Abstract
PURPOSE:
The purpose of this study was to create a clear, standardized test description to rate spasticity severity into four categories according to the definition given by Lance [1], referred to as the Velocity Dependent Measure of Spasticity (VDMS).
METHOD:
Muscle groups of the upper and lower limbs of children with neuromotor disorders were evaluated on their response to passive movement in a fast-versus slow-velocity test condition. The interrater and test-retest reliability were assessed using Gwet’s alpha one (95%-CI) and the percentage agreement.
RESULTS:
Two physiotherapists independently assessed 45 children and youths (age 4–19 years). The interrater reliability of the VDMS was substantial to almost perfect (Gwet’s alpha one: 0.66–0.99,
CONCLUSION:
The VDMS can be recommended as a reliable assessment with a standardized procedure to assess spasticity of the extremities in children with neuromotor disorders.
Introduction
Spasticity is one of multiple physical signs of upper motor neuron syndromes. It is also called spasticity ‘sensu strictu,’ and is described as involuntary muscle hyperactivity triggered by rapid, passive joint movements [2]. As spasticity influences motor function in children with cerebral palsy (CP) and other neuromotor disorders, its management is critical [3, 4, 5].
Therefore, assessments with clear descriptions of how to conduct and rate spasticity are needed in clinical practice. Assessments for children should be practical and efficient to perform so that the test itself is not arduous for the children. The Hypertonia Assessment Tool (HAT) is an assessment that discriminates between the hypertonia subtypes of spasticity, dystonia, and rigidity in children with neuromotor disorders [6]. The HAT characterizes spasticity as a velocity-dependent resistance of a muscle to stretch [7] and highlights that other factors such as a child’s state of alertness, activity, posture, pain, surface contact, or other non-noxious sensory inputs have to be considered when assessing spasticity. For detection of spasticity in all four extremities of children with CP and other neuromotor disorders, the validity of the HAT was considered moderate to good (0.57–0.74) [6, 8]. However, it lacks a scale for rating the severity of spasticity.
In a recent review of spasticity measures in children and adolescents with CP, three groups of assessments were included: 1) clinical assessment tools (Ashworth and modified Ashworth Scale, Tardieu and modified Tardieu Scale, HAT, and the Composite Spasticity Scale) 2) biomechanical assessment tools (Myotonometer, Wartenberg pendulum test, three-dimensional pendulum test, dynamometry, measures using goniometry, inertial sensor, and stiffness tool with robotic-assisted gait orthosis) and 3) neurophysiological assessment tools (electromyography, tonic stretch reflex testing, and the Hoffmann reflex of soleus muscle) [9]. They concluded that there is insufficient psychometric evidence for all of these spasticity measures. While the authors stated that neurophysiological tools provided the most promising results, they questioned the accuracy of the information derived from ordinal-scaled clinical assessments [9]. While these assessments theoretically might be less sensitive to change over time, for example, due to an intervention (i.e., responsiveness), such assessments often provide quick and easy testing in clinical settings. In comparison, implementing neurophysiological measures like electromyography in clinical practice is time consuming, needs special equipment and knowledge, and there remain various factors influencing reliability [10].
In a systematic review, they divided the existing clinical spasticity assessments into three groups [11]. One group of assessments used non-specific velocity to assess spasticity using the Ashworth scale. Another group of assessments referred to as “Tardieu-like scales” used the joint-angle measurement at the onset of resistance to measure spasticity. A third group used a combination of Ashworth- and Tardieu-like scales. They concluded that only the original Tardieu Scale is an appropriate assessment of spasticity in children with CP. However, it is time consuming and lacks standardization of velocities [12]. Further, the absolute measurement errors of the joint angles are high in test-retest and interrater reliability [13]. Regarding the psychometric properties of the Ashworth scales, both the original and modified version have low content validity as they do not test velocity-dependent changes of muscle tone [16], and reliability is also weak [14]. Both the Ashworth and Tardieu test have insufficient descriptions of the testing procedures, rating scales, and instructions [15]. Hence, they were applied in many different ways which negatively affected their reliability and ultimately made their clinical benefit questionable [10].
These reviews of the existing literature demonstrate the need for a test procedure which utilizes both fast and slow passive movement in its administration so that it addresses the commonly accepted definition of spasticity [2]. It should be accompanied by a clear standardized description of the testing procedure as well as consecutive rating.
