Abstract
PURPOSE:
Progressive scoliosis significantly impacts the quality of life in patients with cerebral palsy (CP). Spinal fusion is the mainstay of treatment of progressive spinal curves. The current study aims to identify approaches used by pediatric spine surgeons to optimize care of patients with CP undergoing scoliosis surgery.
METHODS:
A 33-question survey was distributed electronically to 181 POSNA/SRS members with an established interest in pediatric spinal deformity surgery. Eighty one responses were obtained (45%). Using the Delphi consensus guidelines, agreement
RESULTS:
There was a consensus on 15 out of 33 questions (46%). 97% of responders identified nutrition status as a comorbidity which could be optimized. However, the timing and method of obtaining nutritional assessment varied. 92% of the surgeons stated that they used shared decision making with the family but only 22% used a formal decision aid. 83% use antifibrinolytics routinely, 81% used a surgical site infection prevention protocol, 78% obtained preoperative pulmonary consult, and 88% took steps postoperatively to prevent pulmonary complications.
CONCLUSION:
There is significant variability in the current practices of perioperative management of patients with CP undergoing scoliosis surgery. This data can be used in future studies to create a standardized integrated care pathway.
Introduction
Progressive scoliosis is common in patients with cerebral palsy (CP). Prevalence increases with age and in children with quadriplegia with GMFCS level 4 or 5 ambulatory function [1, 2]. Progressive neuromuscular scoliosis (NMS) significantly impacts the quality of life in patients with cerebral palsy. It affects sitting balance, increases the risk of ischial decubiti, costo-pelvic impingement and pain, as well as progression of restrictive lung disease [3]. Once the degree of curvature approaches 50 degrees it typically progresses rapidly [2, 3].
To improve quality of life, surgery is generally indicated when curves reach 50 or more degrees. The goals of surgical intervention are to prevent curve progression, restore spinal balance, facilitate seating and repositioning, decrease pain, and improve patients’ quality of life. Deformity correction in patients with CP leads to improved patient and parent satisfaction and subjective quality of life as well as improvements in cosmesis, sitting-balance outcomes, bodily pain, and social functioning [4, 5, 6, 7, 8].
Patients with neuromuscular scoliosis, including patients with cerebral palsy, are prone to increased rates of perioperative morbidity and complications compared to idiopathic scoliosis patients, which lead to increased hospital stays and costs [9, 10, 11]. A recent meta-analysis found pooled rates of pulmonary, implant, infectious, neurological, and pseudoarthrosis complications at 22.7%, 12.5%, 10.9%, 3.0%, and 1.9% respectively [12]. The increased complication rate compared to children with idiopathic scoliosis is likely multifactorial, stemming from increased comorbidities including seizures, restrictive pulmonary disease, gastro-esophageal reflux disease, poor nutrition, decreased ambulation, and poor cognition. In addition to complications, these factors lead to increased length of stay and hospital costs in patients with neuromuscular scoliosis undergoing spinal fusion surgery [11, 13].
In this patient population, interventions focused on minimizing adverse outcomes of scoliosis surgery in patients with CP could decrease the complication rate and costs associated with this surgery. However, there is currently little consensus and considerable absence of evidence to support best practices on how to manage these fragile patients [14]. There has also been no formalized effort for cost analysis or value stream mapping for this procedure in this patient population which has significant risks of complications and enormous costs. This lack of consensus and clarity leads to variation in surgeon practices and hospital protocols to optimize care in these patients. Previous studies on those with idiopathic scoliosis have shown that perioperative management practices vary between surgeons and institutions [15]. The goal of this study was to explore the spectrum of perioperative protocols used by pediatric spine surgeons to optimize care of patients with cerebral palsy undergoing scoliosis surgery.
Methods
Prior to the start of this study, approval was obtained from our institution’s Institutional Review Board. All procedures performed were in accordance with the ethical standards of the institution where the study was conducted. Informed consent was obtained for all participants.
A questionnaire survey was distributed electronically to 181 members of the Pediatric Orthopedic Society of North America (POSNA), the American Academy of Cerebral Palsy and Developmental Medicine (AACPDM), and the Scoliosis Research Society (SRS) with established interest in pediatric spinal deformity surgery in children with cerebral palsy. Subjects were first identified by orthopedic membership in AACPDM who also perform spine surgery. The secondary identification was by a search of POSNA and SRS members at the primary children’s hospitals at all major metropolitan areas in the United States; those members were then asked to identify the orthopedists at those sites who performed the majority of the neuromuscular spine procedures at their institution.
