Abstract
Tissue engineering holds the promise of providing new solutions for heart transplant shortages and pediatric heart transplantation. The aim of this study was to evaluate the ability of a peritoneal-generated, tissue-engineered cardiac patch to replace damaged myocardium in a heterotopic heart transplant model. Fetal cardiac cells (1 × 106/scaffold) from syngeneic Lewis rats were seeded into highly porous alginate scaffolds. The cell constructs were cultured in vitro for 4 days and then they were implanted into the rat peritoneal cavity for 1 week. During this time the peritoneal-implanted patches were vascularized and populated with myofibroblasts. They were harvested and their performance in an infrarenal heterotopic abdominal heart transplantation model was examined (n = 15). After transplantation and before reperfusion of the donor heart, a 5-mm left (n = 6) or right (n = 9) ventriculotomy was performed and the patch was sutured onto the donor heart to repair the defect. Echocardiographical studies carried out 1–2 weeks after transplantation showed normal LV function in seven of the eight hearts studied. After 1 month, visual examination of the grafted patch revealed no aneurysmal dilatation. Microscopic examination revealed, in most of the cardiac patches, a complete disappearance of the scaffold and its replacement by a consistent tissue composed of myofibroblasts embedded in collagen bundles. The cardiac patch was enriched with a relatively large number of infiltrating blood vessels. In conclusion, cardiac patches generated in the peritoneum were developed into consistent tissue patches with properties to seal and correct myocardial defects. Our study also offers a viable rat model for screening and evaluating new concepts in cardiac reconstruction and engineering.
Introduction
Tissue engineering of viable cardiac patches with a potential to grow and remodel may provide a new solution to the serious problems of heart donor shortage and pediatric heart transplantation (4). However, to advance this clinically relevant strategy, there is a need for animal models to test the scientific and clinical feasibility of tissue engineering.
Much of the encouraging data in the field of cardiovascular tissue engineering have been obtained from small animal models (7,9,11,16). The small-animal model has many advantages, such as the accessibility of inbred animals, availability of autologous-like cell sources, and low cost. Nonetheless, cardiac tissue engineering in the murine model is limited by difficulties in replacing full-thickness left ventricular (LV) free wall. Thus, the rat model of heterotopic heart transplantation offers the ability to operate on an explanted donor organ without using cardiopulmonary bypass or cardiovascular compromise (8).
For full free wall reconstruction, a tissue-engineered patch should be muscular, vascularized, elastic, and ideally possess contractile potential. As this goal is hard to achieve using current in vitro techniques, new strategies are warranted (4,12). The aim of our study was, therefore, to develop a novel cardiac patch to replace damaged heart muscle. Accordingly, a cardiac patch was developed by seeding fetal cardiomyocytes within macroporous alginate scaffolds followed by their vascularization into the rat peritoneal cavity prior to transplantation onto a damaged donor heart. The performance of the novel cardiac patch was tested in a rat model of heterotopic heart transplant, which enables replacement of a full-thickness free wall. Our model can be thus useful for screening and evaluating new strategies and methods of cardiac tissue engineering.
Materials and Methods
This study was performed in accordance with the guidelines of The Animal Care and Use Committee of the Tel-Aviv University, which conforms to the policies of the American Heart Association.
Cardiac Cell Isolation
Fetal cardiomyocytes (16 days of gestation) from Lewis rats were used to seed the alginate scaffold. Hearts were removed and placed in cold dissociation buffer. The ventricles were cut into 1–2-mm cubes and dissociated through gentle stirring in a solution of proteolytic enzymes (RDB, obtained from Israel Institute for Biological Research, Ness-Ziona, Israel), prepared from a fig tree extract. The RDB was diluted 1:100 in Ca2+- and Mg2+-free PBS at 25°C and incubated with the tissues pieces for a few cycles of 10 min each (6). The mixture was centrifuged at 300 × g for 5 min and the supernatant was discarded. The cells were resuspended in Dulbecco's modified Eagle's medium (DMEM) containing 10% horse serum (Biological Industries, Kibbutz Beit Haemek, Israel) and 2% chick embryo extract (Biological Industries) and preplated onto a 60-mm dish for 15 min at 37°C. An aliquot of the nonattached cells, >70% of which were cardiomyocytes (5), was counted and was used for seeding the alginate scaffold.
