Abstract
Transplantation of hepatocytes or bone marrow-derived cells has been shown to ameliorate liver fibrosis in animal models, but no direct comparison of relative efficiency has been made. The aim of this study was to compare the efficiency of a bone marrow-derived clonal mesenchymal stem cell line established by us (rBM25/S3) with that of its adipogenic or hepatogenic differentiation derivative for suppression of rat liver fibrosis. After induction of differentiation of rBM25/S3 cells into adipogenic or hepatogenic cells in culture, we intrasplenically transplanted the three types of cells into rats (3 × 107 cells/rat) before and 4 weeks after initiation of carbon tetrachloride treatment (1 ml/kg body weight twice a week for 8 weeks) to induce liver fibrosis. Undifferentiated rBM25/S3 cells were the most effective for suppression of liver fibrosis, followed by the adipogenic cells and hepatogenic cells. Expression levels of MMP-2 and MMP-9 were also highest in undifferentiated rBM25/S3 cells. These results indicate that bone marrow-derived clonal mesenchymal stem cell lines are useful for further mechanistic studies on cell-mediated suppression of liver fibrosis and that such cell lines will provide information on an appropriate cell source for transplantation therapy for cirrhosis.
Introduction
Liver fibrosis is characterized by accumulation of extracellular matrix (ECM) proteins, including collagen, which occurs in most types of chronic liver disease (2,7). Excess accumulation of ECM proteins results in destruction of the hepatic tissue architecture by formation of a fibrous scar and in deterioration of the function of liver parenchymal cells, eventually leading to cirrhosis with nodules of partially regenerated hepatocytes surrounded by irregularly distributed fibrous septa. Liver cirrhosis affects hundred millions of people worldwide, and the mortality rate is steadily increasing (15). Liver transplantation is the most drastic therapy for cirrhosis with chronic hepatic failure (15), but shortage of donor organs limits applicability of this therapy. Thus, alternative approaches that circumvent the use of the whole organ, including transplantation of cells (34,35), are needed.
Liver fibrosis, once regarded as a relentlessly progressive abnormality, is now considered to be potentially reversible when the causative factors are removed (14,33). To facilitate resolution of fibrosis, transplantation of cells of various origins has been examined. Transplantation of hepatocytes was reported to improve survival and/or liver function in rats with cirrhosis induced by administration of CCl4 and phenobarbital (9,25,26). Sakaida et al. (31) reported that transplantation of freshly isolated unfractionated bone marrow cells reduced CCl4-induced liver fibrosis in mice. They indicated possible involvement of MMP-9, which was expressed at a high level in the bone marrow-derived cells, in the resolution of fibers. Oyagi et al. (28) provided evidence that transplantation of bone marrow-derived mesenchymal cells cultured with HGF, but not those without HGF, improved some hepatic functions and suppressed liver fibrosis in rats injured by CCl4. Several other studies have shown that transplantation of bone marrow-derived mesenchymal stem cells could ameliorate liver fibrosis (6,38), but their effects were marginal or characterization of the cells used was limited. Thus, it remains controversial which type(s) of cells among those derived from the bone marrow shows the most potent suppressive effect on fibrosis, although further evidence that bone marrow cells contribute to regression of liver fibrosis in mice has recently been provided (11,16).
We recently established clonal mesenchymal stem cell lines from rat bone marrow (23). One of these cell lines, rBM25/S3(GFP), grew rapidly (with a doubling time of ~24 h) for at least 300 population doubling levels over a period of 300 days, retaining multipotency to differentiate into hepatogenic, adipogenic, osteogenic, and chondrogenic lineages. When plated on Matrigel with hepatocyte growth factor and fibroblast growth factor-4, the cells differentiated into hepatocyte-like cells with a high efficiency of practically 100% within 7 days. Intrasplenic transplantation of the differentiated cells prevented fatal liver failure in 90%-hepatectomized rats, indicating exhibition of hepatic function by the cells in vivo as well. Considering the extremely high propagation potency of rBM25/S3 cells, such type of cells will be a promising transplantation cell source if proved to have a suppressive effect on liver fibrosis. The present study was undertaken to examine possible effects of rBM25/S3(GFP) and its differentiated derivatives on CCl4-induced liver fibrosis. We found that undifferentiated rBM25/S3(GFP) cells were the most effective for suppressing the fibrosis in the liver.
