Abstract
Spina bifida is a birth defect that commonly causes bowel and bladder dysfunction in children with a significant negative impact on quality of life and emotional wellbeing. Fecal continence improves satisfaction and the quality of life of both children and their caretakers. Bowel management in children with spina bifida is hampered by limited controlled studies and variable practice within different institutions and subspecialists. The goals of a successful bowel management program in children with spina bifida consist of predictable bowel movements, social continence, and eventual independence. Treatment options range from conservative interventions such as diets and oral laxatives that modify stool consistency and transit, to trans-anal irrigations and antegrade continence enemas that facilitate predictable recto-sigmoid emptying and provide a greater degree of independence. In children, the treatment approach should be implemented in the context of the child’s developmental age in order to allow for optimum social integration with their age-appropriate peers. We present a review of a stepwise approach to bowel management in children with spina bifida and the challenges related to the proposed treatment options.
Introduction
The live born prevalence of spina bifida worldwide ranges from 33.8 to 48.4 per 100,000 births [1]. Bowel and bladder dysfunction affects approximately 76% of both children and adults with spina bifida and has a negative impact on their quality of life [2, 3, 4]. Only 23% and 29% of patients with spina bifida aged 5–21 are reported to have bowel and bladder continence, respectively [5]. The impact of abnormal bowel function on quality of life and social activities increases with age [6, 7]. Approximately 60–86% of children with spina bifida are dependent on caretakers for evacuation of their bowels [8, 9]. Parents perceive bowel and bladder dysfunction to be more stressful than motor disability [8].
Abnormalities in the interaction between the extrinsic and intrinsic innervation, colorectal anatomy, sphincter and recto-sigmoid function, colonic transit, stool consistency, and sensation result in bowel dysfunction [10, 11]. Patients with spina bifida have slow colonic transit, abnormal rectal tone, compromised internal anal sphincter function, decreased sensation, and poor external anal squeeze effort [12, 13, 14, 15, 16, 17]. Such alterations of involuntary and voluntary properties of the colon and anorectum are further amplified if the child has underlying cognitive delays. Adolescents and young adults with present hydrocephalus are 9.5 times more likely to have fecal incontinence [18].
The goals of bowel management in children with spina bifida include predictable bowel movements, social continence, and independence. Independence in bowel care is critical, as those who are independent at toileting have been shown to have significantly higher quality of life scores [9]. A successful bowel program can both improve the child’s quality of life and family functioning while decreasing caregiver depression and anxiety [19, 20]. Bowel management should be implemented and modified in the context of the child’s development as well as any underlying renal, neurological, and orthopedic comorbidities.
Randomized controlled studies evaluating the efficacy of different bowel regimens in children with spina bifida are methodologically poor. The studies are limited by small sample size, variable practice amongst subspecialists and institutions, and inconsistent definitions of bowel continence. There are few tools available to assess neurogenic bowel symptoms in children with spina bifida. Two recently proposed items include the pediatric Neurogenic Bowel Dysfunction Score [21] and Adolescent Fecal Incontinence and Constipation Symptom Index [22]; however, these measures are limited by small sample size and need to be validated by larger studies.
Conservative management
The cornerstone to a successful bowel program is a strong relationship between the patient, the caretakers, and the medical team. The treatment should be tailored to achieve predictable bowel movements and social continence with the long-term goal of independence. A stepwise approach is recommended in most cases. See Fig. 1. In conjunction with behavioral modification, we can facilitate recto-sigmoid emptying as well as alter stool transit, consistency, and volume [10, 11]. With regards to behavioral modification, toilet training is an integral part of a child’s developmental and social milestones. The medical team is encouraged to provide the parents and caregivers with the appropriate resources and guidance to implement a scheduled toilet training program [23]. Based on the child’s mobility and development, then a bowel management plan can be devised. We propose a stepwise approach to the bowel management in children with spina bifida (Fig. 1) adapted from the proposed treatment of neurogenic bowel in adults [13, 24].
