Abstract
PURPOSE:
This study explored neuromuscular mechanisms and clinical measures that contribute to countermovement jump performance in survivors of childhood acute lymphoblastic leukemia (ALL CCS) compared to age- and sex-matched peers.
METHODS:
This exploratory cross-sectional observational study examined 12 participants, six ALL CCS and six age- and sex-matched peers (7–16 years). During a countermovement jump, rates of muscle activation of lower leg muscles were measured with electromyography, and joint torques and peak jump height with force plates and a motion capture system. Clinical measures included muscle extensibility, balance, and mobility measured by active ankle dorsiflexion, Bruininks-Oseretsky Motor Proficiency (BOT-2), and Timed Up and Go (TUG) tests.
RESULTS:
Compared to peers, ALL CCS demonstrated reduced gastrocnemius muscle extensibility and tibialis anterior rate of muscle activation, decreased jump height, and poorer performance on the BOT-2 and TUG. Jump height was significantly correlated with clinical measures of the BOT-2 and TUG.
CONCLUSION:
These ALL CCS demonstrated neuromuscular impairments that may impact jump performance, an essential childhood physical activity. Further research is needed to explore intervention strategies to improve the neuromuscular mechanisms that contribute to high-level gross motor skills in ALL CCS.
Introduction
Acute lymphoblastic leukemia (ALL) is the most prevalent pediatric cancer, with survival rates exceeding 90 percent [1]. ALL is commonly diagnosed in children younger than 10 years of age and requires medical treatment lasting two to three years. This is a critical time period for the development of gross motor skills, such as jumping. The skill of jumping is required in many childhood physical activities such as playground activities and sports. However, many survivors of childhood ALL (ALL CCS) experience long-term peripheral and central nervous system (CNS) neurotoxic side effects [2–4] that can lead to poor muscle function [5–13], decreased balance proficiency [14–17], impaired gross motor mobility [5, 11] and reduced physical activity levels [18].
The chemotherapy agents most commonly used in ALL treatment include vincristine, methotrexate, mercaptopurine (6-MP), cytarabine, doxorubicin, busulfan, cyclophosphamide, and L-asparaginase. Specifically, vincristine and methotrexate have known short- and long-term neurotoxic side effects that impact the sensory and motor function of distal muscles of the lower extremities [2, 3] and motor processing speed [19]. These physiological changes negatively impact the capacity to acquire and refine muscle activation patterns [6, 11]. As a consequence, muscle activation takes longer [2, 3], motor timing is slower [20], ankle dorsiflexion range of motion is limited [7, 21–23], and agonist and antagonist muscles co-activate during gross motor tasks such as walking [4, 6].
The deficits in the sensory and motor systems in ALL CCS limit the ability to participate in sports or other recreational activities and to keep up with peers [10]. The development of age-appropriate motor skills is necessary to facilitate participation in physical activity and maintain health-related fitness levels [24]. Deficits in gross motor skills can limit engagement in age-appropriate physical activities and can significantly influence the child or adolescent’s quality of life [25]. Additionally, participation in sports and physical activity is associated with improved general health, disease prevention, social behavior, and psychosocial development [25]. Fewer experiences that stimulate neuromuscular input and output can impair balance control and thus impede participation in physical activities [18]. Studies that investigate the mechanisms of neuromuscular function and gross motor performance in ALL CCS are necessary to develop targeted interventions.
The primary purpose of this study was to explore the neuromuscular mechanisms, including the rate of muscle activation and joint torques, that contribute to countermovement jump performance as measured by jump height in ALL CCS compared to age- and sex-matched peers without ALL. The secondary purpose was to explore the clinical measures of muscle extensibility, balance, and mobility and their contribution to jumping performance. We hypothesized that compared to age- and sex-matched controls, ALL CCS would have lower anterior tibialis and gastrocnemius muscle activation rates, decreased joint torques, and reduced jump height during a countermovement jump. Additionally, we hypothesized that ALL CCS would present with impaired gastrocnemius muscle extensibility and poorer balance and mobility performance, measured by the Bruininks-Oseretsky Test of Motor Proficiency, 2nd edition (BOT-2) and Timed Up and Go (TUG) respectively, as compared to controls. The relationships between neuromuscular mechanisms and clinical measures of muscle extensibility, balance, and mobility were explored. We hypothesized that there would be positive correlations between muscle extensibility, rate of muscle activation, and joint torques with jump height, a positive correlation between jump height with BOT-2 scores, and a negative correlation with TUG time.
