Abstract
PURPOSE:
The aim of the study was to evaluate the use of a novel technique for baclofen delivery using an intrathecal catheter inserted through a lumbar laminotomy with the tip placed at the cisterna magna (supraspinal baclofen (SSB)) for the treatment of severe generalized secondary dystonia.
METHODS:
A cohort study of six individuals (4M/2F, mean±SD = 15±4.86 years) with generalized dystonia unresponsive to oral medications were treated with SSB and followed clinically for 9 years. Intrathecal catheter tips were positioned under fluoroscopic guidance just above the level of the foramen magnum, at the cisterna magna.
RESULTS:
Five of the 6 patients experienced sustained benefit with SSB; the group mean modified Fahn-Marsden scale scores decreased from 95 to 55 (t = 3.29, p = 0.02). One patient suffered complex pump pocket infection, and therefore underwent pump explantation.
CONCLUSION:
Supraspinal baclofen may be an effective method for infusing baclofen into the ventricular system of the brain for treatment of secondary dystonia which is unresponsive to oral therapy.
Introduction
There are limited options for the management of secondary generalized dystonia with the mainstay of treatment being oral medications, including baclofen, tizanidine, benzodiazepines, and dopaminergic agents [1, 2]. Unfortunately, achieving symptomatic benefit with oral therapy often requires high dosages and is limited by central effects, especially sedation. Furthermore, secondary dystonia and childhood-onset dystonia, such as that observed in patients with cerebral palsy (CP), have been reported to be less responsive to pharmacotherapy compared to primary and adult-onset dystonia, which tend to be more focal and less severe [1]. Deep brain stimulator therapy also has been examined with variable effects reported, particularly in patients with secondary dystonia [3].
Filloux and Hallett were the first to suggest supraspinal pathophysiology to underlie secondary dystonia in the setting of brain injury [4, 5]. Thus, more specifically indicating that brain injury causes excessive stimulation in the motor cortical and subcortical pathways and results in secondary generalized dystonia. In combination with poor permeability of most medications across the blood-brain barrier, oral medications, expectedly, have demonstrated limited efficacy in the treatment of generalized dystonia. Therefore, centrally delivered medications are likely to demonstrate greater benefit at lower concentrations compared to oral medications [6].
Early studies describing applications of intrathecal baclofen to treat secondary dystonia report greater therapeutic benefit at significantly lower concentrations when compared with oral baclofen therapy, and hence, also, fewer side effects [7–9]. Furthermore, in line with studies demonstrating that spasticity is improved when the catheter tip is placed nearer to the therapeutic target of interest (with the catheter tip at higher spinal levels for patients with upper extremity spasticity compared to patients with primarily lower extremity spasticity). Later methodologies demonstrated higher catheter tip placement at the cervical level resulted in greater improvement in dystonia symptoms compared to catheter tip placement at thoracic levels [10, 11]. Given that dystonia is generated from pathologic activation of cortical and subcortical pathways, baclofen delivery at a supraspinal level, and in nearer proximity to the ventricular system of the brain, has the potential to provide maximal therapeutic benefit at the lowest drug concentration.
The purpose of the current cohort study is to characterize 9 years of experience utilizing a novel therapeutic approach in which baclofen is infused supraspinally (SSB) into intracranial cerebrospinal fluid via an intrathecal catheter placed at the cisterna magna for treatment of pediatric patients with severe, secondary generalized dystonia which had previously been unresponsive to oral medication.
Methods
Patients
This study was a cohort of 6 patients with secondary generalized dystonia who had previously been unresponsive to oral medications and were treated using supraspinal baclofen (SSB) infusion. The complete cohort of patients who have undergone SSB implantation for secondary generalized dystonia is reported. Written and informed consent for the procedure was provided by the patient’s parent or medical legal proxy. Research study procedures, including analysis and characterization of the group, and publication of subsequent results were approved by the University of California, Irvine Institutional Review Board. Demographic and pre-operative data are presented in Table 1. Prior to treatment, each patient was evaluated by a pediatric physiatrist (KBB), physical therapist, occupational therapist, and neurosurgeon (BC) in a multidisciplinary hypertonicity clinic at Miller’s Children’s and Women’s Hospital. All patients had secondary generalized dystonia, which had been unresponsive to oral medication. The patient with dystonia in Case 2 had previously been unable to tolerate oral medications and was therefore not being treated with them prior to surgery.
