Abstract
Immunoisolation of pancreatic islets is extensively investigated for glycemic control in diabetic experimental animals. We previously reported that subcutaneous xenotransplantation of bovine islets protected by a selective polysulfone membrane successfully controlled glycemia in diabetic rats for up to 20 days. We then wondered whether immunoisolated islets have adequate oxygen supply in this device, where only diffusive transport allows cell function and survival. Here we set up an experimental technique to measure oxygen consumption rate (OCR) using a Clark's electrode inserted in a glass thermostated chamber connected to a data recorder and acquisition system. Bovine islets were isolated from 6-month-old calves, encapsulated in sodium alginate microcapsules or inserted in polysulfone hollow fibers. After 1 and 2 days in culture a series of measurements was performed using free islets (at normal or high-glucose concentration), islets encapsulated in microcapsules, or in hollow fibers. In free islets OCR averaged from 2.0 ± 0.8 pmol/IEQ/min at low-glucose concentration and from 2.5 ± 1.0 pmol/IEQ/min at high-glucose concentration (p < 0.01). OCR in islets encapsulated in microcapsules and in hollow fibers was comparable, and not significantly different from that measured in free islets. Two days after isolation OCR averaged 2.3 ± 0.6 in free islets, 2.3 ± 0.9 in alginate microcapsules, and 2.2 ± 0.7 pmol/IEQ/min in hollow fibers. These results show that OCR by bovine islets is comparable to that previously reported for other species. OCR increases in islets stimulated with high glucose and may be considered as a functional index. Moreover, islet encapsulation in alginate microcapsule, as well as in hollow fiber membranes, did not significantly affect in vitro OCR, suggesting adequate islet oxygenation in these conditions.
Introduction
Outcomes of recent clinical trials on pancreatic islet transplantation in patients with type 1 diabetes open new perspectives in the treatment of this disease (12, 20, 30, 32). However, the shortage of pancreas donors and the need of chronic immunosuppressive therapy remain the major hurdles to the widespread clinical use of pancreatic islet transplantation (13). To overcome these limitations several studies are in progress on isolation of islets from pig and from bovine pancreas, to attempt xenotransplantation. In parallel, immunoisolation devices are investigated to protect islets from rejection without the use of immunosuppression. Regarding the first issue, despite the fact that porcine islets have been considered the best source for xenogenic transplantation, they have been shown to be fragile and their isolation is rather inconsistent with loss of viability and islet fragmentation (28). Recently, it has been shown that bovine islets can be isolated efficiently from pancreas tissue and they demonstrate adequate in vitro and in vivo function (11).
After isolation process and before transplantation, a reliable measurement of islet viability and function is needed, in order to prevent transplantation of inefficient islet preparations in experimental as well as in clinical studies. Because direct evaluation of β-cell viability and function in terms of insulin production is complex and time consuming, we reasoned that evaluation of oxygen consumption could be used to rapidly estimate islet viability after the isolation process. Because pancreatic islets in physiological condition have a dense, glomerular-like vascular architecture, the lack of blood supply may result in functional abnormalities and eventually in cell death (8, 16). In this condition, islets gather nutrients only by passive diffusion. Oxygen consumption by free islets produces concentration gradients that drive oxygen diffusion through the entire islet volume. Therefore, whenever the diffusion is excessively low, the oxygen flux to the inner cells of the islets would be inferior than required, resulting in severe hypoxia, islet functional loss, and ultimately in cell death. If these phenomena take place, oxygen consumption by islet cell would rapidly decrease.
Islet encapsulation in semipermeable matrixes, such as alginate gel or membrane devices (7, 18), have been studied in experimental (19) as well as in clinical setting (37). Both systems must allow adequate diffusion of oxygen and nutrients to maintain islet viability and function but, at the same time, must be selective enough to prevent the permeation of host immune proteins (6, 18). In immunoprotected islets the limitation in oxygen diffusion may be even more important and the possibility to obtain adequate oxygenation, up to the center of the islet, depends from the geometry of the system and oxygen permeability of the encapsulation media. We have previously reported that subcutaneous xenotransplantation of bovine islets protected by polysulfone hollow fibers successfully controlled glycemia in diabetic rats for up to 20 days (9). In the attempt to prolong islet function, we wondered whether immunoisolated islets in this condition still have adequate oxygen supply. We then estimated oxygen consumption rate (OCR) of free and encapsulated bovine islets as indicator of islet viability and function. We here report the technique used to measure OCR in pancreatic islets and the effect of islets encapsulation in sodium alginate capsules and in polysulfone hollow fibers on OCR by bovine islets.
