Abstract
Several reports suggest that islets isolated from younger donor pancreata are of better quality for clinical islet transplantation. The relative inefficiency of the continuous gradient purification process (CGP) is one of the major obstacles to the utilization of these younger donor pancreata. This study demonstrates the benefits of utilizing an additional purification step, rescue gradient purification (RGP), to recover trapped islets and examines the possible superiority of these rescued islets. Seventy-three human islet isolations purified by RGP following CGP were divided into two groups based on age, and the isolation results were retrospectively analyzed (group I: age ≤40, group II: age >40). The quality of islets from both CGP and RGP were assessed by β-cell fractional viability (βFV) and ADP/ATP ratio. Significant increases in the percent islet recovery from RGP and the percent trapped islets in group I compared to group II were observed. Donor age correlated negatively to the percent islets recovered from RGP (R = 0.440) and to the percent of trapped islets (R = 0.511). RGP islets had higher βFV and better ADP/ATP ratio compared to CGP islets. In conclusion, RGP improved the efficiency in the purification of trapped islets, which often come from younger donor pancreata. The better quality of β-cells in RGP islets encourages us to perform RGP, considering the higher quality as well as the quantity of remaining islets.
Introduction
Encouraging success in islet transplantation has been achieved in recent years as a result of progressive improvements in islet isolation methods and immunosuppressive strategies (33,39,40). Islet transplantation may be an ideal therapy for patients with type 1 diabetes because of the less invasive nature of the procedure, fewer complications, and improved quality of life (5,25). However, the procedure is still hampered by an inadequate supply of islet cells and the gradual loss of islet function after transplantation (35,41). Current clinical trials have demonstrated that more than 10,000 IEQ/kg recipient weight are frequently required to achieve insulin independence using current isolation procedures (33,39,40). Eventually, two to three donor pancreata are required to achieve euglycemia (8,33,39,40). Because the availability of donor pancreata is limited, there is an urgent need for improving not only the quantity of islets isolated from a single donor pancreas but also quality of islets.
Pancreata from younger donors have been reported to have superior potentials in β-cell replacement therapy (7). Maedler et al. reported that aging correlated with decreased proliferative activity and enhanced sensitivity to glucose-induced apoptosis (21). It has been reported that pancreas grafts from older donors (age >45 years) was one of the risk factors related to poor glycemic control and premature loss of pancreas function in pancreas transplantation (18). In addition, it has been currently reported that islets from younger donor pancreata demonstrated better functionality (3,42). However, younger donor pancreata have not been ideal for clinical islet transplantation even though there was much evidence to demonstrate the superiority of these islets.
The major obstacle for the utilization of younger donor pancreata is poor islet yields (19,23,43). This may be due to the difficulty in separating islets in younger pancreata from the surrounded acinar tissue without fragmenting the islets (1,30). However, embedded islets were reported to be as functional as nonembedded islets (29). In fact, surrounding acinar tissue may provide necessary protection from mechanical and chemical stress during the islet isolation procedure.
In current human isolation procedure, islets are separated from acinar tissue using a continuous density gradient centrifugation after mechanically enhanced enzymatic digestion. Density gradient centrifugation using the semiautomated COBE-2991 cell processor is currently the most effective method for the purification of human islets (32,33,39,40), and is utilized in the majority of islet processing centers. This method allows us to separate islets from pancreatic digests based on the difference in density between islet cells and other pancreatic tissue. Trapped islets primarily migrate into the higher density layers, causing them to be lost during continuous gradient purification (CGP) in the lower purity layers.
Recently, we have reported that rescue gradient purification method (RGP) could be of assistance in maximizing the number of islet preparations successfully used for transplantation protocols (13). RGP was performed following CGP when CGP resulted in relative low islet yields or when a considerable number of islets were observed in the impure layers, which are generally not considered for transplantation.
In this study, isolation data was retrospectively analyzed to determine which donor or isolation-related factors affect the efficiency of RGP. Our results show that RGP is more capable of purifying trapped islets than CGP, suggesting the benefits of RGP to maximize the islet yield from younger donor pancreata.
Materials and Methods
Human Islet Isolation
Between August 2000 and September 2005, human isolations were performed at the Human Islet Cell Processing Facility of Cell Transplant Center, Diabetes Research Institute, University of Miami. The data of isolations with cold ischemia time more than 12 h were excluded from this analysis. Seventy-three islet isolations performed using the automated method (13,32) with CGP, followed by RGP, were divided into two groups based on the donor age (group I: age ≤40, group II: age >40) and retrospectively analyzed for an association with donor age with multivariate analysis.