Bowel management protocols and techniques implemented by different centers commonly start with conservative treatments that modify the stool consistency and volume. Therapy should aim at avoiding loose and hard stool. Dietary changes in conjunction with fiber and appropriate fluid intake have been shown to improve stool consistency and increase stool frequency in otherwise healthy children [25]. Unfortunately, similar studies are lacking in children with spina bifida.
Stepwise approach for bowel management in children with spina bifida. Adapted from
the proposed treatment of neurogenic bowel in adults [13, 24]. Success at
each step is defined as a bowel program where the child has predictable bowel
movements and social continence with a long-term plan for independence.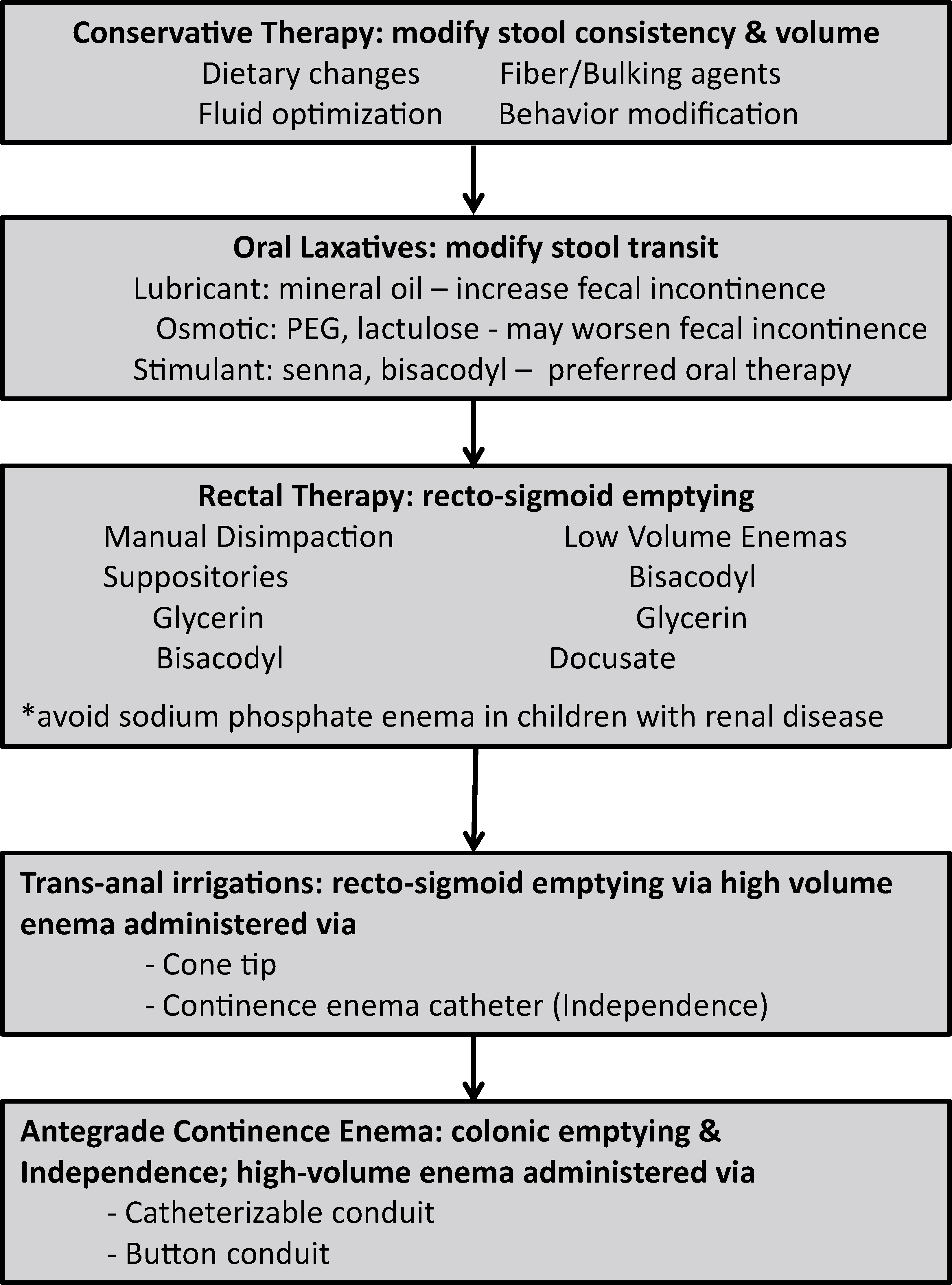
Patients with spina bifida have slow colonic transit [16, 17]. The goal is to increase colonic transit without significantly altering the stool consistency, as liquid stool consistency results in increased fecal incontinence since there is compromised sensation and voluntary ability to squeeze to withhold stool. Lubricant laxatives such as mineral oil should be avoided as they alter stool consistency and increase fecal incontinence. Osmotic laxatives such as lactulose and polyethylene glycol have been shown to increase stool frequency and decrease frequency of fecal incontinence, particularly in the polyethylene glycol group [26]. However, osmotic laxatives at high doses may worsen fecal incontinence. We recommend the use of stimulant laxatives such as senna and bisacodyl to increase colonic transit and facilitate recto-sigmoid emptying [10, 11, 27, 28]. Administering stimulant medications at the same time of day establishes a predictable pattern. Dosing of these medications is typically determined depending on stool output, frequency, and amount of fecal incontinence. Given that stimulant medications increase colonic transit, stool consistency may become too loose and hence dosage should be adjusted accordingly and bulking agents including fiber (e.g., pectin, psyllium) added.
Children may fail oral medication therapy despite rigorous medication administration, as oral medications do not always produce predictable bowel movements. The absorptive capacity and bioavailability of oral medications vary depending on diet and brand of medication. Lubiprostone (a selective chloride channel-2 agonist) and Linaclotide (a peptide agonist of guanylate cyclase 2C) are FDA approved for chronic idiopathic constipation and constipation-predominant irritable bowel syndrome in adults. Although Lubiprostone has shown promising results in healthy children, it has not been studied in children with spina bifida [29]. These medications are most useful in children where stool consistency is difficult to control despite optimization of stimulant oral medications and rectal therapies. As previously reported, in our experience in patients with spinal anomalies and anorectal malformations, we have used Lubiprostone as an adjunct to other stimulant or rectal therapies in order to better regulate stool consistency and potentially achieve a more predictable stooling frequency as its action depends on serum levels [30].
