Abstract
OBJECTIVE:
This study aimed to identify clinical measures that have been used to evaluate function, health related quality of life (HRQoL), and/or satisfaction in children who use lower limb prostheses (LLP). The data reported on psychometric properties for children who use LLP were collected for each measure.
METHODS:
First, PubMed, CINAHL, and Web of Science databases were searched using broad search terms to identify standardized outcome measures of function, HRQoL, and/or satisfaction with treatment used in pediatric LLP research published in 2001 or after. For each of the eligible measures found, a second search was performed to identify psychometric properties (e.g., validity, reliability) assessed with children who use LLP.
RESULTS:
Forty-four standardized outcome measures were identified from 41 pediatric LLP research articles. Five measures (i.e., Gait Outcomes Assessment for Lower Limb Differences, Functional Mobility Assessment, Child Amputee Prosthetics Project- Prosthesis Satisfaction Inventory, Child Amputee Prosthetics Project- Functional Scale Index, and Lower Limb Function Questionnaire) had data on psychometric properties for children who use LLP.
CONCLUSIONS:
Few studies report psychometric data for assessing the overall HRQoL, function, and/or satisfaction for children who use LLP. Further research is needed to validate or create new outcome measures that assess the HRQoL, satisfaction, and/or function of children who use LLP.
Introduction
Approximately 1.6 million people are living with limb loss or limb difference (LLLD) in the United States [1]. Children with LLLD account for 1.6% of this total, or about 25,000 individuals [1]. The rapid growth and development of children necessitates that the treatment plan and componentry of prostheses be routinely and longitudinally evaluated. Outcome measures provide clinicians with information that is essential to this evaluation process. Not only do outcome measures inform the medical team, patient, and caregiver of changes in function, but they may also aid in determining prosthetic componentry.
Outcome measures are standardized instruments that collect observable information on function, satisfaction, or health related quality of life (HRQoL) constructs through patient reported, parent-proxy reported, or clinician-assessed information. The rapid change and development of children’s language and physical capabilities highlight the need for outcome measures to be psychometrically examined before use in children who use lower limb prostheses (LLP). Several validated outcome measures exist for adult LLP users [2], but it is unknown how many exist for children who use LLP. A lack of psychometrically-sound outcomes measures prevents rapid progress in understanding treatment outcomes in children with LLP.
Due to the lack of population-specific outcome measures, clinicians and researchers are faced with having to decide on the suitability of the outcome measure for their pediatric patients with LLP. This lack also likely results in practitioners using a measure that is not validated for pediatrics or not validated in LLP users, or not using a measure at all. Consequently, there is the potential for misinterpretation of outcomes, use of instruments that are not appropriate for children or not sensitive to change in children who use LLP, or a lack of routine standardized outcome measurement for these patients. Clinicians and researchers could benefit from a resource to aid in choosing outcome measures to assess their pediatric LLP patients.
The purpose of this review was (1) to identify standardized outcome measures that measure function, HRQoL, and/or treatment satisfaction in children who use LLP, and (2) to report on the availability of data on the reliability, validity, and responsiveness of those measures. Scoping review methods were undertaken for this work because it involved a broad investigation of the literature and identification of research gaps. In addition, outcomes measures for children who use LLP are an emerging research topic. As such, there are not enough studies reporting data on these measures to inform the certainty of evidence related to their psychometric properties. This scoping review instead aimed to provide groundwork for future research on outcome measure refinement, use, and development in this patient population. It may also serve as a resource to aid clinicians and researchers when selecting among available measures to assess outcomes in their pediatric LLP patients.
Methods
Overview
A scoping literature review was conducted based on methods described by Peters and colleagues [3–5] and followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR) reporting guidelines. The protocol for this scoping review was not registered.
Information sources
A search of PubMed, Cumulated Index to Nursing and Allied Health Literature (CINAHL), and Web of Science was performed on August 4, 2023, to identify a list of outcome measures used in published research with children using LLP. Using the same databases, secondary searches were performed to find published articles describing psychometric assessment(s) of each identified outcome measure with children who use LLP.
Search strategies
To find pediatric LLP research publications, the author team developed search strategies for each database using terms related to amputation, prostheses, and pediatrics (Appendix 1). Following the extraction of outcome measures from eligible articles, secondary search strategies were developed to find published data about psychometric properties for each outcome measure in children who use LLP. Search terms for the secondary search included the name of the outcome measure, terms related to psychometric properties, and terms related to prostheses and pediatrics (Appendix 2). Eligible articles from the secondary search were subjected to forward and backward citation searches to ensure that all published articles with psychometric data for each measure of interest were identified. Results from primary and secondary searches were exported to an Endnote (Clarivate Analytics, Philadelphia, PA) library, and duplicate publications were removed.
Selection
Following both the primary (to identify outcome measures used in pediatric LLP research) and the secondary (to identify published data on psychometric properties for each outcome measure) searches, two authors independently screened search results based on title, abstract, and full text review. Articles deemed eligible by at least one reviewer during title and abstract reviews were included in the next round. If the two reviewers did not reach consensus for eligibility in the full text review round, a third, independent reviewer was consulted. A forward and backward search of each included article’s references was conducted to ensure no eligible articles were missed. However, no additional articles were found during this search.
