Abstract
BACKGROUND:
Alternative splicing is a mechanism to produce different proteins with diverse functions from one gene. Many splicing factors play an important role in cancer progression. PRPF8 is a core protein component of the spliceosome complex, U4/U6-U5 tri-snRNP.
OBJECTIVE:
However, PRPF8 involved in mRNA alternative splicing are rarely included in the prognosis.
METHODS:
We found that PRPF8 was expressed in all examined cancer types. Further analyses found that PRPF8 expression was significantly different between the breast cancer and paracancerous tissues.
RESULTS:
Survival analyses showed that PRPF8-high patients had a poor prognosis, and the expression of PRPF8 is associated with distant metastasis-free survival (DMFS) and post progression survival (PPS). Gene Set Enrichment Analysis (GSEA) has revealed that PRPF8 expression is correlated with TGF-
CONCLUSIONS:
These results have revealed that PRPF8 is a significant factor for splicing in breast cancer progression.
Introduction
Breast cancer is a common cancer, which has a higher morbidity rate in women [1]. The clinical outcomes of breast cancer are closely linked to prognostic parameters, such as tumor size, grade, and lymph node and metastasis status. However, genes involved in mRNA alternative splicing are rarely included in the prognosis. By generating multiple mRNAs from a precursor mRNA (pre-mRNA), alternative splicing greatly diversify the genome coding capacity. Most genes are multiple-exon genes and generate more than one functional protein.