Demographic and preoperative characteristics. Abbreviations: BMI (Body
Mass Index), GMFCS (Global Motor Function Classification System)
Demographic and preoperative characteristics. Abbreviations: BMI (Body Mass Index), GMFCS (Global Motor Function Classification System)
The modified Fahn-Marsden dystonia scale (FMDS) as detailed by Albright was used to grade dystonia. Although the Barry-Albright dystonia scale was developed for assessment of secondary dystonia, the FMDS was selected in the current study due to its documented high responsiveness in interventional studies involving children with CP [13]. Pre-operative and post-operative dystonia scores were evaluated by the same physiatrist (KBB).
The SSB intraspinal catheter and programmable pump placements were placed by the same neurosurgeon (BC) for all patients. All patients had placement of a SynchroMed II Pump with Ascenda catheter (Medtronic, Inc, Minneapolis, MN). Using superficial landmarks, a midline incision was made overlying the spinous processes of L3-L4 and the muscle was stripped down to expose the lamina at that segment. A small laminotomy was performed and a Tuohy needle was used to enter the subarachnoid space. The spinal catheter was passed cephalically under fluoroscopic guidance to a level just above the foramen magnum (within the cisterna magna), and the manufacturer provided stay device was used to secure the distal catheter end prior to being tunneled subcutaneously to the abdomen. A subcutaneous pocket was made in the right lower quadrant of the lateral abdomen to accommodate the programmable pump; 40 ml pumps filled with baclofen (2000μg/ml) were used in two cases, and 20 ml pumps were used in the remaining four. The distal end of the catheter was connected to the pump, which was secured in place with silk sutures in line with manufacturer recommendations.
Post-operatively, patients were admitted and observed in the Pediatric Intensive Care Unit for monitoring. SSB therapy was started at 100μg/day using a simple continuous basal infusion rate and was increased 5% per day as tolerated and until a clinical response was demonstrated. In line with the standard of care following conventional intrathecal baclofen pump placement, oral baclofen was discontinued in conjunction with initiation of SSB. Other anti-spasticity medications, including tizanidine and benzodiazepines, were continued according to the standard of care to minimize withdrawal complications.
Follow up
Patients returned to the clinic for surgical wound check and infusion adjustment approximately 2 weeks after discharge. Subsequently, they returned for infusion rate adjustments with oral medication titration at 4- to 6-week intervals until stable symptom control was achieved. Pump refills were performed at 2- to 6-month intervals depending on pump size and infusion rate. As with intrathecal baclofen, refills were performed percutaneously as an outpatient procedure, and infusion adjustments were made using the handheld Medtronic clinician programmer. All patients have been maintained on a simple continuous infusion.
Analyses
Statistical analyses were performed with R 3.6.1 [14]. Shapiro-Wilk’s test was used to check for data normality and the Wilcoxon rank sum test was used to test for statistically significant change in dystonia score. Statistical significance was preset at p < 0.05. A 30% decrease in dystonia score was considered clinically significant. The STROBE cohort reporting guidelines were used in writing the current manuscript [15].
Results
The perioperative course was uncomplicated for all 6 patients. Although baclofen infusion at upper cervical, supraspinal, and intraventricular levels suggests an increased risk of central depression, including respiratory complications, the group had a remarkably uneventful initial post-operative course, without sedation, seizures, cerebrospinal fluid leak, infection, or hardware malfunction, in the first month following placement. Four of 6 patients were discharged to home by post-operative day 4. The patient in Case 1 underwent SSB pump placement in the last 3 weeks of a 4-month hospitalization for multiple concurrent medical complications related to their dystonia but unrelated to the SSB pump and was discharged to home on post-operative day 19 following completion of medical therapies. The post-operative course of the patient in Case 6 was notable for initial tachycardia and hypertension which had self-resolved by post-operative day 5; they were discharged to home on post-operative day 8.
