Abstract
Respiratory dysfunction is a leading cause of morbidity and mortality in individuals with cerebral palsy (CP). In children and adults with CP, movement and physical function is always affected. Yet, many clinicians overlook potential for impaired movement and function of the diaphragm muscle (DIAm) in individuals with CP. Since individuals with pre-existing respiratory disorders are at greater risk for respiratory complications if they contract COVID-19, understanding potential risks to individuals with CP is important. In this review we present research on respiratory function and DIAm force generation in children with CP. We compare this clinical work to basic science research investigating phrenic motor neuron and DIAm motor unit dysfunction in an animal model with CP symptoms, the spa mouse. Finally, we integrate the clinical and basic science work in respiratory function in CP, discussing potential for individuals with CP to have severe respiratory symptoms from COVID-19.
What is the relationship between respiratory complications and the diaphragm muscle (DIAm) in individuals with cerebral palsy (CP)?
For clinicians who see and diagnose infants and children with CP, a question that frequently arises from families is “will my child be able to walk?”. Yet, for individuals with CP, respiratory dysfunction, not their inability to walk, is the greatest contributor to their morbidity and mortality [1, 2, 3, 4]. CP is a clinically recognized syndrome defined as a permanent disorder of movement or posture that occurs during the development of the fetal or infant brain [5]. The movement difficulties in individuals with CP can be classified based on the limbs that are primarily affected such as hemiplegic, diplegic, and quadriplegic CP and on the type of movement difficulty including spastic (80–85% of individuals), dyskinetic (less than 10% of individuals), and ataxic (less than 5% of individuals), though other subtypes and classifications exist [6, 7, 8, 9, 10]. However, one muscle that is often neglected when discussing impairments of movement and posture is the diaphragm muscle (DIAm). In addition to being the major inspiratory muscle for breathing, the DIAm is also critical for coughing and sneezing which are high force expulsive behaviors needed for airway protection and clearance [11, 12, 13, 14, 15]. In one registry review, 58% of children with CP had a daily cough or wheeze, 10% had obstructive sleep apnea, 40% had a cough with drinking, and 20% had abnormalities on clinical pulmonary exam [16]. Furthermore, adults with CP have a greater risk of respiratory disease than adults in the general population [17, 18]. The DIAm plays a critical role in reducing susceptibility to respiratory infections in two ways: 1) during the pharyngeal phase of swallowing, the DIAm generates a large negative intrathoracic pressure necessary to propel a food bolus through the pharynx and into the esophagus, with failure resulting in aspiration; and 2) coughing and sneezing, where the DIAm contributes to increased intra-abdominal pressure necessary to expel aspirates and phlegm from the airway [11, 19, 20, 21]. Therefore, we propose that we should educate families of children with CP that their child may also have an impaired cough, which can increase their child’s susceptibility to respiratory complications.
What complications are seen with COVID-19?