3D Culturing Within Alginate Scaffolds
The isolated cardiac cells were seeded, at a concentration of 1 × 106 cells per scaffold, within cylindrical alginate scaffolds (6 × 1 mm, diameter × height (Fig. 1A) placed in a 96-well plate. The macroporous scaffolds were prepared from a 1% (w/v) solution of sodium alginate (LVG; NovaMatrix, FMC Biopolymers, Drammen, Norway) cross-linked with a 0.3% (w/v) solution of D-gluconic acid hemicalcium salt, and subjected to a controlled freeze-dry procedure (15). The cells were seeded into the dry alginate scaffolds by dropping the cardiac cell suspension on top of the scaffold. Due to their hydrophilic nature, the scaffolds were easily wetted by the culture medium such that cell seeding was efficient (5). The cultures were incubated in a humidified atmosphere of 5% CO2 and 95% air at 37°C, with daily medium changes. The cell constructs were followed microscopically for 4 days before implantation into the peritoneal cavity. The viability and metabolic activities of the cardiac cell constructs were tested by the MTT viability assay (5). Cell distribution was assessed by histology and hematoxylin-eosin (H&E) staining of thin cross sections of the cardiac constructs.

Cardiac patch characteristics. (A) Fetal cardiomyocytes were seeded in alginate scaffolds (1 × 6 mm, thickness × height). (B) The scaffolds are characterized by highly porous and interconnecting pore structure with 50–100 μm pore diameter (SEM images). (C) By H&E histology after cell seeding, the cells are distributed in the alginate matrix as small clusters (scale bar: 50 μm).
Surgical Procedures
Peritoneal transplantation of the seeded scaffold was performed 4 days after cell seeding in the alginate scaffold and cultivation in vitro. Lewis rats were anesthetized with ketamine (100 mg/kg), xylazine (10 mg/kg), and, under sterile conditions, a small incision was made in the shaved abdominal wall and two or three cell-seeded or empty scaffolds were placed inside the peritoneal cavity of each animal.
One to 4 weeks after peritoneal implantation, heterotopic heart transplantation was performed in Lewis rats (weighting ~250 g), using the technique described by Ono and Lindsey (13). The donor rat underwent a midline abdominal and chest incision separating the anterior chest and the diaphragm; the anterior rib cage was hinged, exposing the heart. UW (Viaspan, DuPont) cardioplegic solution, containing heparin (100 U/10 ml), was administered through the inferior vena cava in order to arrest and preserve the heart. In the recipient rat, a long abdominal incision was performed, exposing the abdominal aorta and the inferior vena cava from a short segment just below the renal arteries to the point chosen for the anastomosis. The abdominal vessels were clamped, the ascending aorta of the donor heart was anastomosed to the abdominal aorta using 8/0 prolene suture, and the pulmonary artery was anastomosed to the inferior vena cava using 10/0 prolene suture. A longitudinal ventriculotomy was performed either on the left or right ventricular free wall and the peritoneal-generated tissue-engineered patch was sutured to seal the defect using 8/0 prolene suture. After declamping the abdominal vessels, the transplanted heart spontaneously resumed contractions and the abdominal cavity was closed using an absorbable 2/0 Vicryl running suture.
Evaluate Remodeling and Contractility by Echocardiography
An echocardiography study designed to evaluate the performance of the heart transplant was performed on eight animals, 7 to 14 days after heart transplantation. Rats were anesthetized as described above, the abdomen was shaved, and the rats were placed supine. Transabdominal echocardiograms were performed with a commercially available echocardiography system equipped with 12-MHz phased-array transducer (S12, Sonos 5500, Hewlett-Packard). The heart was imaged in the 2D mode in at least two planes of the short axis view. LV function was semiquantitatively assessed by an experienced echocardiographer using visual grading of global and regional contraction (1).
Histological and Immunohistochemical Examination
Representative samples of the cell-seeded scaffolds were fixed and immunostained 1 week after implantation in the peritoneum. Four weeks after transplantation, recipient animals were killed by phenobarbital overdose. The hearts were harvested, sectioned, and processed for histological and immunohistochemical examination. Adjacent blocks of the peritoneal-generated cardiac patch or harvested heart were embedded in paraffin, sectioned into 5-μm slices, and stained with H&E or Masson trichrome for collagen. Serial sections were immunolabeled with antibodies against the smooth muscle α-actin isoform (SMA, Sigma, St. Louis, MO), sarcomeric actin (Zymed-Invitrogen, Carlsbad, CA), myosin heavy chain (MHC, Zymed-Invitrogen), or actinin (Sigma) and counterstained with hematoxylin.