Materials and Methods
Reagents
Carbon tetrachloride (CCl4; Wako Pure Chemical Industries, Osaka, Japan) was dissolved with olive oil at a volume ratio of 1:1. Epidermal growth factor (EGF), platelet-derived growth factor (PDGF)-BB (Sigma-Aldrich, St. Louis, MO), and fibroblast growth factor (FGF)-4 (R&D Systems, Minneapolis, MN) were commercially obtained. HGF (Δ5 variant, a mature two-chain form) was kindly provided by Daiichi Pharmaceutical (Tokyo, Japan). Fibronectin and Matrigel were purchased from Sigma-Aldrich and BD Sciences (Bedford, MA), respectively. Type I collagen was prepared from tail tendons of rats as previously described (27). Trypsin (1:250) and type I collagenase were purchased from Difco Laboratories (Detroit, MI) and Sigma-Aldrich, respectively.
Cell Culture
rBM25/S3(GFP) (23) cells were maintained in MAPC medium (18), which consists of 60% DMEM (Nissui Pharmaceutical, Tokyo), 40% MCDB-201 (Sigma-Aldrich), 1× insulin/transferrin/selenium A containing sodium pyruvate (Gibco BRL, Grand Island, NY), 1× linoleic acid/bovine serum albumin (Sigma-Aldrich), 10–9 M dexamethasone (Banyu Pharmaceutical, Tokyo), 10–4 M ascorbic acid 2-phosphate (Sigma-Aldrich), penicillin (100 U/ml), streptomycin (100 μg/ml), and 2% fetal bovine serum (FBS). The cells were labeled in advance with green fluorescence protein (GFP) by transfecting with a plasmid pEGFP-N2 (BD Biosciences) with Trans IT®-LT1 reagent (Mirus, Madison, WI). The labeled cells showed properties very similar to those of the original cells. For inducing differentiation into hepatogenic lineage, the cells were cultivated in MAPC medium supplemented with HGF (20 ng/ml) and FGF-4 (10 ng/ml) on Matrigel as previously described (23). For adipogenic differentiation, cells were inoculated into collagen-coated dishes in the same medium as that described above. Cytoplasmic lipid droplets were detected by Oil Red-O staining.
Induction of Liver Fibrosis with CCl4 and Transplantation of Cells
Eight-week-old male Sprague-Dawley rats were injected twice a week intraperitoneally with CCl4 (1 ml/kg body weight as a 1:1 mixture with olive oil) for 8 weeks to induce liver fibrosis (Fig. 1). rBM25/S3 and its differentiated derivatives were transplanted before and 4 weeks after the initial application of CCl4. Cells were suspended in PBS at 3 × 107 cells/0.7 ml and transplanted into the spleen of each rat under anesthesia with diethyl ether. No immunosuppressive reagent was given after the cell transplantation. All of the experiments were performed according to the institutional ethical code for the care and use of experimental animals.
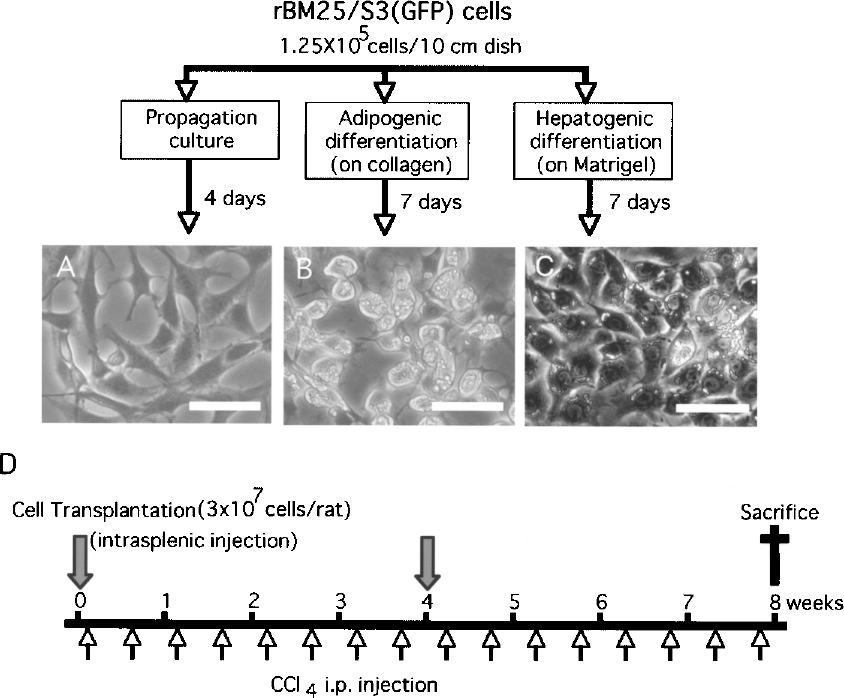
Differentiation of rBM25/S3(GFP) cells and experimental protocol of cell transplantation into rats exposed to CCl4. rBM25/S3(GFP) cells maintained in the propagation medium (A) were induced to differentiate either on collagen (B) or on Matrigel (C) with HGF (20 ng/ml) and FGF-4 (10 ng/ml) for 7 days. Scale bars: 50 μm. (D) Experimental protocol. Rats were intraperitoneally injected with CCl4 (0.5 ml/kg body weight) twice a week for 8 weeks. Control animals were administered a vehicle (olive oil). Undifferentiated or differentiated rBM25/S3(GFP) cells were intrasplenically transplanted at a dose of 3 × 107 cells/rat at indicated time points.
