Abstract
PURPOSE:
To explore the effects of neuroprosthesis use on participation, level of community-based walking activity, safety and satisfaction in children with hemiplegic CP.
METHODS:
Eleven children (mean 9 years 11 months) with hemiplegic CP Gross Motor Function Classification System (GMFCS) Level I and II participated in a 16-week intervention using the Ness L300 neuroprosthesis. Outcome measures included satisfaction and performance with self-selected participation goals (Canadian Occupational Performance Measure (COPM)), level of community-based walking activity (Step Watch Activity Monitor (SAM)), trip and fall frequency (caregiver report) and a satisfaction questionnaire.
RESULTS:
Significant (
CONCLUSION:
Results indicate that daily neuroprosthesis use may improve performance and satisfaction with participation goals and reduce trips. No changes in community-based walking activity were noted. Further study is needed to examine response based on GMFCS levels, across geographical regions and between FES neuroprosthesis and a control group.
Keywords
Introduction
Cerebral palsy (CP) describes a group of permanent disorders of movement and posture that are attributed to non-progressive disturbances in the developing brain [1] with a prevalence rate in the United States of 3.1–3.6 per 1000 [2]. Children with CP are less involved in physical or skill-based community activities than their typically developing peers [3, 4] and demonstrate lower levels of daily walking activity [5, 6]. Hemiplegia accounts for 22.6–40% of all cases of CP [7, 8, 9, 10, 11], often affecting children with relatively high functional and cognitive levels [12].
The International Classification of Functioning, Disability and Health (ICF) framework provides an integrated and universal language for measuring health and disability applied across rehabilitation studies [13]. The ICF framework reflects a dynamic interaction between person and environment with a move toward participation as an important outcome of health [14]. This has resulted in a shift of clinical research focus from body structure and function (BSF) to an increased emphasis on activity and participation outcomes [15]. Outcomes used to measure intervention effectiveness should encompass the impact of what rehabilitation specialists offer at different levels across the ICF [14].
One treatment option for hemiplegia involves wearing an ankle foot orthosis (AFO) to improve ankle position and stability for walking. AFOs may limit active movement, potentially hinder motor learning, and contribute to muscle weakness in CP [16]. Older children are often unwilling to wear an AFO due to the bulk and discomfort [17]. Newer technology such as functional electrical stimulation (FES) neuroprostheses provide an alternative to AFOs for foot drop. Unlike older electrical stimulation devices, these newer devices do not have exposed cords or require repositioning of electrodes with each use [18]. Use of FES neuroprostheses have been shown to increase neuroplasticity, function and quality of life in adults with hemiplegia [19, 20, 21, 22, 23, 24, 25, 26, 27].
To date, studies of pediatric FES neuroprosthesis use have evaluated effects on BSF outcomes [13, 16, 17, 28, 29] using clinic-based measures. Specifically improvements in ankle range of motion (ROM) [28], gait kinematics [16, 17, 29], muscle size [16], muscle control [16, 28], strength [28] and reduced spasticity [28] have been reported. One study [28] assessed safety and reported a reduction in trips and falls during and after FES. Another study assessed satisfaction [29] and found 86% of subjects were so satisfied they chose to continue to use the device.
Clinic-based measures examine a child’s capability (what he can do) in a controlled environment, in contrast to actual performance in an everyday more natural environment [30]. Studies show that children with CP demonstrate differences in performance across settings [31, 32]. Using the Gross Motor Function Measure (GMFM) and parent questionnaires to describe motor performance in 307 children with CP, Tieman [31] found subject’s mobility methods differed across home, school and community settings. Holsbeeke [32] found low correlations between clinic-based measures (GMFM) and real world motor performance (Pediatric Evaluation of Disability Inventory) in 85 young children with CP illustrating the role that environment plays in motor performance. Based on current literature, a gap exists regarding the effects of a neuroprosthesis on participation of children with CP in their natural environment and level of community-based walking activity. The use of new technology, such as a neuroprosthesis, not only requires understanding the changes it imposes on an individual’s capacity in a structured clinical environment; practitioners also need to understand how these interventions affect a child’s community-based activity and participation in daily life and meaningful activities [33].
The purpose of this pilot study was to explore the effects of FES neuroprosthesis use on participation and community-based activity level in children with hemiplegic CP. We further sought to augment existing literature regarding user safety and satisfaction associated with the use of a neuroprosthesis.
Methods
This study was approved by the Institutional Review Board. Written parental consent and participant assent were obtained prior to participation, in accordance with institutional policy.
Design
This study was part of a larger two-factor repeated measures pilot study [34] of FES neuroprosthesis intervention that assessed clinic-based measures of gait and function at baseline and post-intervention with the stimulation turned off and repeated with the stimulation turned on. Also obtained in the larger study, and the focus of this report, were self-selected individualized participation goals, community-based walking activity level, safety data on trip and fall frequency and user satisfaction.