Eligibility criteria
For the primary search of pediatric LLP research, article eligibility criteria included (1) participants were children (<18 years old) who used LLP and had amputation or limb loss at or above the ankle, (2) researchers used standardized outcome measure(s) that assessed function, HRQoL, and/or satisfaction with treatment, and (3) the article was original research or a research protocol published in 2001 or after.
For the secondary search to identify psychometric properties in pediatric LLP samples, article eligibility criteria included (1) participants were children (<18 years old) who used LLP and had amputation or limb loss at or above the ankle, and (2) the article reported psychometric properties for one or more of the outcome measures identified in the primary search. Articles identified in either the primary or secondary rounds were excluded if they did not include human participants or were only published as a poster or abstract.
Data extraction and synthesis of results
Data from articles deemed eligible in the primary search of pediatric LLP research were extracted into an Excel spreadsheet (Microsoft, Redmond, WA) by a single reviewer and checked for accuracy by a second reviewer. Extracted data from primary round articles included the outcome measures used in each study, as well as study characteristics (e.g., sample size, age range, level of amputation, and study design) to characterize the body of research in pediatric LLP. Data extraction forms were tested and edited prior to use.
Data extracted from eligible articles that assessed measurement properties in samples of children who use LLP included characteristics of the studies (e.g., sample size, age range, level of amputation, and study design) and information relating to the report of the measurement’s psychometric properties. Extracted data were checked for accuracy by a second independent reviewer and summarized in tables. Study characteristics, such as demographic information, were collected to assess the generalizability of psychometric data for measurement of children who use LLP. Psychometric data in children who use LLP, when available, were collected. Examples of articles that assess psychometric properties in related populations (e.g., people who use LLP or children) were noted, but details about these studies were not extracted as this information was outside of the scope of this article.
Deviations from the a priori protocol