Results
Overall, 52 syngeneic Lewis rats were included in our study, designed to establish the model and evaluate cardiac patch performance. In a pilot study aimed at establishing the feasibility of blood vessel penetration into the macroporous scaffold and technical aspects of the method, each of two rats were implanted with two to three unseeded (empty) scaffolds, inserted into the peritoneal cavity. Twenty rats were used as “bioreactors” for the peritoneal-generated cardiac patches; two to three cell constructs were implanted per rat. Fifteen heterotopic heart transplantations were performed using 30 rats (15 donors and 15 recipients). In the recipients, the defects were either in the LV (n = 6) or RV (n = 9) wall. Five rats died within 24 h of peritoneal implantation of the seeded scaffolds because of surgical complications. There were no perioperative deaths in the rats that underwent heterotopic heart transplantation and surgical reconstruction, nor were any patch failures observed.
Cardiac Cell Constructs
The alginate scaffolds used in the present study displayed a highly porous structure (>90%) with interconnected pores and pore sizes that ranged between 50 and 100 μm in diameter, as judged by scanning electron microscope (SEM) examination (Fig. 1B). These properties should enable the penetration of blood vessels into the macroporous matrix, as well as its vascularization (14). The alginate scaffolds were sufficiently stable in culture and were easily handled. Seeding of cardiac cells onto the alginate scaffolds was efficient due to the hydrophilic nature of the alginate material, which allowed rapid wetting of the matrix by the culture medium. The viable cells were uniformly distributed throughout the scaffold volume and were mainly located within the scaffold pores in small clusters (Fig. 1C). After 4 days in culture and following validation of cell viability, the cardiac cell constructs were implanted into the peritoneum.
A Peritoneal-Generated Cardiac Patch
The pilot study intended to examine blood vessel entry into the unseeded alginate scaffolds after implantation into the peritoneum. Histological examination of 1–2-week engrafted scaffolds revealed extensive blood vessel in-growth into the entire volume of the scaffold, validating its interconnecting pore structure.
Visual inspection of the peritoneal-generated cardiac patches, examined 1 week after implantation, revealed coverage by a thin connective tissue, enriched with blood vessels (Fig. 2A). The extensive neovascularization into the cardiac patch emerged from the neighboring peritoneal network. Microscopic examination of cross sections of the peritoneal-generated cardiac patch, positively stained for SMA, revealed the presence of connective tissue infiltrated with new large blood vessels, particularly in the periphery of the graft (Fig. 2B). In the center of the patch, the blood vessels were smaller in size (Fig. 2C). Their positive staining for SMA points, however, to pericyte and smooth muscle cell coverage of the vessel, and to its maturity. Myocardial fibers or typical sarcomeres were not visible in detectable numbers in the peritoneal-generated cardiac patch.

Vascularization and tissue engineering of the cardiac patch in the peritoneal cavity for a week. (A) A macroscopic view reveals a well-vascularized peritoneal-generated cardiac patch. Microscopic examination of representative cross sections of the peritoneal-generated cardiac patch, after immunostaining for smooth muscle α-actin (brown color), revealed extensive neovascularization (V) and the population of the patch by myofibroblasts (M). Large new vessels were particularly apparent at the periphery of the patch (B), while in the center, most vessels were small (C).
Heterotopic Heart Transplantation
One month after transplantation of the peritoneal-generated cardiac patch onto heterotropic rat heart, macroscopic examination of H&E-stained cross sections revealed, in most cases, the presence of the cardiac patch sealing the LV or RV defect (Fig. 3). In three cases, the cardiac patch was not identified, either because it underwent complete integration into the ventricular wall (n = 2) or because it was detached (n = 1). Nonetheless, the LV or RV defect was sealed in each case (Fig. 3B). With most of the transplanted cardiac patches, tissue in-growth onto the scaffolds was nearly complete. We identified small acellular areas in the center of only four of nine RV grafts and two of six LV grafts (Fig. 3C).