Recent studies have confirmed multiple splicing factors affect the splicing of critical breast cancer-related genes [2]. PRPF8 is a core protein component of the spliceosome complex, U4/U6-U5 tri-snRNP and contains several WD repeats, which function in protein-protein interactions. It participates in the two sequential transesterification steps of pre-mRNAs during the cut and link of pre-mRNAs. Loss of PRPF8 can lead to the death of mouse embryonic cells [3] and Drosophila cells [4]. Thus, the study aimed to investigate the significance of PRPF8 in breast cancer. Our present study, the data of Oncomine and TCGA (The Cancer Genome Atlas) were used to analyze the expression levels of PRPF8 in normal tissue and carcinomas. The clinical significance of PRPF8 in breast cancer was further explored. GSEA analysis and in vitro experiments have disclosed the possible role of PRPF8 in breast cancer.
Materials and methods
Bioinformatic and statistical analysis
The expression of PRPF8 in different cancer types, between cancer samples and adjacent tissues, is analyzed from GEO and Oncomine database (
Cell culture
MCF-7 and HEK-293T cells were obtained from the National Infrastructure of Cell Line Resource. MCF-7 cells were cultured in
The shRNA sequences
The shRNA sequences
Viral packaging vectors of pMD2 (Addgene, USA) and pPAX2 (Addgene, USA) and pGIPZ (Addgene, USA) were transfected into 293T cells with Lipofectamine 2000. The shRNA sequences were listed (Table 1), for pGIPZ. After 3 days, the medium was purified with 0.45
Primers used for quantitative PCR
Primers used for quantitative PCR
500 MCF-7 cells per well were grown in six-well plates and maintained at 37
(A) The mRNA expression of PRPF8 in different cancer cell lines. (B) The protein levels of PRPF8 are higher in breast cancer than in adjacent normal tissue. (C) PRPF8 expression in the subtypes of breast cancer.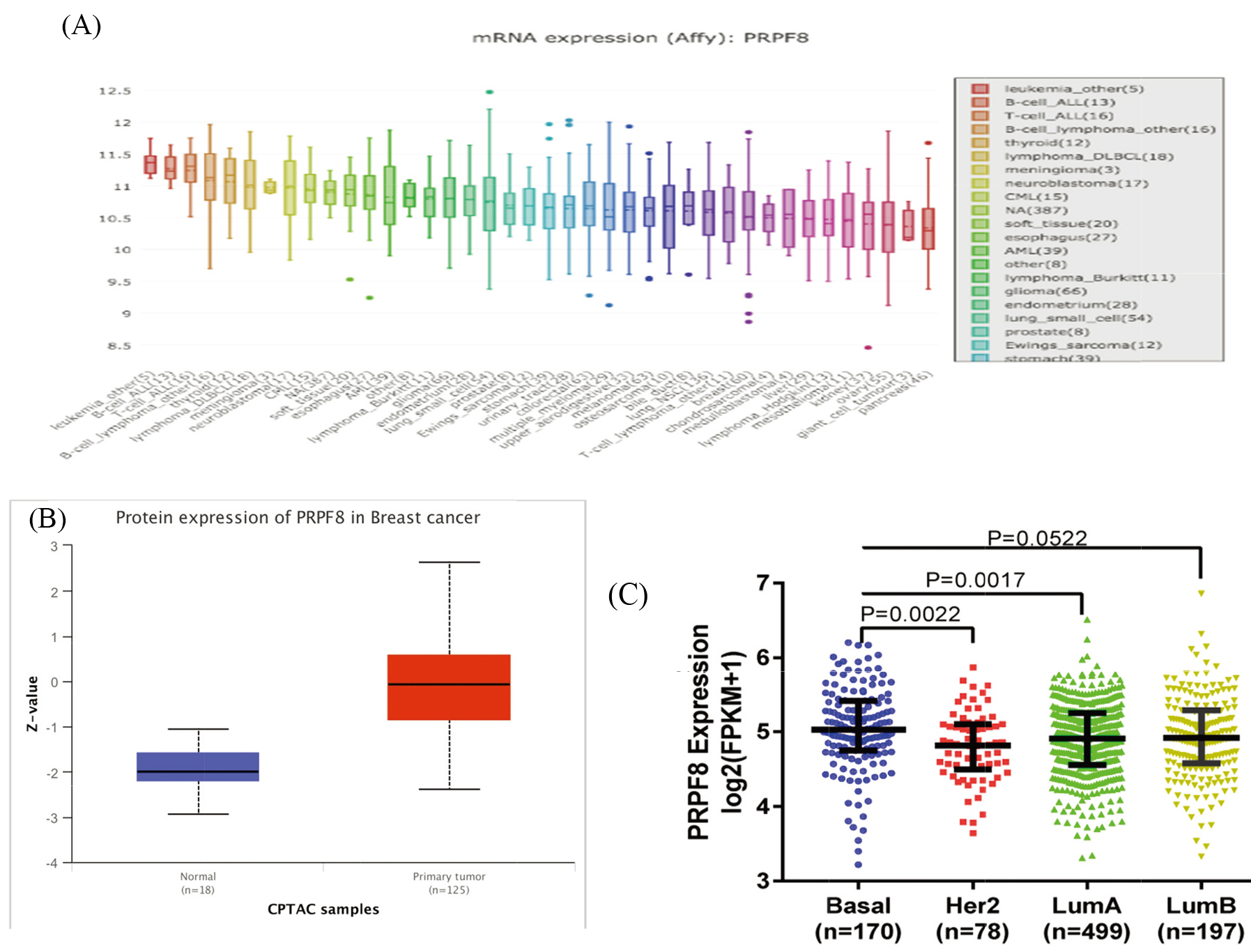