Islet Purification Procedures
Pancreas digestion was performed using Liberase HI (1.4 mg/ml) (Roche, Indianapolis, IN). Islets were purified using a semiautomated cell processor (Cobe 2991; COBE Laboratories, Inc. Lakewood, CO) (11,27) in a refrigerated (4°C) cell processing room. The standard procedure for the purification of human islet preparation consists of CGP performed by loading Ficoll-based density gradients, 1.100 g/ml (osmolality 320–373 mOsm/kg/H2O) and 1.077 g/ml (285–327 mOsm/kg/H2O), in a doughnut-shaped bag using a gradient making device. This was followed by top-loading the pancreatic digest (≤20 ml of tissue/run) in UW solution and centrifuging for 5 min at 2400 rpm (13). Approximately 15 fractions were collected and examined for purity and quantity of islets.
RGP was performed when prepurification yield was sufficient but the CGP resulted in a relative low islet yield. Typically, in this case, a considerable number of islets were observed in the impure layers, generally not considered for transplantation. This additional purification step allowed the recovery and further purification of the islets cells. Briefly, the low purity fractions and pellet were pooled and washed in modified RPMI media by centrifugation (420 × g, 1 min, at 4°C). The tissue (≤30 ml of tissue/run) was then resuspended in stock Ficoll (density 1.132 g/ml; osmolality 420–440 mOsm/kg/H2O) and loaded in the bottom of the COBE bag (13). The discontinuous gradient was obtained by applying the subsequent Euroficoll solutions (Mediatech, Herindon, VA) with density 1.108, 1.096, and 1.037 g/ml (75 ml each). After a 3-min centrifugation at 2400 rpm, four fractions were collected: a 100-ml waste layer, a 1.037/1.096 interface layer usually yielding the purest islets, a 1.096/1.108 interface with less pure islets, and a third 1.108/1.132 interface with the least pure islets (13).
Islet Cell Dissociation
Single cell suspensions were obtained by incubating aliquots of approximately 1,500–2,000 islet equivalents (IEQ) in 1 ml Accutase (Innovative Cell Technologies, Inc, San Diego, CA) at 37°C for 10–15 min, as described previously (12).
Assessment of β-Cell Viability
Single cell suspensions were incubated for 30 min at 37°C in phosphate-buffered saline (PBS) without Ca2+ and Mg2+ with Newport Green PDX acetoxymethylether (NG; 1 μM) and tetramethylrhodamineethylester (TMRE; 100 ng/ml; both from Molecular Probes). After washing, cells were stained with 7-aminoactinomycin D (7-AAD; 10 μg/ml; Molecular Probes). Cell suspensions were analyzed (minimum 3.0 × 104 events) using a FACScan cytometer with the CellQuest-pro software (Becton Dickinson, Mountain View, CA). Apoptosis was evaluated selectively in the NGbright cells. After calculating, dead cells (7-AAD+) were excluded from further analysis, live β-cells (NGbright) were analyzed for mitochondrial membrane potential (TMRE), which allows for discriminating between healthy (TMRE+) and apoptotic (TMRE) cells. The percentage of TMRE+ on each cell subset was decided based on the gate of TMRE unstained samples.
Assessment of Cellular Composition Using LSC/iCys
Dispersed cells from islet preparations were fixed on glass slides with 2.5% paraformaldehyde (Electron Microscopy Sciences, Washington, PA). After incubating with Protein Block (Bio-Genex, San Ramon, CA) and washing (Optimax Wash Buffer; Bio-Genex, San Ramon, CA), cells were incubated for 2 h with the following antibodies: mouse monoclonal anti-C peptide antibody (1:100; Abcam Inc., Cambridge, MA), mouse monoclonal anti-glucagon antibody (1:500; Sigma, St. Louis, MO), rabbit monoclonal anti-pancreatic polypeptide antibody (1:1000; Sigma), and rabbit monoclonal anti-somatostatin antibody (1:500; Sigma). Staining samples were washed with Optimax three times. Cells were incubated for 1 h with either goat anti-mouse (Alexa Fluor 488 goat anti-mouse IgG, 1:200 dilution) or goat anti-rabbit (Alexa Fluor 647 goat anti-rabbit IgG, 1:200 dilution) antibodies, both from Molecular Probes (Eugene, OR). Omission of the primary antibody served as negative control. After washing, 4′,6-diamidino-2-phenylindole (DAPI) was applied to stain cell nuclei. Samples were analyzed using an iCys (CompuCyte, Cambrige, MA). A minimum of 5,000 cells was acquired and analyzed for each sample (12).
