Abstract
PURPOSE:
The aim of this systematic review was to analyze the results of studies on quantifiable physical risk factors (beyond questionnaires) for adolescent low back pain (LBP).
METHODS:
A systematic search was conducted in Medline (OvidSP), Premedline (PubMed), EMBASE, Cochrane, CINAHL, PEDro and PsycINFO. Cross-sectional, prospective and retrospective English language studies on LBP in adolescents aged 10 to 18 years were included.
Results:
Twenty-two mostly cross-sectional studies were included. Trunk muscle endurance in particular seemed to be associated with adolescent LBP, while a possible association of trunk muscle strength and spinal flexibility was less clear.
CONCLUSION:
There is a need for prospective studies on quantifiable physical risk factors for adolescent LBP. Such studies should focus on back and abdominal muscle endurance, possibly in combination with sagittal spinal mobility, sagittal postural alignment and neurodynamics as possible modifiable risk factors for LBP.
Introduction
Back pain starts early in life and its prevalence increases with age, accelerating in the early teens around the age of 12 to 15 [1, 2, 3] and reaching adult prevalence by the age of 18 [1, 4]. As for low back pain (LBP), an eight-year follow up from adolescence to adulthood showed a fourfold increase in the risk of adolescents with LBP for having LBP in adult life. Thus, it was postulated that the focus of research, prevention and treatment in this area should be changed from the adult to the young population [5].
Although there are several specific pathologies that can result in spinal pain in adolescence, a considerable number of cases of adolescent spinal pain, around one third in a recent study [6], are considered non-specific. As for risk factors for non-specific adolescent LBP, some psychosocial parameters, such as back pain of one or two parents [7, 8] as well as anxiety and depression [7, 9] were identified. Indeed, about 10% of the adolescents with LBP showed an increased probability of having sleep disorders and headaches along with the corresponding psychological problems, possibly linked to a dysregulation of the hypothalamic-pituitary-adrenal axis [10]. As for lifestyle factors, TV consumption, computer use and smoking, but not body weight or fitness level, correlated with adolescent LBP [11, 12, 13, 14]. With regard to physical parameters, controversial results were reported that differed from comparable studies in adults and, thus, suggesting that risk factors for adult LBP cannot be extrapolated to adolescent LBP [15]. So far, no physical risk factors for adolescent spinal pain could clearly be defined [11, 16, 17], and the suggestion that psychosocial factors are more important than physical factors for spinal pain in young populations [18] remains disputed. A recent study, found that although psychosomatic symptoms were most strongly associated with 1-month prevalence of adolescent spinal pain, these were followed by factors from the physical and psychosocial domains. Consequently, these authors suggested that the importance of physical risk factors for non-specific adolescent spinal pain may have been underestimated [19].
This systematic review aimed to determine whether there are quantifiable physical risk factors for LBP in adolescence. It focused on adolescents between 10 and 18 years and included prospective, cross-sectional or retrospective studies that used quantifiable measures beyond questionnaires.
Methods
Search strategy
A systematic literature search of studies on risk factors for back pain in adolescents up to and including September 25, 2015 in Medline (OvidSP), Premedline (PubMed), EMBASE, Cochrane, CINAHL, PEDro and PsycINFO was performed by a professional medical librarian from the local university library. The search was repeated on October 24, 2016. The two resulting additional publications from this repetitive search [19, 20] were included in the discussion section.
In a first step, the search was not restricted to physical risk factors as studies on psychosocial risk factors might have investigated physical risk factors as secondary outcomes. The literature search was also not restricted to LBP because the present study was part of a larger project investigating physical risk factors for any type of adolescent back pain. No limits were applied for the publication date of the articles. Medical subheadings (MeSH) were used as search terms. To find the most recently published studies that have not yet been linked with MeSH, keywords were also searched for in the title or abstract. The search strategies can be seen in Appendix A.