(A) PRPF8 protein levels are associated with the OS of breast cancer patients in Kaplan-Meier plots. (B) The mRNA expression of PRPF8 is associated with the DMFS probabilities of breast patients. (C) The mRNA expression of PRPF8 is associated with the PPS probabilities of breast patients. (D) Prognostic analysis of PRPF8 with mRNA expression in luminal A patients. 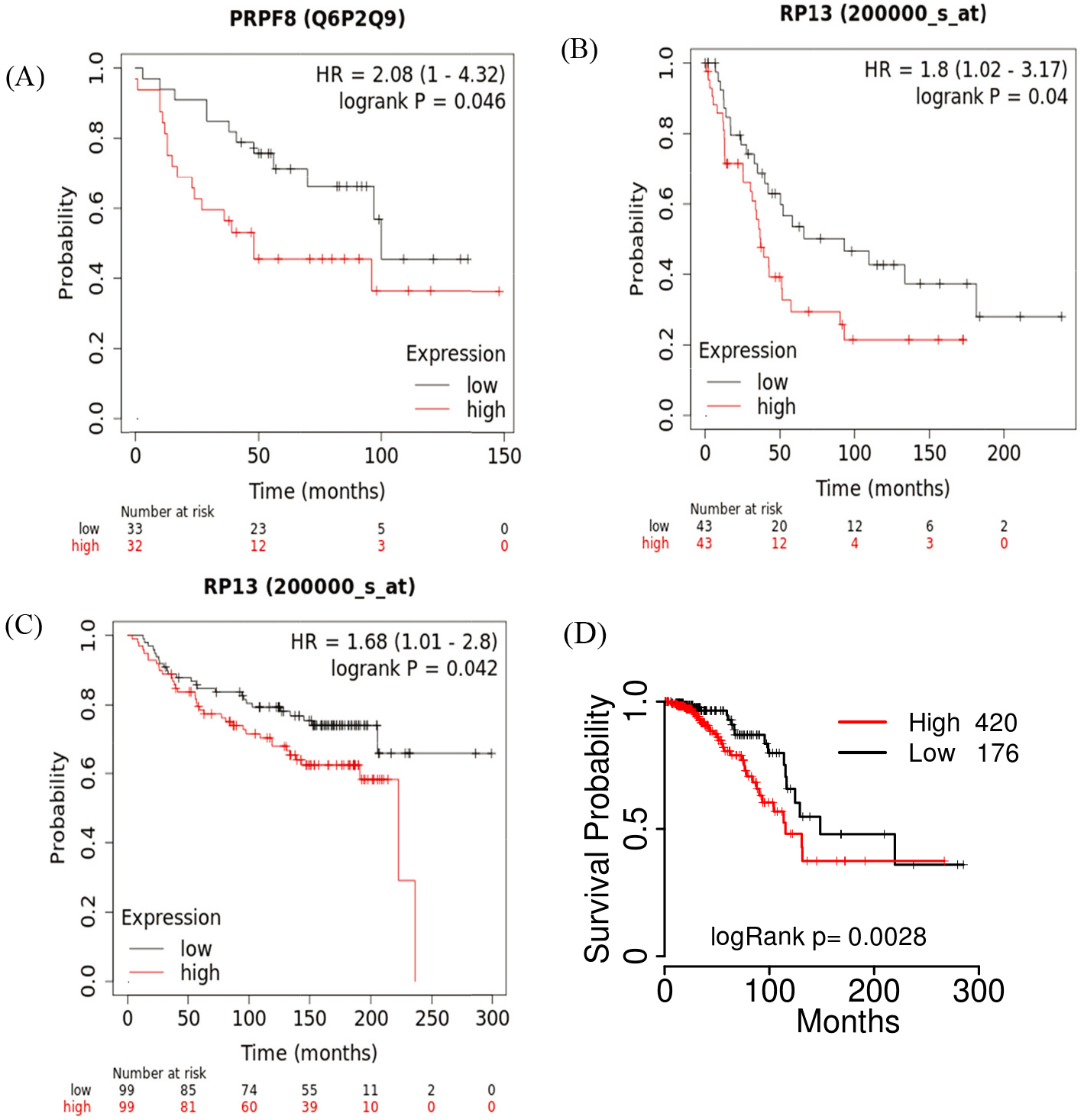
Characterizing the expression of PRPF8 in breast cancer
There are few reports on the expression of PRPF8 in breast cancer. In this study we analyzed the expression levels of PRPF8 in various human tumors from the Oncomine database and Cancer Cell Line Encyclopedia (CCLE) (fold change of
PRPF8 expression is associated with the survival of breast cancer patients
Subsequently, we investigated the association of PRPF8 expression with breast cancer patient survival. In breast cancer, the PRPF8 protein level was associated with the overall survival (OS) according to Kaplan-Meier plots database. Consistent with this result, PRPF8 mRNA expression is also associated with the OS in the Kaplan-Meier plots cohort. In the cohort of (GSE7390), we also found that PRPF8 expression was associated with the PPS (
Inhibition of PRPF8 expression impaired breast cancer cell proliferation
To confirm the function of the PRPF8 in breast cancer, we employed shRNAs to silence PRPF8 expression in breast cancer cells, MCF-7. Western Blot (WB) shown that both shRNAs suppressed the expression of PRPF8 (Fig. 3A). Meanwhile, inhibiting PRPF8 expression, the numbers of colonies decreased (Fig. 3C and D), and the growth curves were significantly inhibited, suggesting that PRPF8 was essential for the maintenance of the proliferation in cancer cells (Fig. 3B).
