Abstract
OBJECTIVE:
In our study, it was aimed to compare vestibulo-ocular reflex (VOR) gain and saccade parameters in HIMP and SHIMP tests between gender, right and left ears, and age groups in healthy adults and to examine the correlation between the tests regarding these parameters.
METHODS:
The study included a total of 100 healthy participants aged 18–65 and without complaints of hearing loss, dizziness, lightheadedness, and/or imbalance. Participants underwent HIMP and SHIMP tests, respectively.
RESULTS:
No significant difference was found in HIMP and SHIMP VOR gain values according to gender and age groups. SHIMP duration was significantly longer in women. VOR gain values were lower in the right ear. HIMP amplitude values were higher and SHIMP amplitude values were lower with increasing age. In older age groups, SHIMP peak velocity and duration values were significantly decreased, while HIMP duration value increased and latency value was longer. In the 1st saccade, a significant difference was obtained between HIMP and SHIMP tests for all saccade parameters. There was a statistically significant positive correlation between the VOR gain values of HIMP and SHIMP tests.
CONCLUSIONS:
The present study showed that VOR gain and saccade parameters obtained in different age groups will be important in determining clinical outcomes in vestibular pathologies.
Introduction
The vestibulo-ocular reflex (VOR) provides visual stabilization and clarity by moving the eye in the opposite direction of the head movement. The Head Impulse Paradigm (HIMP) Test is an objective test battery that evaluates VOR gain and saccade parameters of each semicircular canal with high-speed head movement [2]. VOR gain is the ratio of head speed to eye speed [10]. Rapid eye movements placing a moving object in the visual field are called saccades [23]. Saccades that occur during head movement are called covert saccades, while saccades that occur after head movement are called overt saccades. In individuals with vestibular system pathology, the HIMP test is used to detect covert saccades as they may not be recognized by the clinician during subjective assessment with the head impulse test (HIT) [32]. The HIMP test can also be performed using a video head impulse test (vHIT) device, which allows quantification of the VOR and detection of all compensatory saccades [30].
In 2016, MacDougall et al. developed the Suppression Head Impulse Paradigm (SHIMP) test as a complement and alternative to the HIMP test. Unlike the HIMP test, the individual follows a laser light in the same direction as the head movement during the test. In the SHIMP test, anti-compensatory saccades occur in healthy subjects but not in patients with vestibular pathology. In conclusion, saccades in the SHIMP test are a function of the vestibular system [18].
The normalization studies are important in order to establish normal value ranges in healthy individuals, allowing for comparisons with patient groups and the observation of pathological values [8]. In the literature, normalization studies conducted on healthy subjects have been applied to individuals without a history of otological, neurological, visual, and vestibular diseases [5, 34]. There are a limited number of SHIMP normalization studies in the literature. These studies only evaluated VOR gain and saccade parameters (except for duration) together [6, 25–27].
The aim of this study was to compare VOR gain and saccade parameters (amplitude, peak velocity, duration, and latency) in HIMP and SHIMP tests between gender, right and left ears, and age groups in healthy adults and to examine the correlation between the tests regarding these parameters.
Materials and methods
This study was conducted in the Audiology Unit of the Department of Ear Nose Throat (ENT) and the Neurosensory Laboratory of the Department of Neurology at Ege University Faculty of Medicine Hospital between January and July 2022. The study was approved by the Ege University Faculty of Medicine Medical Research Ethics Committee (Date: 02.12.2021, Decision No: 21-12T/6). Participants were informed about the study verbally and in writing and they signed an informed consent form.
The research group included 100 participants (52 women and 48 men) who agreed to volunteer among the relatives of patients, Ege University students, and hospital staff who declared that they did not have any health problems. The sample of this study was determined using the G*power 3.1 program.
The hearing test and tympanometry test of the participants were performed in the Audiology Unit of the Department of ENT. Videonystagmography (VNG), HIMP, and SHIMP tests were performed in the Neurosensory Laboratory of the Department of Neurology. The tests were administered on the same day by a single experienced audiologist (S.B). Test results were evaluated by an ENT specialist (T.K), a neurologist (N.C), and an experienced audiologist (G.K).
The study included participants aged 18–65 years, whose pure tone hearing thresholds, tympanometry findings, and oculomotor test results in VNG were within normal limits, whose positional tests were negative, and who agreed to volunteer. Participants who reported hearing loss and/or dizziness according to the case report and whose test results revealed hearing loss, any external and/or middle ear problem, BPPV or central vestibular disorder, complaints such as neck hernia, and visual impairment or head trauma that would affect the test were excluded from the study.
The participants were divided into 4 different age groups: 18–25, 26–40, 41–55, and 56–65 years old.
