other studies in small animals, but similar to the clinical situation, reperfusion of the myocardial infarct was allowed (1). The closed-chest model of temporary left coronary artery occlusion in rats used in this study is novel and rarely applied in experimental BMC-CTx. This approach offers good resemblance with the clinical situation when patients with acute myocardial infarction undergo early PCI to achieve prompt revascularisation and reperfusion. Thereby, it allows investigation of the effect of CTx in addition to the beneficial effect of reperfusion therapy. Furthermore, thoracotomy induces cytokine expression, which may influence the results of small-animal studies in which infarct induction and cell injection are performed during open thorax surgery (17). Therefore, ischemia and reperfusion as well as cell injection are performed one week after suture placement during thoracotomy reducing the confounding factor of major surgery. Thirdly, interventional percutaneous cell injection was performed during echocardiographic control, which allowed to assess successful injections, and which allowed visualization of the needle to guide the injections exactly into the functionally impaired myocardial peri-infarction border zone. This procedure was performed for the first time by Springer et al. (21). Their quantitative dye injection studies in mouse hearts demonstrated that guided closed-chest injections and open-chest injections deliver comparable amounts of injectate and prevents erroneous cell injections into the left ventricular cavity. Here, we were able to detect an ultrasound-opaque cell deposition as quality control for intramyocardial delivery (even without the use of a contrast agent), which is impossible during open-thorax cell injections.
Fourthly, and in contrast to recent studies in small animals, we chose reconstructive 3D-echocardiography (r3D-echo) as method for the assessment of left ventricular dimensions, quantification of global and regional cardiac function, and investigation of myocardial viability. Former studies commonly used one- or two-dimensional echocardiography, which is limited by the asymmetric shape of the infarcted left ventricle, although all mathematical models are validated for symmetric ventricles (5,23). Moreover, approximations do not allow quantification of regional dysfunction secondary to infarction. These limitations are overcome here by acquisition of a full-volume dataset of the rat heart to accurately measure left ventricular dimensions, global and regional function, and contractile reserve (6). Previously, it was shown that the accuracy of this method is close to the accuracy of MRI in small animals (4,16), and may therefore help to detect and quantify also small therapeutic effects of CTx. Additionally, and finally, we extended the imaging protocol by inotropic testing for investigation of global contractile reserve and assessment of myocardial viability 3 weeks after CTx (10). To our knowledge this is the first investigation of myocardial viability with r3D-echo after CTx. This method allows valid quantification of regional myocardial dysfunction and global contractile reserve. Furthermore, r3D-echo allows precise differentiation of vital myocardial border zone and identification of nonvital transmural scar tissue noninvasively (6). Ultimately, inotropic stimulation exposed no disguised regional and global left ventricular functional improvement of reperfused but dysfunctional myocardium in animals after BMC-CTx.
In conclusion, we found that closed-chest intramyocardial injection of BMCs is as effective as open-chest injection with comparable (but low) cell persistence at 3 weeks after transplantation. At this distinct time point after transplantation, we did observe neither deceleration of left ventricular dilatation nor improvement of the global and regional systolic function. Infarct size as assessed by echocardiography and histomorphometry also remained unchanged. The results support the notion that the therapeutic efficacy of bone marrow cell therapy after myocardial infarction may be limited and may depend on very distinct conditions that apparently were not chosen in the present study. The closed-chest echo-guided percutaneous technique of cell injection may be particularly useful for repeated and/or sequential cell applications after myocardial infarction.
Footnotes
Acknowledgments
A.G. was supported by the German Heart Foundation. A.Z. received a stipend from the Köln Fortune program of the Medical Faculty of the University of Cologne (to A.Z. and J.M.-E.). K.T. was supported by a research grant of the German Research Foundation (DFG-SFB 656, C3). This study was supported by the Ernst und Berta Grimmke Stiftung (J.M.-E.). We thank Pascal Paschenda, Balakrishnan Karthikeyan, and Rafael Struck for their excellent technical assistance.
