Abstract
Intracerebral transplantation of dopaminergic (DA) cells is currently further explored as a potential restorative therapy for Parkinson's disease (PD). However, before they can be considered for a more widespread clinical use a number of critical issues have to be resolved, including an optimized transplantation protocol. This study has been performed in a rat 6-hydroxydopamine model of PD and is based on the microtransplantation approach. The results demonstrate a reduced survival (threefold) for a single cell suspension of E14 rat ventral mesencephalon compared to a fragment suspension when a metal cannula is used for implantation. However, fragment suspensions result in a more variable graft survival and ectopically placed cells along the implantation tract. When a glass capillary is used for implantation, the survival of the single cell suspension (so-called “micrograft”) improved by fourfold (vs. single cells/metal cannula) and is superior to the combination of the metal cannula and fragment suspension (+40%). The micrografts show a reduced variability in DA neuron survival as well as fewer ectopically placed cells. Moreover, the implantation time can significantly be reduced from 19 to 7 min in micrografted animals without a compromise in DA graft survival and functional behavioral outcome. Using the microtransplantation approach graft size can be tailored effectively by varying the density of the final cell suspension at least between 11,000 and 320,000 cells/μl, resulting in comparable survival of tyrosine hydroxylase (TH)-positive neurons in the range of 2–4%. With this approach no more than 100 surviving TH-positive neurons are necessary to produce functional effects in the amphetamine-induced rotation test. Interestingly, we found that DA micrografts into lesion striatum present 20% higher survival rates of TH neurons in comparison to the intact striatum. In summary, these results provide further evidence for the usefulness of the microtransplantation approach and allow for a more precise and tailored adaptation of the implantation parameters for further studies on DA, and possibly also other neural-, glial-, and stem cell-derived grafts.
Introduction
Transplantation of dopaminergic (DA) tissue was performed initially by the implantation of whole pieces of the ventral mesencephalon (VM) into cortical cavities overlying the striatal complex (5). This has resulted in functional DA grafts but was confounded with a limited accessibility of DA grafted-derived fiber reinnervation only to the close vicinity around the transplants (i.e., the dorsal striatum). Björklund and colleagues (5, 6) introduced the cell suspension approach that significantly improved the placement of DA grafts into deeper brain structures by using stereotactic techniques and a partial dissociation of the VM tissue (7). This method has been widely used over the last two decades and has become a “standard” neurobiological tool in many laboratories (9, 13). We have subsequently further optimized this grafting protocol by introducing a more refined implantation instrument, a more vigorous preparation of single cell suspension, and a multiple placement of so-called micrografts with a volume of 250–500 nl cell suspension per graft deposit (26, 27). This has led to a significant improvement of DA graft survival, reproducibility, and accuracy of graft implantations (21, 22) and a more extensive behavioral recovery in neonatal and adult animal models of Parkinson's disease (PD) (23–25). However, following the original description (26) a number of interesting questions related to the implantation procedure have emerged that are relevant also for the more general and expanding field of stem cell transplantation and therefore prompted further investigations.
The current study was undertaken in order to address the following issues: i) different preparation techniques of the cell suspension grafted with two different implantation cannulas, ii) implantation into the intact versus the lesioned hemisphere, iii) duration time of implantation procedure, and iv) the influence of the final concentration of DA cell suspensions on the survival, tyrosine hydroxylase (TH)-positive fiber outgrowth, graft volume, and functional behavioral recovery following intrastriatal implantation into unilaterally 6-hydroxydopamine (6-OHDA)-lesioned rats.
Materials and Methods
Experimental Design
A total of 66 adult female Sprague-Dawley (SPRD) rats weighing 250–300 g at the beginning of the experiment were used in three different experiments. Animals were housed two or three to a cage in a temperature-controlled environment under a 12-h light/dark cycle with free access to food and water. All animals were subjected to unilateral 6-OHDA lesion and subsequent intrastriatal transplantation. Stereotactic surgeries and transcardial perfusion were performed under general anesthesia with ketamine-hydrochloride and xylazine (rompune). Two weeks after the 6-OHDA lesion, amphetamine-induced (all experiments) and apomorphine-induced (only experiment 1) rotational behavior was examined and the lesioned animals were distributed into equal groups with respect to their scores in the amphetamine-induced rotation test. Five to eight (experiment 2) weeks after 6-OHDA lesion transplantation surgery was performed. The grafts were derived from VM of 14-day-old rat embryos (E14, crown-rump length 10–11 mm). After transplantation, rotation tests were performed at different time intervals. Finally, animals were perfused transcardially and their brains were processed for TH immunohistochemistry and quantitative analysis of the total number of surviving cells per graft.