Head Impulse Paradigm (HIMP) Test and Suppression Head Impulse Paradigm (SHIMP) Test
HIMP and SHIMP tests were performed with an Interacoustics EyeSeeCam vHIT (Interacoustics, A/S Audiometer Allé 1 5500, Middelfart, Denmark). All participants sat on a stationary chair 1.5 meters away from the wall for the application of HIMP and SHIMP tests. After the eye and head calibration phase, the HIMP testing phase began. EyeSeeCam vHIT goggles have an interchangeable portable socketed camera for testing the right and left eye [11]. In our measurements for HIMP and SHIMP tests, the camera was attached to the left eye, and we recorded the left eye. The individual was asked to keep their gaze on the target point in the center of the wall and to maintain a fixed gaze on the target during head movements. The participant’s head was held from both sides and head impulse was performed in the horizontal plane at a 15°–20° angle with a sudden and fast (150–300°/s) motion in an unpredictable direction. Measurements within 700 ms after the head movement were included in the analysis. At least 10 maneuvers were performed separately for each side. For each head movement, saccades caused by other reasons such as blinking were not included in the analysis. Then the SHIMP test was started. Unlike the previous test, the participant was instructed to follow the midline laser light reflected on the wall during the movement (Fig. 1 and 2).
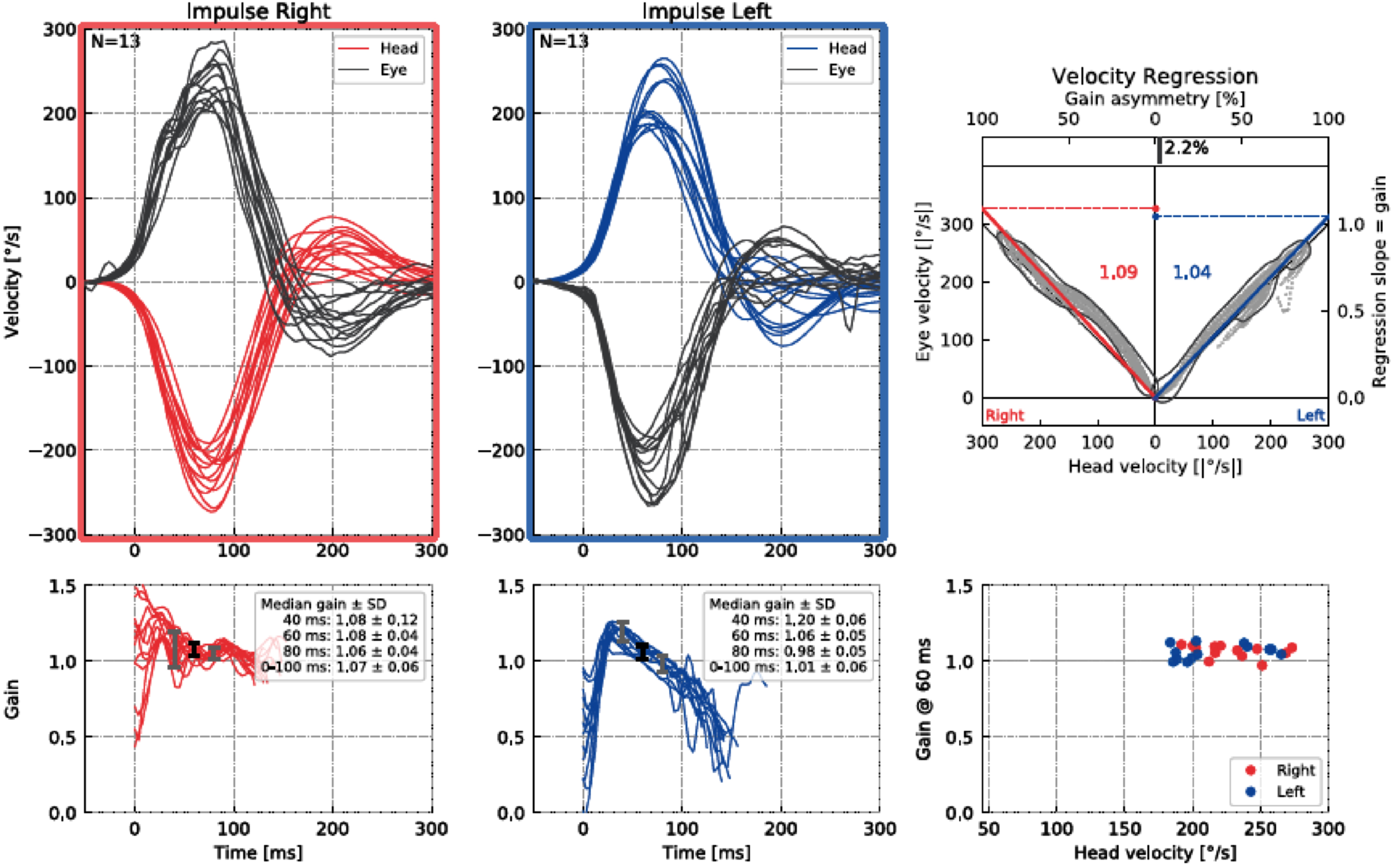
Results of an HIMP test for a participant.