Therefore, in this study the modified Ashworth Scale and the HAT were adapted and combined to create a scale quantifying spasticity of the upper and lower extremity with improved standardization of the assessment and scoring procedure. This adapted assessment was named the Velocity Dependent Measure of Spasticity (VDMS). In addition, the interrater and test-retest reliability of the VDMS was determined.
Methods
The VDMS is composed of the test procedure described in the test manual of the HAT to test spasticity as well as a rating scale that is an adaptation of the Modified Ashworth Scale [6, 17].
A slow and fast movement velocity was defined in accordance with the definition of spasticity [2]. First, the tested limb is moved slowly (slow stretch) two times until the end range of motion is reached. These movements in combination last 4 to 5 seconds, depending the degree of range of motion. Second, the tested limb is moved two times at a fast speed (fast stretch) until the end of the range of motion is reached. The two fast stretch movements in succession last less than one second in total and are repeated after a pause of at least 5 seconds to feel the resistance of the antagonized muscles. Then, the assessor rates the difference in perception of resistance between the slow and fast passive stretches. For example, if a rater perceives the resistance in the fast, passive stretch to be the same as the resistance in the slow, passive stretch, spasticity is rated as zero which indicates that there is no spasticity. Based on the literature and on clinical experience, four categories were defined: 0) no spasticity; 1) mild spasticity; 2) moderate spasticity; 3) severe spasticity (Table 1). This clear definition, and test procedure supports the assessor in rating the spasticity. The categories were first described in German and then translated into English.
Velocity Dependent Measure of Spasticity: Definitions of categories in comparison to the modified Ashworth Scale
Velocity Dependent Measure of Spasticity: Definitions of categories in comparison to the modified Ashworth Scale
Abbreviations: VDMS – Velocity Dependent Measure of Spasticity; MAS – modified Ashworth Scale.
This was a cross-sectional study with repeated assessments. The VDMS was performed three times on each child. On day one, both raters performed the assessment in order to determine interrater reliability. Rater order (rater A second author, rater B first author) was randomized. The VDMS was performed within a one-hour time window after the lunch break with at least a ten-minute break between the two assessments. One of the raters repeated the assessment one week later at the same time of day and in the same room. Due to practical reasons, the decision of which rater would perform the third assessment was not randomized. Twenty minutes were scheduled to perform this assessment. This design ensured that the assessments were performed independently from each other. The raters were physiotherapists with more than 5 and 10 years of experience treating children with neuromotor disorders.
Participants
Children were recruited from the Swiss Children’s Rehab of the University Children’s Hospital Zurich. Inclusion criteria were: diagnosed as having a neuromotor disorder, aged four to 19 years, the ability to follow simple instructions (such as closing and opening the eyes), and the ability to lie in supine position for 20 minutes. Participants were excluded from the study if they had received surgery within the last three months, changed medication (i.e., muscle relaxants) during the measurement period, or were not able to be moved passively. Parents and juveniles aged 14 years and older signed the informed consent form. The study was approved by the ethics committee of the canton of Zurich (KEK-ZH-Nr. 2011-0404) and it complied with the declaration of Helsinki guidelines of good clinical practice. According to Temel et al. [18], a minimum sample size of 30 participants is required (difference between of the overall agreement probability and the chance-agreement probability 0.10, relative error 0.10). If a drop-out rate of 10% was assumed, at least 33 children and adolescents needed to be recruited to adequately power this study.
Assessment procedure
The VDMS was applied in the same test situation as the HAT. The patients were lying in supine position with their heads supported by a pillow and their knees by a positioning roll or a rolled towel. The extremities were tested in a random order. Seven muscle groups were tested per limb moving from proximally to distally. For the upper extremity: shoulder adductors, elbow flexors, elbow extensors, forearm pronators, forearm supinators, wrist flexors, and wrist extensors. For the lower extremity: hip adductors, hip flexors, hip extensors, knee flexors, knee extensors, ankle plantar flexors, and ankle dorsal extensors.