(A) WB shown the inhibition of PRPF8 shRNA in MCF-7 cells. (B) MCF-7 cells growth curves by transduced with PRPF8 different shRNAs. (C) The images of MCF-7 colony formation, which were transduced with different shRNAs. (D) The numbers of colonies after MCF-7 cells were transduced with different shRNAs.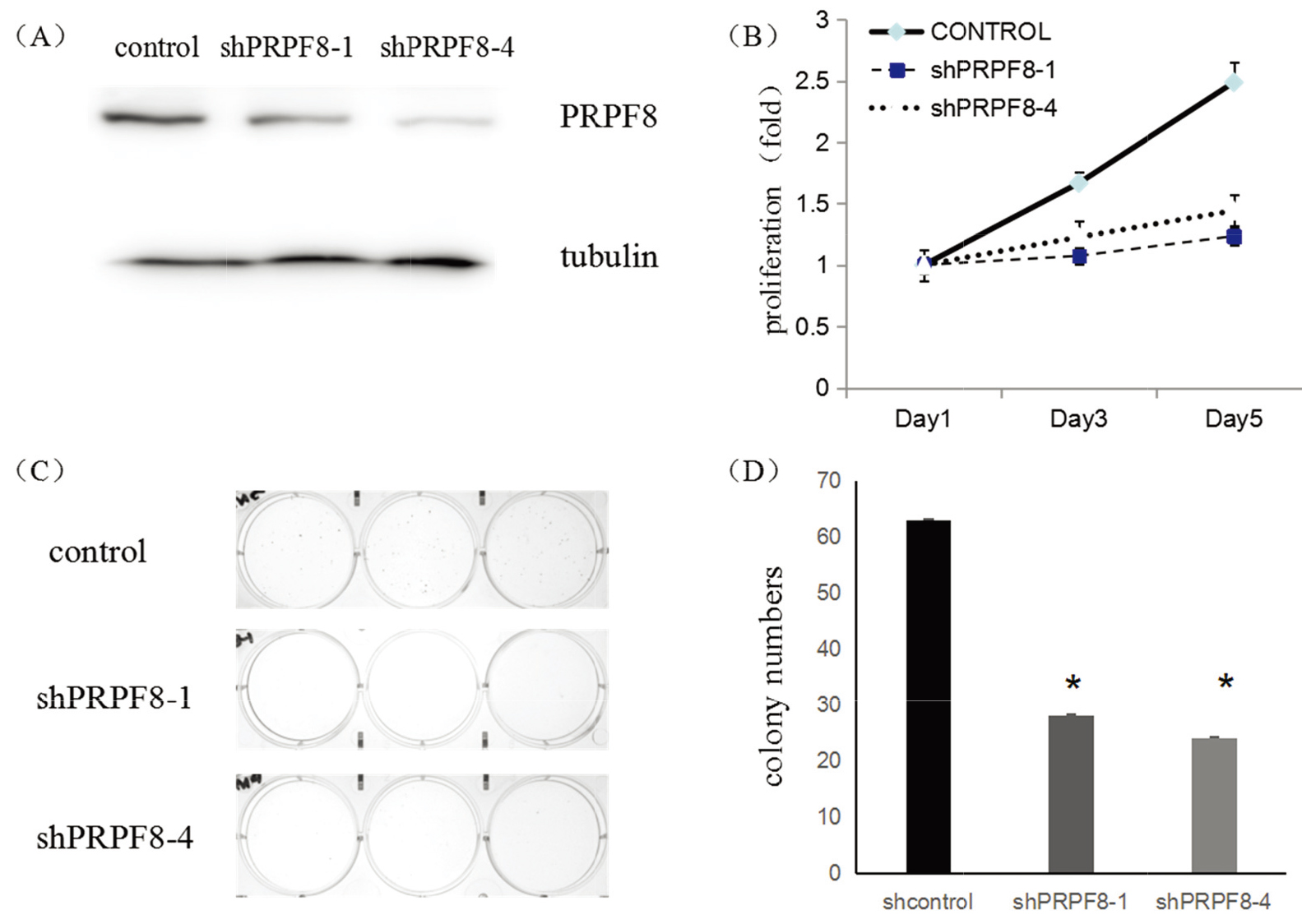
To further explore the functional mechanism of PRPF8 in the breast cancer cell, we used GSEA to analyze the pathways associated with PRPF8 in breast cancer. The results have shown that PRPF8 positively regulates 153 pathways and negatively regulated 31 pathways. The high expression of PRPF8 was correlated with JAK-STAT signaling pathway (ES
(A) GSEA results show TGF-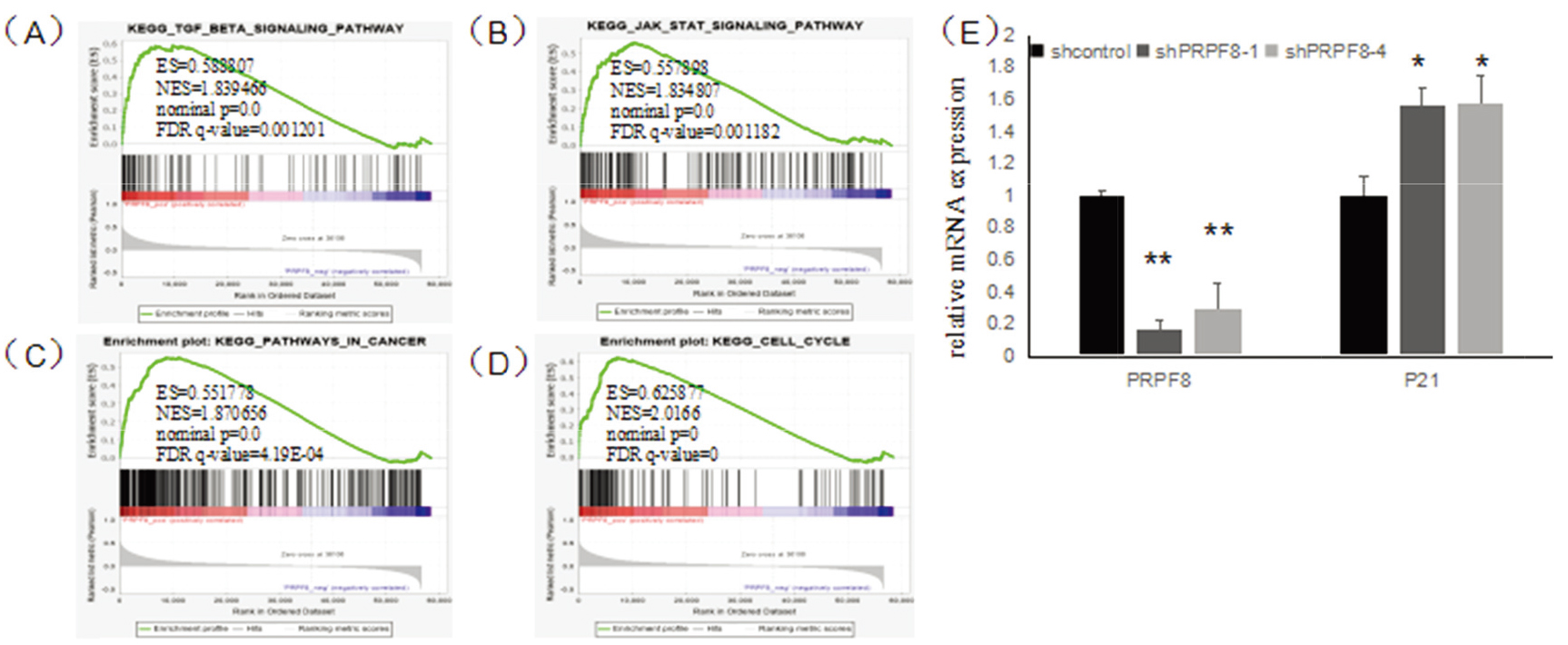
In this study, we investigate the role of PRPF8 in breast cancer. PRPF8 mRNA expression is significantly elevated in breast cancer samples compared with the paracancerous tissue. PRPF8 mRNA is differentially expressed among different breast cancer molecular subtypes, and its levels were inversely correlated with the OS in breast cancer patients. Furthermore, we confirmed the role of PRPF8 in breast cancer with in vitro experiments, which have shown that silencing PRPF8 in breast cancer cells repressed cell proliferation by upregulating