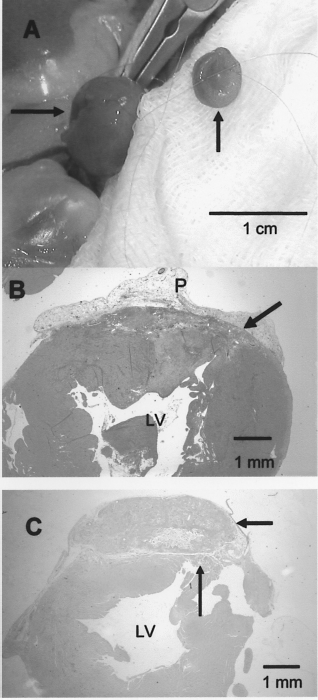
Heterotopic heart transplantation and LV reconstruction. (A) A longitudinal ventriculotomy was performed on the left ventricular free wall (long arrow) and the peritoneal-generated cardiac patch (short arrow) was sutured to seal the defect. (B, C) One month after heterotopic heart transplantation of the cardiac patch, H&E-stained cross sections revealed that the site of LV ventriculotomy (thin arrow) and LV cavity were sealed by the cardiac patch (thick arrow). In (B), a complete integration of the cardiac patch with the LV wall (arrow) is seen; the patch is covered by the remaining peritoneal adhesion (P). In (C), the cardiac patch is still seen with small acellular areas, although most of its area is cell populated. The anchorage site of the cardiac patch to the myocardium is identified (long arrow).
Microscopically, the heart-transplanted cardiac patches were populated with newly formed capillaries and arterioles embedded within a confluent collagen bundle matrix that stained positive for Masson trichrome (Fig. 4). Typical myofibroblasts and infiltrated macrophages were also present. At many anchorage sites, the cardiac patch showed complete integration with the host tissue, with the specimens showing almost complete disappearance of the scaffold (Fig. 4A). The fibroblasts within the cardiac patches were stained intensely for SMA (Fig. 5). Staining for myogenic proteins, such as slow MHC, sarcomeric actin, or actinin was not apparent in the implanted cardiac patches, 4 weeks after transplantation (Fig. 5).
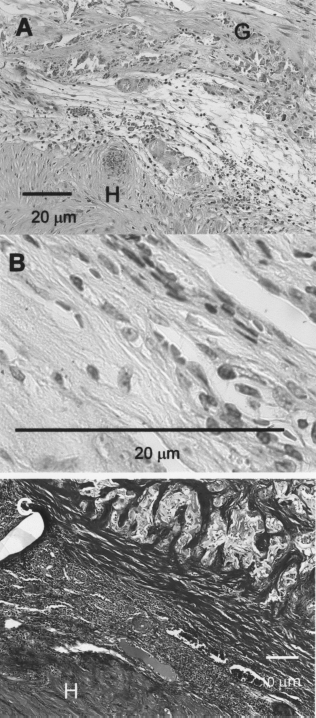
(A) A higher magnification view of the transplanted heart (Fig. 3) concentrates on the border between the transplanted cardiac patch (G) and the host myocardium (H), with some remnant of the alginate scaffold at the anchoring site seen. (B) A closer look at the transplanted cardiac patch reveals that extensive areas are composed of fibroblasts and extracellular matrix. (C) Masson trichrome staining revealed extensive areas of collagen bundles in the patch. The collagen stained blue, while the host myocardium (H) stained deep red.

Immunostaining of the transplanted cardiac patch for cardiac markers. (A) Smooth muscle α-actin immunostaining reveals the patch to be colonized with many myofibroblasts (brown color; 1000×). (B) Immunostaining for actinin revealed positive (brown color) sarcomeric staining and striation (arrows) at the reconstructed host myocardium, while no staining for actinin was found inside the patch (C; 1000×).
Echocardiography
Semiquantitative two-dimensional echocardiographic assessment showed good global LV function of the abdominal transplanted heart without any segmental wall motion abnormalities in the segment of the patch grafting or other areas in seven of the eight animals studied (Fig. 6). LV contractility appeared synchronized and smooth. Only one animal exhibited moderate global LV dysfunction, compatible with a clinical and histological picture of transplant rejection. We did not identify dyskinesis, akinesis, or aneurysm formation in the area of the LV incision repaired by the tissue-engineered cardiac patch.

Two-dimensional echocardiography (short-axis view) of the transplanted, reconstructed LV provides evaluation of global and segmental LV contractility.