Respiratory parameters in pulmonary function testing. Total lung capacity is the amount of air in the lungs after maximal inspiration and is about 4 L to 6 L in adults with volumes varying depending on age, sex, and body composition [93, 94, 95]. Tidal volume is the amount of air inhaled and exhaled during quiet breathing. In healthy adults, tidal volume is between 400–500 ml, approximately 10% of the total lung volume [96]. Vital capacity is the maximal amount of air that can be exhaled after maximal inhalation [97]. Vital capacity is composed of the tidal volume, inspiratory reserve volume, and expiratory reserve volume. Two common measures for vital capacity are the forced vital capacity which is the maximal amount of air that can be exhaled at maximum speed and effort following maximal inspiration, and slow vital capacity which is the maximal amount of air that can be slowly exhaled following maximal inspiration. Forced expiratory volume at 1 second is the maximal amount of air that can be exhaled in 1 second following maximal inspiration [98]. Residual volume is the amount of air remaining in the lungs after maximal expiration.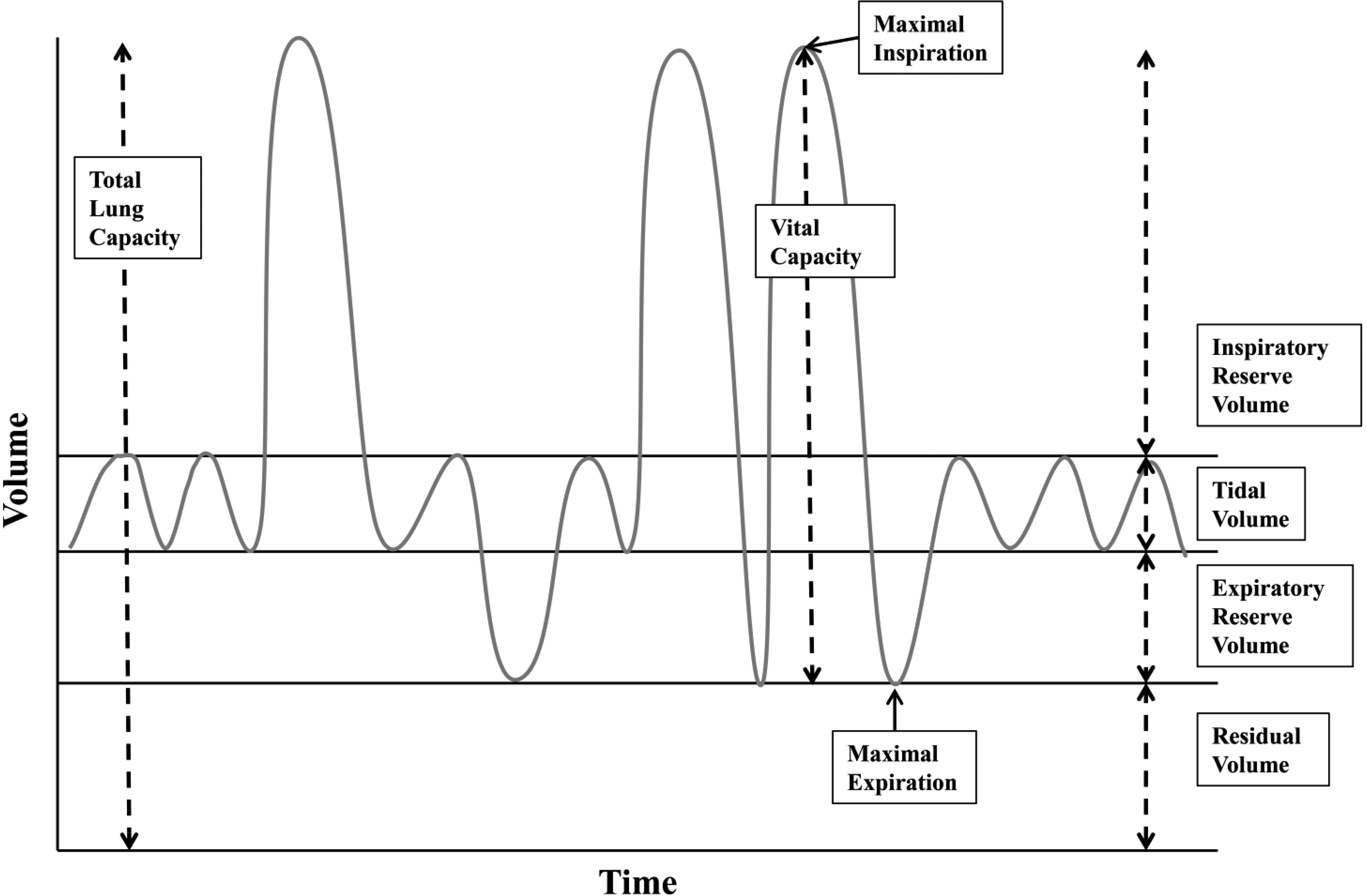
Effective and coordinated coughing and sneezing are particularly important when an individual contracts an acute respiratory illness. More recently, the general population has become aware of the devastating effects of an acute respiratory illness with the outbreak of the coronavirus SARS-CoV-2, now known as COVID-19 which was declared a pandemic by the World Health Organization on March 11, 2020 [22]. The working case definition of COVID-19 is a severe acute respiratory illness with fever and respiratory symptoms such as cough and shortness of breath. While respiratory symptoms are the most devastating, other symptoms have also been reported including gastrointestinal, cardiac, vascular, and neurologic [23, 24]. In a report modeling epidemiologic susceptibility to symptomatic infection and death, the peak age of susceptibility was found to be 60 years and older. Death from symptomatic infections was also found to rise dramatically at this age [25]. Unfortunately, the presence of pre-existing conditions such as respiratory, cardiac, or neuromuscular disorders was not included in this modeling. However, in another report, higher fatality rates were noted in those with pre-existing conditions including chronic respiratory disease, cardiovascular disease, diabetes, and hypertension [26].