Fluorescein Diacetate/Propidium Iodide Viability Staining
Islet preparations were assessed for islet cell viability using cell membrane exclusion dyes as described previously (13). Briefly, 50–100 IEQ islets were transferred in PBS to a 10 × 35-mm counting petri dish. The 0.46 μM of fluorescein diacetate (FDA) and 14.34 μM of propidium iodide (PI) stock solutions were added to the sample, respectively. Using a fluorescent microscope, 50 islets were then assessed for cell viability by estimating the percentage of viable cells (green) versus the percentage of nonviable cells (red) within each islet. The mean and standard deviation of viable cells were then calculated for each preparation.
Glucose-Stimulated Insulin Release
After overnight culture at 37°C, islets (50–100 IEQ) were incubated with 2.8 and 20 mM glucose in RPMI medium supplemented with 1% bovine serum albumin (BSA)/25 mM HEPES for 1 h at 37°C. The supernatant was collected and stored at −80°C for insulin assessment by ELISA (Alpco, Salem, NH, USA). The glucose-stimulated insulin index was calculated as the ratio of insulin released during exposure to high glucose (20 mM) over the insulin concentration released during low-glucose incubation (2.8 mM).
Adenosine Diphosphate (ADP)/Adenosine Triphosphate (ATP) Ratio Assay
The ADP/ATP ratio assay was performed using the ApoSENSORTM ADP/ATP Ratio Assay Kit (MBL, Nagoya, Japan) according to the manufacturer's method. Briefly, 50 IEQ islets were transferred into a 1.5-ml microcentrifuge tube and washed with 1 ml of DPBS. The islet cell suspension was transferred into a 96-well luminometer plate in triplicate. Nucleotide releasing buffer (100 μ1) was added and gently shaken on a microplate shaker for 5 min. ATP monitoring enzyme (50 μ1) was added. The sample was incubated 1 min and read with a luminometer (CENTRO LB960, Berthold technologies, Bad Wildbad, Germany) (Data A). The samples were read at 10 min to measure background levels (Data B). Then, 50 μ1 ADP converting enzyme was added and the samples were read again in 1 min (Data C). ADP/ATP ratio is calculated as: (Data C – Data B)/Data A.
Statistical Analysis
All statistical analyses were performed using SAS 9.1 software (SAS Institute Inc., Cary, NC, USA) and EXCEL software (Microsoft, Redmond, WA). Descriptive statistics are given as means ± SE. Findings were considered statistically significant if the corresponding value was p < 0.05. Methods of analysis include two-sample t-tests, chi-square tests of independence, and multiple linear regression. Methods used in each analysis are described below with their corresponding results.
Results
Islet Recovery From Human Pancreata Purified Using RGP Following CGP
To evaluate the effect of donor age on islet purification, human pancreata were divided into two groups based on donor age (group I: age ≤40, group II: age >40). The donor characteristics and isolation-related variables of the assessed human pancreata are shown in Table 1 and Table 2. The differences between groups I and II were compared using two-sample t-tests and chi-square tests of independence, and were considered statistically significant if the value was p < 0.05.
Donor-Related Variables

Isolation-Related Variables
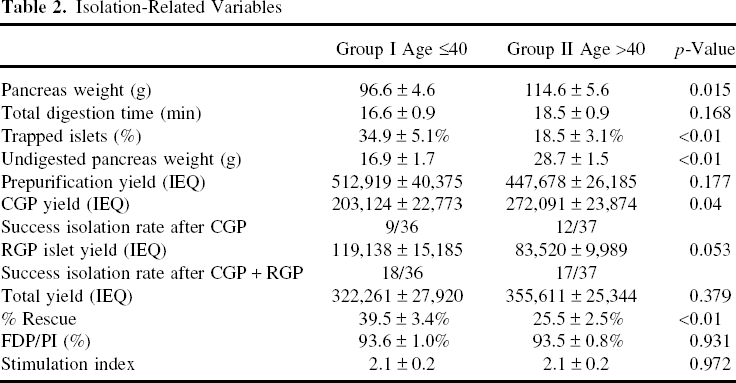
There were no significant differences in BMI, CIT, and gender between the two groups (Table 1). However, pancreas weight of group I was significantly lower than that of group II (96.6 ± 4.6 vs. 114.6 ± 5.6 g, p = 0.015).