Preparation of Tissues and Immunostaining
For preparation of tissue sections, the animals were sacrificed with deep anesthesia and perfused via the heart with 4% paraformaldehyde to flush out blood cells. The right median lobe of the liver and the spleen were resected and incubated in 4% paraformaldehyde overnight. The tissues were then soaked in 30% sucrose for 3 days and frozen with liquid nitrogen until preparation of tissue sections with a cryostat.
Cultured cells or frozen tissue sections (6 μm in thickness) were fixed with cold acetone and treated with a primary antibody [i.e., anti-rat MMP-9 antibody (Dako, Denmark), anti-human α-SMA antibody (Chemicon International, Temecula, CA), or anti-GFP antibody (raised in this laboratory)], followed by a second antibody, FITC- or TRITC-conjugated anti-IgG antibody (Sigma-Aldrich). The samples were mounted with VEC-TASHIELD (Vector Laboratories, Burlingame, CA).
Quantitative Analysis of Liver Fibrosis
Tissue sections of the liver were stained with picrosirius red under conventional conditions. Pictures were taken of five areas per section in three sections per liver selected randomly, and extent of fibrosis was estimated using an image analysis software (Scion image analyzer).
RT-PCR Analysis
Total RNA was isolated by the guanidine/thiocyanate/phenol method, and 1 μg RNA was used for cDNA synthesis. The resulting products were amplified under the following conditions: initial incubation at 94°C for 4 min followed by 30 cycles at 94°C for 30 s, 55°C for 30 s, and 72°C for 30 s, and then a final step at 72°C for 5 min. The primers and expected length of products are shown in Table 1. GAPDH was used as an internal control. The amplified products were subjected to electrophoresis in 1% agarose gels and stained with ethidium bromide.
Primers and Expected Length of Products

Zymography
Cells were washed twice with Opti-MEM (Invitrogen, Carlsbad, CA) and further incubated in 10 ml of the same medium for 24 h. The conditioned medium was harvested, concentrated into 100 μl by centrifugation using a Centricon (Amicon Ultra-15 Ultracell-10k; Millipore, Billerica, MA), and mixed with 50 μl of 3× SDS sample buffer. The samples (10 μl each) were applied onto 8% SDS-polyacrylamide slab gel containing 1 mg/ml gelatin (Sigma-Aldrich). After electrophoresis, the gel was washed with water to remove SDS, soaked in a protein-refolding buffer (2.5% Triton X-100, 10% glycerol, 0.5 mM CaCl2, 100 mM NaCl, 50 mM Tris-HCl/pH 7.4) for 1 h, and incubated in 50 mM Tris-HCl/pH 7.4 containing 0.5 mM CaCl2 for 12 h at 37°C. The gel was then stained with 1% Coomassie brilliant blue G250. The gel was further treated with 10% methanol and 5% acetic acid to destain the gel. The gelatinolytic activity was detected as clear bands on a blue background of the CBB-stained gel. The experiment was done in triplicate.