Interestingly, children appear to have milder symptoms than adults, and many present asymptomatically [27, 28]. There is limited information regarding the impact of pre-existing conditions in children on symptoms or severity of COVID-19. One case series of 34 children who tested positive for COVID-19 reported that none had pre-existing conditions [29]. However, in another study describing outcomes of 48 children with COVID-19 who were admitted to pediatric intensive care units (PICU), 40 (83%) had pre-existing conditions [30]. Of those with pre-existing conditions, 21 (44%) were considered “medically complex” defined as having long-term dependence on technology and/or tracheostomy and/or developmental delay and/or genetic differences and/or having chronic lung disease [30]. Overall, 38% required intubation or tracheostomy ventilation, 13% required extracorporeal therapy (ECMO) with 4% of the children dying with 32% still hospitalized at the time this report was published [30]. While CP was not a diagnosis this study addressed, it seems likely that some in the “medically complex” group would have had CP.
Respiratory and pulmonary function
For children and adults with CP, the potential for respiratory impairment is typically considered in those with severe physical symptoms, such as those in the Gross Motor Functional Classification System (GMFCS) levels IV and V (i.e., those who function primarily from a wheelchair base) [31, 32]. Individuals with severe CP symptoms are more likely to have difficulties with sialorrhea, swallowing, recurrent respiratory infections, hypoventilation in sleep, and need for tracheostomy [33, 34]. Similarly, these same risks are prevalent in the more intensively studied population of individuals with spinal cord injury [35, 36, 37]. For these children, and many others with CP, pulmonary function testing (PFTs) cannot be reliably performed due to severity of motor dysfunction, cognitive impairment, or simply the inability to perform the testing. However, there have been some recent studies addressing pulmonary function in children with mild CP [38, 39, 40, 41]. For review, Figure 1 shows an example of respiratory parameters.