Methods deviated from the a priori protocol in article eligibility criteria. Specifically, non-English articles, gray literature, and research protocols were originally excluded but ultimately included in the final publication in response to reviewer suggestions.
Results
Outcome measures used in pediatric prosthetic research publications
The primary search identified 4,387 articles after removing duplicates (Fig. 1). A total of 44 different standardized outcome measures were used to assess children who used LLP across the 41 articles that met eligibility criteria. Collectively, the standardized outcome measures assessed function (19 measures), HRQoL (13 measures), or satisfaction (1 measure) or were multi-domain instruments (11 measures) that included assessments of function, HRQoL, or satisfaction as one of the subdomains (Table 1). Over 80% of the standardized measures used in the 20-year period of this review were described in only one or two research publications each.
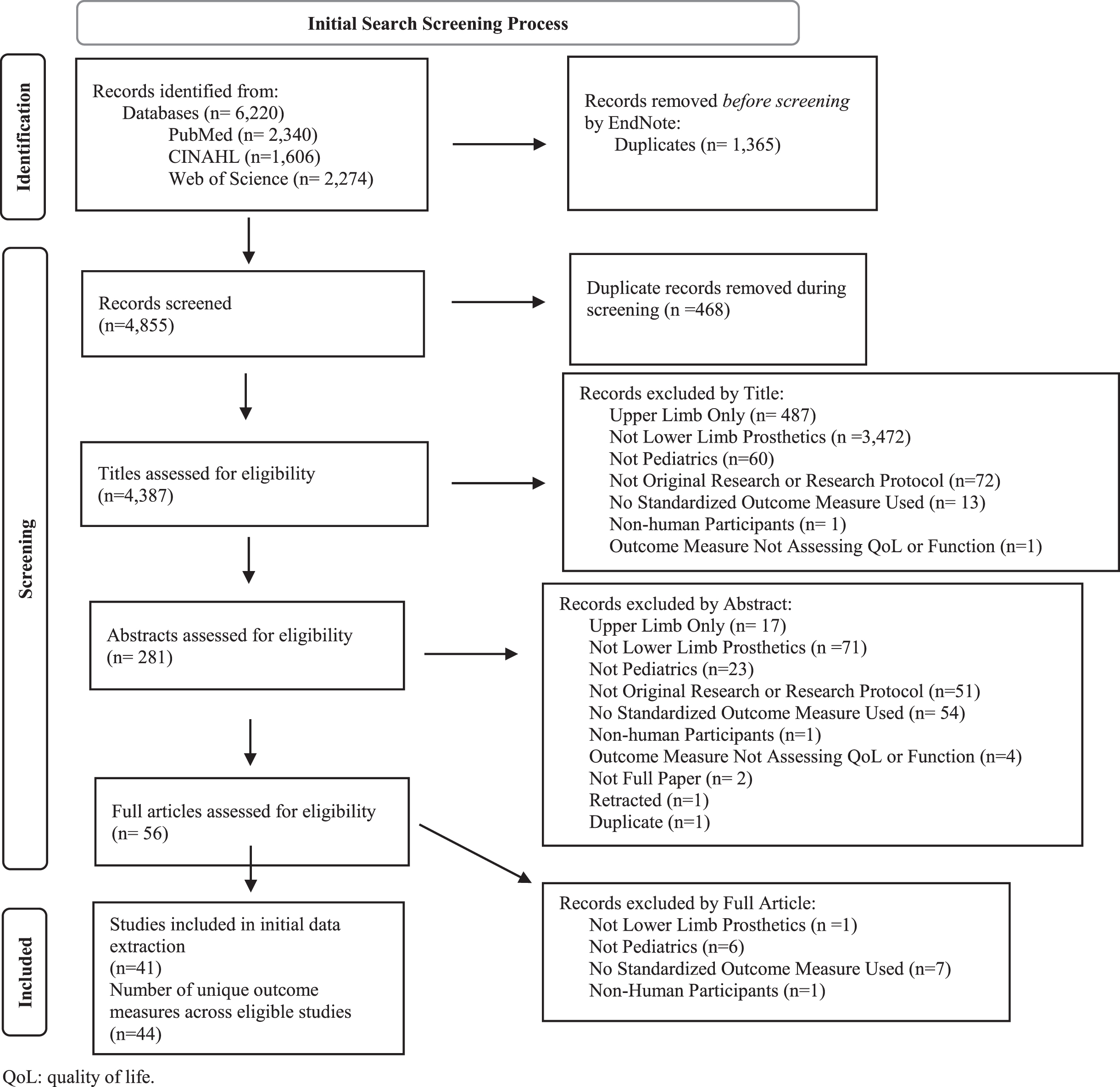
PRISMA 2020 Flow Diagram [98] for the primary search. The aim of the primary search was to identify outcome measures that were used in pediatric lower limb prosthetics research from 2001-present. The primary search resulted in the identification of 41 studies that described use of 44 unique outcome measures.
Outcome measures identified in pediatric lower limb prosthetics (LLP) publications and the populations for whom psychometric data is available
Secondary searches identified publications that reported on the psychometric properties of outcome measures extracted from articles found in the primary search. These secondary searches resulted in a total of 104 articles after removing duplicates. Following title and abstract review, 10 articles were retained for full-text review. Five articles examining five separate outcome measures met eligibility criteria and were included in the full data extraction (Fig. 2, Table 2). Measure and psychometric properties of the five measures are summarized below.
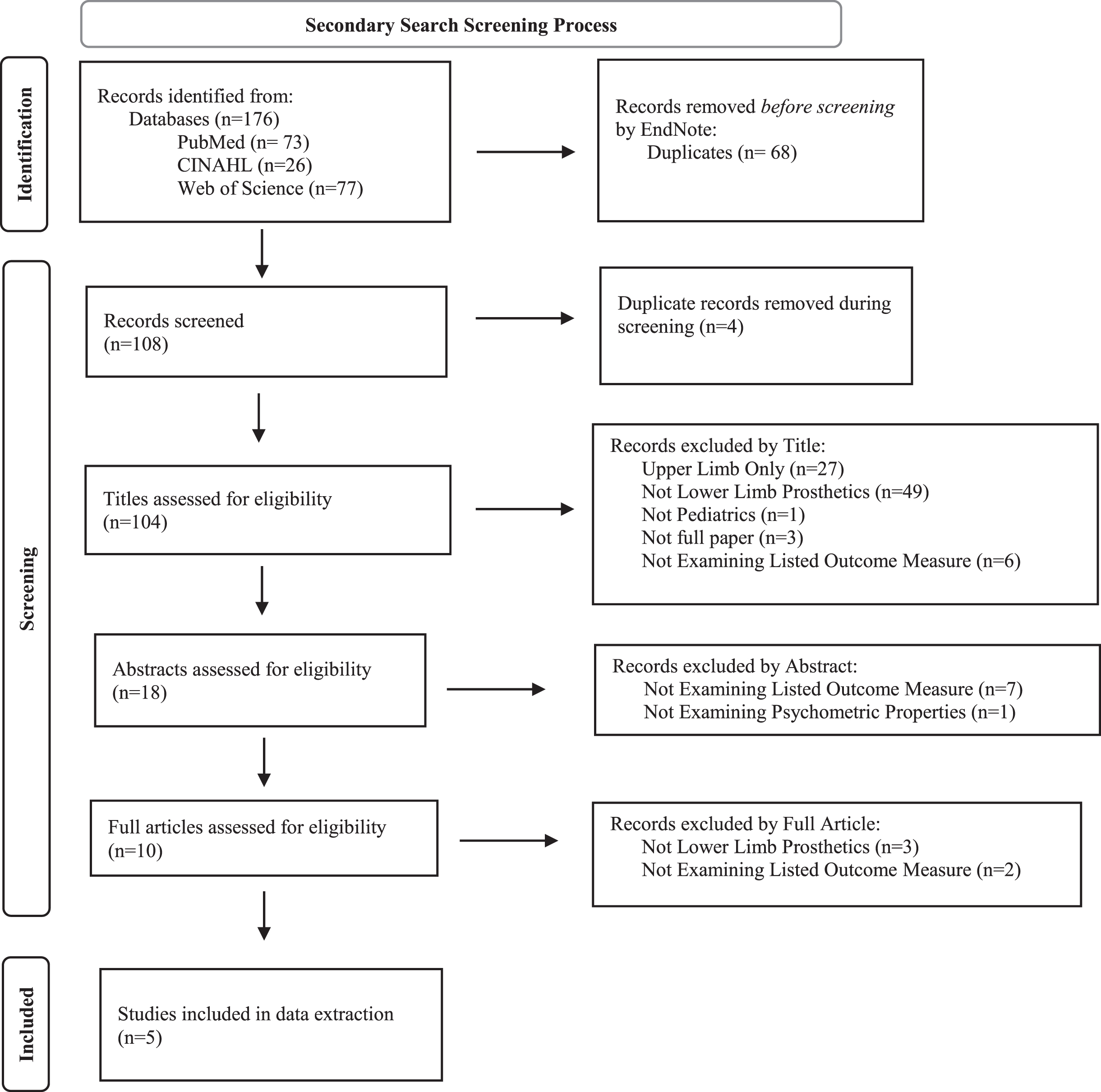
PRISMA 2020 Flow Diagram [98] for the secondary search. The aim of the secondary search was to identify articles that describe the psychometric assessment of outcome measures in children who use lower limb prostheses. The secondary search resulted in the identification of five eligible articles that described psychometric properties for five unique outcome measures.