Inclusion criteria
A study was included if it was a cross-sectional, a retrospective or prospective cohort study in English and investigated back pain in adolescents from age 10 to 18. This age range was chosen because there is some evidence that puberty is a risk factor for back pain in the young [15] with pubertal development starting at 9.5 years for girls and at 10 years for boys [21]. If a study covered a wider age range, it was included only if the participants’ mean age was within the age limits of this review. In addition, to be included in this review, the studies had to use quantifiable measures and not be restricted to questionnaires only. Studies were excluded if they focused on back pain of a specified pathology such as scoliosis, Scheuermann’s disease, spondylolisthesis, disc degeneration, hypermobility, coccydynia, fibromyalgia, posttraumatic or postoperative back pain, radiographic studies., Also excluded were particular populations for example athletes, or disabled children or exclusively on lifestyle factors such as computer use, school bag weight, body weight, and sport activities. Studies that focused on possible genetic background for adolescent LBP were also excluded. Lastly, only studies that explicitly investigated back pain were included, while studies on the sacro-iliac area or on musculoskeletal pain in general were excluded.
Flow diagram of the study selection process.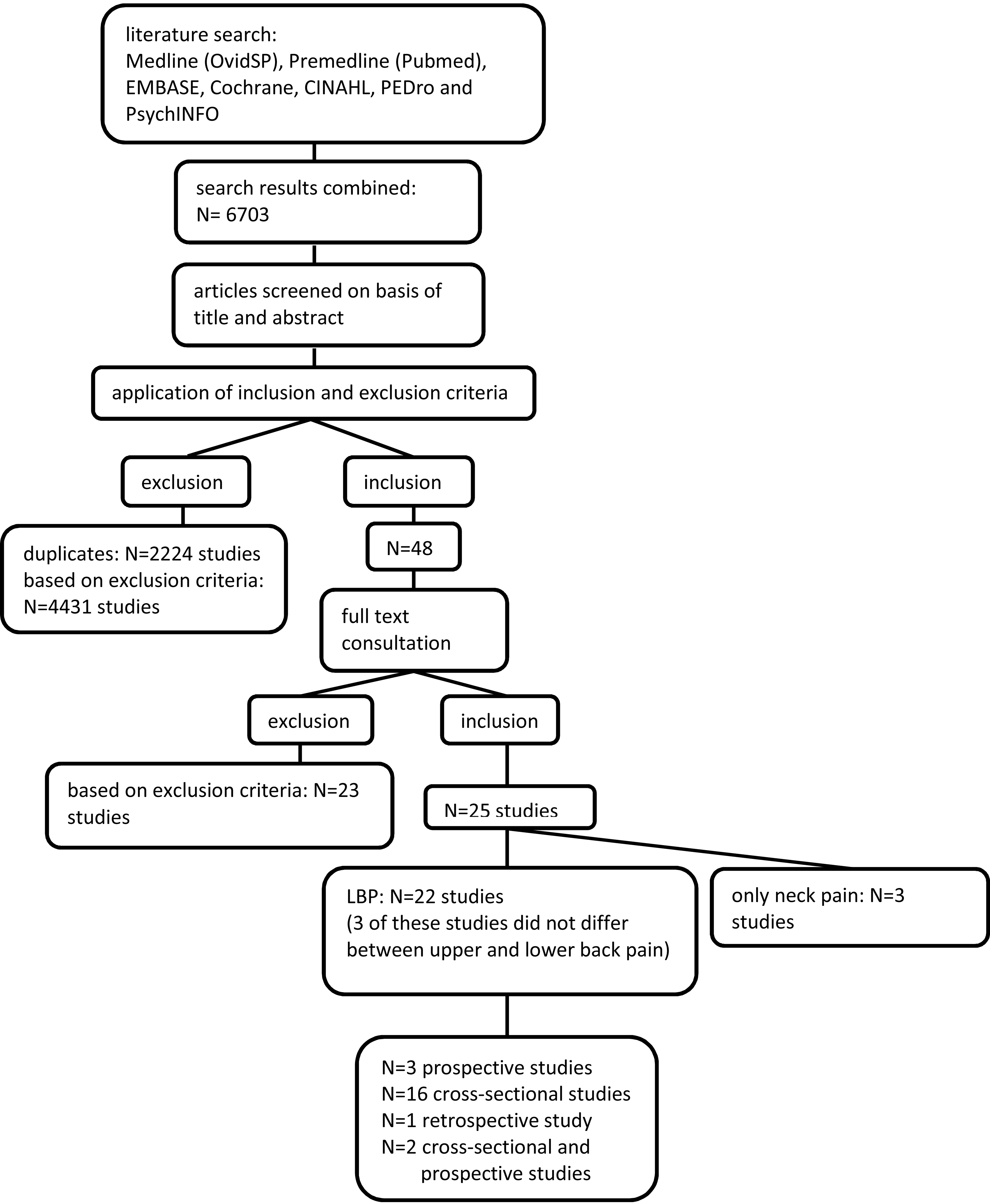
In a first step, titles and abstracts of the articles were screened by two authors (TP and BW) according to the inclusion and exclusion criteria listed above. In a second step, the same two authors each screened the full text of the remaining articles for eligibility. The full texts were also retrieved if no abstract was available or the selection could not be made on the basis of the abstract. In two consensus meetings, some discrepancies regarding eligibility of the articles were resolved. A third author (KH) was consulted in case no consensus could be reached for giving the final decision. In a last step, only studies that focused on LBP were selected for the present review. Figure 1 depicts the search flow.
Quality assessment