Methods
Study participants
In this exploratory cross-sectional observational study, ALL CCS participants were recruited from the Department of Pediatric Hematology/Oncology at the University of Maryland Medical Center. Individuals were asked to participate if they were between 6 and 17 years of age and had completed medical treatment for ALL between one and 60 months prior to enrollment. Participants with a previous diagnosis of any neurological disorder preceding the ALL diagnosis were excluded from the study. Age- and sex-matched controls were recruited through the University of Maryland, School of Medicine, Department of Physical Therapy & Rehabilitation Science. The study was approved by the University of Maryland, Baltimore Institutional Review Board. Informed consent and assent were obtained from all participants and their guardians.
Data collection
Participant demographic information was collected including sex, age, height, weight, and level of participation in recreational activities as measured by the Children’s Activity Participation and Enjoyment (CAPE) diversity score. The CAPE diversity score is the number of activities the child has reported participating in over the past four months. For ALL CCS, age of diagnosis, duration of on-treatment time, and years off-treatment were also collected.
Participants performed three trials of a countermovement jump, which is a valid and reliable functional measure of the capacity to produce lower extremity force [26]. The participants stood with each foot on two adjacent force platforms (AMTI, Inc., Newton, MA, USA) in an upright position, arms at the side, and hands slightly anterior to the hips. Following a demonstration, participants were instructed to “jump as high as you can”. The countermovement jump was performed by a downward movement into a squat position and then an immediate push off of the supporting surface into a jump. The participants rested one minute between each trial.
Measurements of the rate of muscle activation were collected using surface electromyography (EMG) with a TeleMyo™ DTS 24-channel wireless EMG system (Noraxon, USA Inc., Scottsdale, AZ, USA). Dual electrodes with a 2 cm inter-electrode distance were placed bilaterally over the muscle bellies of the soleus, tibialis anterior, and lateral gastrocnemius muscles according to SENIAM recommendations [27]. To obtain measurements of jump height and lower extremity joint torques, reflective markers were placed on the bilateral acromia, lateral epicondyles, ulnar styloid processes, greater trochanters, lateral condyles, lateral malleoli, 2nd metatarsal heads, inter-aural axes, and on the top of the head. Kinematic data were recorded with a ten-camera motion capture system (VICON, Los Angeles, CA, USA) at 150 Hz, and ground reaction forces were measured using dual force plates at 600 Hz and resampled to 120 Hz.
Clinical measures of muscle extensibility, balance, and mobility were collected. Gastrocnemius muscle extensibility was measured while in a sitting position with the knee in full extension. Using a goniometer, active ankle dorsiflexion was measured starting from a full plantarflexed position and averaged across 2 trials. Balance was measured by the BOT-2 balance subscale, which assessed balance during standing and walking in a series of pre-determined positions that alter the base of support (BOS). Items were administered and scored according to standardized procedures. Mobility was assessed using the TUG, a measure of the time it takes to rise from a chair, walk three meters, turn around, walk back to the chair and sit down. Participants completed two trials and the lowest time was used for analysis. To ensure consistency, a single experienced examiner completed the testing trials for each outcome measure across all of the participants. When using standardized procedures, the clinical measures used in this study are valid and reliable, including established intra- and inter-rater reliability in children with and without physical impairments [28–33].