As a group, mean FMDS scores decreased from 15.8±4.7 to 7.0±1.7 points from immediately pre-operatively to current across the 6 patients (Table 2). Among the 5 patients who continued with SSB therapy, change in mean FMDS scores from 15.0±4.7 to 7.0±1.7 points was statistically significant (U = 51, p = 0.0043). Two have since been titrated off all previous scheduled anti-spasticity medications. The patient in Case 4 had previously been poorly managed on a combination oral regimen at dosages well-above maximum dosing recommendations and now has significantly improved dystonia symptoms on SSB combined with standard dosing scheduled tizanidine. The patients in Case 2 and Case 3 experienced such significant functional improvement in symptoms that both elected for replacement of the programmable pump at 7 and 6 years, respectively, to continue SSB therapy. The patient in Case 1 died of an unrelated cause. At time of death in Case 1, they continued to demonstrate benefit from SSB with clinically significant improvement in dystonia score compared to pre-operative score.
Postoperative outcomes. For Case 1, current FMDS score, follow-up in months,
and SSB dose in ug/day are provided from the last documented evaluation
prior to death and are marked by parentheticals to differentiate from
current values documented for current patients. Abbreviations: FMDS
(modified Fahn-Marsden dystonia scale), SSB (supraspinal baclofen)
Postoperative outcomes. For Case 1, current FMDS score, follow-up in months, and SSB dose in ug/day are provided from the last documented evaluation prior to death and are marked by parentheticals to differentiate from current values documented for current patients. Abbreviations: FMDS (modified Fahn-Marsden dystonia scale), SSB (supraspinal baclofen)
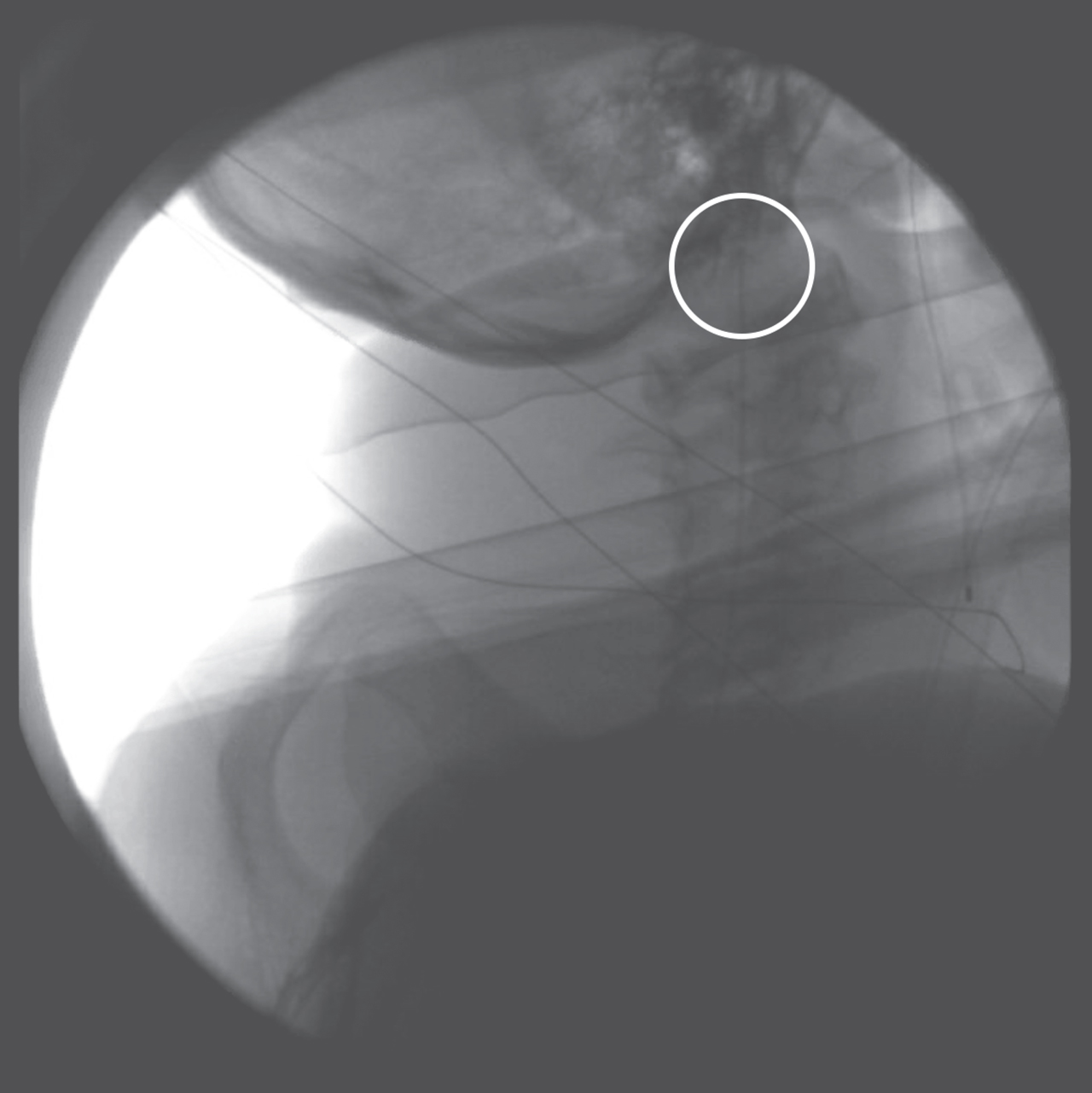
Intraoperative view of catheter tip placement at foramen magnum. White circle highlights location of catheter tip.
The patient in Case 6 previously had suboptimal response to high-dose intrathecal baclofen infusion with the catheter tip at T5 in addition to high-dose oral anti-spasticity medications. At the time of end-of-battery-life, the decision was made for intrathecal catheter tip reimplantation to a supraspinal location concurrent with pump replacement, and the patient did well. Interestingly, when the catheter tip was at T5, the patient was noted to have improvement in lower extremity spasticity symptoms and concurrent worsening of dystonia symptoms. Following