Quality assessment was performed by two authors (SR and BW) and a consensus meeting was held after individual ratings to clarify possible disagreements. Raters’ agreement was quantified using Cohen’s kappa, calculated using IBM SPSS Statistics 21. The quality assessment was based on the “Critical appraisal form for quantitative studies” by Law et al. [22]. Because no interventions were followed, questions referring to any intervention were removed; e.g. as done in a comparable review [23]. In their place, question 14 (estimates of random variability of data) was added from the Downs and Black checklist [24]. Moreover, two questions about biases [23] and about the adequate description of the assessments [23, 24] were included (questions 4 and 7, Appendix B). All questions were either answered by YES (
Data extraction
One author (TP) extracted the following information from each article: study design, number, age and gender of participants; physical risk factors that were investigated; assessments and tests that were used, and the results.
Results
Study selection
The selection process is illustrated in Fig. 1. The database searches resulted in a total of 6703 articles of which 2224 were duplicates. After screening the titles and abstracts for the inclusion and exclusion criteria, 48 articles were considered potentially suitable for this review. After reading the full texts, 25 studies remained that fulfilled the inclusion and exclusion criteria. In a last step, three studies that solely focused on neck pain were excluded. This resulted in 22 studies identified as suitable for this review. Three studies were prospective, 16 cross-sectional and one study was retrospective. Two studies consisted of both a cross-sectional and a prospective part. Three of the 22 studies [26, 27, 28] investigated ‘back pain’ and did not differentiate between lower and upper back, while the remaining focused explicitly on LBP.
Quality of the selected studies
After the consensus meeting, the two reviewers agreed on the scores of 19 articles. In 3 articles [29, 30, 31], there was a difference of one point in the item covering the report of clinical importance of the study results, which was deemed acceptable. The average of both ratings was used for further analysis. The resulting
Quantifiable physical risk factors
The results for all outcome variables are shown in detail in Table 1 and summarized per outcome variable in Table 2. In this text section, due to the large amount of information gained, we will only present summarised information on the main results for the sake of clarity.