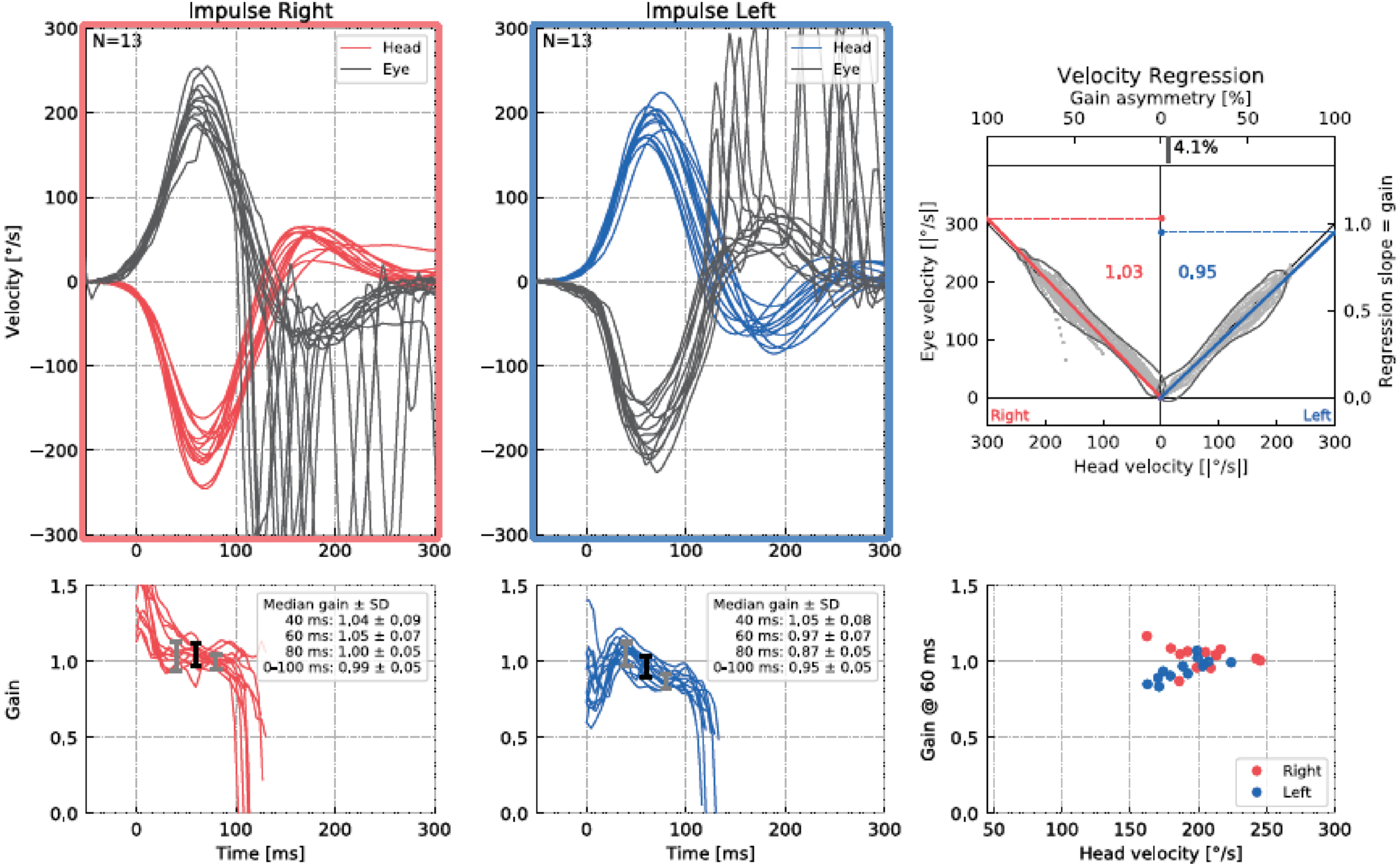
Results of an SHIMP test for a participant.
In our study, VOR gain (40 ms, 60 ms, 80 ms and regression gain), saccade amplitude (°), peak velocity (°/sec), duration (ms), and latency (ms) parameters were used to evaluate HIMP and SHIMP tests (Fig. 3 and 4). For all saccades, the amplitude is the area under the curve based on eye velocity [3]. Peak saccade velocity is defined as the velocity at the point where the anti-compensatory saccade reaches its maximum [16]. Duration refers to the time between the beginning and the end of the saccade [17]. Saccadic latency is defined as the time between the onset of the saccade and the appearance of the saccade response [4] (Fig. 5). More than one saccade can occur in a single head impulse (1st saccade, 2nd saccade,...) [11].