reimplantation of the catheter tip for SSB infusion, they were seen with persistent good control of spasticity symptoms and improved control of dystonia symptoms thus resulting in a decrease in their oral anti-spasticity medications. At maximum therapeutic benefit, the patient was maintained at 586μg/day SSB with scheduled tizanidine 8 mg/day. Unfortunately, they suffered both tip migration and pump pocket infection approximately 2 months following supraspinal catheter tip re-implantation and had multiple operations for wound washout and replacement of the programmable pump, which ultimately failed. The ensuing post-operative course was further complicated by wound dehiscence, and the pump was explanted.
The novel supraspinal baclofen infusion technique shows promise to be a safe and effective treatment for patients with severe secondary dystonia who are unresponsive to oral medication. The current report appears to be the first to describe a method for baclofen delivery into the upper cerebrospinal fluid using an intraspinal catheter placed at the cisterna magna for the treatment of severe secondary dystonia.
The described SSB infusion method is a variant of the previous intraventricular baclofen (IVB) infusion system by Albright and colleagues [6, 16]. In IVB, a ventricular-peritoneal shunt catheter is endoscopically placed into the third ventricle, and the distal end of catheter is subcutaneously tunneled to the abdomen and connected to a programmable pump. IVB provides baclofen directly to the ventricular system, while SSB infuses baclofen into the cerebrospinal fluid at the cisterna magna with secondary cortical suffusion, thus more directly treating the pathophysiology underlying secondary dystonia. In both IVB and SSB, intraventricular baclofen delivery was demonstrated to improve dystonia symptoms in patients with severe secondary dystonia that had previously been unresponsive to oral medication and/or intrathecal baclofen. Compared to IVB, SSB decreases both surgical and hardware complexity by supraspinal baclofen infusion through intrathecal placement of the catheter tip at the cisterna magna. This circumvents the unconventional use of a ventriculoperitoneal shunt for drug delivery, and decreasing potential additional risks associated with the VP shunt catheter disconnection from the pump. By utilizing an intrathecal approach for supraspinal baclofen infusion, the SSB method conveys a decreased risk of intracranial injury compared to IVB.