Trunk
There was concordant evidence from several cross-sectional studies that reduced endurance of trunk extensor muscles [32, 33, 34] as well as of trunk flexor muscles [31, 34] was associated with LBP. This finding was slightly challenged by one study that found both reduced and greater abdominal and back muscle endurance in girls with LBP [28] and one low-quality study that found no association between abdominal muscle endurance and adolescent LBP [33]. The evidence for trunk strength was conflicting with two studies that investigated different aspects of trunk strength and found controversial results [26, 35]. As for trunk mobility, there was evidence from one prospective study that sagittal range of motion (ROM) might be associated with adolescent LBP [35]. In contrast, when measuring either spinal mobility in flexion or extension, the results were conflicting with several studies using different clinical tests and finding markedly controversial results [31, 34, 45, 36, 37, 38]. High lumbar sagittal mobility combined with low lumbar extension strength (high mobility/strength ratio) emerged as a risk factor for LBP from one prospective study of high quality [37]. Regarding functional trunk stability, the results were controversial [38, 39]. Trunk asymmetry, in contrast, was consistently reported to be a risk factor for LBP in three studies, one of which was cross-sectional [38], one retrospective [8] and one prospective [40].
Lower limb
There was evidence from two studies that lower limb endurance was associated with LBP [32, 33], which was supported by a third study that found an association between lower limb power and LBP in girls [28]. Three studies reported that changes in hamstrings flexibility might be associated with more severe-LBP [34], particularly in boys [28, 41], where both, reduced [28, 41] and enhanced [28] flexibility was associated with LBP. There was, however, one low-quality cross-sectional study that found no association between hamstrings flexibility and severe LBP [42]. For the hip, one cross-sectional study presented decreased hip flexion with knees extended as a risk factor for recurrent LBP [31], while another cross-sectional study reported that decreased hip flexion and internal rotation were associated with LBP in boys [41]. Fur-
Characteristics of the studies included and their outcome variables
Characteristics of the studies included and their outcome variables
LBP
Summary of the results per outcome variable
Prospective studies are shown in bold. LBP
thermore, hip extensor weakness was found to be a risk factor in one low quality cross-sectional study [33].
Sitting height emerged as a risk factor for adolescent LBP from one cross-sectional [30] and one prospective [40] study, although its importance was minor [40]. For sitting posture, contradictory results were reported. A weak association between greater slump degree and LBP that was observed in one cross-sectional study [27] was contradicted by another study that found differences in sitting posture only when the adolescents were sub-classified [29]. Posture alignment in stance was concordantly stated to be a risk factor for LBP by three studies. Non-neutral alignment emerged from one prospective study [43] and an increased pelvic displacement angle [44] and sway back posture [45] were demonstrated in two cross-sectional studies as risk factors for LBP in adolescence for boys, but not in girls [46]. No association of local spinopelvic parameters, such as vertebral level of the lumbar apex, pelvic tilt or sacral inclination [44], and of general motor competence, assessed by a test for neuromuscular development [28], with adolescent LBP were found.
Discussion
The aim of this systematic review was to summarize the current knowledge about quantifiable physical risk factors for LBP in adolescents between 10 and 18 years. Prospective, cross-sectional or retrospective studies that used quantifiable measures beyond questionnaires were included. There is wide consensus in the literature that psychosocial factors are associated with LBP in adolescents [10, 18, 47, 48]. The role of mechanical factors was reported to be minor, although many studies restricted mechanical factors to body weight, school bag weight or physical activity [18]. However, there is some evidence that the importance of physical risk factors might have been underestimated [19]. Indeed, this review revealed that there is conclusive evidence that some quantifiable physical parameters might put adolescents at risk for LBP. This accounts particularly for back muscle endurance concordantly reduced in adolescents with LBP, which was in line with a recent review by Lardon et al. [49]. There was the exception of one study that reported both reduced and enhanced back muscle endurance to be associated with LBP, but only in girls [28]. This study reported, however, small associations of all investigated factors to LBP and was the only one that did not use the Biering-Sorensen test as assessment for back muscle endurance. Similarly, reduced endurance of the abdominal muscles was, with