Example of the interpretation of the Velocity Dependent Measure of Spasticity categories. If the passive range of motion (pROM) of knee extension is 120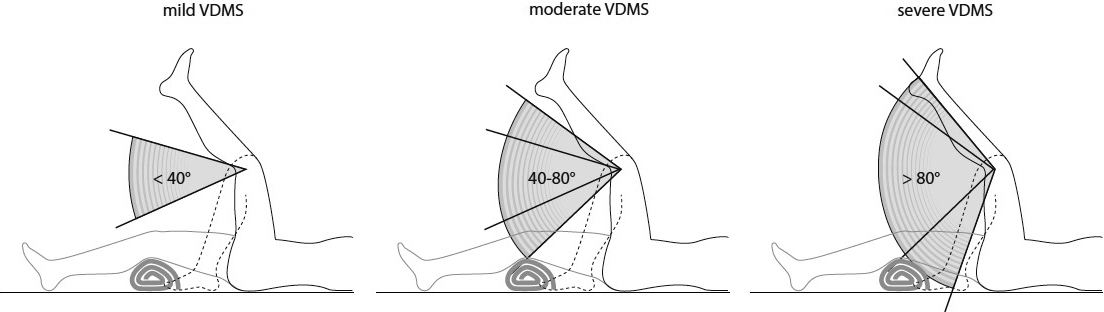
The test procedure defined by Jethwa et al. [6] was followed: “Support the limb against gravity. Move the joints of the limb throughout the child’s full range starting with the joint in full flexion or adduction, moving to full extension or abduction, and then returning to flexion or adduction, twice slowly and twice as quickly as possible.” For the upper extremity: “shoulder adduction and abduction – begin with shoulder in full adduction; elbow flexion and extension – begin with elbow in full flexion; forearm pronation and supination – begin with forearm in full pronation; wrist flexion and extension – begin with wrist in full flexion.” For the lower extremity: “hip adduction and abduction – begin with hip in full adduction; hip flexion and extension – begin with full extension; knee flexion and extension – begin with knee flexed with the hip in 90
For some muscle groups, a low occurrence of spasticity was expected based on the results of the reliability study for the HAT [8]. Therefore, Gwet’s first-order agreement (Gwet’s AC1) with 95 confidence intervals (95%-CI) was applied, as this statistical method was shown to be less influenced by prevalence [19, 20]. Gwet’s AC 1 was interpreted as follows:
As previously mentioned, children with different neuromotor disorders were included, as they were representative of those treated in our rehabilitation center. As noted in a previous study, children showed signs of spasticity in at least one muscle group in 57% of the left upper limbs, 42% of the right upper limbs, 76% of the left lower limbs, and 59% of the right lower limbs [20]. Given our patient population, a lower occurrence of spasticity in certain muscle groups and a higher occurrence in others was expected.
Results
Forty-six children (32 boys, 16 girls) were recruited for this study. Due to the illness of one of the raters, one child (GMFCS Level IV; spastic CP) performed only a single measurement. Data from this child were excluded from the analysis. Ages ranged from 4y 2mo to 18y 10mo. Twenty-seven of the children used a wheelchair for mobility and 19 were ambulatory. Additional patient characteristics of all 46 children are presented Table 2. The GMFCS Levels of the 22 children with spastic CP were: level I,
Patient characteristics
Patient characteristics
Number, age and WeeFIM values apportioned by the four categories of diagnosis. The maximal WeeFIM mobility score is 35 points; the maximal WeeFIM total score is 126 points. Abbreviations: CP – cerebral palsy; y – years; mo – months; SD – standard deviation; WeeFIM – functional independence measure for children.
The VDMS was applicable for all children. It took 7–12 minutes to assess all four extremities in each child. No pain or adverse event occurred during the VDMS testing. Figure 2 shows the frequencies of the VDMS categories (no, mild, moderate, and severe) per tested muscle group and participant (x-axis) from rater A and rater B.
