Abstract
We report the case of a six-year-old girl with Moyamoya disease who presented with bilateral internal carotid artery malignant infarct following encephaloduroarteriosynangiosis (EDAS). During her neurorehabilitation, she developed gradually worsening dystonic spasms with opisthotonic posturing, tachycardia, tachypnea and desaturation. This rare life threatening movement disorder was diagnosed as status dystonicus based on the history and clinical presentation. Status Dystonicus occurs commonly in children and the etiology is often diverse. It occurs in patients with preexisting dystonia or following an acute central nervous system insult of varied etiology. Status dystonicus is usually precipitated by one or more triggering factors. Rarity and lack of objective criteria for diagnosis often delays the management thereby increasing the risk of mortality and morbidity. Here, we discuss the challenges faced in the diagnosis and management of a child with denovo status dystonicus.
Introduction
Status dystonicus refers to episodes of “increasingly frequent and generalized dystonia which had necessitated urgent hospital admission” [1]. It occurs commonly in children between 5–15 years of age [2]. Status Dystonicus usually occurs in central nervous system disorders such as hypoxic ischemic encephalopathy, inflammatory or autoimmune disorders and encephalitis, non-inflammatory disorders such as drug-induced movement disorder, metabolic disorder such as glutaric aciduria, and vascular disease including Moyamoya disease [3]. The frequently reported triggering factors are pain, surgery, fever, infection and calculi, gastroesophageal reflux disease, constipation, abrupt initiation, abrupt withdrawal or change in medications including dopamine receptor blockers, haloperidol, lithium, tetrabenazine and clonazepam [4].
Case report
A six-year-old girl was diagnosed with Moyamoya disease in September 2011 and she underwent encephaloduroarteriosynangiosis (EDAS) in September 2013. Post-operatively, she developed bilateral internal carotid artery (ICA) infarct, for which left frontotemporoparietal decompressive craniectomy was done (Fig. 1). On transfer to the Physical Medicine and Rehabilitation ward, her Glasgow Coma Scale score was 8/15 and the level of consciousness by Ranchos Los Amigos (RLA) was stage II. Tracheostomy tube was in situ and she was haemodynamically stable. Spasticity of all four limbs with brisk muscle stretch reflexes, and extensor plantar responses were observed.
(a) T 2 weighted image at the level of basal ganglia shows multiple, tiny, low intensity rounded flow voids in bilateral basal ganglia (black arrows), suggestive of collateral vessels in basal cisterns & basal ganglia. (b) T2 weighted image showing hyperintense signal in the bilateral internal watershed area more on the right side. (c) MR Angiography reveals narrowing of right ICA with complete cut off the supra clinoid ICA. Complete cut off of the left supra-clinoid ICA. (d) Post-operative CT brain showing left decompressive craniectomy with multiple areas of hypodensity in both the cerebral hemispheres suggestive of bilateral ICA infarcts.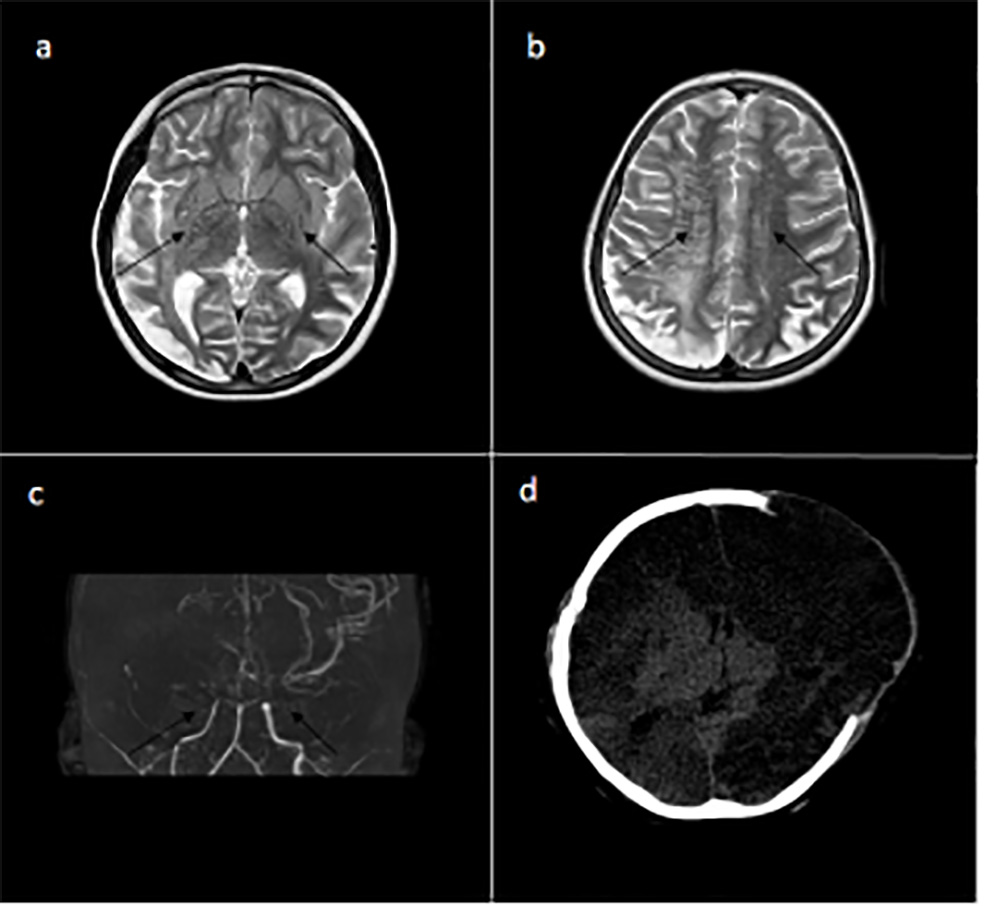