Data analysis
The onset of muscle activity was identified when the EMG amplitude was greater than two standard deviations above the baseline activity, which was calculated 500 milliseconds preceding movement onset. The rate of muscle activation was calculated as the EMG amplitude, from baseline to peak, divided by the time from onset to peak muscle activity. Values were normalized by dividing by the peak EMG amplitude of each muscle during the task. There was no significant difference between the right and left limbs for either group; therefore, all values for the right and left limbs were averaged and the mean value of the trials were reported. EMG signals were collected with MyoResearch XP software (Noraxon, USA Inc., Scottsdale, AZ, USA) at a 1500 Hz sampling rate and later exported to Spike 2 V6.03 software (Cambridge Electronic Design, Cambridge, UK) for analysis. After subtracting off the mean DC bias, EMG data were rectified and high-pass filtered at 20 Hz.
The joint torque for the hips, knees, and ankles was estimated by inverse dynamics as described by Zatsiorsky [34]. Peak joint torque of the hips, knees, and ankles was identified as the maximum value during the jump acceleration phase from the lowest point of the center of mass (COM) to toe-off from the force plates. The mean joint torque from the trials was normalized to the height and weight of the participant. Ground reaction forces and kinematic data were filtered using a 10 Hz low-pass 4th order Butterworth zero-lag filter, MATLAB filtfilt (MathWorks, Natick, MA, USA). Data were bandpass filtered between 16–500 Hz. All values for the right and left limb were averaged because there were no significant differences between the right and left limb for either group.
Countermovement jump height, measured in the sagittal plane from whole-body motion capture, was calculated from the body COM. The COM was estimated using the method described by Jensen [35]. Jump height was calculated as the mean of the difference between peak COM height during the jump and COM height at the start of the jump. The jump height was normalized by the height of the participant.
Statistical analysis
Statistical analysis was performed using SPSS version 26.0 (IBM Inc., Chicago, IL, USA). Descriptive statistics were reported as mean±standard deviation (SD). Normality of data was assessed using skewness, kurtosis, and the Shapiro-Wilk test. Nonparametric statistical methods were used because the variables demonstrated skewed distributions. Muscle extensibility, the rate of muscle activation, peak joint torques, jump height, BOT-2, and TUG were tested for between-group significance using a Mann-Whitney U. Correlation comparison between clinical measures and the jump variables was determined using the Spearman’s correlation coefficient. Since the nature of this study was exploratory, no multiplicity corrections for the number of statistical tests were utilized. All comparisons were made at the α< 0.05 level of significance.
Results
A total of 12 children participated in this study, six ALL CCS (3 males, 3 females), and six age- and sex-matched controls. Descriptive characteristics of individual participants are listed in Table 1. ALL CCS mean age was 11.52±3.27 years, and the control group was 10.92±3.55 years. There were no significant differences in age, height, weight, or reported level of participation, as measured with the CAPE, between the groups. The mean age of diagnosis was 5.93±3.08 years, time on treatment for ALL CCS was 35.16±5.74 months, and time from completion of medical intervention (off-treatment) for the ALL CCS was 27.50±17.24 months.
Participant Characteristics of Study Population
Participant Characteristics of Study Population
While performing a countermovement jump, ALL CCS demonstrated a significantly reduced rate of muscle activation of the tibialis anterior muscle as compared to controls (p = 0.030) (Fig. 1), but no significant differences in the rate of activation of the soleus (p = 0.328) or lateral gastrocnemius (p = 0.247) muscles. Of the three lower extremity joints, there were no significant between-group differences in the amount of peak torque that was developed at the ankles (p = 1.00), knees (p = 0.485), or hips (p = 0.818) during the acceleration phase of the countermovement jump (Fig. 2B-D). ALL CCS presented with significantly reduced peak jump height compared to controls (p = 0.041) (Table 2 and Fig. 2A).