Discussion
We demonstrate the use of a peritoneal-generated cardiac patch to replace a full-thickness section of the ventricle free wall in a rat model of heterotopic heart transplantation. The cardiac patch was developed by seeding fetal cardiomyocytes within macroporous alginate scaffolds, followed by implantation into the rat peritoneum cavity prior to transplantation onto the heterotopic heart. The construction of the cardiac patch in vivo resulted in a consistent tissue due to the extensive cell in-growth from the host and the extensive vascularization of the patch. Such cardiac patches may thus possess the mechanical and elastic properties and tissue consistency required to repair LV defects.
The use of the peritoneum for tissue engineering offers several advantages, including the maintenance of cell construct sterility and contribution to two critical properties of the cardiac patch, namely elasticity and mechanical strength, traits that are necessary for the long-term success of the cardiac patch. By growing the neotissues in the peritoneal cavity, serving as a “natural bioreactor,” it is possible to create an environment that duplicates conditions required for the development of normal tissue. The neotissues are formed with greater collagen content, making them better suited for the active condition of the cardiac work load. Our results also showed that the peritoneal-generated cardiac patch was colonized and populated by autologous myofibroblasts and an extensive blood vessel network that could add to the firmness and confluence of the tissue-engineered cardiac patch. Another advantage of using the peritoneal cavity as a bioreactor for tissue engineering is the creation of a cardiac patch from autologous cells in the host environment.
The peritoneal-generated cardiac patch was tested in the heterotopic heart model, which enables the replacement of a full-thickness wall. The model was initially developed by Krupnick et al. (8), who employed scaffolds made of poly(tetrafluoroethylene), poly(lactide) mesh, and type I and IV collagen hydrogel and seeded with mesenchymal stem cells. To facilitate implantation of the cell construct into the myocardium, the construct in their study was attached to an identically sized support layer composed of either a biodegradable or synthetic material. In our model, the tissue engineering of the cardiac patch in the peritoneal cavity resulted in a well-built patch, which obviates the need for any such back-up support layer. Furthermore, our results reveal that the peritoneal-generated cardiac patch was able to seal the ventricular (LV or RV) defect, enabling good global functioning of the wall.
An interesting finding of this study is that the peritoneal-generated cardiac patch lacks the characteristic features of myocardium, namely no striation has been demonstrated in the cardiac patch. Still, the patch was successful in sealing the defect in the LV wall and further maintaining the global contractility of the heart. There are several possible explanations for this observation: 1) the peritoneal-generated cardiac patch, with its high content of collagen and myofibroblasts, forms an elastic patch that can seal the defect and not affect the global contraction of the heart; 2) the patch, in addition to sealing the defect in the myocardium, may secrete growth factors that can contribute to maintenance of the viable remaining myocardium; and 3) the cardiac patch helps preserve the thickness of the ventricular wall. These explanations have been offered previously both by us (6,9,10) as well as by others (7,11,16) to explain the beneficial effects of cell- or tissue-engineered cardiac biograft transplantation in a rat model of infarction.
In the present article, the strategy of tissue engineering has been employed for correcting and sealing a defect in the myocardium. This challenge calls for a rigorous approach aimed at achieving prevascularized consistent tissue. We generated such tissue by using the peritoneal civility as our bioreactor. This approach has been previously used by Campbell et al. (3) to re-create vascular replacements. In that report, the conduits were grown in the peritoneal cavity of the same animals in which they are grafted (thereby ensuring no rejection), in the short time of 2–3 weeks. It was subsequently shown that the peritoneal-grown conduits are almost indistinguishable from the native vessels. The same group (2) recently showed that myofibroblast tissue produced in the peritoneal cavity is sufficiently plastic to permit differentiation of cells into bladder, vas deferens, or uterine smooth muscle.
In summary, we have applied a viable small animal model to test tissue-engineered cardiac patch generated in the peritoneum. Our model is handy, inexpensive, and relatively easy to establish. We believe that this model may open new perspectives in cardiac tissue engineering research. Cardiac patches may thus be developed to replace infarcted myocardium or to reconstruct congenital heart defects, thus avoiding the need for organ transplantation.
Footnotes
Acknowledgments
The research was supported by a grant from the Israel Science Foundation (793/04). Smadar Cohen holds the Claire & Harold Oshry Professor Chair in Biotechnology.