Overall, patients responded favorably to SSB. Collectively, the group demonstrated both a clinically and statistically significant decrease in FMDS scores post-operatively. While the FMDS has been reported to be a relatively sensitive measure for capturing changes [13], measures that assess degree of functional deficit and level of disability were not followed and thus quantifying the benefits for activities of daily life in this study are limited. Although the group did anecdotally report benefits in daily life after SSB implantation, future studies on this method will certainly benefit from a more rigorous assessment scheme to adequately capture changes across the ICF framework of function and disability [17]. For the current cohort, the overall functional benefits from SSB are partially reflected in their subsequent ability to wean off oral medications. As SSB infusion rates were increased and dystonia symptoms improved, the patients were also able to tolerate substantial decreases in their total oral medication regimens with associated decrease in central sedating effects. As an exemplar, the patient in Case 6 previously had an intrathecal catheter with the tip placed at T5 without improvement in dystonia symptoms. However, following reimplantation of the catheter tip above the foramen magnum, their overall dystonia symptoms improved in conjunction with reduction in oral medication dosing. However, further studies will be helpful in assessing the degree to which SSB infusion differs from intrathecal baclofen delivery at upper cervical levels for improvement of dystonia symptoms.
None of the patients experienced respiratory depression either in the immediate or extended post-operative period. To minimize the risk of central effects with supraspinal baclofen delivery directly into the fourth ventricle, adjustments to infusion rate were limited to 5% in a 24-hour period. In conjunction with relatively slow infusion rate increase and decrease in oral medications, patients did not reach a stable state of dystonia symptom control until several months of SSB infusion. Further studies are needed to determine the maximal permissible rate of infusion increase, particularly in the monitored immediate post-operative period, to accelerate the time frame in which patients achieve stable symptom control.
There was one patient who unfortunately had multiple complications which ultimately led to pump removal. At 2-months following reimplantation of the catheter tip above the foramen magnum, the patient in Case 6 presented to hospital with baclofen withdrawal. They subsequently had multiple operations for replacement of the catheter tip, pump pocket wound washout, and pump replacement. In the setting of previous placement of a pump system and multiple operations to correct subsequent complications, this patient was at increased risk of infectious complications [18]. Unfortunately, following pump removal, the patient was not able to achieve dystonia symptom control on oral medications and experienced significant functional decline following removal. In order to minimize the number of operations and risk of infectious complications, it is critical to define therapeutic goals during counseling with patients who are considering a baclofen pump system in order to precisely select the optimal placement of the catheter tip at the initial implantation.
Supraspinal baclofen infusion is a promising novel technique as a safe mode of administration. It is effective for those with severe, functionally limiting, secondary generalized dystonia which was previously unresponsive to oral medications and intrathecal baclofen infusion. While the relatively small sample size is a limitation of the current study, the improvements demonstrated by the group with SSB represent important groundwork towards validating this novel intervention for severe secondary dystonia. Additional studies are still needed to substantiate the findings, refine infusion parameters, and specify patient selection criteria. In addition, long-term follow-up in a larger patient group is needed to determine if SSB for dystonia demonstrates decreased responsiveness with chronic infusion as seen in intrathecal baclofen for spasticity management [19]. Regardless, the current report identifies supraspinal baclofen as a promising novel approach for the treatment of severe secondary dystonia, a condition of significant functional implications and limited therapeutic options.
Footnotes
Acknowledgments
Intramural funding for this research was provided by the Department of Physical Medicine and Rehabilitation at the University of California Irvine Medical Center.
Conflict of interest
The authors have no conflicts of interest to report.