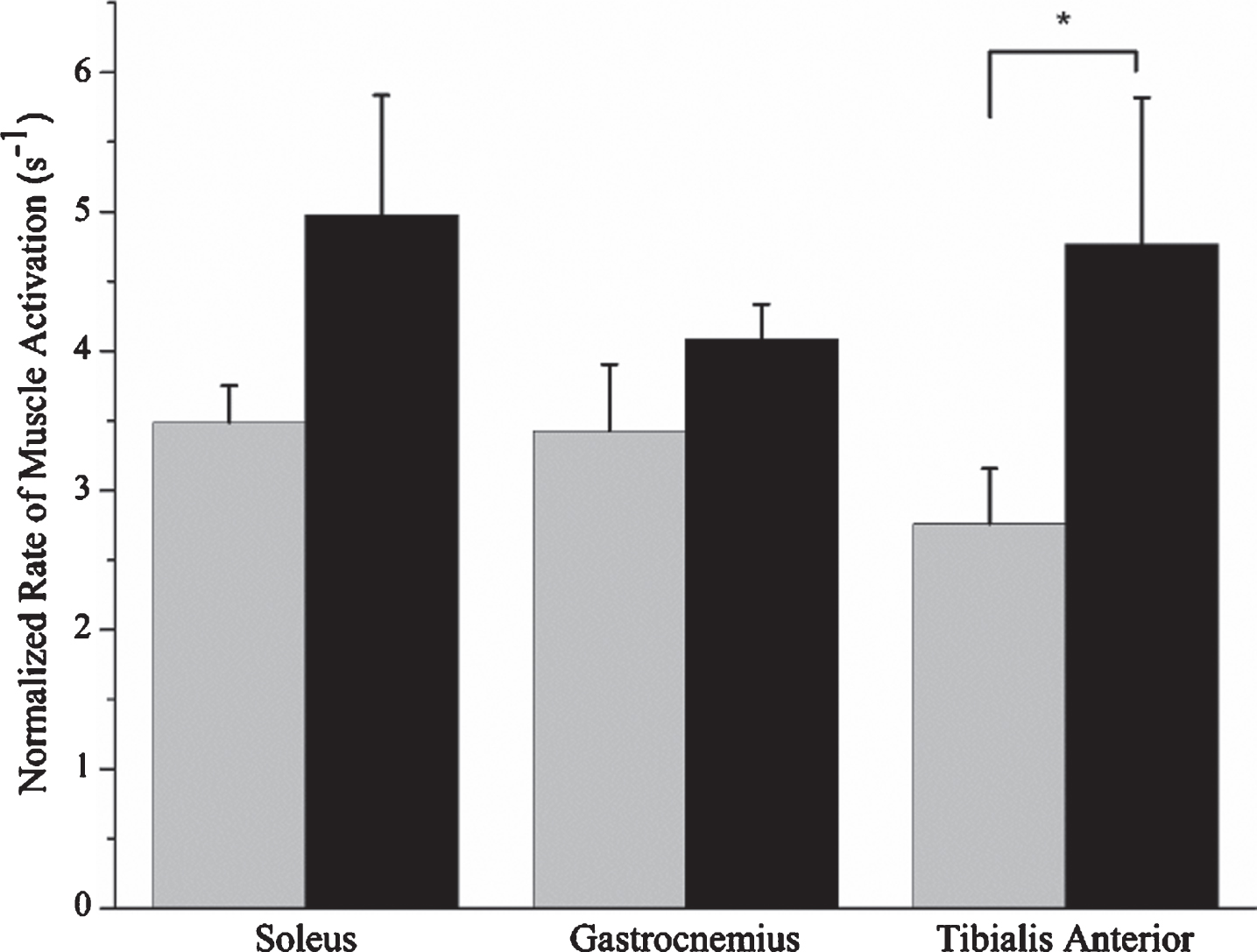
Rate of muscle activation of ALL CCS (gray bar) and controls (black bar) for the soleus, gastrocnemius, and tibialis anterior muscles. Expressed as the mean and standard error. *P < 0.05 for Mann-Whitney U.