Rectal therapies
If the child is not socially continent with diet or medical therapy, then rectal therapies should be implemented. Rectal therapies include manual disimpaction and rectal medications such as suppositories and enemas. Manual disimpaction (digital stimulation) in the setting of controlled constipation is commonly used in the spina bifida population. It stimulates the ano-colonic reflex and facilitates recto-sigmoid evacuation [33]. In patients with greater hand dexterity, this method is preferred because it can allow for independence and is also less time consuming [34]. Rectal medications such as over-the-counter suppositories include glycerin (pediatric, adult) and bisacodyl. Water-based suppositories and polyethylene glycol-based bisacodyl suppositories are reported to be more effective than those with an oil base since they are quicker to be absorbed by the rectal mucosa [10, 13]. There are a number of over-the-counter and prescription enemas such as sodium phosphate, glycerin, bisacodyl, mineral oil, and docusate-based enemas. Caution should be taken when having parents purchase enemas over the counter. Brand names and ingredients vary and may lead to confusion, inappropriate administration, and risk of complications. This is especially critical in children with spina bifida who may have abnormal renal function. In those with spina bifida, over-the-counter or prescription “saline” or sodium phosphate containing enemas should be avoided due to reports of hyperphosphatemia, hypocalcemia, and hypokalemia [31, 32]. Avoidance of the use of magnesium-based products in patients with severe constipation is because of the theoretical risk of hypermagnesemia and hypocalcemia [11]. An anal plug is a containment product that resembles a tampon and upon insertion into the anal canal provides a temporary seal with the goal of decreasing fecal incontinence. There is limited evidence to recommend the routine use of anal plugs in the prevention of fecal incontinence in children with spina bifida [35]. A recent Cochrane review of anal plugs in children and adults highlights the small sample size, high drop out rates, and the potential for bias in the literature [36]. The most common side effects reported within the spina bifida population were discomfort and displacement of the plug, especially within more ambulatory children [37, 38]. The smaller pediatric studies support the use of anal plugs as an intermittent and adjuvant therapy to an already established bowel management plan [37, 38, 39].