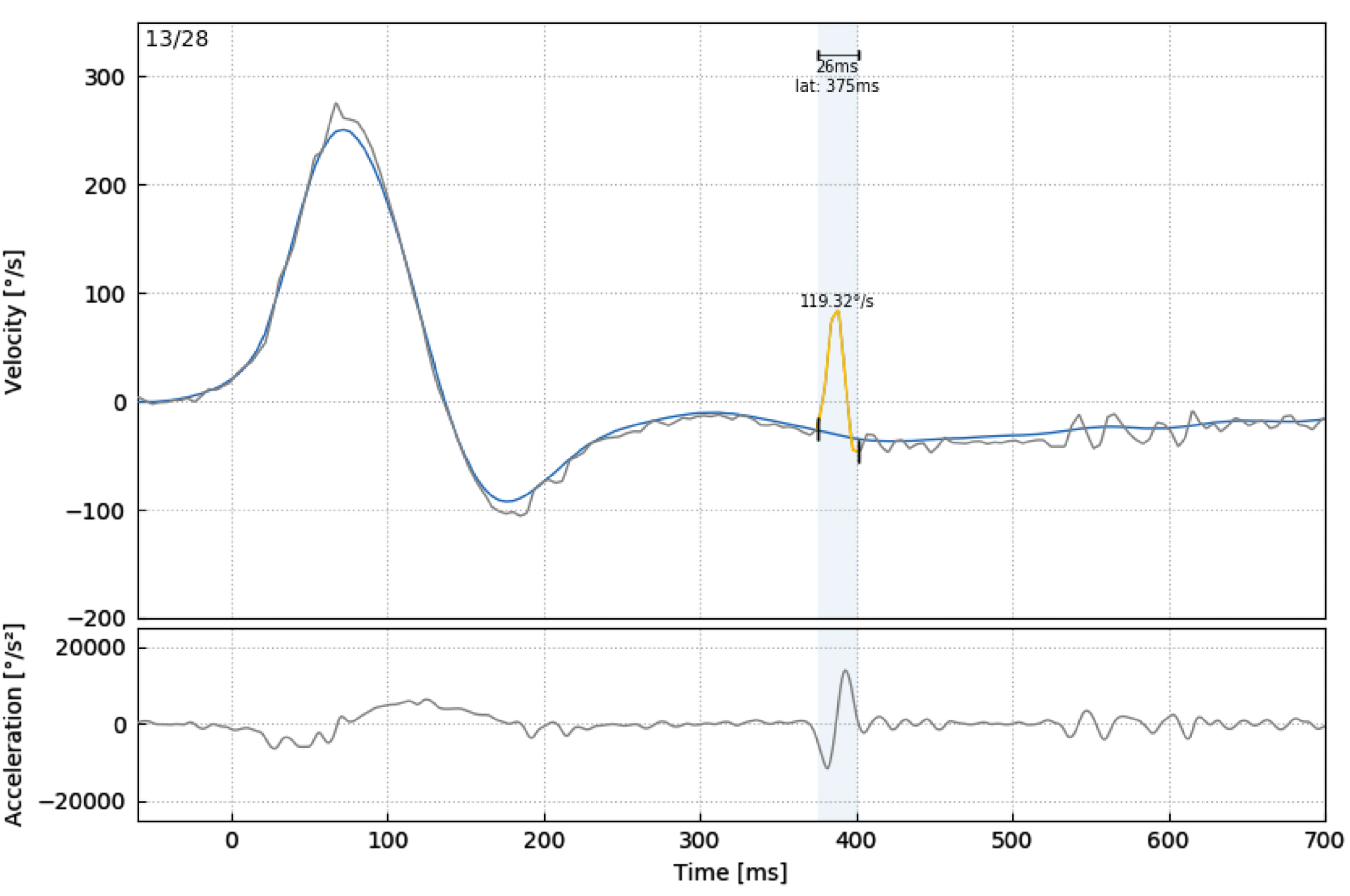
Analysis of a participant’s HIMP first saccade parameters (peak velocity (°/sec), duration (ms), and latency (ms)).
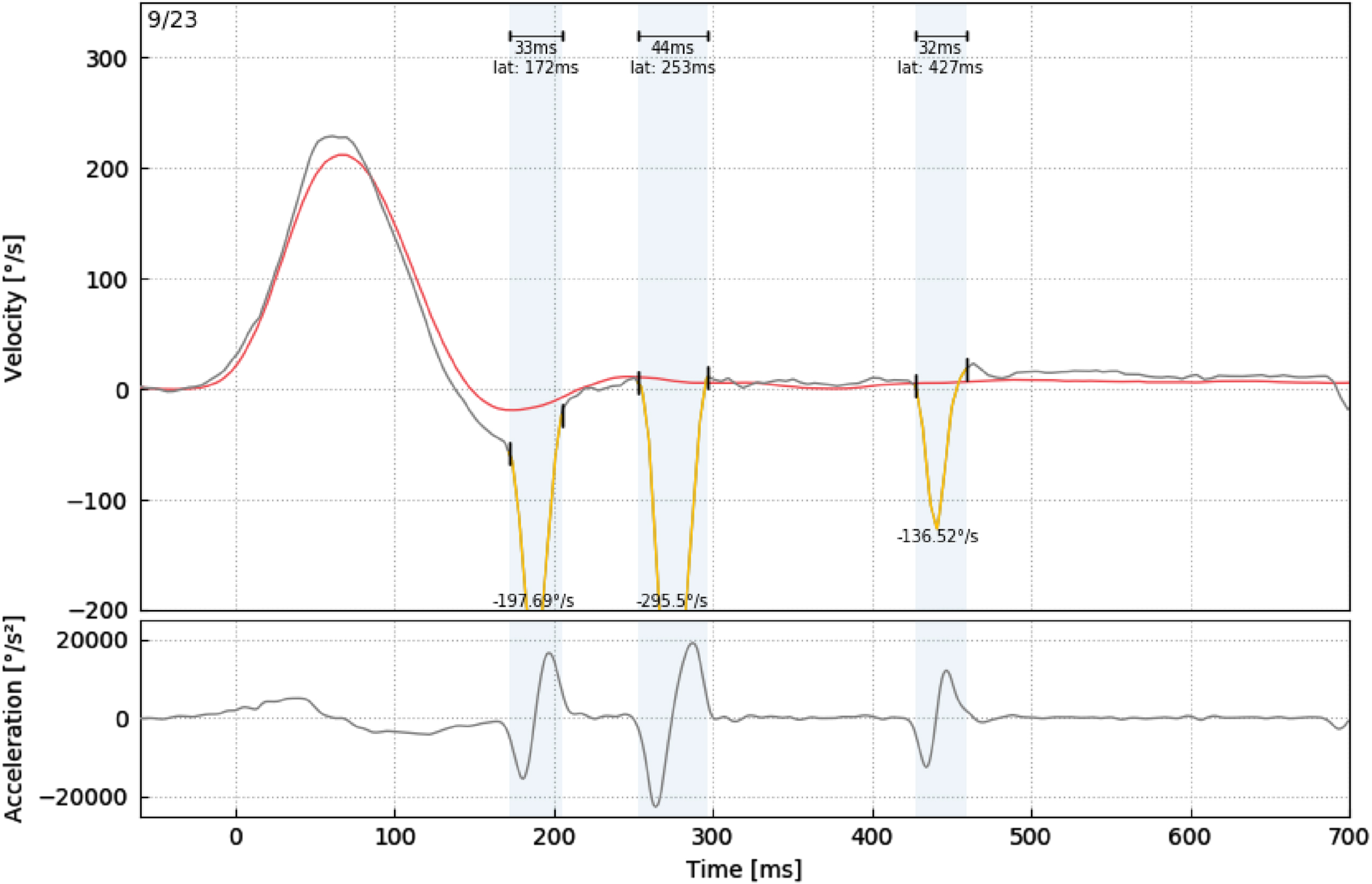
Analysis of a participant’s SHIMP 1st, 2nd, and 3rd saccade parameters (peak velocity (°/sec), duration (ms), and latency (ms)).
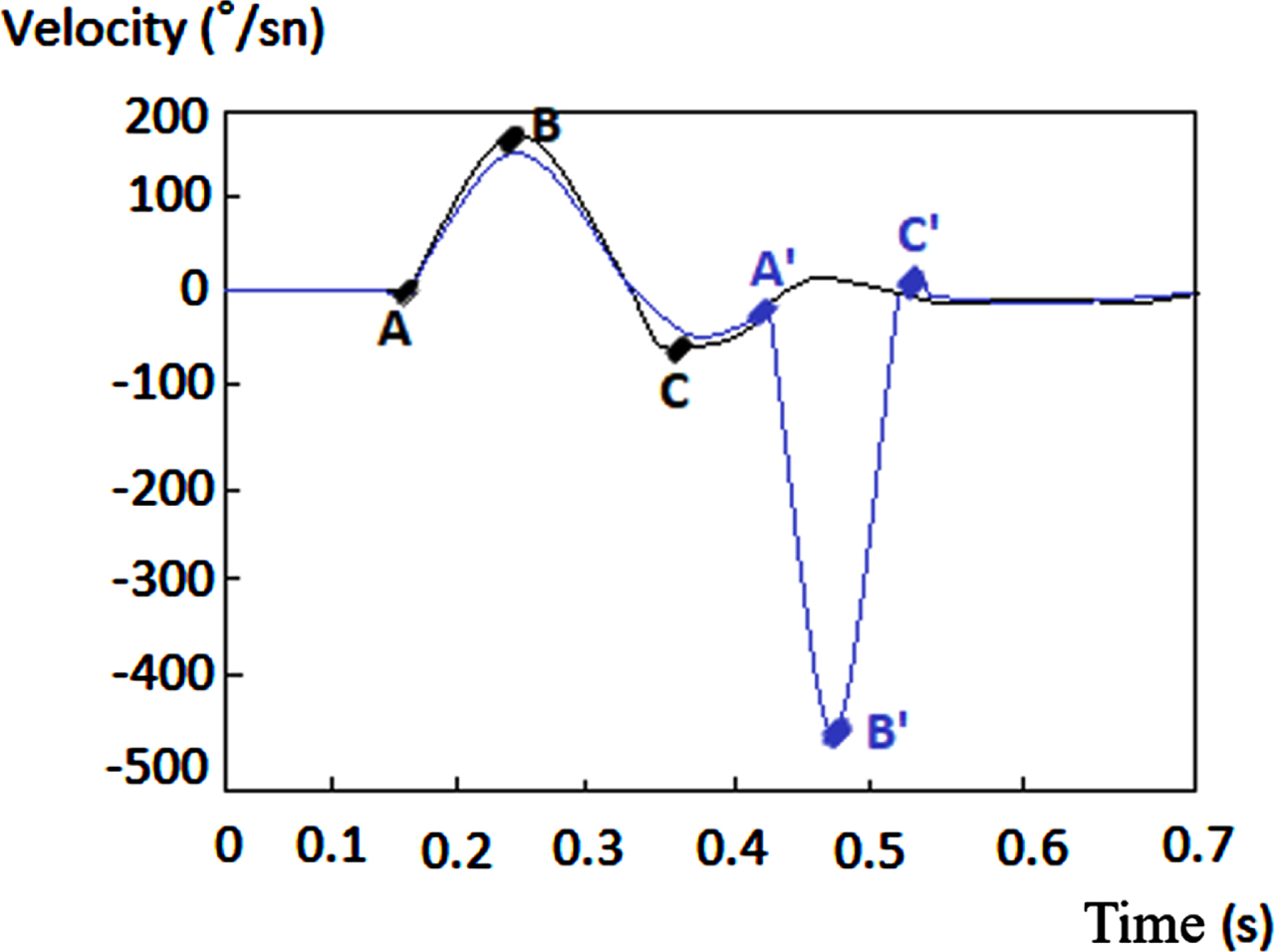
A: Onset of VOR eye movement, A’: Onset of the saccadic eye movement, B: VOR peak velocity, B’: Saccadic peak velocity C’: End of saccadic eye movement A’-C’: Saccade duration, A’-A’: Saccade latency.
To avoid spontaneous saccades or blink disturbances during head impulses, saccades with a minimum amplitude of 0.5° and occurring within the 700 ms analysis window following head impulse were included in the evaluation. Saccades that did not comply with this and were identified as artifacts were not included in the analysis. While the 1st and/or 2nd saccades occurred in the HIMP test after the head movement, the 3rd saccade was also observed in the SHIMP test in most participants.
Data analysis was performed with SPSS (Statistical Program in Social Sciences) 25 program. The Kolmogorov Smirnov Test was used to check whether the data obtained were suitable for normal distribution. Comparisons in independent paired groups were made with the Mann Whitney U test since the normality assumption was not met. Comparisons in independent