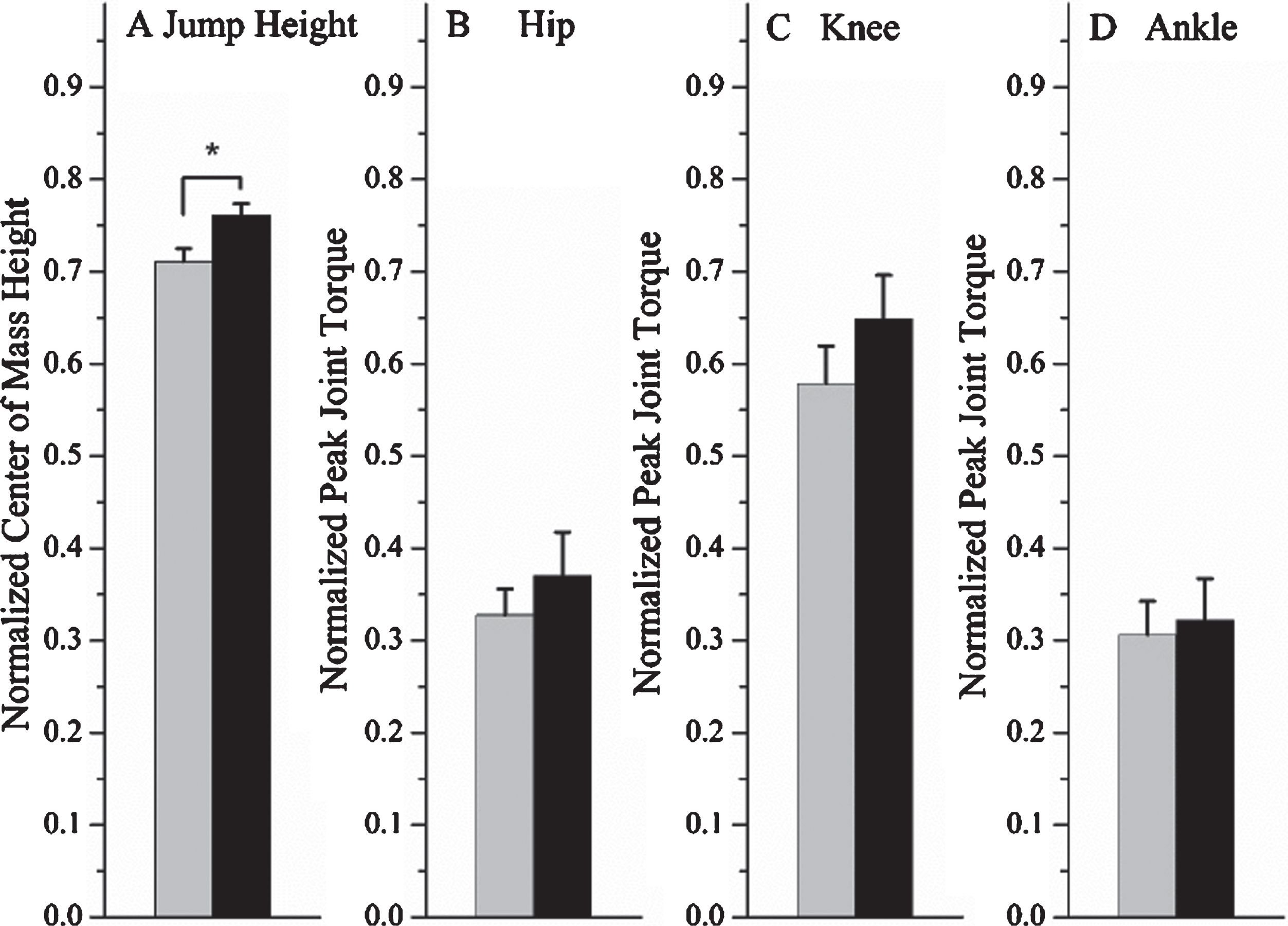
The jump height (A) and peak joint torque of the hip (B), knee (C), and ankle (D) of the ALL CCS (gray bar) and age- and sex-matched controls (black bar). Expressed as a mean and standard error. *P < 0.05 for Mann-Whitney U.
Outcome Measures of Survivors of Childhood ALL (ALL CCS) and Controls
*P < 0.05 for Mann-Whitney U.