Study characteristics for publications that assessed psychometric properties of outcome measures for pediatric participants who use lower limb prostheses (LLP)
aFunding provided by the Government of Ontario, the Canadian Institutes of Health Research, the University of Toronto Open Fellowship, SickKids Continuing Professional Development Fund, and Holland Bloorview Graduate Student Support. bFunding source not reported. cFunding provided by grants from the Shriners Hospitals for Crippled Children Research Fund and the Milo Brooks Foundation for Limb Deficient Children. dFunding provided through “private fundraising by individual researchers.”
One article [6] examined psychometric properties of the GOAL-LD, a patient-reported outcome measure assessing six domains including gait function/mobility, pain/discomfort/fatigue, physical activities/games/recreation, gait appearance, use of braces/assistive devices, and body image/self-esteem. The GOAL-LD was adapted from the original Gait Outcomes Assessment List (GOAL) [7] for use with children who have lower limb differences. The article included 25 children, aged 9–18 years, who were recruited from limb reconstruction clinics [6]. However, only one participant used LLP (Table 2). This study reported on face and content validity, which were assessed through cognitive interviews with children who had lower limb differences and their parents, and through online surveys with healthcare professionals (i.e., pediatric orthopaedic surgeons, physical therapists, and nurse practitioners). Items in the questionnaire with at least 90% acceptance by respondents were included with a total of 45 items in the final instrument (Table 3). Further psychometric assessments (e.g., test-retest reliability and construct validity) were reported in a follow-up article but were not included in this review because the sample for the follow-up article did not include children who use LLP [8].
Measurement characteristics for publications that assessed psychometric properties of outcome measures for pediatric participants who use lower limb prostheses (LLP)
Measurement characteristics for publications that assessed psychometric properties of outcome measures for pediatric participants who use lower limb prostheses (LLP)
aAdditional validity assessments in participants with lower limb differences, but no evidence that children who used LLP were among the participants in this study sample [8].
One article [9] examined the psychometric properties of the FMA, a performance-based outcome measure with three patient-reported items examining functional mobility. The FMA was originally developed for patients with osteosarcoma aged 13 years or older [9]. Scores from the Timed Up and Go (TUG), Timed Up and Down Stairs (TUDS), and the 9-minute walk test (9MWT), as well as heart rate (HR) during each activity, Borg Rating of Perceived Exertion (RPE), Wong and Baker FACES pain scale, assistive device use, and other patient-reported items are combined in the FMA to produce a maximum score of 70. Authors of this study also collected physiological cost index (PCI) to compare each item of the FMA for the control and LLP user groups to assess known groups validity. The study included 37 participants, aged 8–19 years, 25 of whom had amputations or limb differences at varied levels. They reported good ability to discriminate between control and LLP user groups in overall FMA score and the individual sub-scores of the TUG, TUDS, and 9MWT items (p < 0.01), providing evidence of known groups validity. No difference was found between the two groups in FACES, RPE, or PCI for any item of the FMA (Tables 2, 3).
Child Amputee Prosthetics Project- Prosthesis Satisfaction Inventory (CAPP-PSI)
One article examined the psychometric properties of the CAPP-PSI parent-proxy reported outcome measure assessing HRQoL [10]. This measure is one of the scales within the CAPP and looks specifically at satisfaction with the prosthesis and service. CAPP-PSI has three subscales: parent-rated child satisfaction with the prosthesis (proxy measure), parent satisfaction with the prosthesis, and parent satisfaction with service. The study included 97 upper and lower limb prosthesis users, aged 1–17 years, with 57 participants using LLP. This study reported on face validity, construct validity, and internal consistency (i.e., item-to-item and item-total correlations). Face validity was assessed by expert clinician review. For convergent construct validity, child and parent satisfaction scales one year later were significantly correlated with parent ratings of prosthesis appearance on a visual analogue scale when the device was new (r = 0.52 and r = 0.48, respectively). However, only parent satisfaction was significantly correlated to their rating of prosthesis appearance one year later (r = 0.40). Parent and child satisfaction with prosthesis scales were correlated with prosthesis wear and use (r = 0.37–0.56). For divergent construct validity, satisfaction with service was not well correlated with prosthesis wear and use. Internal consistency reliability was reported for item-to-item correlations (Cronbach’s alpha = 0.8–0.9) and item-total correlations (r = 0.5–0.8) (Tables 2, 3).