In a study involving children approximately 5 to 12 years of age, 35% with CP could not perform the task of breath holding [42]. Those with CP who could hold their breath were only able to do so for less than half the amount of time as typically developing children, likely related to a significantly smaller inspiratory volume [42]. Furthermore, the ability to breath hold and the volume of inspired air was further impaired in children with choreoathetoid CP [42]. In a study of children with hemiplegic and diplegic CP (GMFCS I-III, mean age
DIAm pressure generation
Activation of the DIAm serves to create a negative intra-thoracic pressure and a positive intra-abdominal pressure, with the trans-diaphragmatic pressure (
Diaphragm motor unit development timeline in rodents. Phrenic motor neuron (PhMN) and diaphragm (DIAm) developmental timing in rodents. Rodent gestation is approximately 18–20 days. PhMN development begins around embryonic day 10 (E10), with the phrenic nerve contacting the DIAm around the same time. For the DIAm, myoblasts form around E12, rapidly transitioning to immature embryonic and neonatal muscle fibers with emergence of Type I and IIa muscle fibers. Type IIb and/or IIx DIAm fibers are the last to emerge and do so around postnatal day 14 (P14). Type I, IIa, IIb, and/or IIx are the complement of fiber types present by maturity. 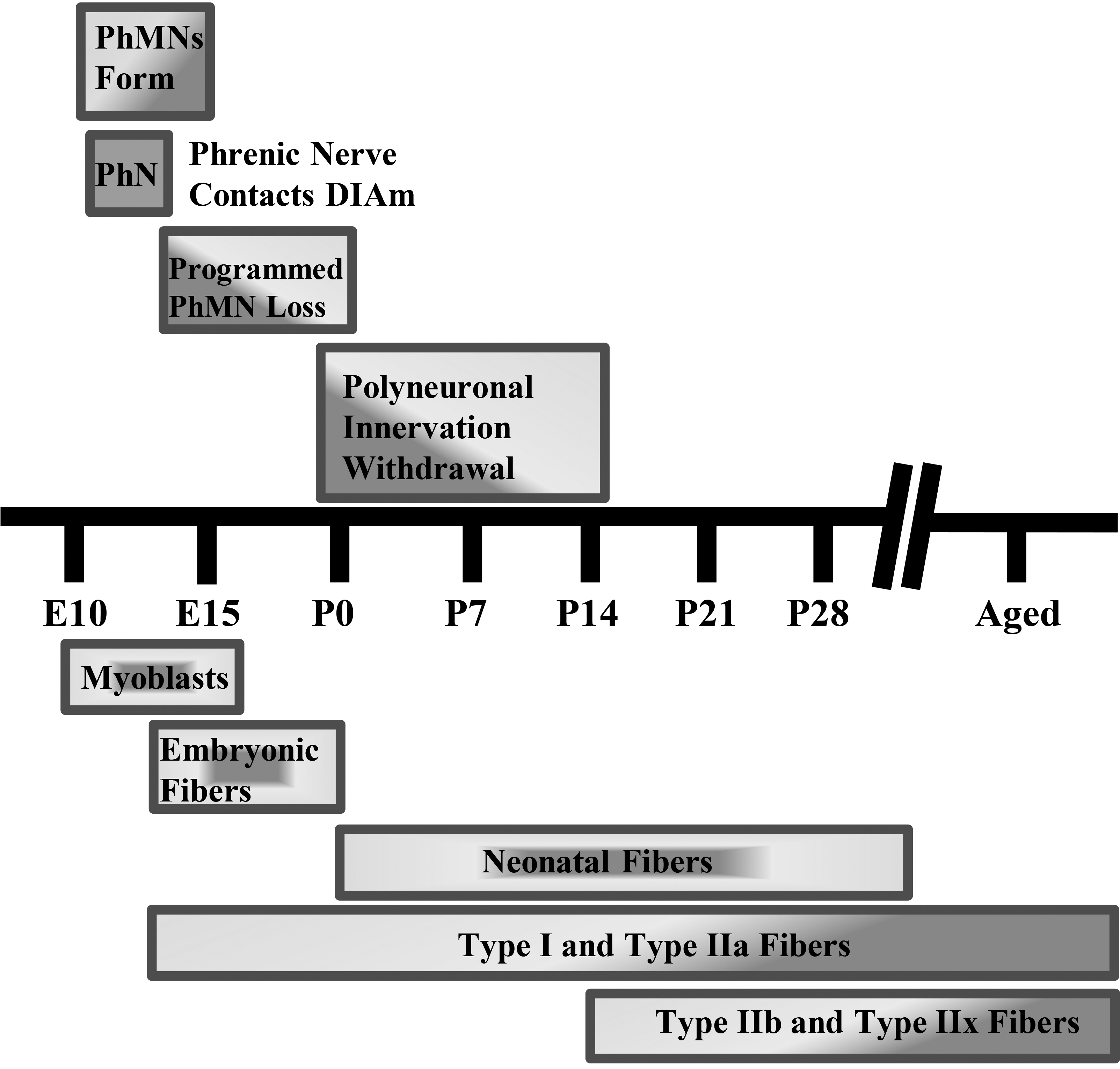
The DIAm is not only important for sustaining ventilation and intermittent coughing and sneezing, it also plays an important role in swallowing, preventing aspiration, and reducing the risk of aspiration pneumonia. Swallowing is a complicated process that includes precise coordination of muscles from the mouth to the DIAm. During swallowing, the DIAm is important in generating negative intrathoracic pressure to facilitate propulsion of a food or liquid bolus. Peak DIAm activation during swallowing appears to occur just prior to the start of the pharyngeal phase [55]. In anesthetized, spontaneously breathing cats, negative esophageal pressure (due to DIAm activation) increases by over 40% during the pharyngeal phase of swallowing [19]. Prior to phrenic motor neuron and DIAm activation during swallowing, there is increased activation of inspiratory pre-motor neurons in the dorsal medulla of the brain in anesthetized, spontaneously breathing cats [20]. Thus, the negative intrathoracic pressure generated by the DIAm contributes to the rapid movement