ALL CCS had significantly less gastrocnemius extensibility (p = 0.015) and lower scores on the BOT-2 balance subscale (p = 0.004), and they took longer to complete the TUG (p = 0.002) than the controls (Table 2). There was a strong correlation between the time off-therapy with the BOT-2 balance scale (rs = 0.93; p = 0.008), but not between the rate of muscle activation of the gastrocnemius muscle, peak jump height, gastrocnemius extensibility, or TUG time. There were moderate correlations between peak jump height with the BOT-2 (rs = 0.78, p = 0.003) and TUG time (rs = –0.61, p = 0.037) (Fig. 3). Gastrocnemius extensibility was negatively correlated with the time to complete TUG (rs = –0.67, p = 0.017).
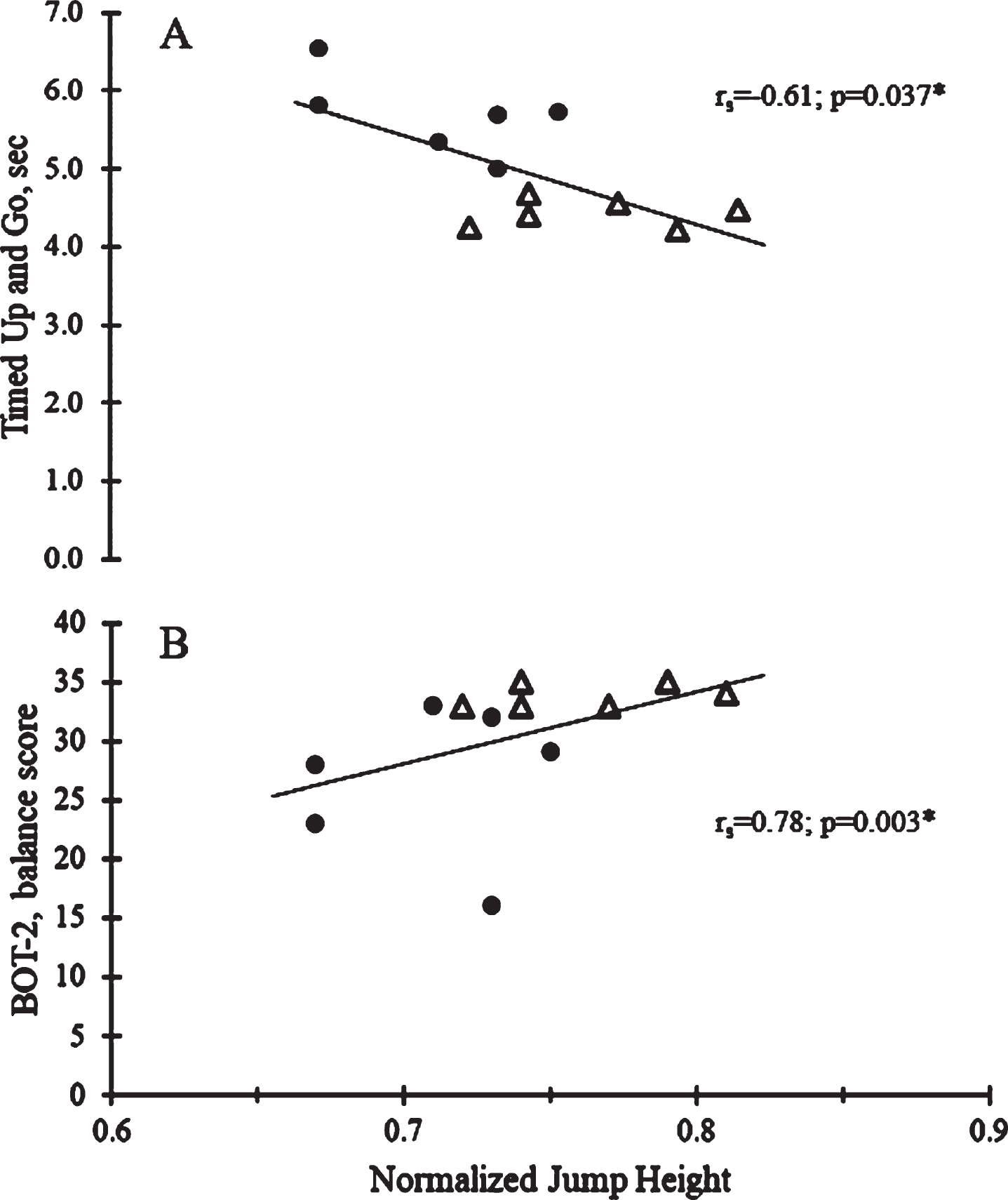
Relationships between jump height versus (A) Timed-Up and Go (TUG) and (B) Bruininks-Oseretsky Test of Motor Proficiency 2 (BOT-2), balance subscale in ALL CCS (filled circles) and controls (open triangles).