Although prepurification yield in both groups were comparable (group I vs. II: 512,919 ± 40,375 vs. 447,678 ± 26,185 IEQ; p = 0.177), postpurification yield from CGP in group I was significantly lower (group I vs. II: 203,124 ± 22,773 vs. 272,091 ± 23,874 IEQ; p = 0.040). The percent rescue in group I, which is calculated by islet yield from RGP/total islet yield, was significantly higher when compared to group II (36.8 ± 3.3% vs. 24.9 ± 2.7%; p < 0.01).
The overall percent rescue was positively correlated with the percent trapped islets (R = 0.584) (Fig. 1A). Donor age was negatively correlated with the percent rescue (R = 0.440) and with the percent trapped islets (R = 0.511) (Fig. 1B, C).

(A) Correlation analysis between percent rescue and percent trapped islets. (B) Correlation analysis between percent rescue and age. (C) Correlation analysis between percent trapped islet and age.
In addition, the success rate of islet isolation was assessed in each group, defined by the following formula: Total islet yield/pancreas weight >3000 IEQ/g and purity >30%. While the success rate in group I increased by RGP from 25% (9/36) to 50% (18/36), that in group II increased from 32.4% (12/37) to 45.9% (17/37) (Table 2). The overall rate of success from RGP, in 52 cases where CGP alone did not result in success (27 and 25 cases in group I and II, respectively), was 28.9% (15/52) (95% CI = 17.1%, 43.1%), and was not statistically different between age groups. Moreover, the percentage of trapped islets was significantly higher in group I (group I vs. II: 34.4 ± 5.2% vs. 17.3 ± 3.2%; p < 0.01). The results indicated that RGP was more efficient in the purification of trapped islets, which often come from younger donors rather than older donors.
There were no significant differences between groups in FDA/PI and stimulation index.
Furthermore, the relationship between islet yield (both RGP and CGP) and percentage of trapped islets was assessed using multiple linear regression simultaneously adjusting for pre-purification yield and age.
Results indicated that percent trapped islets each have an independent positive association with RGP islet yield (Fig. 2A), and that donor age adjusted for percent trapped islets was not significant (Fig. 2B). An increase of 100,000 IEQ in prepurification yield was associated with a significant increase in islet yield (p < 0.0001). An estimated 20% increase in RGP yield was associated with each 100,000 IEQ increase in prepurification yield. An increase in the percent trapped islets was associated with a significant increase in RGP yield (p = 0.004). An estimated 10% increase in RGP yield was associated with each 10% increase in percent trapped islets.

(A) Correlation analysis RGP yield and trapped islet (%). (B) Correlation analysis between RGP islet yield and donor age. (C) Correlation analysis between CGP yield and % trapped islet. (D) Correlation analysis between CGP yield and age.
Analysis indicates that donor age and percent trapped islets each have a significant independent association with CGP islet yield adjusted for prepurification yield (Fig. 2C, D). Donor age was positively associated with CGP yield (Fig. 2D). Results of analysis estimate a 33% increase in expected CGP islet yield comparing donors over the age of 40 to donors under the age of 40 (p = 0.03). Adjusted for donor age and prepurification yield, percent trapped islets was significantly negatively associated with CGP yield (Fig. 2C). Results of analysis estimate an expected 10% decrease in CGP yield associated with each 10% increase in percentage of trapped islets (p < 0.0001).
Comparison Between CGP and RGP Islets
Our data indicate that RGP following CGP has substantial benefits in the purification of trapped islets. However, the effects of the RGP procedure regarding islet quality should be addressed. Cell viability was not altered by the addition of a RGP step (13). The quality of islets has been evaluated by in vivo assessment of islet potency using diabetic nude mice, islet viability test using FACS, and insulin stimulation index. Therefore, the quality of β-cells from RGP following CGP was examined precisely.
β-Cell Fractional Viability
To investigate β-cell-specific viability islets were stained and dissociated with the following: Newport green, TMRE, and 7-AAD. The stained islet cells were analyzed by FACS. There was no apparent difference in the percentage of apoptosis in whole cell subsets. However, RGP islets had an 11% higher viability in β-cells compared to those from CGP islets (p < 0.01) (Fig. 3A).