CAPP- Functional Scale Index (CAPP-FSI)
One article examined the psychometric properties of the CAPP-FSI parent-proxy reported outcome measure assessing function [11]. This measure is another scale within the CAPP; it looks specifically at the function of children who use a prosthesis. Items in the instrument assess the extent to which the child engages in a range of activities and whether they use their prosthesis to complete the activity. The study included 65 upper and lower limb prosthesis users, aged 8–17, with 43 participants using LLP. CAPP-FSI has two subscales: upper extremity function and lower extremity function (Table 2). This study reported on face validity, construct validity, and internal consistency (i.e., item-to-item and item-total correlations). Face validity was assessed by expert clinician review. Known groups construct validity was assessed by examining prosthesis use for a given task in a category (upper or lower extremity) between upper and lower limb loss or difference groups. Children with LLLD were significantly more likely to endorse “uses prosthesis” for lower extremity items on the CAPP-FSI (p < 0.0001) compared to children with upper limb loss or difference. With respect to internal consistency reliability assessments, the measure had moderate to good task-to-task consistency (Cronbach’s alpha = 0.58–0.96), and all item-total correlations for retained items were r > 0.25 (Table 3). Two subsequent measures were derived from the CAPP-FSI specifically for toddlers [12] and preschoolers [13].
Lower Limb Function Questionnaire (LLFQ)
One article examined construct validity and test-retest reliability of the LLFQ, a patient-reported outcome measure of overall function [14]. This study included 40 participants, aged 13–25 years, nine of whom used LLP. Convergent construct validity was assessed by comparing the LLFQ to other functional performance tests (i.e., 6-Minute Walk Test (6MWT), TUG, obstacle course distance traveled, oxygen consumption during the 6MWT and obstacle course, and gait parameters during the TUG). LLFQ mean score was significantly correlated with distance traveled on an obstacle course (r = 0.62) and some of the TUG gait parameters: velocity (r = 0.66), stride length (r = 0.76), stance phase percent of gait cycle (r=–0.699), single-support phase percent of gait cycle (r = 0.70), and double-support phase percent of gait cycle (r=– 0.695). LLFQ mean score was not significantly correlated with 6MWT scores, overall TUG scores, or oxygen consumption during the obstacle course or 6MWT. Test-retest reliability was reported to have an intraclass correlation coefficient (ICC) of 0.79 (Tables 2, 3).
Discussion
Results from this scoping review demonstrated that 44 self-reported and performance-based measures have been used to assess function, HRQoL, and satisfaction in recent pediatric LLP research. There were almost as many outcome measures as the number of pediatric LLP research articles (41), indicating a wide variety of measures in use. The variation in standardized outcome measures used across these research publications presents challenges to clinicians and researchers who attempt to synthesize findings across pediatric LLP research studies. Such synthesis is particularly important for clinicians determining outcomes and treatment decisions for children who use LLP, given that sample sizes are often small in each individual research study. Consistency in the standardized outcome measures used across research studies would facilitate synthesis of pediatric LLP research information to support evidence-based practice. This is highlighted by the recently published recommendation from the International Society of Prosthetics and Orthotics, which provided recommendations for specific standardized outcome measures to be used in assessing adults with lower limb absence [15]. A potential opportunity then arises for the development of core outcome sets or consensus in outcomes of importance in pediatric LLP research. A lack of validated outcome measures is seen in multiple medical disciplines (e.g., pediatric orthopedics [16]) and diagnoses (e.g., cerebral palsy [17]).
Of the 44 outcome measures identified, only five had publications that described the assessment of validity and/or reliability in children who use LLP. However, the data provided in these studies did not demonstrate a complete psychometric assessment for any of these standardized outcome measures, meaning that the publications did not describe validity and/or reliability assessments that could guide clinical use of these measures. Furthermore, the articles that assessed measurement properties for these instruments included relatively few children who use LLP. The age range of the participants was typically skewed towards older pediatric participants. No data of responsiveness or normative data were found for any measure examined.
There are many reasons for this dearth of publications in pediatric LLP outcome measures, which may include recruiting difficulties, few clinicians and researchers specializing in pediatric LLP, heterogeneity of this population, and a lack of emphasis on outcome measure use. The relatively small number of pediatric LLP users available to participate in these studies may increase the difficulty in recruiting participants for validation studies. This limitation is compounded by even fewer pediatric specialized clinicians and researchers with the experience and expertise to conduct these studies. Much of the research in outcome measures for LLP users has been funded by the Department of Veterans Affairs and Department of Defense, which understandably prioritize research for adults who use LLP. These factors contribute both to the paucity of literature in general and to the lack of outcome measures that appropriately assess children who use LLP and that provide appropriate assessment by clinicians who care for them. For example, while multiple measures are available to assess comfort or pain in children, only two studies [9, 18] used single-domain outcome measures specific to comfort or pain. This is surprising given how integral comfort and pain are to overall quality of life and how frequently they are given as reasons for visits to the prosthetist’s office. Further, condition-specific assessments of comfort, such as the Socket Comfort Score (SCS) [19, 20], were not found through the initial search. Assessment of socket comfort is a critical aspect of prosthetic care. Thus, measures like the SCS could provide valuable clinical information when treating pediatric LLP patients. However, further research is needed to support use of these condition-specific measures for children who use LLP.