Statistical Analysis
Data are expressed as means ± SD. Significance in difference was determined by Student's t-test following Bonferroni correction (threshold adjusted to p < 0.005).
Results
rBM25/S3(GFP) and its Differentiation Derivatives
rBM25/S3(GFP) cells rapidly grew on fibronectin-coated dishes in the presence of EGF and PDGF-BB (Fig. 1). They showed properties characteristic to mesenchymal stem cells with concomitant expression of some adipogenic genes at moderate levels (23). rBM25/S3 (GFP) cells could be induced to differentiate into hepatocyte-like cells under conditions described previously (23) [i.e., on Matrigel substratum with HGF (20 ng/ml) and FGF-4 (10 ng/ml)]. The cells expressed albumin, cytochrome P450 (CYP) 1A1, CYP1A2, glucose 6-phosphatase, tryptophane-2,3-dioxygenase, tyrosine aminotransferase, hepatocyte nuclear factor (HNF) 1α, and HNF4α. Intrasplenic transplantation of the differentiated cells prevented fatal liver failure in 90%-hepatectomized rats, indicating that the differentiated rBM25/S3(GFP) cells exhibit hepatic function in vivo as well.
When rBM25/S3(GFP) cells were cultivated in the same medium but on a collagen matrix, the cells more prominently exhibited adipogenic properties (Fig. 2). Lipid droplets progressively accumulated in the cytoplasm (Fig. 2A). Among the adipocyte-related genes examined, adiponectin, leptin, and resistin were newly induced when the cells were cultivated on collagen matrix (Fig. 2B). Expression levels of G3P acyltransferase, adipocyte lipid-binding protein, adipsin, and lipoprotein lipase increased under the differentiation conditions. PPAR-γ and fatty acid synthase were expressed already in undifferentiated rBM25/S3(GFP) cells and the expression levels remained unchanged.

Differentiation of rBM25/S3(GFP) cells into adipocyte-like cells on a collagen matrix. (A) Accumulation of lipid droplets in the cytoplasm demonstrated by Oil Red-O staining (counterstained with hematoxylin). Scale bars: 50 μm. (B) RT-PCR analysis of expression of adipocyte-related genes, such as peroxisome proliferator-activated receptor gamma (PPAR-γ), fatty acid synthase, glycerol-3-phosphate acyltransferase, adipocyte lipid-binding protein, adiponectin, adipsin, leptin, lipoprotein lipase, and resistin. GAPDH was used as an internal control. The cells were cultured for the indicated days.
Suppression of Liver Fibrosis by Transplantation of rBM25/S3(GFP) Cells
It is well known that biweekly administration of CCl4 induces severe fibrosis in the liver of rats. The treatment retarded increase of body weight during the observation period in the young rats we used (Fig. 3). We compared effects of transplantation of the undifferentiated original rBM25/S3(GFP) and the two types of differentiated derivatives. The retardation of increase in body weight was partially recovered by intrasplenic transplantation of rBM25/S3(GFP) cells, with the most prominent effect exerted by the undifferentiated cells, followed by the adipogenic cells and hepatogenic cells (Fig. 3).
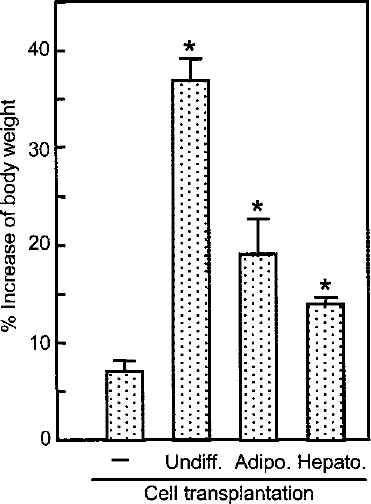
Percent increase in body weight during the observation period compared to body weight at the beginning of the experiment. Values are means ± SD for three rats. Statistically significant difference from the value of the untransplanted rats (–) was verified by Student's t-test following Bonferroni correction. The threshold of significant difference was <0.005. *p < 0.0001.