The survey included 33 questions targeting surgeons’ current practices and approaches to pediatric NMS surgery, especially in children with cerebral palsy. Members of the Complex Care Committee of the AACPDM and the authors composed the questions; since this was the first survey of its kind on this topic, the questionnaire was not validated. This committee is a multidisciplinary group that focuses on the care of medically complex children and adolescents, and is comprised of pediatric orthopedic surgeons, physical medicine and rehabilitation physicians, pediatricians, neurologists, internal medicine/pediatric experts, nutritionists, nurses, therapists, and researchers. All responses were evaluated by the senior author as well as two well established pediatric orthopedic surgeons, (with predominantly cerebral palsy related practices) who were consulted to develop the questionnaire and review the results. Questions focused on identifying perioperative management practices of each surgeon. The questions are listed in Fig. 1. When appropriate, an open-ended response “other (please specify)” was included to accommodate additional comments or choices not listed as options. The questionnaire survey was then reviewed and approved by the complex care committee of the AACPDM before it was sent out to the targeted recipients to ensure quality and clarity of the questions.
(a) Preoperative survey questions; (b) Perioperative and postoperative survey questions.
Statistics: Descriptive statistics were used for data analysis. Using the Delphi consensus guidelines, agreement
Response rates and surgeon demographics: A total of 81 responses were obtained, representing a 45% response rate. There was a large variance in the number of NMS patients seen yearly at their respective centers. On average, each center saw 199 (range, 100–800, standard deviation 65) patients with cerebral palsy and scoliosis per year; the mean number of operative cases per site was 63 (range 10–112, standard deviation 32). Ninety percent of the surgeons were in academic practices and 10% were in private practice settings.
There were several areas of agreement and discordance amongst responders. Using a cutoff of 75% agreement for concordance, there was responder consensus on 15 out of 33 questions (Fig. 1).
Several questions addressed peri-operative protocols and team approaches. Seventy seven percent of surgeons reported that they have specific pre-operative protocols in place for the management of patients with cerebral palsy undergoing scoliosis surgery (Fig. 1). Similarly, 75% of surgeons had post-operative protocols in place. Ninety two percent of surgeons reported using some method of shared decision-making with the family. However, only 22% used a standardized decision aid to assist them with clinical decisions. Sixty percent reported having a dedicated multidisciplinary team or hospitalists to co-manage patients peri-operatively.
Surgeon’s perceptions of comorbidities impacting pre-, intra-, and post-operative care in patients with cerebral palsy undergoing scoliosis surgery.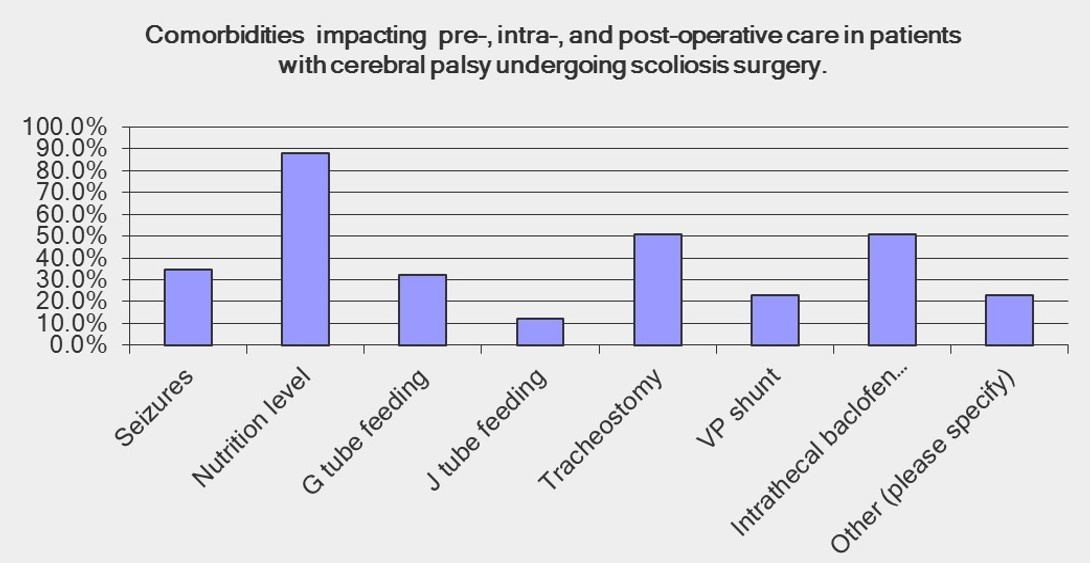
Respondents were given a series of questions concerning the management of comorbidities (Fig. 2). Eighty eight percent of responders chose nutrition level as a comorbidity which significantly affects perioperative care, with 97% indicating that nutritional status is a comorbidity which can be optimized. Eighty four percent performed a nutritional status evaluation before surgery. However, there was no clear consensus as to the timing of the nutritional assessment; twenty six percent evaluate nutrition at 12 weeks, 36% at 6 weeks and 13% immediately preoperatively. Additionally, the measurements used to evaluate the assessment of nutritional status varied. Albumin/prealbumin was used to assess nutritional status by 44% of surgeons, 16% use absolute lymphocyte count, and 14% use intake assessment.