(A) β-Cell fractional viability assessment of CGP or RGP islets. Five human islet preparations were utilized to evaluate the effect of RGP on islet cell viability. After gating the 7-AAD- cell population, the percentages of TMRE+ cells in NGbright (β-cells) were analyzed. β-Cell fractional viability from RGP was significantly higher when compared to CGP. (B) Analysis of cellular composition in CGP or RGP islets. There was no significant difference in β-cell content assessed by LSC/iCys between CGP and RGP islets. Data are presented as mean ± SD of five independent islet preparations. (C) ADP/ATP ratio assay of CGP or RGP islets. To assess the quality of CGP and RGP islets, ADP/ATP ratio assay was performed. ADP/ATP ratio in RGP islets was significantly lower than CGP islets. Data are presented as mean ± SD of five independent islet preparations.
Cellular Composition Assay
In general, RGP was performed to recover islets from the heavy density layers. In order to examine the differences between CGP and RGP islets in terms of cellular composition, islet cells were stained with anti-C-peptide, glucagon, somatostatin, and PP-antibody and analyzed by LSC/iCys. There were no significant differences between the two groups (Fig. 3B), indicating the islets recovered by CGP and RGP have a similar β-cell content in the islets.
ADP/ATP Ratio Assay
The ADP/ATP assay could be a rapid and potent predictor of islet quality (10). In order to compare the quality of CGP and RGP islets, the ADP/ATP ratio assay was performed. The ADP/ATP ratio was significantly better in the RGP islets than in the CGP islets. The results suggested that the quality of whole islet cell subsets from RGP might higher than those from CGP (Fig. 3C).
Discussion
The quality and quantity of islets transplanted into patients with type 1 diabetes is critical to achieve a successful islet transplantation. Despite significant improvements in both the procurement and preservation of human pancreata, as well as in isolation technology (2,6,9,14,15,17,36,46), insulin independence still requires transplantation from multiple donors under current protocols. This eventually raises the issue of the shortage of pancreata. There is an urgent need to expand the age criteria of donors utilized for islet transplantation (16,31). The present study showed that RGP was more effective in the purification of trapped human islets than CGP, resulting in improved isolation outcomes from younger donors. The quality of β-cells in RGP islets was significantly better when compared to those in CGP islets. The data indicate that RGP will be of assistance in expanding donor criteria and achieving more successful isolation outcomes.
Donor age is significantly associated to islet isolation outcomes (19,23,26). In general, younger donors have less medical history and are not exposed to as many pathogens. Younger donor islets were also reported to be of a high quality and functionality (3,42). However, it is still difficult to consistently recover sufficient islet yields from younger donors using the current islet isolation methods (3,19,23). One of the major reasons is the technical difficulty in obtaining highly purified islets from younger donor pancreata. In many cases, islets from younger donors remain mantled and embedded by surrounded acinar cells after enzyme digestion. These trapped islets are difficult to separate from nonendocrine cells using CGP (3,28,30). The data demonstrate the inefficiency of CGP to separate trapped islets and its negative effects on islet yields from younger donor pancreata. When many trapped islets are obtained after the digestion process due to poor enzyme digestion and/or donor factors such as younger age, RGP should be considered as an additional purification step to separate the islets trapped in exocrine tissue.
Our data showed that the percentage of trapped islets in isolations from younger donor pancreata significantly increased. Considering pancreas structure, collagens are the major fibrous proteins constituting the islet–exocrine interface. The islet–exocrine interface of the human pancreas is comprised of type I, III, IV, V, and VI collagens (11,44,45). Also, it has also been reported that the total collagen content increases with age in the human pancreas (4). This increase of total collagen in the pancreas may be responsible for the variability seen with age in regards to enzymatic digestion.
In the human islet purification procedure, islet cell separation is based on differences in density or velocity of sedimentation. However, exocrine tissue density is intrinsically variable, depending on the secretory status of the acinar cells. Therefore, exocrine tissue is more sensitive to ischemic (22) and mechanical stresses (38), inherent in the islet isolation procedure. These factors could effectively cause exocrine enzyme discharge or impair cell viability, leading to cellular swelling and edema. The failure of the islet purification process often results from this acinar tissue swelling (20), leading to a migration of acinar tissue from higher density to lower density layers.
In the current islet isolation, large-scale continuous purification using the COBE2991 cell processor, with top loading, is the gold standard (33,39,40). Top loading has the potential advantage of keeping the islet cells in a physiological medium for the maximum possible time and minimizing centrifugal forces (20). On the other hand, top loading increases cell aggregations, which decrease the efficiency of purification. RGP is a discontinuous gradient purification method with bottom loading. The disadvantages of discontinuous gradients include a lower effective cell load and an accumulation of cells at the interfaces, which can cause aggregation (20).