multiple groups were analyzed by the Kruskal Wallis test. In consideration of the expected increase in the p-value as the number of comparisons rises in variables with detected differences, Bonferroni-corrected p-values have been utilized and calculated as ’(0.05/number of pairwise comparisons)’ [1]. After the Kruskal-Wallis test, the values obtained through the Mann-Whitney U test are compared with the calculated Bonferroni-corrected p-values to reach a decision. Pairwise comparisons were made to calculate which age groups were different in the comparison. Since the number of age groups in the study was 4 (18–25, 26–40, 41–55 and 56–65) and the number of comparisons was 2, it was calculated by
Results
The study included a total of 100 participants, 52 females and 48 males, and the mean age of participants was 39.62 years (range: 18–65 years). The participants were divided into four different age groups: 18–25, 26–40, 41–55, and 56–65 years and there were 25 participants in each age group (Table 1).
Demographic features of the participants
Demographic features of the participants
SD = standard deviation.
No significant difference was found in HIMP VOR 40 ms, 80 ms, and regression gain, and saccade parameters and SHIMP VOR 40 ms, 60 ms, 80 ms, and regression gain, saccade parameters values according to gender (p > 0.070). SHIMP 1st and 3rd saccade durations (ms) in the right ear (48.68±8.51 ms and 40.22±9.91 ms, respectively) and 1st, 2nd, and 3rd saccade durations (ms) in the left ear (49.75±9.42 ms, 41.74±6.57 ms and 37.5±7.11 ms, respectively) were significantly longer in women (p < 0.045).
Between right and left ears
Right ear HIMP VOR gain value (60 ms) and SHIMP VOR gain values (60 ms, 80 ms and Regression) were lower than those of the left ear (p≤0.001) (Table 2).
Comparison of HIMP and SHIMP VOR Gain values between right and left ears
Comparison of HIMP and SHIMP VOR Gain values between right and left ears
Mann Whitney U Test Value, p value; statistical significance, *p < 0.05; There is a statistically significant difference between the groups.
There was no statistically significant difference between age groups in the VOR gain values of HIMP and SHIMP tests (Table 3).
Comparison of HIMP and SHIMP VOR gain values by age groups
Comparison of HIMP and SHIMP VOR gain values by age groups
Kruskal Wallis Test Value, p value; statistical significance, *p < 0.05; There is a statistically significant difference between the groups.
Right ear HIMP 1st saccade amplitude (°) value had a larger amplitude in the 56–65 age group compared to the 26–40 age group (Table 4). In contrast, right ear SHIMP 2nd saccade amplitude (°) and peak velocity (°/sec) values had larger amplitude and higher peak saccade velocity in the 18–25 age group compared to the 26–40 and 41–55 age groups (Table 5).