Eighty one percent of those surveyed reported having a specific NMS protocol for preventing surgical site infection (SSI) which differs from the protocol used in adolescent idiopathic scoliosis (AIS) patients. Eighty three percent used antifibrinolytics routinely and 76% used the same method of optimizing blood management as in patients with adolescent idiopathic scoliosis. Eighty eight percent took steps postoperatively to prevent pulmonary complications.
Several questions addressed medication management. Only 34% percent of respondents altered seizure medications preoperatively. Fifty-three percent of surgeons recommended medications to protect gastric mucosa and optimize gastrointestinal motility postoperatively. In patients with a baclofen pump, 92% opt to carefully work around the catheter surgically, and only rely on catheter reinsertion by an orthopedic surgeon or neurosurgeon as needed.
Surveyors were asked a series of questions regarding preoperative clearance. In patients with ventriculoperitoneal shunts, 52% of surgeons obtained a preoperative neurosurgical evaluation. Seventy-eight percent of surgeons consulted pulmonary before surgery. Only 35% ordered a gastrointestinal evaluation preoperatively.
Various preoperative testing protocols were surveyed. Only 28% used serum laboratory values to assess vitamin D and calcium levels preoperatively. Testing for urinary tract infections was done by 60% of surgeons. Forty-two percent assessed dental health preoperatively.
Surgeons were asked about intra-operative spinal cord monitoring. Fifty-three percent of surgeons monitored somatosensory evoked potentials and motor evoked potentials intra-operatively on every child where signals could be obtained, while 30% monitored every child regardless. Seventeen percent of surgeons monitored only children with lower extremity functional ability.
Finally, pain management varied considerably amongst responders. Sixty-one percent reported using a nurse or family-controlled PCA. Fifty-nine percent use IV morphine and valium. Twenty percent use an intraoperative epidural injection. Forty-nine percent use a specific pain scale to assess postoperative pain.
Preoperative
Currently, there are no consensus guidelines in place to optimize outcomes in patients with cerebral palsy undergoing scoliosis surgery. Surgeons have adopted practices based on their training, experience, and the policies of their institution. Seventy-five percent of surveyed surgeons reported having either preoperative or postoperative management protocols in place for the treatment of patients with CP undergoing scoliosis surgery. However, the wide distribution of responses regarding specific interventions reveals that protocols vary substantially. Establishing such a guideline is not without precedence. Mullender et al. recently published a consensus guideline focused on scoliosis surgery in patients with spinal muscular atrophy (SMA) and Duchenne’s muscular dystrophy. It provides a range of recommendations regarding issues such as respiratory function monitoring, spinal cord monitoring, and specific indications for surgery [17]. The first step in creating a similar surgical guideline for CP scoliosis surgery is to identify concordant and discordant practices amongst surgeons. Ultimately, this may allow for standardization of practices and improvement of outcomes. The goal of this study was to explore the spectrum of perioperative protocols used by pediatric spine surgeons to optimize care of patients with cerebral palsy undergoing scoliosis surgery. By identifying concordant and discordant practices, the information gathered by this study may be used as a focus for future discussion and for initiating future investigations in the development of best practice guidelines for cerebral palsy scoliosis surgery. Ultimately, this knowledge could be used to design prospective multicenter collaborative studies to optimize outcomes, reduce complications and healthcare costs in patients with cerebral palsy undergoing spine deformity correction surgery.