6-OHDA Lesion Surgery
All animals received two stereotactic injections of 6-OHDA using a 10-μl Hamilton syringe into the right mesostriatal pathway at the following coordinates (in mm with reference to bregma and dura): i) 2.5 μl of 6-OHDA hydrochloride (Sigma H4381, 3.6 μg/μl in 0.2 mg/ml L-ascorbate-saline) at AP −4.4, LAT −1.2, VERT −7.8, tooth bar −2.3; ii) 3 μl of 6-OHDA at AP −4, LAT −0.8, VERT −8, tooth bar +3.4. The injection rate was 1 μl/min and the cannula was left at the injection place for additional 4 min before slowly being retracted.
Transplantation Surgery (Fig. 1 and Table 1)

Schematic overview illustrating the design of experiment 1 [modified from Dunnett and Björklund (11)]. VMs of 14-day-old rat embryos were dissected (1. Step) and prepared either as a fragment suspension (FS) or single cell suspension (SS), as described in Materials and Methods (2. Step). The FS was implanted (3. Step) only with the metal cannula in one group (MC-FS, n = 6). The single cell suspension was implanted either via the metal cannula (MC-SS, n = 8) or via the glass capillary (GC-SS, n = 7). All three graft groups received two trajectories of two 0.5-μl deposits each into both the lesioned (right) and nonlesioned (left) striatum (3. Step). CPU, caudateputamen unit; LV, lateral ventricle; CC, corpus callosum.
Parameters for the Intrastriatal Transplantation of Nigral Cell Suspensions

FS, fragment suspension; SS, single cell suspension.
Experiment 1: Cell Preparation, Implantation Cannula, and Degree of DA Denervation
Five weeks after lesion surgery, DA-rich cell suspensions were prepared from the VM of E14 SPRD rat embryos according to the modified microtransplantation approach (26, 27) and based on the cell suspension technique described by Björklund et al. (7). Three experimental graft groups were assigned according to the implantation protocol as following: metal cannula + single cell suspension (MC-SS); metal cannula + fragment suspension (MC-FS); glass capillary + single cell suspension (GC-SS). The metal cannula had an outer diameter (OD) of 460 μm and an inner diameter (ID) of 260 μm; the OD of the glass capillary was 50–70 μm. The dissected VM pieces were incubated in 0.1% trypsin (Worthington, Freehold, NJ), 0.05% DNase (SigmaDN-25; Sigma, St. Louis, MO), and DMEM (Dulbecco's modified Eagle's medium) at 37°C for 20 min followed by four rinses with 0.05% DNase/DMEM. Two different cell suspensions were prepared: single cell suspension and fragment suspension. For the single cell suspension 25 VMs were incubated followed by a mechanical dissociation using 1-ml and 200-μl Eppendorf pipette tips (about 15 strokes each) and a centrifugation at 600 rpm for 5 min. The remaining pellet was resuspended with 0.05% DNase/DMEM in a final volume of 125 μl (5 μl per VM). The final cell suspension contained 113,000 cells/μl and the viability was more than 95%, as determined by trypan blue dye exclusion assay. For the fragment suspension, 16 VMs were mechanically triturated only with the 1-ml Eppendorf pipette tip (15 strokes) and resuspended in 80 μl 0.05% DNase/DMEM (5 μl per VM). The final cell count of the fragment suspension revealed 50,000 cells/μl, but this represents only the percentage of single cells within the suspension. In addition, about 50% of the suspension remained in the “tissue fragment” state due to incomplete dissociation where cell numbers cannot reliably be counted.
Using the two implantation instruments, the stereotactic implantation was performed bilaterally into the striatum placing two deposits (one medial and one lateral) of 0.5 μl each along two trajectories of each suspension. A total of 21 unilaterally 6-OHDA-lesioned rats were allocated to the three different graft groups and implanted using the parameters provided in Table 1.
Experiment 2: Duration of Implantation
Twenty rats with a good rotational asymmetry score [amphetamine >6 turns per minute (tpm)] were split equally in respect to their rotational scores in two experimental groups: TX19min and TX7min (n = 10 each). Eight weeks following lesion surgery and rotation tests, DA-rich single cell suspension were prepared from the VM of E14 rat embryos, as described above. Briefly, a total of 140 μl of cell suspension was prepared with a final cell concentration of 165,000 cells/μl and viability greater than 96%. In both groups the single cell suspension was implanted with the glass capillary (OD 50–70 μm) connected to a 2-μl Hamilton microsyringe into the right lesioned hemisphere, both into the striatum and nucleus accumbens along a total of three trajectories, as outlined in Table 1. The injection rate was 0.5 μl/min. In group 1 (TX19min) each graft deposit injection was followed by a 1-min waiting period in addition to a 3-min waiting period after the final deposit of each tract (“slow hand”). This resulted in a total implantation time of 19 min per hemisphere. In group 2 all the graft deposits were injected subsequently with the same injection rate, but in contrast, no additional waiting was added after the injection shortening the overall implantation time to 7 min per hemisphere (“fast hand”).