Alternative splicing is one of the mechanisms to increase protein diversity [5, 6, 7, 8, 9]. Recently, with the better understanding of alternative splicing process [10, 11, 12, 13, 14, 15], it has been found that abnormal expression of splicing factors is closely related to many diseases. Many splicing factors play an important role in cancer [16, 17, 18, 19, 20, 21, 22, 23], including in breast cancer [24, 25, 26]. PRPF8 is the core component of the ribonucleoprotein (RNP) complexes in the spliceosome and participates in splice-site recognition, branch-point formation and catalysis process [27, 28, 29]. Whether PRPF8 plays a role in breast cancer is not known.
In this study, we demonstrate that PRPF8 is critical for breast cancer cell survival. Firstly, PRPF8 is elevated in breast tumors compared with the normal tissue (Fig. 1B). Second, PRPF8 was related to OS, PPS, and DMFS in breast cancer patients (Fig. 2A, B&C). More importantly, silencing of PRPF8 slowed down breast cancer cell growth and reduced the colony formation of MCF-7 cells (Fig. 3). Therefore, we found PRPF8 plays an important role in breast cancer.
Conclusion
Our study provides evidence that splicing factor PRPF8 is critical for breast cancer cell survival and has the potential prognostic value in breast cancer. PRPF8 may achieve its functions in breast cancer by modulating
Footnotes
Conflict of interest
The authors report no conflict of interest.
Funding
This work was supported by funds from the Natural Science Foundation of Heilongjiang Province (Grant Number H2017039).