the exception of one low-quality study, associated with LBP in three cross-sectional studies, two of which focused on adolescents with recurrent or continuous LBP. However, as no prospective study was found in this context, no conclusion about causality can be drawn. The relevance of trunk muscle strength became less clear. Only one cross-sectional study found some association between LBP and the ratio of flexion and extension peak torque in boys [26]. Thus, in accordance with the review of Lardon et al. [49], who reported no association between back muscle strength and adolescent LBP, the measurement of trunk muscle strength seems to be of minor importance. Accordingly, the results regarding spinal mobility were rather diverse as were the measurement methods that were used (Schober test, fingertip-floor distance, flexicurve). In a recent prospective study by Aartun et al. [20], both methods, the fingertip-floor distance as well as the Schober test (with dichotomized results), could not predict future LBP. Interestingly, one prospective study of high quality reported a combined measure of strength and mobility, more precisely a high lumbar mobility-extension strength ratio to be predictive for adolescent LBP [37]. However, in fact, the authors conducted a modification of the Sorensen test, which means that the proposed measure is rather a combination of back muscle endurance and spinal mobility. Thus, it can be hypothesized that the decrease in back muscle endurance is a risk for adolescent LBP, which becomes more accentuated when spinal mobility is enhanced. The results of the study by Salminen et al. [34] seem to further support this hypothesis as they found decreased endurance of back muscles and abdominal muscles along with increased flexion mobility in the adolescents with recurrent/continuous LBP. Unfortunately, they did not combine these measures. Trunk asymmetry, assessed by forward bending, was consistently associated with LBP in this review, but did not have a predictive value in the study by Aartun et al. [20]. This test is broadly used in scoliosis screening, and was reported even in this context to have limited positive predictive and sensitivity values [50]. Thus, its application in the field on non-specific adolescent LBP seems of minor value.
Lower limb performance was associated with LBP in three cross-sectional studies. Accordingly, knee extensor strength was significantly smaller in adult runners with chronic LBP when compared to control runners [51]. Furthermore, adult patients with chronic LBP who reported psychologic distress and a high pain level showed reduced quadriceps muscle torque, probably as a result of increased inhibition of muscle activity [52]. Given these findings, a prospective study in adolescents that includes quadriceps strength might be of interest. The situation in terms of hip mobility and hamstrings flexibility seems more complex as the measuring methods differed substantially. Jones et al. [31] tested hip flexion with the knee extended, which is comparable to the passive straight leg test used by Salminen et al. [34] for assessing hamstrings flexibility. Both studies dealt with a subpopulation of adolescents with more severe LBP and found a positive association between reduced flexibility and recurrent LBP. However, it was shown via ultrasonography that the straight leg raise score is not determined by hamstring elongation [53], but rather by altered neurodynamics such as sliding capacity of the nerve roots and the spinal cord in the thoracolumbar region [54]. Thus, it might rather be impaired neural sliding than hamstrings tightness that could be a risk factor for adolescent LBP. The limited importance of hamstrings flexibility is underlined in the present review of two studies by Sjölie [41] and Harreby et al. [42] that measured hamstrings flexibility via knee extension deficit in 90 degrees of hip flexion and found contradicting results. Furthermore, in the study done by Perry et al. the use of sit-and-reach tests for assessing hamstrings flexibility was not recommended [55] or only with reservations [56].
Anthropometric measures and sitting posture seem to play, at most, a minor role in the context of adolescent LBP, while alignment in stance seems more important. In the prospective study by Smith et al. [43], any deviation from the neutral posture (sway, flat, hyperlordotic) was a risk for back pain. The three cross-sectional studies by Dolphens et al. [44, 45, 46] reported sway-back posture to be associated with LBP in boys, while the investigated local spinopelvic parameters, such as apex of lumbar lordosis and pelvic orientation in the sagittal plane did not show a relationship. In contrast, in a recent study of this group, higher lumbar lordotic apex and an increase in pelvis retroversion were the two physical factors with the highest odds for LBP [19]. This corresponds well with findings in adults with chronic LBP, where a greater proportion of patients with a low sacral slope and a long but small lumbar lordosis were reported when compared to healthy controls [57].