Experiment 3: Cell Density of the Final Suspension
Twenty-five rats with a good rotational score (amphetamine >6 tpm) were split equally with respect to their rotational scores in five experimental groups (n = 5 each) and named after the final cell concentration per μl of the cell suspension of each group multiplied by 1000 (Table 2): TX11, TX23, TX52, TX108, and TX320. A total of 200 μl of a single cell suspension was prepared from the VM of E14 rat embryos, as described above. The initial cell number was 320,000 cells/μl and the viability greater than 97%. Then 100 μl of this suspension was diluted 1:1 with 0.05% DNase/DMEM for the further four consecutive steps resulting in a total of five cell suspensions with 320,000, 108,000, 52,000, 23,000, and 11,000 cells/μl, respectively, as determined by the trypan blue exclusion method. The observed deviations from a theoretical halving of the cell counts are most likely due to sampling variations of the individual cell suspensions. Animals in each graft group received four deposits of 0.5 μl along two implantation tracts in the dorsal striatum. Coordinates are given in Table 1.
Influence of the Density of the Final Cell Suspension on DA Micrograft Survival

Based on 10% of TH-positive neurons in the original cell suspension.
Drug-Induced Rotational Behavior
After lesion and transplantation surgery, amphetamine- and apomorphine-induced (only Experiment 1) rotations were performed in automatic rotometer bowls according to Ungerstedt and Arbuthnott in 1970 (34). When both tests were performed the animals were given first 0.05 mg/kg apomorphine SC (hydrochloride hemihydrate, Sigma A4393; diluted in saline) and right and left full body turns were monitored over 40 min. Four days later, the animals were injected IP with 2.5 mg/kg d-amphetamine (sulfate, Sigma A5880; in ascorbate-saline) and the rotational behavior was recorded for 90 min.
Immunohistochemistry
Ten weeks after the transplantation, animals were deeply anaesthetized with ketamine/xylazine and perfused transcardially with 50 ml of 0.9% saline followed by 300 ml of 4% paraformaldehyde in 0.1 M phosphate buffer (PB) (pH 7.4). Their brains were postfixed overnight, dehydrated in 20% sucrose/0.1 M PB, and cut in serial coronal sections (30–40 μm thick) with a freezing microtome. In order to assess the graft survival, every third section was processed for TH immunohistochemistry (1:2500, anti-TH, Sigma) using the ABC kit and DAB for visualization as previously described (22).
Microscopic Analysis
In all animals the number of TH-positive neurons of each graft was counted under the microscope (20× magnification) using the formula of Abercrombie (1) to calculate the total number of surviving cells per graft. Utilizing the CAST-Grid software (Version 1.10, Olympus Denmark, Copenhagen, Denmark) the volume of the intrastriatal grafts was measured. In one series of sections the area of the grafts was delineated on the computer screen at 4× magnification. The result of the measured area was multiplied by 90/120 μm (30/40-μm sections × 3 series) to estimate the total volume [counted number of points × A (p) × number of series × thickness of sections].
Statistical Analysis
Results are expressed as means ± SEM for the different transplantation groups. Comparisons between the groups were performed by using one-factor or two-factor analysis of variance (ANOVA) followed by a Student-Newman-Keuls post hoc test, or by a nonparametric Mann-Whitney U-test with Bonferroni corrections for multiple comparisons when appropriate. The level of statistical significance was set at p < 0.05. For the evaluation of the number of surviving cells and graft volumes an average between the medial and lateral graft in each animal was used.
Results
Experiment 1: Cell Preparation, Implantation Cannula, and Degree of DA Denervation
Surviving DA grafts were found in all animals and in both hemispheres. Typically, both deposits along the injection tract had fused into an elongated graft oriented along the dorsoventral aspect of the striatum, as illustrated in Figure 2. Grafts in the lesioned hemisphere demonstrated moderate (MC-SS, Fig. 2A) to substantial (MC-FS and GC-SS, Fig. 2B and C) TH-positive fiber reinnervation of the striatum. Interestingly, surviving TH-positive neurons were dispersed as single cells throughout the transplant in the SS groups, whereas more cluster like survival pattern were observed in the FS group. Grafts placed in the intact left hemisphere tended to be smaller but present with similar patterns of TH-positive neuron survival as on the lesioned side (Fig. 2D–F).