There were several interesting findings regarding preoperative care in our survey. The majority of surveyed surgeons identified nutrition as the comorbidity which most impacts perioperative care and the factor that could be better optimized. However, the approaches taken to manage this risk are diverse. There was considerable variability in the timing and content of preoperative nutritional assessments, and some surgeons do not obtain a nutritional evaluation at all. This is likely explained by a lack of high quality evidence on which to base such decisions [14, 18]. A recent systematic review by Wessner et al. found that there were few high quality studies addressing nutrition in pediatric surgical patients, with no recommendations given higher than Grade D evidence [11]. Vitale et al. reported that patients with NMS should have a preoperative nutritional assessment by measuring albumin and prealbumin, but this was done by expert panel consensus [19].
Efforts at family-centered care, and involving the family more in key health decisions are an important evolution of health care in the 21st century. However, the degree to which CP-focused surgeons seek family involvement and use shared decision aids varies. Only a minority of surgeons reported the implementation of some kind of a formal decision tool to help families in surgical planning, despite findings that such tools may result in improvements in families’ knowledge gain, satisfaction, and decisional conflict [20]. Also, the opinions of the effectiveness of the shared decision making tool from the patients’ and families’ perspective was not captured in this study and could differ from that of the surgeon.
A limited number of surgeons alter seizure medications such as Depakote preoperatively. This is likely explained by conflicting evidence regarding the extent to which valproic acid affects bleeding risk [21, 22]. There are no definitive guidelines to aid decisions regarding seizure medication management prior to NMS surgery, although a study by Sarwark et al. recommended that these medications need to at least be evaluated prior to surgery [23].
Urinary tract infection is a modifiable risk factor for surgical site infection which is not routinely tested for by CP surgeons. Two level IV studies suggest that the organism causing UTIs in this patient population is frequently found to be the same organism cultured from subsequent surgical site infections [15]. Vitale et al. included preoperative urine cultures in his guidelines for high-risk pediatric spine surgery patients [19].
Our survey demonstrated practice variance in several additional areas of preoperative care such as G tube management, bone health optimization, and dental health assessment. There is a paucity of published data analyzing these issues amongst patients with CP undergoing spinal surgery.
Intraoperative
The majority of surgeons use a protocol for SSI prevention in CP scoliosis surgery that differs from their AIS protocol. This is likely due to evidence that surgical site infections (SSIs) amongst AIS surgical patients occur at a lower rate and tend to be gram positive organisms such as Staphylococcus aureus. In contrast, nearly half of NMS SSIs contain at least one gram-negative organism, with Pseudomonas aeruginosa being the most common [24]. Although this survey did not address specific antimicrobial protocols used, McLeod et al. found that such practices vary across the United States of America [24]. By expert consensus, Vitale et al. recommended that patients should receive perioperative IV cefazolin along with an IV gram negative antimicrobial. Adherence to such regimens should be standardized [19].
Surgeons were divided in their use of somatosensory evoked potentials (SSEPs) and motor evoked potentials (MEPs), likely due to conflicting literature regarding the reliability of spinal cord monitoring in patients with NMS [25]. Sarwark et al. concluded that while multimodality monitoring is useful in children with NMS, there are not reliable data to show benefit in patients with cerebral palsy with severe cognitive impairment [23].
Antifibrinolytics were used routinely by the majority of surgeons, a trend supported by recent data. In a systematic review, Wang et al. found that antifibrinolytics such as transexamic acid (TXA) reduce perioperative blood loss, with no evidence that use of such agents is a risk factor for adverse events [26].
Vitale et al. recommended additional intraoperative guidelines including limiting operating room traffic when possible, not using UV light in the OR, using intraoperative wound irrigation, and using vancomycin powder in the bone graft and/or surgical site [19]. These particular practices were not assessed in our survey but are important points to consider in the creation of future guidelines.