In our protocol, the digests were collected and suspended in the Euroficoll (1.132 g/ml) prior to centrifugation. Hyperosmolar Euroficoll with a high glucose content and viscosity prevents acinar tissue from being edematous. Although the incubation of digests in Euroficoll increases the density of both endocrine and exocrine tissue, the latter is affected much more than the former (20). Therefore, the difference in the density between endocrine and nonendocrine tissue might allow RGP to separate trapped islets from nonendocrine cells more effectively than CGP.
It has been reported that Ficoll exposure to islets negatively affects function (37). However, in previous studies, no differences in the quality of islets obtained by CGP and RGP, with both in vitro and in vivo assays, has been demonstrated (13). During the additional purification, islets obtained by RGP are exposed to Ficoll gradient again, as well as additional centrifugal stress. Therefore, the viability of RGP islets is assumed worse. Unexpectedly, β-cell viability of islets through RGP is significantly better when compared to CGP.
There are several reasons to possibly explain this increased viability. During islet purification by continuous density gradients, pancreatic tissue fractions with different densities (1.077–1.100 g/ml) were collected. RGP is usually performed to recover islets from impure and/or pellet fractions, which usually came from relatively heavier density layers. In many cases, β-cell viability of islets from impure layers is better when compared to the purest layer (unpublished data). Therefore, β-cell viability of RGP islets might be better even after additional purification because they have higher β-cell viability initially. These data encourage the performance of RGP even when the number of islets remaining in the impure layer is low. It is also possible the surrounding tissue protects the β-cell from exposure to the mechanical and chemical stress during islet isolation. RGP islets typically consist of a higher proportion of these trapped islets.
This study also compared β-cell content of CGP and RGP islets. There was a tendency for the β-cell content of RGP islets to be higher than that of CGP islets (not significant). Significant stress created during islet isolation causes substantial damage to islet cells. β-Cells are more sensitive to noxious stimuli than other cells comprising islets (24,27,34). Therefore, relatively lower β-cell content in islet cells may imply that islet cells were exposed to greater stress. Taken together, higher β-cell viability and β-cell to α-cell ratio in RGP may suggest trapped islets to be of a higher quality.
ADP/ATP ratio of RGP islets is also significantly lower than CGP islets. However, ADP/ATP ratio was affected by the level of non-β-cells such as acinar, ductal, and α-cells contained in islet preparations. Therefore, it is very difficult to compare β-cell viability of islet preparations that have different islet purities by ADP/ATP ratio. In this study, the cellular composition of islets from both groups was very different. RGP islets usually contained a higher amount of nonendocrine cells. Despite the fact that it is difficult to conclude which islets have better β-cell viability using ADP/ATP ratio assay, the overall viability of whole cell subsets in RGP islets was significantly better even after the additional purification step. These results also support the concept that RGP causes marginal effects in islet cell viability.
Although our data demonstrate RGP is effectively capable of recovering extra islets, especially trapped islets, the cost effectiveness of RGP should be taken into consideration. In general, RGP has been performed when more than 50,000 IEQ are remaining in the impure fractions (<30%). However, our data demonstrate the quality of islets recovered by RGP to be significantly higher. This seems to encourage factoring in the higher quality of the remaining islets, as well as the quantity of remaining islets, into the decision to perform RGP.
This study demonstrated the ability of RGP to effectively purify trapped islets, which can improve islet yield. Additionally, the quality of β-cells in RGP islets was shown to be significantly better when compared to CGP. These findings suggest that RGP is capable of improving isolation outcomes from young donor pancreata, which subsequently may have a higher potential for β-cell replacement therapy.
Footnotes
Acknowledgments
This work was supported in part by National Institute of Health grants for General Clinical Research Center 5 U01 DK70460-02, 5 R01 DK025802-23, Islet Cell Resources grant 5U42RR016603, Juvenile Diabetes Research Foundation International grants 4-2004-361, and the Diabetes Research Institute Foundation. The authors are grateful to the members of the Human Cell Processing facility, Preclinical Cell Processing Laboratory of the Cell Transplant Center, Clinical Islet Transplant Program, General Clinical Research Center, Imaging Core at the Diabetes Research Institute, Administrative Offices at the Diabetes Research Institute, and Organ Procurement Organizations for the continuous enthusiasm and support to our program.