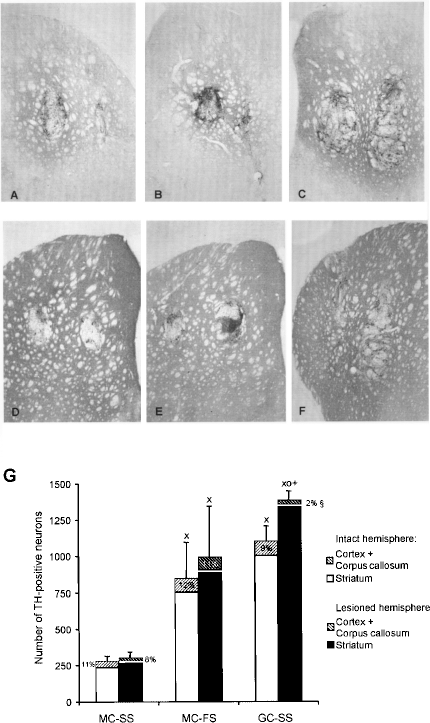
(A–F) Photomicrographs of TH-stained coronal sections at the level of the striatum showing representative examples of the three graft groups (A and D: MC-SS; B and E: MC-FS; C and F: GC-SS) both for the lesioned (upper row, A–C) and nonlesioned (lower row, D–F) hemisphere. In general, DA grafts in the 6-OHDA-lesioned striatum appeared larger and contained more numerous TH-positive neurons than their counterparts in the normal striatum. Additionally, GC-SS grafts provide for the most robust graft survival and extensive TH-positive reinnervation compared to the two other graft groups. (G) Corresponding quantitative analysis of the number of TH-positive neurons in the three graft groups in both the lesion (dark columns) and nonlesioned (white columns) striatum. The separated part in each column represents the percentage of TH-positive neurons that was found outside the striatum “ectopically” located along the injection tract in the overlying corpus callosum or cortex. Note the bigger variability of DA survival in the FS graft group compared to the two SS grafted groups. X: significant difference from MC-SS; O: significant difference from MC-FS; §: significant difference from MC-SS and MC-FS. +: significant difference from the contralateral normal side. MC, metal cannula; GC, glass capillary; SS, single cell suspension; FS, fragment suspension. Error bars represent SEM.
Figure 2G shows that the highest survival rate was achieved in the GC-SS grafted animals, both on the lesioned as well as on the intact side. In this group 1385 ± 63 TH-positive neurons survived on the lesioned side, 1.4 times as many as in the MC-FS (994 ± 351) and 4.6 times as many as in the MC-SS (302 ± 41) grafted groups. Whereas this difference between GC-SS and MC-SS was statistically significant in parametric post hoc tests (p < 0.01), the comparison to the MC-FS was compromised by a higher variance of the results due to 10 grafts with a lower DA survival rate (between 50 and 1064 TH-positive neurons/graft) and two grafts with an higher DA survival rate (2918 and 3524 TH-positive neurons/graft). Therefore, we undertook a post hoc comparison by the nonparametric Mann-Whitney U-test, which confirmed the significant differences in survival rates of the MC-FS group (416, 305, and 1039 TH-positive neurons/graft; i.e., median, lower, and upper quartiles) in comparison to both MC-SS (1412, 1304, and 1534 TH-positive neurons/graft; i.e., median, lower, and upper quartiles) and GC-SS (269, 239, and 384 TH-positive neurons/graft; i.e., median, lower, and upper quartiles) groups at the p < 0.01 level (U = 25).
In addition, the variability in DA graft survival observed after implantation of the FS was significantly higher compared to both groups grafted with the SS [p < 0.001, F(11, 11) = 68.77, and 26.71 in comparison with the MK-SS and GC-SS groups, respectively) (Fig. 2G). As 0.2 of one VM was grafted into each trajectory this represents a survival of 1,450 (MC-SS), 4,968 (MC-FS), and 6,646 (GC-SS) TH-positive neurons per VM, respectively. The number of TH-positive neurons that were found at extrastriatal sites along the implantation tract (i.e., cortex and corpus callosum) amounted to 11 ± 11% in the MC-FS group, 8 ± 9% in the MC-SS, and a significantly lower 2 ± 2% for the GC-SS grafted animals (p < 0.05).
There was an overall slightly increased survival of TH-positive neurons in the lesioned versus the intact hemisphere (+20%) that reached a significant level in the GC-SS group with 1,385 ± 219 versus 1,100 ± 107 TH-positive neurons in the lesioned and intact striatum, respectively (+26%, p < 0.05, t-test).