Limitations
Narrowing this review to quantifiable physical risk factors led to some limitations. By limiting the age of the adolescents from 10 to 18 years, we might have missed some interesting studies on slightly older adolescents. Indeed, a recent prospective study in adolescents aged 19 years at baseline, studied proprioceptive control using muscle vibration and found that an ankle-steered control strategy was a risk factor for developing recurrent mild LBP [58]. Furthermore, we excluded studies on physical activity as this was mostly asked via questionnaires. However, some studies measured aerobic capacity as did the study by Perry et al. This study reported an association between greater aerobic capacity and LBP in the last month [28]. Such an association however disappeared when the model was adjusted for back muscle endurance [59]. Thus, a recent review hypothesized that the aerobic capacity could be a proxy measure for back muscle endurance and both measures could be two manifestations of body build with a genetic background [49]. Indeed, the exclusion of genetic studies is another limitation of the present review. Although there are not many such studies, they focus, in a broader sense, also on quantifiable physical risk factors. For example, Skouen et al. found in 1004 adolescents that genetic variants in an adrenergic candidate gene (beta-2 adrenergic receptor) were associated with chronic comorbid neck and low back pain [60]. This might further support the hypothesis of a dysregulated hypothalamic-pituitary-adrenal axis being involved in chronic pain in adolescents [10]. A last limitation that should be mentioned is the focus on correlations between quantifiable physical risk factors and LBP. Correlations do not infer causation. Therefore, it might well be that in longitudinal studies investigating treatment effects through modification of physical risk factors will not result in changes in LBP as seen in adults with LBP [61]. Nonetheless, this review is a first step in identifying modifiable physical risk factors that should be further studied, possibly in prospective studies.
Conclusions
This review demonstrated 1) correlations between LBP and back and abdominal muscle endurance, and to a lesser extent, with sagittal spinal mobility, sagittal postural alignment and neurodynamics; and 2) a need for prospective studies of high quality that assess pos- sible causality between physical risk factors and adolescent LBP. Based on the results of this study, it is recommended that such studies focus on back and abdominal muscle endurance, possibly in combination with sagittal spinal mobility, sagittal postural alignment and neurodynamics as possible modifiable risk factors for the development of LBP in adolescence.
Footnotes
Acknowledgments
We thank Martina Gosteli, information specialist at the Main Library of the University Zurich, for conducting the systematic literature search.
Conflict of interest
The authors have no conflict of interest to report.
Appendix
Appendix A: Search strategies
Appendix B: Quality assessment form
Adapted from the “Critical appraisal form for quantitative studies” by Law et al. [22] using the checklist by Downs and Black [24] and the form by Prins et al. [23]:
TOTAL: Maximal 14 points for cross-sectional studies and 15 points for prospective studies.
STUDY PURPOSE
Was the purpose stated clearly?
Yes/No
LITERATURE
Was relevant background literature reviewed?
Yes/No
DESIGN
Was the design appropriate for the study question?
Yes/No
Were there any biases (random/nonsystematic error or measurement bias/systematic error) that may have influenced the results (apart from reliability and validity of the outcomes)?
Yes/No
SAMPLE
Was the sample described in detail?
Yes/No
Was sample size justified?
Yes/No
OUTCOMES
Were the methods of outcome measurement described sufficiently?
Yes/No
Were the outcome measures reliable?
Yes/No
Were the outcome measures valid?
Yes/No
RESULTS
Results were reported in terms of statistical significance?
Yes/No
Were the analysis method(s) appropriate?
Yes/No
Clinical importance was reported?
Yes/No
Drop-outs were reported?
Yes/No/Not applicable
DATA VARIABILITY
Does the study provide estimates of the random variability in the data for the main outcomes?
Yes/No
CONCLUSIONS AND IMPLICATIONS
Conclusions were appropriate given study methods and results?
Yes/No