Frequencies of Velocity Dependent Measure of Spasticity categories per participants (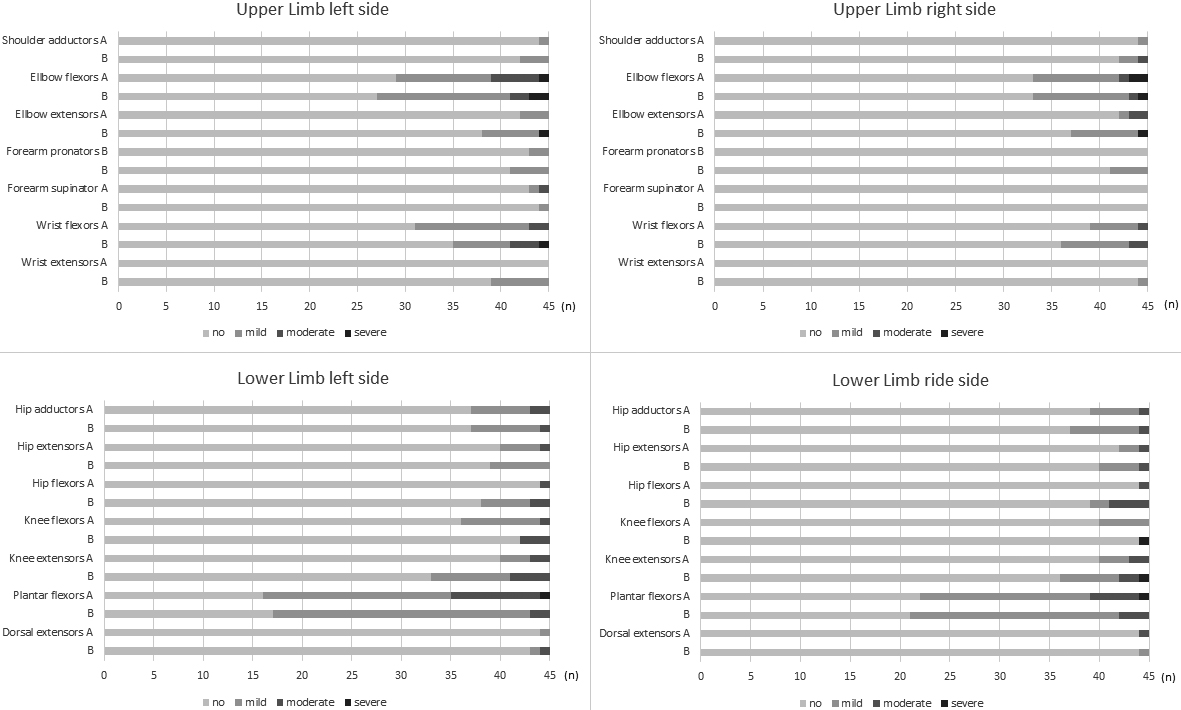
The interrater reliability results are based on data from 45 children who were tested on the same day by rater A and B. The Gwet’s AC1 including the 95%-CI were ‘almost perfect’ for all muscle groups of the upper extremity except the elbow flexors which were rated as ‘moderate’ and the wrist flexors which were ‘substantial’ (Table 3a). The percentage agreements were in line with the Gwet’s AC1.
Interrater and test-retest reliability of the Velocity Dependent Measure of Spasticity
Interrater and test-retest reliability of the Velocity Dependent Measure of Spasticity
a) % Agreement and Gwet’s alpha one coefficient between raters A and B of two consecutive assessments;
Comparable results were found for the lower extremity with ‘almost perfect’ agreements for all muscle groups except the knee extensors which were rated as ‘substantial’ and the plantar flexors which were ‘moderate’ (Table 3a).
Due to practical circumstances, two children could not be measured a second time. Rater A tested 28 children twice, and rater B tested 15. One child had to be excluded due to change of medication, therefore 42 children were included in this analysis.
The percentage agreement and the Gwet’s AC1 including the 95%-CI showed ‘almost perfect’ agreements for all muscle groups of the upper extremity except for the wrist flexors of the left side which showed ‘substantial’ agreements (Table 3b). Likewise, in the lower extremity for all muscle groups ‘almost perfect’ agreements were found except for the plantar flexors which showed ‘substantial’ agreements on both sides (Table 3b).
Discussion
The aim of this study was to determine the reliability of the ordinal-scaled VDMS. This is a standardized, clearly described measure to rate spasticity in children with neuromotor disorders. The test procedure followed the description of velocity-dependent increase in muscle resistance to passive stretch. The interrater as well as the test-retest reliability showed ‘substantial’ to ‘almost perfect’ agreement for all muscle groups included in the test protocol. One explanation for these favorable results could be that a clear description of the testing procedure along with the interpretation of the results to rate the spasticity severity level was provided.
Previous studies used different statistical analyses for quantifying the reliability of the various spasticity assessments. Therefore, a comparison of the results is challenging since they are dependent on the distribution of the data and the applied statistical method. Previous studies often used Intraclass Correlation Coefficients (ICC) even for ordinally-scaled spasticity assessments. Flamand and colleagues [21] compared the interrater reliability of spasticity measurement tools applied in children and youths with CP: the interrater ICCs of the Ashworth Scale varied between 0.54–0.80, while ICCs for the modified Ashworth Scale were between 0.27–0.87. For the Tardieu scale, the percentage agreement of the measured angles (elbow, knee, and ankle) was 42%–100%. The ICC lay between 0.22–0.74 for the modified Tardieu scale. The interrater reliability of the Australian Spasticity Assessment Scale (ASAS), another ordinal scale to assess spasticity, was quantified with the weighted kappa and percentage agreement, and showed similar results compared to our findings [21].