Materials and Methods
Islet Preparation
Islets were isolated from bovine pancreas (11) as previously described (29). Briefly, the main pancreatic duct was cannulated and perfused with collagenase P solution (Boerhinger-Mannheim, Mannheim, Germany) and the pancreas was placed into a perfusion chamber at 37°C. When optimum digestion time was reached, the chamber was flushed with cold (4°C) Hank's balanced salt solution (HBSS, Gibco InVitrogen Corporation, Paisley, Scotland). The digested tissue was collected and purified by centrifugation on Histopaque gradient (1.077 g/ml, Sigma). Islets, >95% purity, were cultured in M199 medium (total amino acids concentration 1.06 g/L, glucose concentration = 5.5 mM), supplemented with bovine serum and glutamine (0.3 g/L) in a cell incubator at 37°C in humidified atmosphere with 5% CO2. Islet yield was determined by counting the total number of islets using an inverted microscope equipped with a calibrated grid. Islet number was expressed as islet equivalents (IEQ, the number of 150-μm-diameter islets having the same total volume of the preparation), as previously described (29), to take into account islet volume variability among different isolations.
Islet Encapsulation in Microcapsules and in Polysulfone Hollow Fibers
After 24 h in culture, for the production of capsules, islets were suspended in a solution of 1.7% sodium alginate (Manugel DMB, Monsanto pic, Surray, UK) at a concentration of 3 IEQ/μl. The islet–alginate mixture was then extruded through an air jet droplet generator into a solution of 100 mM CaCl2 solution. Gel beads resulted to have diameter ranging from 800 to 950 μm (Fig. 1A). For islet encapsulation in polysulfone hollow fibers alginate solution was inserted in the polysulfone hollow fibers (490 μm inner diameter and 70 μm membrane thickness, Fresenius Medical Care, Bad Hamburg, Germany), using a perfusion system to allow uniform loading of islet solution among the fibers at a concentration of 50 IEQ/μl (Fig. 1B). After perfusion, fibers were sealed and immersed into 100 mM CaCl2 solution for 10 min to allow alginate gelification, and then immersed in culture medium.

Light microscopic images of bovine islets in alginate microcapsules (A) or inside polysulphone hollow fiber (B). Diagram of the key components of the chamber used for OCR measurements (C).
Oxygen Consumption Measurements
For the measurement of OCR we designed a thermostated chamber (Fig. 1C) made with two compartments: the inner part, in which the measurements are carried out, and an external water jacket that enables temperature changes in operative conditions. The inner chamber was equipped with a Clark's electrode (3, 31). Briefly, measurements were based on the classical Polarographic method, in which a potential difference is created between a platinum cathode and a silver anode to permit oxygen reduction. In the presence of oxygen an electric current is generated, which is converted into a tension signal digitally recorded.
The system was preliminarily calibrated using distilled water at 37°C, with continuous stirring by a small magnetic bar, in which atmospheric oxygen concentration was assumed to be 215 μM (24). Null oxygen concentration was obtained saturating the solution in the chamber using nitrogen gas. After 10 min of nitrogen gas perfusion recording was constant and assumed to correspond to zero oxygen concentration. Oxygen consumption rate (OCR) per IEQ was calculated from the slope of oxygen concentration recording versus time by linear regression analysis (as representatively shown in Fig. 2A) using the formula
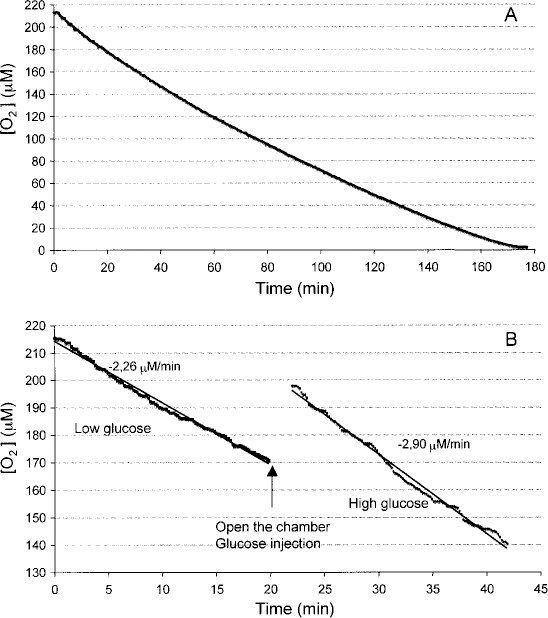
Recording of oxygen concentration in an islet preparation until the limit of zero oxygen was reached (A). Representative changes in oxygen concentration versus time in an islet suspension at low- and high-glucose concentration (B).