Additionally, the demographics and causes of limb loss are different and more varied in children than adults. Congenital limb deficiency, trauma, and cancer diagnoses are the leading causes for amputation in children, while vascular disease is the primary etiology resulting in amputation in adults [1]. Congenital limb difference in children is noted in 0.079% of overall births and varies greatly in cause [21]. Many of these are idiopathic in nature and present clinically in a number of different ways [22]. These etiological differences of amputation can be associated with distinctions in overall activity level and functionality. Children typically have a higher activity level and physically change more rapidly than adults. Because of this preconception, insurance more readily covers prostheses for children as they outgrow them. Consequently, the expectation for adults is that prostheses are replaced at most every five years with the need for additional medical justification, unless significant functional or physical change has occurred sooner. This stereotypical difference in activity level and standardized insurance coverage may have led to a deemphasis of outcome measures in the pediatric LLP population.
This review highlights the need for further testing and/or development of standardized outcome measures to support the holistic and multidisciplinary care of children who use LLP. Routine assessment using standardized performance-based and patient-reported outcome measures of function, HRQoL, and satisfaction may identify areas for improvement in prosthetic treatment and overall health that might otherwise go unnoticed. Standardized outcome measures that assess patients, both clinically and scientifically, should have evidence of psychometric testing (reliability, validity, and responsiveness) in the population of interest to assure that they are appropriate for clinical use. Normative data would also be necessary when interpreting results. This study demonstrates the variability in standardized outcome measures used and the lack of psychometric evidence to support the use of outcome measures to assess children who use LLP.
Until this evidence is developed, other factors need to be considered. When faced with limitations in standardized outcome measures well-suited for use with children who use LLP, some clinicians and researchers will consider using outcome measures validated only in pediatric patients not using LLP or only in adults with LLP. For example, the psychometric properties of the 6MWT have been evaluated in various pediatric populations [23] and in adult LLP users [24]. It could be argued that the 6MWT may be appropriate for use in a child who uses LLP, can follow instructions, and has adequate attention span, even though there is not yet psychometric evidence supporting its use in this specific population. The fact that there is sufficient evidence in not just one but two similar populations may be sufficient to support its use clinically for children who use LLP. It also gives substantial support for a future validation study in children who use LLP. Limited research funding for this population may require careful consideration of which outcome measures should be studied for further validation. If a measure has sufficient evidence available in pediatric populations or adult LLP users, then it may be more advantageous to focus future studies on measures with less evidence overall.
In some cases, it is reasonable that prosthetic-specific measures designed for adults may be applied to children who use LLP. Measures like the Amputee Mobility Predictor (AMP) may be considered for future research with older children (i.e., teenagers) who use LLP to determine age-specific applicability of the measure. Researchers must be careful in choosing their methods of assessment based on the evidence available. Clinicians must use good clinical judgement (e.g., Does my patient have the cognitive capacity to understand the instructions?, Is my patient tall enough to stand and sit in the chair?) when deciding which outcome measures are best for their specific patients in the absence of evidence. Although the AMP does not have psychometric evidence in children who use LLP, a practitioner who is familiar with and comfortable administering the measure may decide to use it to assess older children who use LLP in order to track longitudinal changes in function and areas for improvement.
Limitations
This scoping review has limitations. First, a scoping review protocol was developed a priori but not registered. Registering a scoping review a priori is important to mitigate the risk for reporting bias. All deviations from the a priori protocol were reported in the methods section to increase transparency and minimize this risk. The lack of standardized language across the pediatric LLP literature made the initial search and screening process difficult. Terms that have been used to describe an internal prosthesis (e.g., endoprosthesis) were mixed and inconsistently used. Since this article examined only the use of external LLP, this inconsistent language was difficult to navigate. In addition, the wording of some article titles and abstracts may have caused them to be misunderstood as not meeting inclusion criteria. To reduce the impact of this inconsistent language on the results, two reviewers independently screened each article at each phase of the screening process. In addition, a conservative approach was taken to be as inclusive as possible in the title and abstract rounds of the screening process. The use of controlled vocabulary was used in the search strategy to be as inclusive as possible. Another limitation of this article is that it only examined the outcome measures used in published literature over the past 20 years. Thus, measures that are only being used clinically but not in recent research were not included in the review. This decision was made because outcome measures that are being used in the literature are likely to have the most evidence for use in pediatric LLP. This also provided objective and repeatable criteria to help determine inclusion or exclusion of a measure for further examination. New standardized outcome measures are being developed that may be well-suited to children who use LLP (e.g., LimbQ Kids [25], Pedi CHAMP [26]). However, these instruments were not yet published at the time of writing this article. These measures, and future ones yet to be developed, will hopefully begin to close the gap in the literature on outcome measures well-suited to measurement in children who use LLP.