Apomorphine-induced rotations revealed a significant recovery in all three groups at 4 and 8 weeks postgrafting (Fig. 3A). However, the degree of improvement was only moderate in the range of 22% (GC-SS), 21% (MC-FS), and 13% (MC-SS) from pregrafting rotational asymmetry. In contrast, all groups demonstrated a robust and complete reversal of the amphetamine-induced rotational asymmetry already 2 weeks following transplantation (Fig. 3B), in agreement with the morphological data shown above. Eight weeks postgrafting there was a consistent overcompensation with 9.4 ± 2.0 (MC-SS), 6.0 ± 1.7 (GC-SS), and 6.7 ± 2.2 (MC-FS) contralateral tpm.

Apomorphine-induced (A) and amphetamine-induced (B) rotation scores revealed a significant reduction in all three grafted groups. Whereas the net amphetamine-induced rotational asymmetry is fully compensated by 2 weeks posttransplantation, there is only a moderate partial compensation for apomorphine-induced turning even after 8 weeks postgrafting. V: significant difference from the previous time point; O: significant difference from postlesion; X: significant difference from MC-SS (p < 0.05). TX, transplantation. Error bars represent SEM.
Experiment 2: Duration of Implantation
DA grafts were seen in all three implantation tracts in both TX19min and TX7min groups. In the striatum 818 ± 105 and in the nucleus accumbens 512 ± 113 TH-positive neurons survived in the TX19min group. In the TX7min group there were 800 ± 161 and 376 ± 134 TH-positive neurons in the striatum and in the nucleus accumbens, respectively, and this was nonsignificantly different from the TX19min group (Fig. 4A). Furthermore, the number of ectopically placed TH-positive neurons within the corpus callosum and the cortex was 48.4 ± 16.1 for the TX19min and 47.8 ± 12.1 for TX7min grafted groups (n.s.).

(A) Number of TH-positive neurons following the transplantation of E14 VM single cell suspensions into 6-OHDA-lesioned rats comparing a “slow hand” (TX19min) and “fast hand” (TX7min) injection speed, both for the number of cells per target side (striatum und nucleus accumbens) and for the ectopically found TH-positive neurons (i.e., in the overlying corpus callosum and cortex). (B) Amphetamine-induced rotation scores revealed a full compensation already 3 weeks postgrafting and overcompensation (contralateral turning behavior) at 6 weeks postgrafting in both groups. Interestingly, statistical analysis revealed no significant differences between the two groups in all parameters, indicating a comparable outcome between both techniques. *Significant difference from postlesion; +significant difference from 3 weeks posttransplantation. Error bars represent SEM.
Functional recovery in the amphetamine-induced rotation test was seen after 3 weeks postgrafting with 0.55 ± 1.09 ipsilateral tpm for TX19min animals and 1.43 ± 1.05 contralateral tpm for the TX7min group (p < 0.01 compared to pregrafting values, n.s. to each other) (Fig. 4B). At 6 weeks postgrafting this resulted in 5.3 ± 1.2 (TX19min) and 5.1 ± 1.5 (TX7min) contralateral tpm (n.s. to each other) (Fig. 4B).
Experiment 3: Cell Density of the Suspension
Similar to the grafts observed in the two previous experiments, the two implants along each injection tract had fused into a single elongated graft whose size and appearance were clearly correlated to the density of the cell suspension (i.e., to the number of implanted DA neurons) (Fig. 5). Grafts from the groups TX11–TX52 appeared most commonly as small strands of TH-positive neurons without a clear graft mass and very little displacement of the striatal neuropil and transcending corticospinal fiber bundles (Fig. 5A–C). In contrast, TX108 and TX320 grafts exhibited a “typical” DA graft phenotype with a visible graft volume composed of non-TH-positive compartments intermingled with numerous dispersed clusters of TH-positive neurons, preferentially located within the graft periphery (Fig. 5D–F). Concomitantly, the radius of the TH-positive fiber innervations of the host striatum increased from group TX11 continuously up to group TX320.

TH immunohistochemistry illustrating VM micrografts of 11,000 (A), 23,000 (B), 52,000 (C), 108,000 (D), and 320,000 (E, F) cells/μl. The smaller sized grafts (11,000–52,000 cells/μl, A–C) demonstrated a more even distribution of TH-positive cells throughout the grafts. In contrast, the larger sized grafts (108,000–320,000 cells/μl) have a more bulky appearance with cluster of TH-positive cells (+, D) intermingled with core areas devoid of TH-positive neurons (#, D). The different size of the graft volumes and the accompanying mass effect can easily be depicted through the displacement of the fiber bundles from the internal capsule that delineate the graft–host border (*, B and F).