Trans-anal irrigations (TAI)
Due to poor intra-anal tone, children with spina bifida may not be able to
retain a suppository or an enema. For these patients, we recommend TAIs where high volume
enemas are administered rectally using either a cone tip or a continence catheter with a
balloon. The cone-enema has a cone shaped tip that is inserted at the anus and held in place
by the patient or caretaker. This allows for administration and retention of high volume
enemas without leakage. This system is best used in children with a very patulous anus and
low intra-anal pressures, where retention of a catheter would otherwise be very difficult.
The continence enema catheter is a #24/26 French non-latex urinary Foley. The tip of the
catheter has a balloon that is inflated to volumes of 30–60 mL in order to secure the
catheter within the anal canal and administer the high volume enema. The above-mentioned
methods continue to require parental or caregiver support to administer the enema that is
gravity dependent. Children with low intra-anal pressures who can retain a rectal balloon
during the enema administration would benefit from this system. The
Peristeen
In children and adults, TAIs have been shown to improve constipation, decrease fecal incontinence, and increase overall satisfaction and quality of life in both patients and their caregivers [19, 40, 41, 42]. Success rates using TAIs in children with spina bifida are reported between 66–87.5% [19, 41, 43, 44]. Variability in success rates may be due to lack of standardization of definitions and reporting of fecal continence between the studies. In addition, some studies include patients who have functional and organic fecal incontinence where the success rates of each group are not discussed separately. Bischoff et al. reported a 95% success rate with their TAI-based bowel management program; however, only 12 of 212 children had spina bifida and their specific success rate was not disclosed [45]. Despite their high success rate, TAIs require parental and caregiver support for administration. Matsuno et al. reported that only 23.1% of the children with spina bifida were able to independently perform TAIs, versus 66.7% observed in patients with a Malone antegrade continence enema (MACE) [43]. Their findings are in agreement with other studies, where the overall independence rate of TAI varies between 12%–20% [9, 34, 46]. Two long-term follow up studies assessed the efficacy of trans-anal irrigations in a mixed population of children and adults with neurogenic bowel. They reported lower success rates of 50% and 67% at follow up [42]. These lower success rates may be secondary to decreased independence when receiving TAIs, poor available resources for the transition of care to adulthood, and increased patient reliability on their caretaker for support. Vande Velde et al. reported that adults who were followed for bowel management since they were children were more likely to be continent than those who were lost to follow up during transition of care [46].
Pediatric studies in the use of Peristeen
The enema solution and the additives vary between centers and subspecialists. There are no standardized guidelines regarding enema solutions, additives, and weight-based dosing [53]. Commonly used enema solutions include tap water, normal saline, and GoLytely [54]. In our experience, we prefer the use of normal saline (10–15 mL per kg) [10]. However, parents should be educated about the proper ratio of non-iodized salt to tap water given the inadvertent risk of hypo-or-hypernatremia [55]. In our centers, we recommend mixing 1000 mL of tap water with 1.5 teaspoons of non-iodized salt using a measuring spoon. Tap water enemas have been safely used in a multitude of studies in the literature [41, 49, 56, 57]. In our centers, we recommend the use of tap water enemas with caution given the risk of electrolyte shifts and hyponatremia [58, 59]. Risk of water intoxication and hyponatremia may be increased in children who receive multiple consecutive enemas and in those with megacolon/colonic dysmotility that predisposes them to increased risk of incomplete rectal emptying and fluid retention. Different additives can also be added to the main solution as an adjunct and have been shown to improve fecal incontinence in children and adults with neurogenic bowel [54, 60, 61, 62]. Common additives are glycerin, castile soap, and bisacodyl. Additives should be added with caution due to lack of manufacturing standards and weight-based dosing recommendations. Of note, phosphate-based additives should be avoided, particularly in children with spina bifida with abnormal renal function given the increased risk of hyperphosphatemia, hypocalcemia, and hypokalemia [31, 32]. Chemical colitis has been reported with the use of castile or glycerin soaps and therefore caution should be taken when increasing the concentration of these additives in the base solution [63]. Signs of chemical colitis include bloody and mucoid stools with or without associated abdominal pain. The enemas should be discontinued and the patient should be evaluated urgently by a pediatric gastroenterologist. Complications that may be associated with trans-anal irrigations include pain on insertion, emotional distress, catheter expulsion, burst of balloon, leakage, abdominal pain, and lastly bowel perforation which is very rare with a risk of 2 per million [64, 65].