where IEQ is the number of islet equivalents in the chamber and V the solution volume in the chamber. OCR of free islets was assessed immediately after isolation, and 1 and 2 days later. Islets of four different preparations were used for these measurements. The number of islets in each sample was calculated on the basis of total islet number per isolation and the ratio between sample volume and the volume of islet suspension. We estimated that each sample contained on average 997 IEQ (650–1200 IEQ). Islets were centrifuged for 2 min at 800 rpm, the supernatant was removed, and the pellet was suspended in 200 μl of prewarmed (37°C) M199 medium serum free (glucose concentration = 5.5 mM). The islet suspension was added to the chamber and, after sealing, thermal equilibration at 37°C was allowed for about 30 s. Each measurement lasted about 30 min. For OCR determination at high glucose, 25 mM glucose concentration was used, adding concentrated glucose solution in chamber at the end of basal evaluation. OCR of encapsulated islets was assessed immediately within 2 h after encapsulation in microcapsules and in hollow fibers (the day after the isolation process) and 1 day after encapsulation. Also for this series of measurements, islets of four different preparations were used for both encapsulation techniques. Each sample placed in the chamber for OCR measurement, using either microcapsule and hollow fibers, contained on average 1060 IEQ (670–1500 IEQ). During all measurements the suspension was stirred using a magnetic plate at about 50 rpm. In some experiments, after OCR measurements, free and encapsulated islets were counted under the microscope and resulted to be on average 96% of the value estimated before OCR measurement.
Statistical Analysis
All results are expressed as mean ± SD. Statistical significance was determined using analysis of variance (StatView software, SAS Institute Inc., Cary, NC, USA) and specific comparisons between groups were calculated using t-test with Bonferroni correction (38). The level of statistical significance was assumed when p < 0.05.
Results
We initially estimated the OCR of free bovine islets in suspension during a 3-day period after isolation. A recording of a representative experiment is shown in Figure 2A, in which O2 concentration was estimated for up to complete consumption. As shown in the figure, O2 change with time was rather constant even for low levels of O2 that were reached in more than 160 min. The small decrease in the rate of change in O2 concentration at the end of the observation period was likely due to mechanical damage of islets as a consequence of prolonged stirring. As shown in Figure 3, estimated mean OCR averaged 1.39 ± 0.38 pmol/IEQ/min immediately after isolation and numerically increased to 2.28 ± 0.64 pmol/IEQ/min at the end of the observation time (n = 4). The expected oxygen consumption by the electrode in this condition is less that 3% and assumed to be negligible (3). To investigate the effect of changes in glucose concentration on OCR by free islets, the day after isolation aliquots of islet preparation were suspended in M199 at low glucose (5.5 mM) and OCR measured in this condition; then glucose level was increased to 25 mM. The rise in glucose concentration significantly increased OCR, as shown in the representative recording reported in Figure 2B. Calculated OCR reported in Figure 4 and in Table 1 averaged 1.98 ± 0.79 and 2.49 ± 0.97 pmol/IEQ/min at low- and high-glucose level, respectively (p < 0.01, n = 12).

Mean OCR of free islet suspensions measured the day of islet isolation (0), the day after the isolation process, and 2 days after. Data are mean ± SD (n = 4).

Mean OCR of free islet suspensions measured the day after the isolation process at low- and high-glucose concentration (n = 12). Data are mean ± SD. *p < 0.01, low glucose versus high glucose.
O2 Consumption Rate by Free Islets in Suspension at Low and High Glucose, and Related Absolute and Percentage Increase

OCR, oxygen consumption rate; ΔOCR, changes in OCR upon glucose stimulation.
p < 0.01 versus OCR at 5.5 mM glucose.
The results on the effect of islet encapsulation on OCR are reported in Fig. 5. In islets encapsulated in alginate microcapsules, at a density of 3 IEQ/μl, the day of encapsulation OCR averaged 2.12 ± 0.87 pmol/IEQ/min and the following day it averaged 2.26 ± 0.95 pmol/IEQ/min. The difference was not statistically significant. Both values were also comparable to mean OCR estimated for free islets. OCR by islets embedded in polysulfone hollow fibers averaged 1.75 ± 0.57 pmol/IEQ/min the day of encapsulation and 2.21 ± 0.69 pmol/IEQ/min the following day (n = 4), but the difference did not reach statistical significance (Fig. 5). Mean OCR by islets embedded in the hollow fibers was comparable to mean OCR measured in freely suspended islets. The linearity of O2 changes with time during OCR measurements in islets encapsulated in microcapsules and in hollow fibers was comparable to that observed in free islets suspensions.

Mean OCR of free suspension, of islet embedded in microcapsules, and in hollow fibers measured 1 or 2 days after the isolation procedure. Data are mean ± SD.