The mean number of surviving TH-positive neurons increased progressively as shown in Figure 6 and Table 2. In parallel, the graft volume enlarged from 0.014 ± 0.02 mm3 (TX11) up to 0.308 ± 0.05 mm3 (TX320), as shown in Figure 6B. There was a highly significant correlation between the numbers of surviving TH-positive neurons and the graft volume across all groups, as illustrated in Figure 6C (p < 0.001; r2 = 0.965). Interestingly, there was a small but nonsignificant trend towards an increased TH-positive cell density in smaller grafts (i.e., 3,560 ± 430 TH-positive neurons/mm3 in the TX11 group versus 2,875 ± 123 TH-positive neurons/mm3 in TX320 grafted animals) (Fig. 6D). Similarly, smaller grafts seemed to promote TH-positive neuron survival with a survival rate between 3.3% and 3.9% (TX11–TX52) compared to around 2.7% for the TX108 and TX320 grafted groups, but this was a nonsignificant difference.

Number of TH-positive neurons (A), graft volume (B), relationship between these two parameters (C), density of TH-positive neurons per mm3 (D), and the relative percentage of survival of TH-positive neurons (E) in the five different cell concentration graft groups. Graft survival (A) and volume (B) were directly related to the cell concentration tested and there was a linear correlation between both former parameters across all groups (r2 = 0.965, p < 0.001, C). Note that both the cell density per mm3 (D) and the relative survival rate of TH-positive neurons were nonsignificantly different across the wide range of cell concentrations tested in this study. *Significant difference to all other graft groups; #significant difference from graft group 11,000 and 23,000 cells/μl (p < 0.05). Error bars represent SEM.
The unilateral 6-OHDA lesion resulted in an amphetamine-induced rotational asymmetry of 8–10 ipsilateral tpm, as shown in Figure 7A. The fastest recovery was observed in the TX320 group with a significant reduction already 2 weeks postgrafting down to 5.0 ± 0.5 ipsilateral tpm (p < 0.05), with a further improvement at 4 weeks posttransplantation to 5.0 ± 2.6 contralateral tpm (p < 0.001), which remained stable at 7 weeks. Animals from the TX108 group exhibited their first signs of functional recovery at 4 weeks postgrafting with a significant reduction to 2.9 ± 1.7 ipsilateral tpm (p < 0.001) and a complete amelioration of rotational asymmetry at 7 weeks (0.4 ± 1.6 contralateral tpm). In contrast, there was only a nonsignificant small trend of recovery in the TX52 and TX23 grafted animals and no signs of functional grafts effects in the TX11 group.

Time- and cell concentration-dependent reduction of amphetamine-induced rotational scores (A) as observed until 7 weeks postgrafting. Whereas the TX320 graft group shows a partial compensation already after 2 weeks and an overcompensation of rotational scores after 4 weeks postgrafting, there was a slower recovery with finally a full compensation in the TX108 graft group and only a delayed and partial recovery in the TX52 graft group. No signs of behavioral recovery were seen in the smallest sized graft groups (e.g., TX23 and TX11). Error bars represent SEM. (B) Correlation between the degree of reduction in the amphetamine-induced rotation test at 7 weeks postgrafting and the number of surviving TH-positive neurons per graft across all groups tested. It seems that less than 100 TH-positive cells could not provide any functional benefit, between 100 and 500 TH-positive neurons resulted in a variable degree of partial to full recovery and more than 500 surviving TH-positive neurons reached a “functional plateau” at the level of full recovery to a up to 40% of overcompensation of the rotational score.
Figure 7B demonstrates the relationship between the survival of TH-positive neurons and the percentage of functional improvement in the amphetamine-induced rotation test at the final 7 weeks postgrafting time point. There was an overall good correlation between numbers of surviving TH-positive neurons per striatum and functional improvement from 100 to around 500 TH-positive neurons (p < 0.001). Fewer than 100 TH-positive neurons did not seem to induce any effect on amphetamine-induced rotations, between 200 and 500 TH-positive neurons per grafted striatum could result in a variable rate of recovery ranging from modest to almost complete recovery. More than 500 TH-positive neurons resulted in a complete restoration or even overcompensation of rotational asymmetry. Interestingly, the degree of overcompensation represented maximally 50% of the ipsilateral rotational scores postlesion and did not increase further with higher TH-positive cell numbers up to 2,200 TH-positive neurons/graft.
Discussion
During the course of clinical and experimental studies on neural transplantation for PD and other neurodegenerative diseases over the last two decades, the “optimal grafting approach” has remained as one of the key questions that needed to be addressed in order to further advance the field of neural repair from an experimental strategy towards a clinical cell therapy (9, 13, 17, 31). In this context the results of the present study extend previous observations that the microtransplantation approach offers an efficient, reliable, and precise technique for quantitative studies on the influence of grafting parameters onto the in vivo survival, differentiation, and functional capacity of fetal DA neuron-rich grafts in a rat model of PD. The results demonstrate the importance of the preparation of the cell suspension, the choice of the implantation cannula, the degree of DA denervation, the duration of the implantation procedure, and the density of the final cell suspension for DA grafts in unilaterally 6-OHDA-lesioned adult rats.