Extent of liver fibrosis was examined after staining with picrosirius red (Fig. 4). In the control rats not treated with CCl4, collagen fiber was marginally observed only around central veins in the liver (Fig. 4A). Application of CCl4 for 8 weeks resulted in marked accumulation of collagen fibers, which was nearly completely abrogated by transplantation of the undifferentiated rBM25/S3(GFP) cells before and 4 weeks after initiation of CCl4 treatment (Fig. 4C and E). Liver fibrosis is considered to be due to activation of hepatic stellate cells (4,8,19). In accordance with this notion, α-smooth muscle actin, a marker of activated stellate cells, was induced in association with fiber accumulation (Fig. 4D), which was completely abrogated by transplantation of the undifferentiated rBM25/S3(GFP) cells (Fig. 4F). Contrary to our expectation, the cells differentiated either on collagen or on Matrigel showed a less efficient suppressive effect on the fibrosis (Figs. 4G and H). Quantitation of levels of fibrosis by computer-assisted image analysis confirmed the observation (Fig. 5). Transplantation of the undifferentiated rBM25/S3(GFP) cells most effectively suppressed CCl4-induced liver fibrosis followed by the adipogenic and hepatogenic cells. Difference in levels of fibrosis in each combination among the different experimental groups was statistically significant (p < 0.0001).

Suppression of CCl4-induced liver fibrosis by transplantation of rBM25/S3(GFP) cells. Liver sections of the rats treated under the conditions described in the legend for Figure 1 were stained with picrosirius red (A, C, E, G, and H; scale bars: 200 μm) or with an antibody against α-smooth muscle actin, a marker of activated stellate cells (B, D, and F; scale bars: 100 μm).
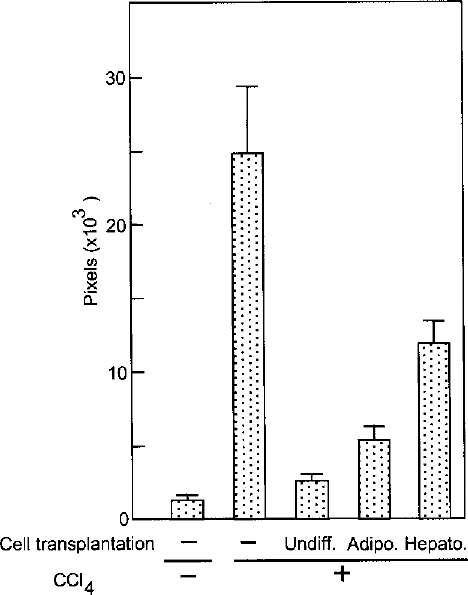
Quantitative estimation of the extent of liver fibrosis. Red signals in the liver sections stained with picrosirius red were collected from five areas/section in three sections/liver and expressed in pixels. Difference between each combination among the groups was statistically significant (p < 0.0001) as examined by Student's t-test following Bonferroni correction (the significance threshold was <0.005).
Expression of MMPs in rBM25/S3(GFP) Cells in Culture and In Vivo
Because some members of the MMP family are known to be involved in matrix remodeling and regulation of fibrosis in the liver (1,10,12), we examined expression of MMPs in rBM25/S3(GFP) and its differentiation derivatives. RT-PCR showed that undifferentiated rBM25/S3(GFP) cells expressed MMP-2, -9, and -14 (Fig. 6A). The expression of MMP-9 in rBM25/S3(GFP) cells was confirmed by immunostaining (Fig. 6B). MMP-2 mRNA was detected but at lower levels in the adipogenic and hepatogenic derivatives. MMP-9 mRNA was not detected in either type of cells. Expression of MMP-14 was not affected by induction of differentiation. Zymography revealed that undifferentiated rBM25/S3(GFP) cells had a higher level of proteolytic activity of MMP-9 than did the adipogenic and hepatogenic derivatives (Fig. 6C). Seven days after intrasplenic transplantation of undifferentiated rBM25/S3(GFP) cells, GFP and MMP-9 double-positive cells were detected in the spleen (Fig. 7A–C). While no MMP-9-positive cells were observed in the normal liver, MMP-9-positive cells appeared in the liver of rats treated with CCl4 for 8 weeks (Figs. 7D–I). After transplantation of undifferentiated rBM25/S3(GFP) cells, many GFP and MMP-9 double-positive cells were observed in the liver (Figs. 7J–L). These results indicate that expression of MMP-9 in the transplanted cells was sustained over the 8-week experimental period.