of the bolus of food or water from the mouth to the esophagus. If a bolus, portion of a bolus, or saliva is inadvertently aspirated, a reflex is triggered that causes a sudden deep inspiration followed by a forceful cough to clear the irritant (i.e., the aspiration reflex) [56]. The DIAm is the key muscle in the aspiration reflex involved with both the deep inspiration and forceful cough. In individuals with CP, if DIAm neuromotor control and force generation is impaired, then the efficacy of the aspiration reflex is also impaired, with dire consequences. An effective cough is not only critical for airway clearance in aspiration, but also at times of respiratory infection. Thus, without an effective cough, one is at greater risk of respiratory infection and when a respiratory infection occurs, impaired airway clearance potentially results in a prolonged course of recovery.
What are the mechanisms for DIAm impairment in CP?
DIAm and phrenic motor neurons during development
For the vast majority of individuals with CP, the developmental injury or abnormality that results in physical impairments occurs in utero or in the early postnatal period. The timing of this developmental injury or abnormality is not only when the brain and spinal cord are vulnerable to disruption, but also motor neurons and motor units, including phrenic motor neurons and DIAm motor units [57, 58, 59, 60, 61, 62, 63, 64]. In fact, in the original description of CP, it was termed a “cerebro-spinal disorder” in acknowledgement of the importance of motor units to the etiology and pathophysiology of CP [65, 66].
In mammals, the absolute number of motor neurons in the spinal cord reaches a maximum at commencement of the third trimester, after which a process of developmental loss occurs into the early perinatal period until the optimal number of motor neurons is achieved (Fig. 2) [59, 60, 64]. However, changes in inputs to motor neurons during this developmental period can dramatically alter the final number of motor neurons [67, 68, 69, 70, 71]. During the perinatal period and into maturity, motor neurons, including phrenic motor neurons, undergo differential growth which appears to correspond with the emergence of motor unit diversity [62]. The differential growth of motor neurons is necessary as a range of motor neuron sizes are required to carry out different motor behaviors and provide for an orderly, graded recruitment of different motor unit types (Fig. 3).