In a previous study the agreement between the two assessors for the occurrence of a spastic catch (item 4 of the HAT) was low [8], while in the current analysis of the VDMS the agreement was high. The description of item 3 of the HAT was used to assess spasticity: “Velocity Dependent Resistance to Stretch” [8]. It was assumed that differentiating between the appearance of a ‘spastic catch’ and ‘no appearance of a spastic catch’ is more challenging than rating the difference in resistance that occurs when applying slow versus fast passive stretch. In the descriptions of the VDMS categories, the term ‘catch’ is not included, which is in line with the definition of spasticity: namely involuntary muscle hyperactivity triggered by rapid, passive joint movements [2]. Both the Modified Ashworth Scale and the ASAS include ‘spastic catch’ in their rating system as well [17, 21].
Study limitations
The agreement between the two assessors testing a child on the same day was lower for some muscle groups (elbow flexors, wrist flexors, and plantar flexors) than the test-retest reliability (testing the child by the same rater with one week in between). Several explanations could account for this. As both raters used the VDMS for the first time in this study, more experience in using the tool and discussions between raters might be needed to increase the agreement. Another reason could be the influence of the first test on the findings of the second test that followed ten minutes later. The first assessment could already induce a reduction of the spasticity level due to mobilization of the muscle groups. This is supported by the finding of a significantly lower VDMS score during the second test compared to the first in three muscle groups (the elbow extensors, the knee flexors, and the knee extensors of the left side) (Bland-Altman test with Wilcoxon signed rank test; bias [i.e., systematic difference] of 0.05, 0.21, and 0.24 respectively. See supplementary data, Appendix II). For the other muscle groups, the biases were low and not significant. The disparity between rater A and B was greatest for VDMS categories “mild” and “moderate” in the elbow flexors and plantar flexors (Fig. 2). It was assumed that the tested resistance was close to one-third of the passive range of motion.
Despite the heterogeneity of the group of patients included, which reflects the patient population in our rehabilitation center, we did not find spasticity in several muscle groups (e.g. shoulder adductors, forearm pronators, forearm supinators, and dorsal extensors). From a statistical point of view, this has to be considered a limitation. It would be desirable to include a sample of children with higher levels of spasticity in these muscles. From a clinical point of view, it is important to discriminate between the hypertonia subtypes (spasticity, dystonia, and rigidity) and exclude high resistance to passive stretch caused by non-contractile structures (22–25). Therefore, finding a good agreement in the absence of spasticity is also a valuable result and indicates the ability to avoid an overestimation of the prevalence of spasticity.
It is apparent that the VDMS can be easily integrated when using the HAT, which is advantageous to determine not only the category of hypertonia but also the severity of spasticity in the different muscle groups.
Conclusion
The VDMS is a well-described and reliable assessment procedure available to score spasticity in various muscle groups. The VDMS is easy to apply in children starting at age four presenting with various neuromotor disorders and heterogeneous sensory-motor as well as cognitive impairments. Further investigation is indicated to substantiate its contribution.
Footnotes
Acknowledgments
The authors wish to thank the children and their parents for their participation in this study. The support of the physiotherapists and all the members of the pediatric research team was also highly appreciated, notably Jeffrey Keller for proofreading the manuscript.
This work was in part sponsored by a grant from the Mäxi-Foundation. We further acknowledge the Zurich Center for Neuroscience (ZNZ).
Conflict of interest
The authors have no conflict of interest to declare.
Supplementary data
Appendix I. User Manual and Score Sheet for the Velocity Dependent Measure of Spasticity (VDMS).
The children in the pictures of the user manual are not patients and were not involved in the study. The children and parents have given their consent for the photos to be used for the test sheet in the user manual.
Appendix II. Wilcoxon signed rank test of the first versus the second measurement and the bias with upper limit of agreement and lower limit of agreement.
Appendix III. STROBE Statement-checklist of items that should be included in reports of observational studies.
The supplementary files are available to download from http://dx.doi.org/10.3233/PRM-200704.