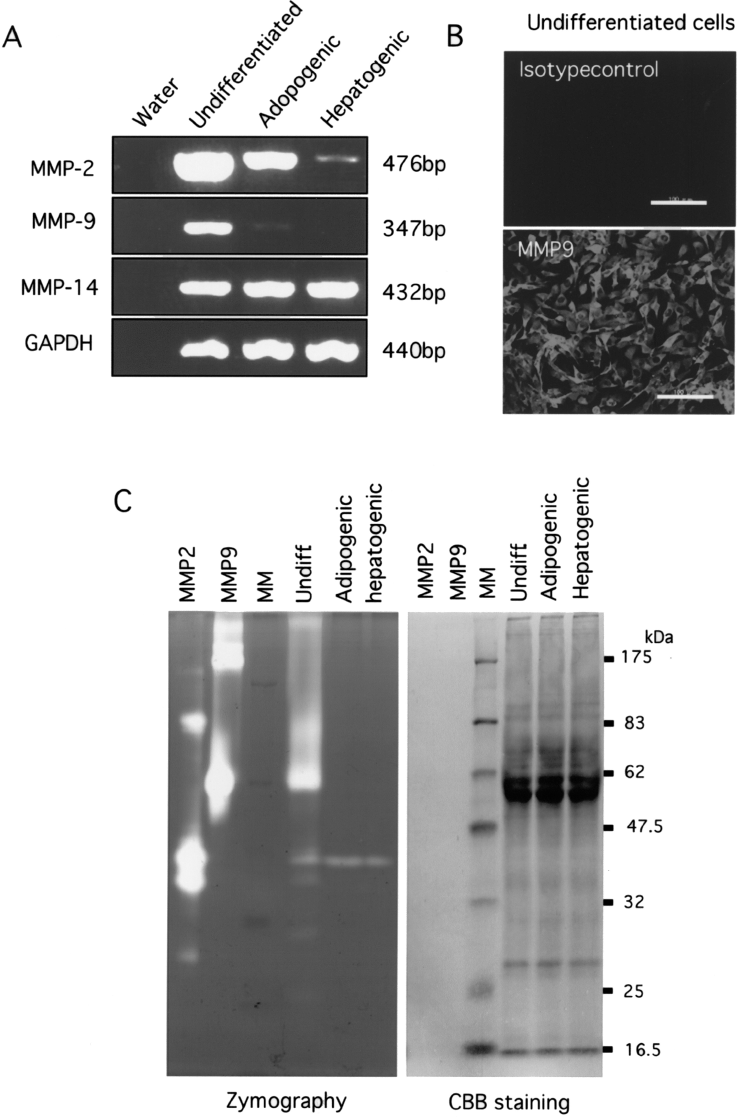
Expression of matrix metalloproteinases in rBM25/S3(GFP) cells in culture. (A) Expression of MMP-2, MMP-9, and MMP-14 in undifferentiated and differentiated rBM25/S3(GFP) cells as determined by RT-PCR. GAPDH was used as an internal control. (B) Immunocytochemistry for MMP-9 in undifferentiated rBM25/S3(GFP) cells. Scale bars: 100 μm. (C) Zymography of undifferentiated and differentitated rBM25/S3(GFP) cells. MMP2 and MMP9, authentic recombinant proteins; MM, molecular marker.

Immunostaining for MMP-9 in the spleen (A–C) and liver (D–L) after transplantation with rBM25/S3(GFP) cells. Tissue sections were stained with DAPI (A, D, G, and J), anti-GFP antibody (B, E, H, and K), or anti-MMP-9 (C, F, I, and L). (A–C) The spleen at 7 days after transplantation with rBM25/S3(GFP) cells; (D–F) the normal liver; (G–I) the liver of rats treated with CCl4 alone fixed at the end of the experiment; (J–L) the liver of rats treated with CCl4 and transplanted with rBM25/S3(GFP) cells fixed at the end of the experiment. Scale bars: 200 μm (A–F), 100 μm (G–L).
Discussion
Liver fibrosis/cirrhosis is a complex process involving progressive degeneration of hepatic parenchymal cells, inflammation, and activation of mesenchymal cells, stellate cells in particular, resulting in aberrant accumulation of fibers (2,7,15). Because damage to hepatic parechymal cells is mostly a triggering event in human and experimental liver fibrosis, it is reasonable to expect that transplantation of hepatocytes may not only improve the hepatic function but also interfere with the vicious spiral and eventually ameliorate liver fibrosis (34). In fact, a number of studies have shown that transplantation of hepatocytes can function to correct liver failure in cirrhotic animals and prolong their survival (3,9,21,25). For future clinical application of this approach, however, immune rejection of nonautologous hepatocytes remains as a critical issue to be solved, although even xenogeneic hepatocytes have been shown to survive after transplantation and to restore hepatic function in certain experimental settings (25).