Influence of the Preparation of the Cell Suspension, Choice of the Implantation Cannula, and Degree of DA Denervation
This is the first in vivo study that directly compares the “classical” preparation of a fragment cell suspension (so-called “macrografts”) with the more traumatic preparation of a single cell suspension (so-called “micrografts”). It could clearly be observed that the more vigorous preparation of a single cell suspension resulted in an approximately fourfold higher loss of DA neuron survival when a regular metal cannula (OD 470 μm) is used for the implantation. This is consistent with our own studies (26, 27) and in vitro studies by Barker and colleagues (2). In the latter, it was demonstrated that the mechanical trauma impinged on the VM tissue during repeated and more vigorous tituration (i.e., to achieve a more homogenous single cell suspension) could result in greater cell loss and impaired DA neuron survival in cell culture. Importantly, we and others (3) observed that cell numbers and concentrations of the final cell suspension can only reliably be estimated by counting methods using completely dispersed single cell suspensions. If fragment suspension is prepared containing clumps of cells together with single cells, counting may only represent approximately 50% of the actual total cell number in a given fragment suspension, as observed in the present study. This is because cell clumps cannot reliably be counted. In the latter case, it seems more appropriate to document the amount of VM pieces and the total volume of the cell suspension and the final graft volume implanted in order to deduct the number of cells per graft indirectly by computation methods (Table 3).
Transplantation Parameters Relevant for the Report on DA Neuron Graft Survival

Modified from Nikkhah and colleagues (26).
However, the disadvantage of the more “harmful” single cell preparation technique has also some considerable advantages. First, it produces a more homogenous cell suspension that results in more consistent graft survival and DA neuron cell numbers, making comparative analysis more meaningful not only across the experimental groups within one study but also across several consecutive studies (27). Secondly, it allows the use of a syringe driver-based microtransplantation instrument, as previously described in detail (26). This study shows that the reduced implantation trauma based on the use of fine glass capillaries (OD 50–70 μm) instead of metal cannulas (OD 470 μm) not only compensates for the detrimental effects of the more vigorous tituration process but also enhances the net survival of DA neurons by about 40% in addition to a more reproducible graft survival. This is consistent with previous reports showing that the reduction of the implantation cannula size is beneficial for the number of surviving DA neurons (8, 32) as well as for a reduced glial (12, 26) and immunological response (18) of the host. Recently, more homogenous single cell suspensions have been introduced into clinical transplantation programs for PD and resulted in a favorable outcome both in terms of morphological graft features and clinical outcome (19, 20).
Interestingly, the present study has demonstrated a significant (+26%) increased DA graft survival in the DA-denervated striatum compared to the normal side, which corroborates previous investigations by Kirik and colleagues (16) and Roedter and colleagues (29). Both studies were performed by using the current microtransplantation technique. In contrast, a similar study by Doucet and colleagues (10) using the “conventional” macrografting technique has failed to see this survival-enhancing effect of the DA lesion on grafted DA neurons, although they observed an increase of cell body size and TH-positive fiber outgrowth from the implanted DA neurons on the DA-denervated side. In retrospect, this seems largely due to the highly variable graft survival in this study, which may have masked the biological effect of the DA denervation and underlines the usefulness of the microtransplantation approach for quantitative transplantation studies. Thus, DA lesions may induce growth promoting changes of the striatal micromilieu. In fact, Nishino and colleagues (28) have demonstrated a better differentiation of mesencephalic neural progenitor cells into DA neurons in the DA-denervated compared to the normal striatum. The underlying cellular and molecular changes responsible for these effects are highly complex and have been addressed previously (9, 16).