Conclusion
The amount of literature assessing children using LLP is limited overall, as is the literature presenting psychometric data for outcome measures assessing this population. Further research is needed to validate the current outcome measures in pediatric LLP. Clinicians may be especially interested in those measures that are separately validated in both pediatric non-LLP and adult LLP users. If a measure is not found to be valid in children who use LLP, then the development of new measures may be warranted to ensure that clinicians and researchers are able to assess the constructs of interest in this population. Once the necessary evidence is available, a set of outcome measures for children who use LLP can be developed to help clinicians and researchers make informed decisions about the measure they choose to assess their patients.
Footnotes
Funding
Funding for this work was provided by the generous donors of Gillette Children’s Specialty Healthcare.
Conflict of interest
The authors have no conflict of interest to report.
Ethical considerations
This study, as a literature review, is exempt from Institutional Review Board approval.
Appendix 1 – Search Strategy Table for Primary Search
| PubMed | ||
| Search | Field Code | Search term(s) |
| 1 | MeSH Terms | artificial limb OR amputation |
| 2 | Title/Abstract | “limb deficiency” OR “limb difference” OR “congenital difference” OR “congenital deficiency” OR “limb deficiencies” OR “limb differences” OR “congenital differences” OR “congenital deficiencies” OR prosthesis OR prosthetic* OR prostheses |
| 3 | 1 OR 2 | |
| 4 | MeSH Terms | pediatric OR children |
| 5 | Title/Abstract | child OR kid OR kids OR toddler* OR infant* OR teen* OR adolescent* OR “young adult” OR “young adults” |
| 6 | 4 OR 5 | |
| 7 | Title/Abstract | “total hip” OR “total joint” OR valve* OR dental OR teeth OR tooth OR penile OR ocular OR cochlear OR ossicular OR stapes OR palate OR eye OR eyes OR ear OR ears OR arthroplasty OR endoprosthesis OR endoprostheses OR animal* OR veterinary |
| 8 | 3 AND 6 NOT 7 | |
| CINAHL (EBSCO) | ||
| Search | Field Code | Search term(s) |
| 1 | SU | artificial limb OR artificial limbs OR amputation OR limb prosthesis |
| 2 | TI | “limb deficiency” OR “limb difference” OR “congenital difference” OR “congenital deficiency” OR “limb deficiencies” OR “limb differences” OR “congenital differences” OR “congenital deficiencies” OR prosthesis OR prosthetic* OR prostheses OR “artificial limb” OR “artificial limbs” |
| 3 | AB | “limb deficiency” OR “limb difference” OR “congenital difference” OR “congenital deficiency” OR “limb deficiencies” OR “limb differences” OR “congenital differences” OR “congenital deficiencies” OR prosthesis OR prosthetic* OR prostheses OR “artificial limb” OR “artificial limbs” |
| 4 | 1 OR 2 OR 3 | |
| 5 | SU | pediatrics OR children |
| 6 | TI | child OR kid OR kids OR toddler* OR infant* OR teen* OR adolescent* OR “young adult” OR “young adults” OR pediatrics OR children |
| 7 | AB | child OR kid OR kids OR toddler* OR infant* OR teen* OR adolescent* OR “young adult” OR “young adults” OR pediatrics OR children |
| 8 | 5 OR 6 OR 7 | |
| 9 | TI | “total hip” OR “total joint” OR valve* OR dental OR teeth OR tooth OR penile OR ocular OR cochlear OR ossicular OR stapes OR palate OR eye OR eyes OR ear OR ears OR arthroplasty OR endoprosthesis OR endoprostheses OR animal* OR veterinary |
| 10 | AB | “total hip” OR “total joint” OR valve* OR dental OR teeth OR tooth OR penile OR ocular OR cochlear OR ossicular OR stapes OR palate OR eye OR eyes OR ear OR ears OR arthroplasty OR endoprosthesis OR endoprostheses OR animal* OR veterinary |
| 11 | 9 OR 10 | |
| 12 | 4 AND 8 NOT 11 | |
| Web of Science | ||
| Search | Field Code | Search term(s) |
| 1 | TI | artificial limb OR artificial limbs OR Amputation OR “limb deficiency” OR “limb difference” OR “congenital difference” OR “congenital deficiency” OR “limb deficiencies” OR “limb differences” OR “congenital differences” OR “congenital deficiencies” OR prosthesis OR prosthetic* OR prostheses |
| 2 | AB | artificial limb OR artificial limbs OR “limb deficiency” OR “limb difference” OR “congenital difference” OR “congenital deficiency” OR “limb deficiencies” OR “limb differences” OR “congenital differences” OR “congenital deficiencies” OR prosthesis OR prosthetic* OR prostheses |