Diaphragm motor unit properties. A full complement of motor units is required for the diaphragm (DIAm) to effectively perform activities ranging from eupneic breathing to cough/Valsalva (A).Smaller motor neurons innervate the fatigue resistant Type I and IIa muscle fibers creating slow (S) and fatigue resistant (FR) motor units needed for sustaining eupnea (A,B). Larger motor neurons innervating Type IIx and IIb muscle fibers form the fatigue intermediate (FInt) and fast fatigable (FF) motor units, respectively (B). FInt and FF motor units are recruited for high force behaviors, such as those needed for an effective cough (A). In animal models of CP, there are fewer larger phrenic motor neurons with reduced specific force generation, which coincides with reduced DIAm pressure generation in humans with CP, suggesting impairment of high force behaviors (A).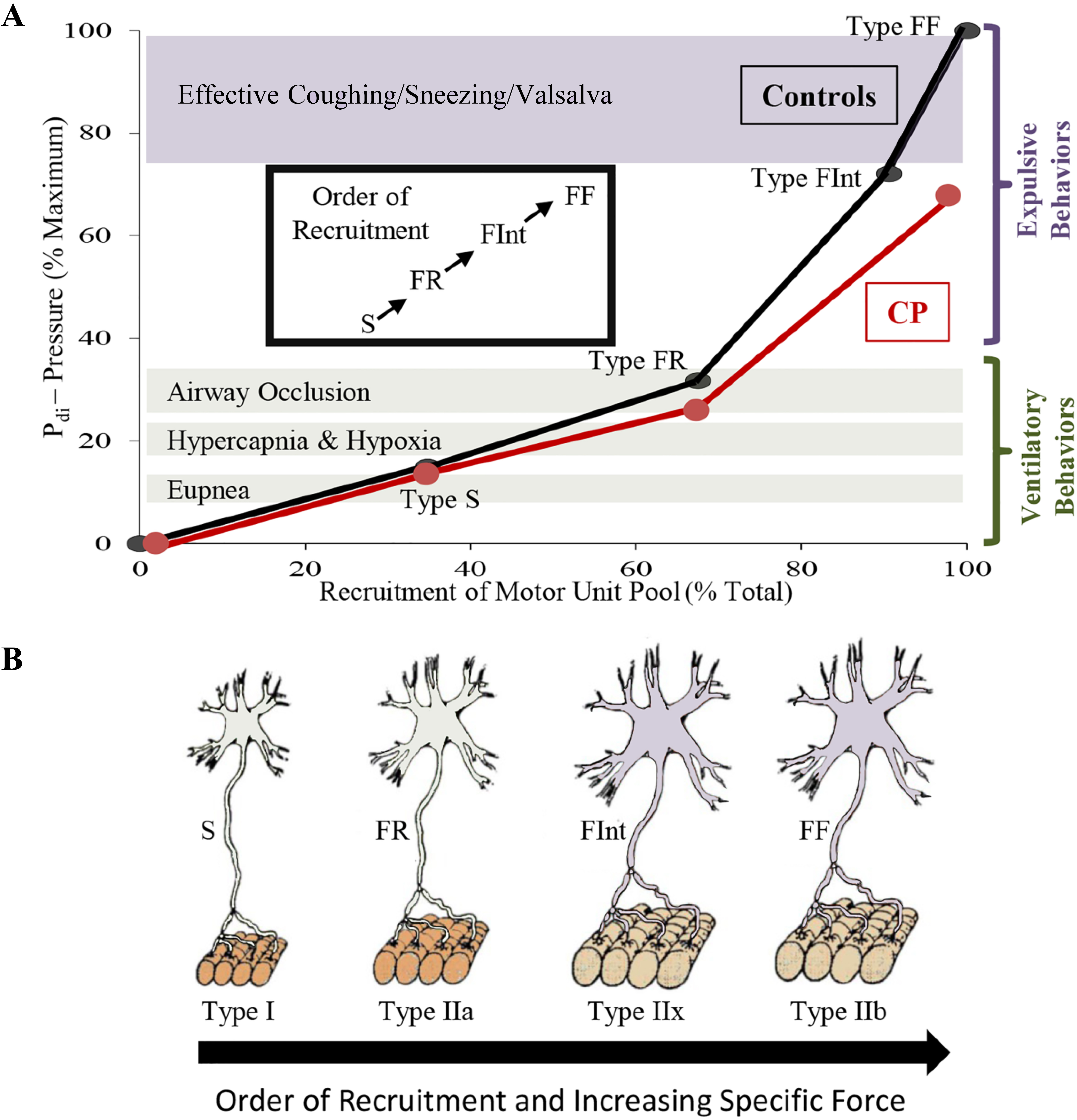
Coinciding with motor neuron development, myoblasts transition to myotubes and finally to immature (embryonic) muscle fibers (Fig. 2). Innervation of DIAm by the phrenic nerve appears very early during myoblast migration and myotube formation. Initially several phrenic motor neurons contact a single developing DIAm fiber (polyneuronal innervation); thus, developing DIAm fibers are influenced by several motor neurons (neurotrophic influence) (Fig. 2). The developmental loss of motor neurons coincides with the loss of polyneuronal innervation of muscle fibers such that by the second postnatal week in rodents, each muscle fiber is innervated by only a single motor neuron (Fig. 2) [64, 72]. During the perinatal period and into maturity, muscle fiber type transitions from embryonic to neonatal to adult fiber types with the emergence and growth of more fatigable fast twitch (type IIx and/or IIb) muscle fibers being the final event to occur [64, 72, 73, 74]. In the adult DIAm, type I, IIa, IIx and/or IIb fibers are all present in varying proportions depending on species. Type I and IIa DIAm fibers comprise fatigue resistant slow (type S) and fast (type FR) motor units, respectively, which are innervated by smaller, more excitable phrenic motor neurons (Fig. 3). These fatigue resistant type S and FR DIAm motor units are recruited for inspiratory behaviors including eupnea and breathing stimulated by hypoxia and hypercapnia (Fig. 3). Type IIx and/or IIb DIAm fibers comprise fast fatigue-intermediate (type FInt) and fast fatigable (type FF) motor units, which are innervated by larger, less excitable phrenic motor neurons. These more fatigable type FInt and FF DIAm motor units generate greater specific force (force normalized for muscle fiber cross-sectional area) and are only recruited to generate the higher pressures required for effective coughing, sneezing or Valsalva-type maneuvers that are typically of short duration (Fig. 3) [64, 75].