Duration of the Implantation Procedure
The time necessary for the completion of the implantation procedure is of considerable interest under both experimental and clinical conditions. Traditionally, a “slow hand” technique is preferred, consisting of a slow injection rate (1 μl/min) and a waiting period (typically 3–5 min) after each injection, accumulating to a considerable transplantation time spent for each patient or animal, especially when multiple trajectories and deposits along each trajectory are combined. Our results show that this “slow hand” procedure is not absolutely necessary when a more refined implantation cannula is used. We did not observe any differences in DA graft survival and functional effects or misplacement of DA neurons along the injection tract, whether at more superficial implantation sites like the striatum or deeper implantation sites like the nucleus accumbens, when the “fast hand” technique was applied (immediate injections and no waiting time after the injections). There are only a limited number of previous studies investigating perigrafting technical aspects. When Sinclair and colleagues (30) used the macrotransplantation technique they observed improved DA graft survival and behavioral recovery only after delaying the injection of the graft suspension for more than 1 h after lowering of the cannula into the striatum. They hypothesized that this effect is due to toxic changes in the striatal micromilieu including glutamate, aspartate, DA, and potassium releases that resolve within 1 h after the injection injury. Importantly, these negative effects are maybe aggravated with larger implantation cannula and substantially are silenced when a glass capillary is used, as in the present study. However, we cannot exclude completely that a similar delayed injection (1–3 h), as performed by Sinclair and colleagues (30), could further improve the survival of DA micrografts. Similarly, hypothermia (32–33°C) of hemiparkinsonian rats during implantation improves DA neuron survival in macrografts, as shown by Karlsson and colleagues (15). Whether this points in a similar direction towards trauma-induced mechanisms that trigger DA neuron apoptotic pathways directly following the implantation [see also (9)] and which can be alleviated, at least partly, by the microtransplantation approach raises an interesting scientific hypothesis for further investigations.
Importance of the Density of the Final Cell Suspension
For single graft deposits the number of surviving DA grafts neurons can effectively be calibrated by a dilution series of the cell suspension and using the current microtransplantation technique. There was a progressive decrease both in DA neuron numbers and DA graft volume when decreasing the cell number per μl of the final cell suspension stepwise from 320,000 down to 11,000 cells/μl. Furthermore, we did not observe any significant difference between the density of DA neurons per mm3 nor in their respective survival rate, which was in the range of 2–4% and thus within previously reported values (9, 13, 31). In a previous study, examining fixed cell concentrations and different implantation volumes, it was shown that 0.5-μl grafts containing 50,000–75,000 cells seemed to provide the optimal choice (26) for DA graft survival and morphological integration. Therefore, the total volume per graft deposit has a significant influence on DA grafts whereas the different cell concentrations provide different absolute numbers but relatively similar survival percentages of DA neuron survival, at least within the wide range of cell density tested in this study. Similar findings were found in another recent study by Johansson and colleagues (14) that reported a constant survival rate of 4–5% DA neurons in two experimental groups receiving either 100,000 or 250,000 VM-derived cells per graft in the same graft volume. However, DA neuron survival in grafts decrease significantly (3–4%) when the cell suspension is below 50,000 cells/μl and decrease even more in 25,000 cells/μl concentration (2–3%) according to Terpstra and colleagues (33). However, one caution was raised by a study by Bartlett and colleagues (4) that demonstrated an impaired graft survival when the cell concentration was raised as high as 400,000 cells/μl compared to 50,000 cells/μl.
The results from this study on micrograft-derived DA neuron survival from the different cell concentration groups provide further evidence for a nonlinear relationship with the extent of functional recovery in drug-induced rotation tests. A variable degree of behavioral recovery was observed with 100–500 surviving DA neurons per grafted animal, whereas all animals with more than 500 surviving DA neurons exhibited a full recovery in the amphetamine-induced rotation test. These results provide a useful orientation for further studies on neuroprotective and neurotrophic substances for DA grafts as to which range of graft size graft-induced functional effects can be further promoted. Interestingly, similar effects of graft size were also detected in the time-dependent appearance of behavioral recovery. The largest grafts (320,000 cells/μl) showed a partial recovery already in 2 weeks and a full compensation 4 weeks postgrafting, whereas animals from the TX108 and TX52 groups showed a more protracted recovery period, with no signs of functional recovery in the two smaller sized graft groups (i.e., TX23 and TX11 groups).
Conclusion and Perspective
Following the original description of the microtransplantation approach for DA cell suspension grafts in a rat model of PD (26), several of its aspects have entered into subsequent experimental and clinical transplantation studies in PD and other neurological diseases (17, 19, 27, 35). The current study has now revealed further novel insights into how the survival, differentiation, and functional behavioral recovery of DA grafts are influenced by transplantation parameters such as the preparation of the cell suspension, the choice of the implantation cannula, the degree of DA denervation, the duration of the implantation procedure, and, finally, the density of the cell suspension. Therefore, the microtransplantation approach offers a unique opportunity to promote our current attempts to tailor cell therapy strategies to the individual restorative challenges present in experimental and potentially also clinical conditions of PD.
Footnotes
Acknowledgments
We warmly thank Jürgen Wittek and Johanna Wessolleck for their excellent technical work. The authors also would like to thank Manuela Schätzle for excellent secretarial assistance. This study was supported by grants from the Deutsche Forschungsgemeinschaft (Ni 330), the Graduate School Freiburg (DFG 843) and the German Parkinson Foundation.