Bone marrow is the most fertile tissue harboring various stem cells (17,24,29). Transplantation of unfractionated bone marrow cells (31) or bone marrow-derived mesenchymal stem cells (6,38) has been shown to suppress liver fibrosis. It is not clear, however, whether bone marrow cells or bone marrow-derived stem cells themselves have an antifibrosis function or whether they can exhibit an antifibrosis function only after differentiation into hepatocytes or some other types of cells in an in vivo environment. Bone marrow cells have been demonstrated to differentiate into hepatocytes in vivo after transplantation into the liver (22,27,32,37).
rBM25/S3 provides a unique opportunity for assessing relative efficiency of suppression of fibrosis among different types of cells. rBM25/S3 is a clonal cell line retaining stable properties during a propagation period of over 300 PDL (23). When transferred onto Matrigel in a medium containing HGF and FGF-4, rBM25/S3 cells readily and homogeneously (~100%) differentiated into hepatocyte-like cells expressing albumin and HNF4α (23). The cells appear functional also in vivo because transplantation of the cells rescued 90%-hepatectomized rats with otherwise fatal liver failure. On a collagen matrix, rBM25/S3 cells showed predominantly adipogenic phenotypes (Fig. 2). These differentiation events were appreciable within 4 days and fully proceeded by the 7 culture days. We compared possible suppression of CCl4-induced liver fibrosis by these three different types of cells derived form the clonal cell line. Undifferentiated rBM25/S3 cells were the most effective in suppression of liver fibrosis, followed by the adipogenic cells and the hepatogenic cells (Figs. 4 and 5). It should be noted that even the hepatogenic derivatives significantly suppressed CCl4-induced fibrosis (p < 0.0001).
It is still not clear why and how the undifferentiated rBM25/S3(GFP) cells most effectively suppressed liver fibrosis. Expression levels of MMP-2 and MMP-9 were highest in undifferentiated rBM25/S3 cells (Fig. 6). MMPs have been shown to be involved in regulation of liver fibrosis but in somewhat contradictory manners depending on the experimental setting, stage of fibrosis, and molecular species (10). MMPs, including MMP-2 and MMP-9, have been shown to be induced coincidently with inflammatory cytokines in the liver of rats at an early stage after exposure to CCl4 (20). Proliferation and activation of stellate cells were less prominent and resulting fibrosis was more strongly suppressed in either MMP-9- or MMP-13-deficient mice than in wild-type mice (30,36). On the other hand, transduction of MMP-1 by an adenovirus has been shown to promote fibrinolysis in the liver (13). Sakaida et al. (31) showed that MMP-9 was strongly expressed in transplanted bone marrow cells, resulting in suppression of CCl4-induced liver fibrosis. It is possible that differential expression of MMP-2 and MMP-9 observed in rBM25/S3(GFP) cells plays some role in ameliorating fibrosis induced by CCl4. In addition, expression of adiponectin was induced when rBM25/S3(GFP) cells were cultured in the differentiation conditions into adipogenic cells (Fig. 2). Adiponectin was shown to reverse the activation of and keep the quiescent state of hepatic stellate cells, thereby inhibiting liver fibrosis (5).
In summary, rBM25/S3 cells have an extremely high proliferation potency (>300 PDL) while stably retaining their original properties. The cells can be readily differentiated into hepatocytes within 7 days with ~100% efficiency. The original rBM25/S3 cells are suitable for treating liver fibrosis and their hepatogenically differentiated derivative can rescue liver failure (23). Use of the different cell types of the same clonal origin or their mixture for different pathological conditions in animal models will certainly contribute to a better understanding of the nature of liver dysfunction and to the development of new cell therapeutic strategies against various liver diseases.
Footnotes
Acknowledgments
The authors declare that they do not have anything to disclose regarding conflict of interest with respect to this manuscript. This work was partly supported by a grant from the Ministry of Education, Culture, Sports, Science and Technology (No. 18590269).