Properties of motor neurons and their motor units have not been well studied in CP, in part due to the lack of animal models with physical symptoms similar to individuals with CP [66]. A second major barrier has been the enormous animal husbandry hurdles that have to be overcome in some of the perinatal injury/hypoxia animal models, which unlike the majority of individuals with CP have very poor survival with death before adolescence [66]. While animal models of CP do not completely recapitulate human disorders, they can provide insight regarding mechanisms underlying the symptoms of CP.
Although CP is a neuro-developmental disorder, there is only one study that has estimated the impact of CP on the final number of motor neurons in adults. In this study, an electrophysiological technique, the Motor Unit Number Index (MUNIX), was used to estimate the number of motor units in the hypothenar muscle of adults with and without CP [76]. Interestingly, this study estimated that there are 21% fewer hypothenar muscle motor units in adults with CP compared to adults without CP (168
Recently, we investigated motor neuron loss and motor unit properties in a mouse model of CP symptoms, the spa mouse. The spa mouse has a homozygous insertion of LINE-1 in the beta subunit of the glycine receptor gene resulting in a splicing error of this subunit [79]. The spa mouse is smaller in size than wild type and heterozygote littermates, and most importantly, it develops symptoms of CP such as hypertonicity and exaggerated startle response by 2–4 weeks of age [80]. This developmental period coincides with the postnatal emergence of symptoms in humans with CP. In a recent study evaluating the number of motor neurons innervating the tibialis anterior muscle in adult spa mice, we found 61% fewer motor neurons compared to wild type mice [81]. In addition, the surviving motor neurons in spa mice were smaller, reflecting a disproportionate loss of larger motor neurons [81]. In another recent study, we found that there are
In both the tibialis anterior muscle and DIAm in spa mice, we found that there was increased susceptibility to neuromuscular transmission failure compared to wild type mice [83, 84, 85]. With neuromuscular transmission failure, the neural signal from the motor neuron fails to evoke muscle fiber contraction, a form of fatigue [86]. In these studies, we found that in spa mice neuromuscular transmission failure in the tibialis anterior and DIAm was apparent even during initial nerve stimulation, and that it worsened during repeated nerve stimulation, especially at higher frequencies of stimulation [83, 84, 85]. Also, these studies also showed that there were no differences in the total number of muscle fibers in the tibialis anterior muscle [83] or in the DIAm (preliminary observation) in spa mice, and that each muscle fiber is innervated (i.e. neuromuscular junctions are present). Thus, with fewer motor neurons and the same number of innervated muscle fibers, the innervation ratio (i.e., number of muscle fibers innervated by a single motor neuron) was greater for both tibialis anterior and DIAm in spa mice. An expanded innervation ratio suggests that the increased susceptibility to neuromuscular transmission failure in spa mice reflects a failure of action potential propagation at the increased number of axonal branch points [86, 87, 88, 89].