Discussion
We set up and used for the present study a simple laboratory method for rapid measurement of OCR in free or encapsulated islets, as a quantitative method for islet preparation quality assessment. The system that we assembled allowed us to measure OCR by bovine pancreatic islets in different experimental conditions. We estimated an average value of 1.98 ± 0.79 pmol/IEQ/min for OCR by free islets in basal condition. This is the first time that OCR of bovine islets is determined and the values we obtained are comparable to other estimates previously reported in literature for other species (15, 36). Thus, the rate of oxygen consumption was found to range from 1.5 to 4.2 pmol/IEQ/min in mouse pancreatic islet (5, 14, 17, 25), from 2 to 3 pmol/IEQ/min in rat islets, and from 2.5 to 3.3 pmol/IEQ/min in human islets (15, 36), and these values are reported to increase during metabolic activity induced by elevated glucose concentrations (34).
We also studied the OCR in bovine islets exposed to a low- and high-glucose concentration to assess islet function beside viability. Metabolism of glucose triggers insulin secretion, with an acceleration of β-cell activity (21, 22, 26, 27), including insulin production. Despite the fact that results are not always consistent, it is generally accepted that oxygen consumption increases upon glucose stimulation in pancreatic islets, providing evidence for an accelerated rate of β-cell metabolism, likely the basis for increase in insulin secretion (23, 35, 39). We have previously reported that bovine islets stimulated with glucose significantly increase insulin production (11). In the present study we did measure that mean OCR in bovine pancreatic islets at low-glucose concentration was significantly increased when islets were exposed to high-glucose concentration, confirming that OCR increase is likely related to increase in insulin production. We actually estimated a mean increase in OCR of 0.51 ± 0.30 pmol/IEQ/min between low and high glucose, as reported in Table 1, corresponding to an average increase of 27% over low-glucose OCR. This result is in line with that reported by Sweet et al. (33, 34) for mice and human islets, confirming for bovine islet the same index of functionality. Thus, the measured increase in OCR in response to changes in glucose concentration may provide a useful and rapid in vitro assay to assess functionality and viability of islet preparations during experimental and clinical transplantation procedures.
We also measured OCR by encapsulated islets to evaluate whether the immunoisolation barriers represents an obstacle to the diffusion of oxygen, limiting normal islet function and viability. We tested two types of immunoisolation devices, alginate microcapsules (850–900 μm O.D.) and polysulfone hollow fibers (490 μm I.D., 70 μm wall thickness). Microcapsules are frequently used in studies on islet transplantation in diabetic rats and have been used also in preliminary studies in humans (4, 9). Hollow fibers are also used as immunoisolation device in attempt to obtain a more stable barrier with time. We recently demonstrated that this is a valid system for immunoisolation, even if we observed central necrosis in islets explanted from the rat, suggesting potential hypoxic condition (9).
There is theoretical and experimental evidence (1, 2, 5) that oxygen is a limiting factor for islet viability and function under immunoisolation conditions. Even if alginate gel is highly permeable to oxygen, the distance between islets in the capsules and oxygen source outside the capsule, the capillary microcirculation, may arrive to more than 400 μm. Similarly, for islets in the hollow fibers, in the worst case, there is a distance of 315 μm between the central part of the islet and the outside part of the device, and the diffusion of oxygen is theoretically limited by this distance. In our experimental conditions both immunoisolation barriers allowed recording OCR values comparable to that measured in free islets. This indicates that, at least for high external oxygen concentrations, oxygen diffusion is not significantly limited by the immunoisolation media. This is consistent with a frequently reported long-term function of islets in alginate capsules implanted in the peritoneal cavity of diabetic rats (10). OCR by islets encapsulated in polysulfone hollow fibers also indicated that there are no major obstacles to oxygen diffusion; thus, oxygen consumption was similar to that of free islets. Despite these results, as mentioned previously, we observed that islets implanted in rats subcutaneously in hollow fiber device had rather short-term duration, less than 20 days. Our present results would indicate that if O2 is present outside the immunoisolation barrier, there should be are no limitations in its transport to the living tissue. However, we can speculate that in subcutaneous space, where venous partial O2 pressure is expected to occur, not adequate O2 availability may develop independently from the immunoisolation device.
In conclusion, our present study demonstrates that in bovine pancreatic islets OCR is comparable to other species and it significantly increases upon glucose stimulation. In in vitro conditions islet encapsulation in alginate gel and in polysulfone hollow fibers allows adequate transport of nutrients to maintain islet function and viability, making the use of these immunoisolation strategies for transplantation worth to investigate.
Footnotes
Acknowledgments
Polysulfone hollow fibers were kindly provided by Fresenius Medical Care (Bad Hamburg, Germany). This study was supported in part by the EU project BARP+ (No. NMP3-CT-2003-505614).