Antegrade continence enemas (ACE)
The developmental stage, degree of dexterity, and comorbid conditions of a child with spina bifida may impede him or her from being able to independently self-administer trans-anal irrigations. In order to achieve greater independence both for the child and the caretaker, antegrade continence enemas are the next recommended step. An antegrade continence enema (ACE) is administered via a surgically constructed catheterizable conduit or a button conduit between the skin and the colon. The procedure may be done surgically (open or laparoscopic), percutaneously by interventional radiology, or endoscopically in conjunction with laparoscopy. In children with spina bifida, this procedure may be done in conjunction with a Mitrofanoff appendicovesicostomy as the appendix may be deemed adequate to split in order to provide a separate conduit for urine and stool access [66, 67, 68].
Children with spina bifida who use an ACE have reported increased satisfaction
and improved quality of life scores [9, 69]. Success rates have been reported between
69–93% [9, 54, 70, 71, 72] with full
continence in 33–71% [9, 62, 70, 72]. Unfortunately, definitions of success and
continence are not standardized between studies. In a mixed cohort of 117 patients, when
compared to baseline, children with myelodysplasia (
The antegrade flushes are very similar in concept and composition as the retrograde flushes. The base solution usually consists of tap water, normal saline, or GoLytely. As previously discussed, additives can be added to facilitate recto-sigmoid emptying. Mugie et al. demonstrated in their large cohort of patients that children whose irrigation solution contained stimulants were significantly more likely to be continent [62]. The antegrade flushes are administered through a catheterizable conduit or a cecostomy button over a 15–20 minutes period followed by toilet sitting for approximately 30–40 minutes. Similar to the TAI, the ACE is ideally administered around the same time each day. Depending on the patient and their response to therapy, some may be able to skip a day or two in between flushes. The goal of ACE is to provide independence and bowel management. However, depending on the patient’s development, cognition, and dexterity, this may not be possible. In children with truncal or upper body hypotonicity or spasticity, specialized chairs and toileting systems may be used for support during toilet sitting. In their cohort of children with spina bifida, Wide et al. reported that only 31% always and 6% occasionally had bowel movements independently [9]. This is critical in this population because the group also reported that those who toilet-sit independently have significantly higher quality of life [9]. On the other hand, Vande Velde et al. 2007 reported that 62.5% of patients were able to administer ACEs independently vs. 12.5% who used TAI [34]. It is unclear if the patients required any help for toileting. Appropriate patient selection and setting appropriate patient and caretaker expectations following ACE surgery is critical. In addition, the degree of patient’s independence at toileting should be assessed at each clinic visit with goals of identifying and resolving possible barriers.
Other surgical interventions
Sacral nerve stimulation (SNS) has been widely used for the treatment of adult fecal incontinence [79]. Studies examining the utility and safety of SNS in children with spina bifida are limited [80, 81].
The role of segmental resection, Malone appendicostomy combined with segmental resection, and diversion colostomy in children with spina bifida is very limited and has not been investigated in this population. Diverting colostomies [13] are used in adult patients as a last resort treatment option. However, some patients may continue to have abnormal colostomy output due to underlying abnormalities in the spinal innervation of the hindgut and may require retrograde or antegrade irrigations of their colostomy.
In summary, children with spina bifida commonly have bowel dysfunction that has a significant impact on their quality of life and emotional wellbeing. Fecal continence has been associated with increased satisfaction and quality of life of both children and their caretakers. The goals of a successful bowel management program are predictable bowel movements, social continence, and eventual independence. A stepwise approach should be implemented in the context of the patient’s developmental age to allow for optimum social integration with their peers.
Footnotes
Conflict of interest
The authors have no conflict of interest to report.