Comparison of HIMP amplitude, peak velocity, duration and latency values between age groups
Kruskal Wallis Test Value, p value; statistical significance, *p < 0.05; There is a statistically significant difference between the groups.
Comparison of SHIMP amplitude, peak velocity, duration and latency values between age
Kruskal Wallis Test Value, p value; statistical significance, *p < 0.05; There is a statistically significant difference between the groups.
Right ear HIMP 1st saccade latency (ms) values were longer in the 56–65 age group compared to the 18–25 and 26–40 age groups (Table 4). Similarly, SHIMP 1st saccade latency (ms) values in the right and left ears were longer in the 56–65 age group compared to the 26–40 age group (Table 5). In the left ear, HIMP 1st saccade duration (ms) value was significantly longer in the 41–55 age group than in the 26–40 age group (Table 4).
Left ear SHIMP 2nd saccade duration (ms) value was significantly longer in the 18–25 age group compared to the 26–40 age group (Table 5).
Right ear SHIMP VOR gain values (60 ms, 80 ms, and Regression) were lower than those of the HIMP test. In the 1st saccade, significant differences were obtained between HIMP and SHIMP tests for all saccade parameters. Right and left ear 1st saccade amplitude (°), peak velocity (°/sec), duration (ms), and 2nd saccade duration values were significantly greater in the SHIMP test than in the HIMP test. In both ears, 1st saccade latency (ms) values were significantly longer in the HIMP test than in the SHIMP test (p≤0.001) (Table 6).
Comparison of VOR gain, amplitude, peak velocity, duration and latency values between HIMP-SHIMP tests
Comparison of VOR gain, amplitude, peak velocity, duration and latency values between HIMP-SHIMP tests
Mann Whitney Test Value, p value; statistical significance, *p < 0.05; There is a statistically significant difference between the groups.
There was a highly statistically significant positive correlation between HIMP and SHIMP tests in all VOR gain values (40 ms, 60 ms, 80 ms and Regression) (r = 0.749, r = 0.717, r = 0.728, r = 0.823, p = 0.001, respectively).
In the 1st saccade duration (r = 0.221, p = 0.027) and latency values (r = 0.297, p = 0.049) a weak positive statistically significant relationship was found between HIMP and SHIMP tests.
Discussion
The HIMP and SHIMP tests are test batteries that evaluate the lateral semicircular canal and provide quantitative data on the amplitude, peak velocity, latency, duration and interaural asymmetry of saccades that occur with VOR gain. In our study, the normalization of HIMP and SHIMP tests in terms of age groups and VOR gain and saccade parameters in the right and left ears and the correlation of the tests with each other were evaluated in healthy adults.
In studies conducted in healthy individuals, gender did not have a significant effect on HIMP VOR gain values, which is consistent with our study [20, 34]. Treviño-González et al. found that the HIMP VOR gain values (80 ms) in the right and left ears were significantly higher in men compared to women [29]. SHIMP saccade duration is the time between the beginning and the end of the saccade [17]. In our study, only the SHIMP duration value was significantly longer in women.
In previous studies, the VOR gain value was significantly higher for rightward head impulse than for leftward impulse [5–7, 34]. The gain asymmetry here depends on which eye is recorded. Weber et al. obtained a higher VOR gain value on the side of the recorded eye [31]. Differences in reflex latency occur when only one eye is recorded. For example, when the head is turned to the right, the right eye travels a greater distance to stay on target, resulting in higher ocular velocity and higher VOR gain [19–21, 29].
In some studies, hand dominance of the operator has been cited as one of the reasons for the right-left gain asymmetry, that is, right-handed users unintentionally apply more force in leftward swings, which makes the leftward head impulse faster [5, 20]. In our study, the tests were performed by the right-handed person and the left eye was recorded. As a result, the left VOR gain value was higher than the right (HIMP VOR 60 ms and SHIMP VOR 60 ms, 80 ms and Regression). This suggests that the main factor in directional gain asymmetry is the eye being recorded.
In our study, there was no statistically significant difference between right and left ears in amplitude, peak velocity, duration, and latency parameters of the HIMP and SHIMP tests. In previous studies, no significant difference was observed between the right and left ears in peak saccade velocity, which is consistent with our study [6, 16]. Unlike our study, there are also studies that have obtained significantly different findings in saccade parameters between right and left ears. On the other hand, in some studies, saccade amplitude has been found to be larger, peak velocity faster and with earlier latency [6, 24]. Janky et al., on the other hand, obtained a longer HIMP latency value in the right ear and stated that this was possibly due to the longer nerve pathway. They suggested that saccade latency would affect its sensitivity in the diagnosis of unilateral vestibular loss [12].
Consistent with the literature, in our study, HIMP and SHIMP VOR gains were not affected by aging and no significant difference was observed between age groups [12, 34]. In some studies, horizontal canal VOR gain was significantly decreased in individuals over 70 years of age [11, 26]. Even if there is a loss of vestibular receptor cells and nerves with aging, it is thought that the functional vestibular eye reflex is relatively preserved, especially until the age of 70, by being controlled by the cerebellum [13].