In preliminary work, we found that maximum DIAm specific force is reduced by
The reduction in the number of phrenic motor neurons in spa mice is similar to the age-related loss of phrenic motor neurons observed in rats. Old rats (24 months old) have 22% fewer phrenic motor neurons with the surviving phrenic motor neurons being 19% smaller compared to young adult rats (6 months old) [90]. In congruence with the tibialis anterior muscle and preliminary DIAm studies in spa mice, [83, 84, 85] old rats display increased failure of neuromuscular transmission upon initial stimulation and greater failure of neuromuscular transmission following repetitive phrenic nerve stimulation [91]. Old rats also showed a 22% reduction in the apposition of pre-synaptic terminals at the post-synaptic endplate of neuromuscular junctions innervating type IIx and/or IIb DIAm fibers compared to type I and IIa fibers [91]. This suggests that in old rats the loss of larger phrenic motor neurons results in denervation of type IIx and/or IIb fibers. In contrast, in spa mice, the extent of apposition of pre- and post-synaptic elements of neuromuscular junctions was not different in type IIx and/or IIb DIAm fibers when compared to wild type. Thus, the loss of phrenic motor neurons may occur much earlier during the perinatal period before the emergence of type IIx and/or IIb fibers (Fig. 2). In the DIAm of older rats, the increased susceptibility to neuromuscular transmission failure may also reflect increased axonal branching resulting in failure at branch points, and also a disturbance at the neuromuscular junction. Despite the loss of phrenic motor neurons and impairment in neuromuscular transmission, the
Conclusions
Children with CP, regardless of the severity of their locomotor symptoms, have impairments in respiratory function due to impaired DIAm pressure generation that continue into adulthood. In an animal model of CP symptoms, there are fewer large phrenic motor neurons which comprise more fatigable DIAm motor units, and are required for airway clearance behaviors such as coughing and sneezing. In addition, neuromuscular transmission at these more fatigable motor units is impaired further limiting the performance of these more forceful behaviors of the DIAm. Thus, this animal work provides insight to the pathophysiology of impaired respiratory function in individuals with CP. Interestingly, the pattern of a loss of larger motor neurons in concert with reduced effectiveness of neuromuscular transmission in the spa mouse model of CP symptoms has a remarkable resemblance to observations with aging. Advanced age and pre-existing respiratory comorbidities are significant risk factors for respiratory complications from COVID-19. Thus, if an individual with CP contracts COVID-19 and develops respiratory symptoms, they should be counseled that they could be at higher risk of progression to severe respiratory symptoms. In order to reduce the risk of contracting COVID-19, we need to be vigilant in encouraging and supporting families of children with CP and individuals who have CP to practice physical distancing, good hand hygiene, minimizing trips to public places such as stores and restaurants, and thorough cleaning of any equipment that is used outside the home (i.e. walkers and wheelchairs).
Footnotes
Acknowledgments
This work was supported by National Institutes of Health grants R01-AG044615 (GCS), R01-HL146114 (GCS), an Australian National Health & Medical Research Council CJ Martin Early Career Fellowship (MJF), and Mr. and Mrs. Richard and Rosemary Crandall (JEB).
Conflict of interest
The authors have no conflicts of interest to report.