| 3 | 1 OR 2 | |
| 4 | TI | pediatric* OR children OR child OR kid OR kids OR toddler* OR infant* OR teen* OR adolescent* OR “young adult” OR “young adults” |
| 5 | AB | pediatric* OR children OR child OR kid OR kids OR toddler* OR infant* OR teen* OR adolescent* OR “young adult” OR “young adults” |
| 6 | 4 OR 5 | |
| 7 | TI | “total hip” OR “total joint” OR valve* OR dental OR teeth OR tooth OR penile OR ocular OR cochlear OR ossicular OR stapes OR palate OR eye OR eyes OR ear OR ears OR arthroplasty OR endoprosthesis OR endoprostheses OR animal* OR veterinary |
| 8 | AB | “total hip” OR “total joint” OR valve* OR dental OR teeth OR tooth OR penile OR ocular OR cochlear OR ossicular OR stapes OR palate OR eye OR eyes OR ear OR ears OR arthroplasty OR endoprosthesis OR endoprostheses OR animal* OR veterinary |
| 9 | 7 OR 8 | |
| 10 | 3 AND 6 NOT 9 | |
Appendix 2 – Search Strategy Tables for Secondary Search
| Pediatric Prosthetics | ||
| PubMed | ||
| Search | Field Code | Search term(s) |
| 1 | Title/Abstract | [name of outcome measure] |
| 2 | Title/Abstract | psychometric* OR valid* OR reliab* |
| 3 | 1 AND 2 | |
| 4 | MeSH Terms | artificial limb OR amputation |
| 5 | Title/Abstract | “limb deficiency” OR “limb difference” OR “congenital difference” OR “congenital deficiency” OR “limb deficiencies” OR “limb differences” OR “congenital differences” OR “congenital deficiencies” OR prosthesis OR prosthetic* OR prostheses |
| 6 | 4 OR 5 | |
| 7 | MeSH Terms | pediatric OR children |
| 8 | Title/Abstract | child OR kid OR kids OR toddler* OR infant* OR teen* OR adolescent* OR “young adult” OR “young adults” |
| 9 | 7 OR 8 | |
| 10 | 3 AND 6 AND 9 | |
| CINAHL (EBSCO) | ||
| Search | Field Code | Search term(s) |
| 1 | TI | [name of outcome measure] |
| 2 | AB | [name of outcome measure] |
| 3 | 1 OR 2 | |
| 4 | SU | psychometrics |
| 5 | TI | psychometric* OR valid* OR reliab* |
| 6 | AB | psychometric* OR valid* OR reliab* |
| 7 | 4 OR 5 OR 6 | |
| 8 | 3 AND 7 | |
| 9 | SU | artificial limb OR artificial limbs OR amputation OR limb prosthesis |
| 10 | TI | “limb deficiency” OR “limb difference” OR “congenital difference” OR “congenital deficiency” OR “limb deficiencies” OR “limb differences” OR “congenital differences” OR “congenital deficiencies” OR prosthesis OR prosthetic* OR prostheses OR “artificial limb” OR “artificial limbs” |
| 11 | AB | “limb deficiency” OR “limb difference” OR “congenital difference” OR “congenital deficiency” OR “limb deficiencies” OR “limb differences” OR “congenital differences” OR “congenital deficiencies” OR prosthesis OR prosthetic* OR prostheses OR “artificial limb” OR “artificial limbs” |
| 12 | 9 OR 10 OR 11 | |
| 13 | SU | pediatrics OR children |
| 14 | TI | child OR kid OR kids OR toddler* OR infant* OR teen* OR adolescent* OR “young adult” OR “young adults” OR pediatrics OR children |
| 15 | AB | child OR kid OR kids OR toddler* OR infant* OR teen* OR adolescent* OR “young adult” OR “young adults” OR pediatrics OR children |
| 16 | 13 OR 14 OR 15 | |
| 17 | 8 AND 12 AND 16 | |
| Web of Science | ||
| Search | Field Code | Search term(s) |
| 1 | TI | [name of outcome measure] |
| 2 | AB | [name of outcome measure] |
| 3 | 1 OR 2 | |
| 4 | TI | psychometric* OR valid* OR reliab* |
| 5 | AB | psychometric* OR valid* OR reliab* |
| 6 | 4 OR 5 | |
| 7 | 3 AND 6 | |
| 8 | TI | artificial limb OR artificial limbs OR Amputation OR “limb deficiency” OR “limb difference” OR “congenital difference” OR “congenital deficiency” OR “limb deficiencies” OR “limb differences” OR “congenital differences” OR “congenital deficiencies” OR prosthesis OR prosthetic* OR prostheses |
| 9 | AB | artificial limb OR artificial limbs OR “limb deficiency” OR “limb difference” OR “congenital difference” OR “congenital deficiency” OR “limb deficiencies” OR “limb differences” OR “congenital differences” OR “congenital deficiencies” OR prosthesis OR prosthetic* OR prostheses |
| 10 | 8 OR 9 | |
| 11 | TI | pediatric* OR children OR child OR kid OR kids OR toddler* OR infant* OR teen* OR adolescent* OR “young adult” OR “young adults” |
| 12 | AB | pediatric* OR children OR child OR kid OR kids OR toddler* OR infant* OR teen* OR adolescent* OR “young adult” OR “young adults” |
| 13 | 11 OR 12 | |
| 14 | 7 AND 10 AND 13 | |