A month after admission in the rehabilitation ward, she developed low grade fever and subsequently had frequent generalized dystonia involving the neck, trunk and lower limbs with opisthotonic posturing. Dystonic spasms were associated with tachypnea, tachycardia and desaturation up to 90% requiring oxygen supplementation. These episodes were variable in frequency, intensity and duration. Retrocollis and opisthotonic posturing of the trunk were absent during sleep, but worsened with rise in temperature or crying.
Differentials considered were neuroleptic malignant syndrome, generalized tonic seizures and status dystonicus. Serum electrolytes, renal parameters, calcium and phosphorus were normal. Creatine phosphokinase (CK) level was elevated (2891 u/L) indicating rhabdomyolysis. CT brain showed no new parenchymal or basal ganglia changes. Ultrasonography abdomen showed no features of pyelonephritis or calculi. There was no epileptiform discharges observed on the EEG recording. Diagnosis of status dystonicus in our case was established based on the clinical observation of frequent and generalized dystonia with complications such as desaturation and exhaustion and also excluding other differentials. The child was treated with trihexyphenidyl and clonazepam but there was no significant clinical improvement. Midazolam infusion was initiated at a dose of 1 mcg/kg/min and the dose was gradually titrated up to 5 mcg/kg/min till good muscle relaxation was achieved. Following midazolam infusion, dystonic spasms reduced significantly, saturation improved to 96%, tachypnea and tachycardia were settled. Tetrabenazine and baclofen were also added. Adequate hydration and nutrition were maintained. An attempt to taper the midazolam infusion after a symptom free interval of 5 days had resulted in rebound status dystonicus thereby necessitating titration of the midazolam dose to 7.5 mcg/kg/min. In addition to the midazolam infusion, the doses of baclofen, trihexyphenidyl, tetrabenazine and clonazepam were titrated. Oral diazepam was also added to facilitate tapering of midazolam infusion once the dystonic storm was well controlled. Few myoclonic jerks were observed for which levetiracetam was added. Midazolam infusion was gradually tapered and stopped. Haloperidol was given for short duration to combat the agitation and restlessness. She was discharged on baclofen, clonazepam, tetrabenazine and levetiracetam. There was no recurrence of dystonic spasms after discharge as confirmed by periodic telephonic conversations with parents until June 2017.