This study explored the neuromuscular mechanisms that contribute to countermovement jump performance in ALL CCS compared to age- and sex-matched peers. Our results showed that, compared to peers, this sample of ALL CCS demonstrated deficits in gross motor performance with a decreased jump height, reduced gastrocnemius muscle extensibility, lower BOT-2 balance scores, and increased time to complete the TUG. The neuromuscular deficit of the reduced tibialis anterior rate of muscle activation was also noted in ALL CCS compared to peers. Jump height was significantly correlated with clinical measures of the BOT-2 and TUG.
Jumping is essential to successfully complete many tasks of daily life, play, and sports such as ballet, soccer, volleyball, and basketball. Jump performance is often used as an indicator of athletic potential [36, 37] and requires an ability to rapidly activate the muscles for balance control and coordination. In this study, ALL CCS demonstrated deficits in the ability to achieve jump height similar to their age- and sex-matched peers. This may limit their ability to participate in the same activities as children their own age with typical development. Järvelä et al. (2010) previously identified deficits in vertical jump height in long-term survivors of ALL that were 11 to 21 years post-diagnosis [18]; however, they did not explore the relationships of the deficits in jump performance with clinical measures of extensibility, balance, and mobility.
Potential underlying mechanisms that contribute to the deficits observed in higher-level gross motor tasks such as jump performance are treatment-related side effects, including chemotherapy-induced peripheral neuropathy (CIPN), that result in sensory and motor deficits. CIPN as commonly experienced by ALL CCS limits the somatosensory information received by the CNS [2]. As a result of medical treatment, ALL CCS demonstrate loss of lean muscle mass [38, 39], loss of ankle dorsiflexion range of motion [7, 21–23], delays in cortical processing [19], and demyelination in the motor pathways [3]. Wright et al. (2017) found that the majority of ALL CCS with CIPN that were on average 11.2 + 5.7 years of age had abnormal EMG firing patterns in the anterior tibialis (76%) and the gastrocnemius muscle (94%) during walking [6]. CIPN leads to reduced ankle active and passive dorsiflexion range of motion, a common effect of ALL medical treatment [7, 21–23] that has been described as a significant correlate to jumping performance [40]. However, our results demonstrated that gastrocnemius muscle extensibility did not correlate with jump height in this sample of ALL CCS. One possible reason for the difference may be that we measured the gastrocnemius extensibility with the knee in extension, which may not fully capture the functional length of the muscle during the jumping task. Secondly, the countermovement jump utilizes the stretch-shortening cycle, which is a rapid eccentric contraction followed by a concentric contraction, and may contribute to jump height [41, 42]. At the lowest point of the jump, the knees are flexed and the ankles are dorsiflexed. Assessment and exercise interventions that aim to measure and enhance the stretch-shortening cycle, such as stretching and eccentric and concentric muscle training, could be useful in ALL CCS to improve jump performance.