Consistent with our study, previous studies have reported that aging significantly increased the lateral canal HIMP saccade amplitude, albeit slightly [3, 24]. It is thought that the observation of larger amplitude saccades in elderly individuals may be due to impaired saccade generation mechanism with decreased VOR function or incompatible interaction between paramedian pontine reticular formation and vestibular nuclei [4]. In addition, the effect of aging on SHIMP amplitude was also examined in our study and SHIMP 2nd saccade amplitude value in the right ear was higher in the youngest age group unlike HIMP 1st. Sayed et al. found that SHIMP saccade amplitude decreased with aging in healthy subjects [15].
In our study, HIMP and SHIMP 1st saccade latency values were found to be longer with aging. In contrast to these findings, other studies have not found a significant difference in saccade latency according to age and they thought that compensatory saccade latency is a learned behavior [3, 27].
Consistent with previous studies, no significant difference was found between age groups in terms of the HIMP peak saccade velocity [12, 33]. Pogson et al. found a small increase in HIMP peak velocity in the elderly group, which they thought might be due to the fact that they did not include non-saccadic impulse in the analysis [24]. In our study, the SHIMP 2nd saccade peak velocity was found to be significantly faster in the youngest age group. Lee and Kim found that SHIMP peak saccade velocity decreased with increasing age of above 70 years [16]. Although there is no study evaluating saccade duration in the literature, in our study, HIMP 1st saccade duration was longer and SHIMP 2nd saccade duration was shorter with increasing age.
Consistent with our study, SHIMP VOR gain was significantly lower than HIMP VOR gain in studies in patient groups and healthy controls, and a significant correlation was obtained between the tests [6, 27]. One reason for the lower SHIMP VOR gain is that a delay in VOR suppression on the visual target was observed during high-speed head impulse. Another reason could be that VOR inhibition is a contributing factor to the SHIMP test [16, 25].
In the studies, while VOR gain values between HIMP and SHIMP tests were compared, saccade parameters were explained to determine the normal value range. Because compensatory and anti-compensatory saccades are not directly related to each other, there was no need for comparison [26]. Since the logic of HIMP and SHIMP tests is different, we expect a difference in their results. We think that the comparison of HIMP-SHIMP saccade parameters has an important place in terms of revealing the findings supporting this difference and showing the differences between the tests.
In our study, the amplitudes of anti-compensatory saccades in the SHIMP test were larger than compensatory saccades in the HIMP test. Therefore, it is thought that it will be easier to observe changes in anti-compensatory saccades and detect pathologic VOR with the SHIMP test [26]. Saccade amplitude was found to be a highly sensitive and specific marker for distinguishing between patients and controls in the study by Macdougall et al. [18]. However, the HIMP test cannot discriminate between the saccade amplitudes of the normal ears of patients with unilateral vestibular loss and the saccade amplitudes of healthy individuals, whereas the SHIMP test can [18]. In future studies, research on sensitivity and specificity between patient and healthy groups can be further enhanced. Macdougall et al. found that the latency of SHIMP saccades was significantly longer than HIMP saccades in patients with bilateral vestibulopathy. Therefore, they emphasized the importance of performing SHIMP test in patients with vestibular loss [18].
In our study, no significant correlation was found in the saccade amplitude of HIMP and SHIMP tests. Roh et al. interpreted this as the presence of covert saccades that may decrease the amplitude of overt saccades [26]. In our study, on the other hand, a weak positive and statistically significant correlation was found between HIMP and SHIMP tests in the first saccade duration and latency values. Janky et al. found that the group with vestibular pathology had a significantly lower mean vHIT gain, higher mean corrective saccade frequency and saccade peak velocity, and earlier saccade latency compared to the control group. Therefore, it is emphasized that the interpretation of corrective saccades in vHIT together with the gain parameter in the determination of vestibular pathology will increase the sensitivity of the test [12].
Conclusion
Since the saccade parameters in HIMP-SHIMP tests showed a significant difference with aging in our study, it is recommended that age range should be kept wider in future studies. Our study contributes to the literature in the sense that normative values of all saccade parameters in HIMP and SHIMP tests have not been examined and these two tests have not been compared previously. Future studies should investigate the clinical outcomes of these parameters in vestibular pathologies.
We think that the interpretation of corrective saccades together with the gain parameter in the determination of vestibular pathology will increase the sensitivity of the test and the accuracy of the vestibular diagnosis and therefore more studies should be conducted.
Since the saccades obtained in the SHIMP test have the potential to detect residual vestibular function, further studies on its usability in vestibular rehabilitation are recommended.
Informed Consent
Written informed consent was obtained from all participants who participated in this study.
Ethics Committee Approval
Ethics committee approval of the study was obtained from Ege University Medical Faculty Medical Research Ethics Committee with the decision number 21-12T/6 on 02.12.2021.
Footnotes
Acknowledgments
This manuscript is produced from the Audiology and Speech Disorders MSc thesis of Suheda Baran under the first thesis supervision of Prof.Dr. Tayfun Kirazli, M.D. and the second thesis supervision of Asst.Prof. Gulce Kirazli, Ph.D.