Lack of objective diagnostic criterion or clinical scoring system, often delays the diagnosis and intervention in status dystonicus. An objective definition would help in early identification of the status dystonicus/dystonic storm and also to differentiate from other similar presenting clinical conditions. The important differentials often confused with status dystonicus are neuroleptic malignant syndrome, malignant hyperthermia, serotonin syndrome, paroxysmal autonomic instability with dystonia and acute dystonic reactions [4]. Close neurological monitoring and exclusion of other differentials are often helpful in establishing the diagnosis of status dystonicus [5]. Diagnosis of status dystonicus becomes more challenging in cases where there is no previous history of dystonia. It is evident from our case that status dystonicus can occur denovo in patients without preexisting extrapyramidal involvement. Infection has been identified as an initial trigger in about 50% of patients with status dystonicus [4]. Although it is clinically difficult to differentiate whether fever is the cause or effect of dystonic spasms, the onset of fever before the dystonic storm and presence of laboratory markers favors an underlying infection. In our patient, the status dystonicus was probably triggered by an underlying urinary tract infection. Hence, it is mandatory to screen for infection in all children manifesting with status dystonicus.
A clinical observation of generalized dystonia and the frequency and severity of dystonic spasms may help in making a prompt diagnosis, grading severity and assessing the potential complications. Early diagnosis, identification of the trigger factors and prompt treatment improved the outcome in our patient. Recent phenomenology divides episodes of status dystonicus into tonic (mainly sustained contractions and abnormal postures) or phasic (rapid and repetitive dystonic contractions) phenotypes [6]. The majority of the cases as seen in our case, present with a tonic phenotype [2]. Predictors of poor outcome include male gender, tonic phenotype and relapsing progressive course with partial or no recovery [2]. Although tonic phenotype was observed, favorable response to treatment was documented in our case.
Anticholinergics or dopamine transmission antagonizing medications are often preferred in the management of patients with status dystonicus [2]. Others agents that have been tried in management of patients with status dystonicus are trihexyphenidyl, benzodiazepines, baclofen, levodopa, haloperidol, gabapentin, chlormethiazole, carbamazepine, valproate and botulinum toxin [2, 7]. Severe cases with poor response to medications should be managed in a stepwise approach, which includes urgent admission, monitoring of serum electrolytes, CPK and renal function, identifying and managing the triggers, intravenous hydration, antipyretics, analgesics, sedation and muscle relaxants. Midazolam is often used for sedation in view of muscle relaxant property, rapid onset action and short half-life [1, 2]. Propofol, muscle relaxants and barbiturates have also been used in refractory cases. Intubation and ventilation may be needed in case of airway compromise or respiratory failure. Comfort and sleep are to be ensured [1, 4]. Episode of dystonic storm and clinical course after a dystonic storm is unpredictable and can result in complete or partial recovery, rebound status and even death [8]. Our patient was managed with supportive measures, elimination of the trigger factors, and anti-dystonic medications. Dystonic storm gradually decreased with midazolam infusion, but rebound status dystonicus occurred on tapering midazolam infusion. This was managed appropriately with restarting midazolam infusion followed by a slow taper, administration of diazepam through nasogastric tube and optimization of other medications. Throughout the entire period of status dystonicus and phase of complete resolution, she was managed in the ward for 40 days.
Similar to status epilepticus, status dystonicus may not respond to conventional treatment which is referred to as “refractory status dystonicus”. Refractory status dystonicus needs more aggressive treatment in the form of general anesthesia administered in an intensive care unit [7]. Temporary measures such as deep sedation and muscle paralyzing agents is reserved for cessation of severe dystonia [9]. Intrathecal baclofen, deep brain stimulation, pallidotomy and thalamotomy have also been tried in refractory status dystonicus [9]. Deep brain stimulation has been proposed to be an effective treatment approach in the management of patients with status dystonicus [8].
Conclusion
Diagnosis and management of status dystonicus are often challenging. An objective definition of status dystonicus would be of great value in establishing an early diagnosis. Intensive care management, anti-dystonic medications and intravenous sedative agents are recommended for management of dystonic storm.
Footnotes
Conflict of interest
The authors have no conflict of interest to report.