Postoperative
About half of the surgeons surveyed use a multidisciplinary team or hospitalists to co-manage their patients postoperatively. There is some evidence that this may reduce variation in outcomes and improve quality, though with increased initial costs [27]. Surgeons also varied on methods used to optimize pain control and to assess pain. Other postoperative guidelines not addressed in our survey but proposed by previous authors include using impervious dressings and minimizing postoperative dressing changes prior to discharge [19]. Other postoperative strategies to investigate include frequent positional changes and specialty beds to prevent decubitus ulcers, use of drains, and postoperative antibiotic timing.
Pain assessment in this population can be quite challenging due to co-existing intellectual and communicative disabilities. Efforts to better understand how to optimize postoperative pain control, especially after orthopedic surgery in children with CP, have been undertaken with some moderate success [28, 29]. A multi-disciplinary pain team of orthopedics, physical medicine, nursing, and anesthesia can minimize pain and result in improved patient satisfaction.
Although there is some published literature that may be relevant to many of the questions in this survey, it is clear that the quality of this literature is poor [11]. Therefore, it is not surprising that there is a general lack of consensus among the participating surgeons on many of the questions of this survey. This study is only the first step in that it establishes lack of consensus and variability in practice. The next steps would be to perform a systematic review of the literature, organizing the evidence and creation of evidence based consensus guidelines. Similar best practice guidelines (BPG) have been published by the Quality, Safety and Value Committee of the POSNA regarding prevention of surgical site infection following high risk pediatric spine surgery. These guidelines will then need to be validated. Further, prospective multicenter research studies would need to be conducted to see if these initiatives optimize care of children with CP who have scoliosis surgery, resulting in reduced health care costs. Also, the purpose of this initial survey was to assess current practices by pediatric orthopedists who traditionally have been the attending physicians in these areas of care. Expanding this survey to other members of the typical multi-disciplinary team, such as intensivists, pulmonologists, hospitalists, and physiatrists would be a logical next step in this research.
Study limitations
There are several limitations to this study. An electronic survey is subject to non-response bias. Our response rate of 45% is low, but is common for a survey of this type published in the literature. There is no scientifically proven minimally acceptable response rate although a response rate of 60% has been used as a threshold of acceptability by some. There is increasing consensus that the degree to which the sampled respondents differ from the survey population as a whole (i.e., nonresponse bias) is central to assessing the representativeness of a survey, rather than response rates [30].
The field of pediatric spinal surgery is relatively small, especially those with experience in caring for children with cerebral palsy and scoliosis. Hence, it is assumed that the respondents were representative of the group of spinal surgeons surveyed. However, this assumption may have led to bias in the survey results. A number of design-based strategies were used to optimize the participation rates of the survey [30]. The use of multiple choice questions may not have captured the most accurate possible response to each given question but is necessary in a broad response survey to assess trends. Our survey attempted to assess a wide range of perioperative variables, although certain practice variances could have been missed. Despite these limitations, the opinions of a large number of spinal surgeons were incorporated. Although reduction in variability of practice has been effective in optimizing patient care in other specialties, this has not been proven in children with cerebral palsy and scoliosis. This survey represents a summary of surgeon responses and is essentially anecdotal. It also assumes that the variability in practice is due to lack of evidence on best practices and not due to lack of awareness of the evidence. However, surveys of this type are essential to identify variability in practice and highlight issues that could be improved to optimize patient care. The information gathered by this study may be used as a starting point for future discussion in the development of best practice guidelines and to design multicenter collaborative studies, optimize outcomes, as well as reduce complications and healthcare costs in patients with cerebral palsy undergoing spine deformity correction surgery.
There is significant variability in the current practices of pediatric spine deformity surgeons with regard to optimizing perioperative management of patients with CP having scoliosis surgery. There was a clear consensus that specific perioperative protocols should be in place for this fragile population in order to manage blood loss, infection risk, nutritional status and pulmonary complications. However, there were several areas of discordance. This is likely due to a lack of high quality evidence available to guide decisions. The data in this study can be used to develop best practice guidelines and collaborative prospective multicenter studies to optimize care, improve outcomes, and to reduce complications and healthcare costs in patients with cerebral palsy having scoliosis surgery.
Footnotes
Acknowledgments
The authors would like to acknowledge the contributions to this project by POSNA, AACPDM, and SRS member participants, as well as those of the AACPDM complex care committee. No external support or research funding was received for the execution of this project.
Conflict of interest
The authors have no conflicts of interest to report.