Balance is an underlying control process that is necessary for smooth, coordinated movement and is essential for walking, running, and jumping. Consistent with previous observations, our findings identified that ALL CCS had poorer balance proficiency as measured by the BOT-2 balance subscale [14–17] and took longer to complete the TUG compared to age- and sex-matched peers [5, 11]. Our results also identified that time off-treatment had a positive correlation with BOT-2 balance performance. Gilchrist and Tanner (2018) reported significant balance deficits in children and adolescents during and after ALL treatment, and these deficits improved with time from completion of medical treatment. However, deficits in balance performance still persisted compared to expected age-related outcomes [14]. Furthermore, balance deficits persist into adulthood, placing ALL CCS at increased risk for falls and early frailty [7]. Previous studies in ALL CCS have examined static or dynamic balance and found deficits in both. However, both components of balance are necessary for executing higher-level motor tasks such as running and jumping [7, 44]. Balance is an important and integral part of daily activities of living. Thus, remediating these deficits early is necessary to avoid the long-term consequences as found in the ALL CCS.
We found that balance and mobility were associated with jump height. Granacher and Gollhofer (2012) suggest that measurements of static and dynamic balance control in children are independent of muscle strength [43]. Our findings of jump height deficits in this sample of ALL CCS may represent a combination of static and dynamic balance deficits in addition to deficits in the ability to generate muscle forces quickly. In numerous published studies, ALL CCS presented with decreased strength of the knee [7–10, 13] and ankle [6, 13]. Therefore, we expected that joint torques of at least one lower extremity joint would differ between ALL CCS and age- and sex-matched peers [45]. Although ALL CCS presented with lower knee and hip torques, no significant differences were detected during the countermovement jump. Therefore, the muscle strength of hip, knee, and ankle extensors may not be the sole contributor to the observed deficits in the countermovement jump height in this ALL CCS sample. Balance impairments in ALL CCS may contribute to poorer jump performance and landing control [46], which could increase the risk of injury. Hence, this study supports the continued need for exploration of jump-related assessments and exercise interventions targeting balance and concentric and eccentric muscle actions in ALL CCS.
The small sample size and absence of specific chemotherapy exposure, secondary medical complications, or measure of peripheral neuropathy in this study limit the ability to assess the effects of individual chemotherapy agents. However, it is important to note that the variety of participants and clinical presentations in this study highlights the importance of individualized assessment and intervention for the spectrum of survivorship in ALL CCS. Further studies with a larger sample size are required to comprehensively examine the neuromuscular mechanisms that contribute to gross motor performance and to address the effects of targeted therapeutic interventions.
Implications for clinical practice and future research
Identifying neuromuscular mechanisms that contribute to gross motor performance will assist in the development of evidence-based therapeutic interventions for ALL CCS, who experience long-lasting sensory and motor deficits that affect activity and participation. Methods to measure kinematic and kinetic data while performing gross motor skills are commonly used to identify movement system impairments, but they are expensive and not readily available in most pediatric clinics. However, the measurement of jump height could be used as an indicator of the ability to generate muscle forces quickly and static and dynamic balance control in clinical settings in order to monitor rehabilitation progress. Changes in jump height have practical consequences as Vescovi and McGuigan (2008) report associations between countermovement jump height as well as sprint time and agility performance among female high school and college athletes [37]. Assessment of gross motor performance, such as jumping, is also beneficial when exploring motor development skills in children and adolescents during and after completion of medical treatment for cancer [46, 47]. The results of this exploratory study highlight the importance to assess and treat the effects of balance proficiency on gross motor skills in ALL CCS. An important goal of rehabilitation is to improve ALL CCS physical activity and participation through interventions that target movements and age-appropriate skills. Physical activity is important to the development of healthy children and has been suggested to improve well-being, cardiovascular capacity, strength, bone health, and physical performance in ALL CCS [48, 49].
Conclusions
The results from this study identified that ALL CCS demonstrate neuromuscular impairments that impact jump performance, an essential childhood physical activity. Countermovement jump height was related to balance proficiency and mobility performance. Further research needs to be performed to explore intervention strategies to improve the neuromuscular mechanisms that contribute to high-level gross motor skills in ALL CCS.
Footnotes
Acknowledgments
We would like to thank all the participants and their caregivers for their valuable time.
Conflict of interest
The authors have no conflicts of interest to report.
Disclaimer
The views expressed in the submitted article are our own and not an official position of our institutions.