Inclusion and exclusion criteria
Inclusion and exclusion criteria
Given the exploratory nature of this study, sample size estimation was not
performed for these outcomes. Participants included the same 11 individuals with
hemiplegic CP (mean age 9 years 11 months) enrolled in the larger study evaluating the use
of FES neuroprosthesis on clinic-based measures of gait and function reported
previously [34] Inclusion and exclusion
criteria are presented in Table 1. Upon enrollment,
demographic information, GMFCS classification level, brace wear, and range of motion for
the affected ankle were obtained. The majority of participants (
Measures
Canadian Occupational Performance Measure
Performance and satisfaction with individualized participation goals were assessed at baseline and post with the Canadian Occupational Performance Measure (COPM) [35], a standardized client-centered semi-structured interview designed to detect change in a client’s self-perception of occupational performance over time [36]. At enrollment, the caregiver and/or child (dependent on willingness to contribute) identified up to four goals where they anticipated positive outcomes following neuroprosthesis use. The performance and satisfaction scores for each goal are summed and averaged to result in a score up to 10. The same caregiver identified and rated the goals at the initial interview and post assessment. Caregivers and participants did not view previous results. Overall scores generated from the COPM were utilized for analysis.
Step Watch Activity Monitor
Community-based walking activity level was measured using the Step
Watch Activity Monitor (SAM) (Orthocare Innovations Oklahoma City, OK), a light- weight
motion sensor device worn around the ankle that records step counts from a single
leg [37]. It is a valid and reliable
instrument to measure physical activity in CP [5, 38]. The SAM was fitted to the
lateral side of the uninvolved ankle and calibrated to the child’s height and walking
pattern. Previous studies report on SAM data capture ranging from 3 [37] to 7 days [5].
The child was instructed to wear the SAM for three typical days in the week (consecutive
days not required) prior to beginning the protocol and for three days during the last
week of intervention during all of their waking hours except when bathing or swimming.
Step activity data were analyzed to obtain mean values of total daily step activity,
percentage of daily time active, percentage of daily active time spent in low intensity
(1–15 steps/min), moderate intensity (16–40 steps/min), and high intensity
(
Falls and trips
A fall and trip questionnaire was completed as a measure of safety. At baseline and post, the parent with child input completed a short questionnaire regarding trips and falls experienced in the previous month. In addition, parents were asked to report any falls or trips that occurred during the entire study period that included any injury requiring medical attention.
Caregiver and child satisfaction
A satisfaction questionnaire [27, 39] was completed by the
caregiver and separately by the child (
Intervention
The Ness L300 Foot Drop System manufactured by Bioness Inc. (Valencia CA, USA) was utilized for this study. The system consists of three components that communicate via radio frequency signals: 1) cuff with integrated stimulation unit and electrodes, 2) a gait sensor placed in the shoe that detects the force under the foot using a force sensitive resistor and 3) a control unit. Algorithms analyze the gait sensor data to detect heel strike and toe off in real time, and use this information to control stimulation of the ankle dorsiflexors. The protocol described previously [34] consisted of a 4 week accommodation phase followed by 12 weeks of recommended daily use of 6 hours day. Details of the protocol included screening and enrollment, baseline assessment within 2 weeks of screening, a four week accommodation period consisting of seven PT visits for the purpose of learning to use the device and gradually increasing wear time, 12 weeks of daily use including monthly well check PT visits, and a post assessment. The manufacturer’s protocol for home use was followed in setting stimulation parameters, providing maintenance and skin care guidelines, performing the initial fitting, and providing instruction for the accommodation period [40]. No additional PT outside of the described protocol was provided during the study protocol. Families were instructed to keep a diary noting hours of use each day, skin integrity and other concerns. The data reported here represent satisfaction and performance with participation goals, community-based walking activity level, and caregiver report on safety and satisfaction with the neuroprosthesis device.
Data analysis
Study data were collected and managed using Research Electronic Data
Capture (REDCap) [41]. All analyses were
completed with SAS 9.3 with an alpha level of
Results
Results were completed for data on 11 participants (mean age 9 years, 11 months). Due to an L300 firmware error that was not detected prior to enrollment, data from the neuroprosthesis device were not available to accurately calculate wear time or number of steps taken per day while wearing the device. Based on sufficient completion of the diary by eight caregivers, four individuals averaged six or more hours of wear per day and four averaged 3–6 hours per day. Three participants documented greater than 60 days of wear, four between 30–60 days of wear, and one less than 30 days of wear during the 12 weeks.
COPM performance and satisfaction scores were distributed normally and assessed
with the paired t test. Significant improvements were noted in COPM performance scores with
a mean difference of 2.44 points (95% CI
Perfomance and Satisfaction with Canadian Occupational Performance Measure (COPM)
goals at Baseline and Post FES neuroprosthesis intervention. 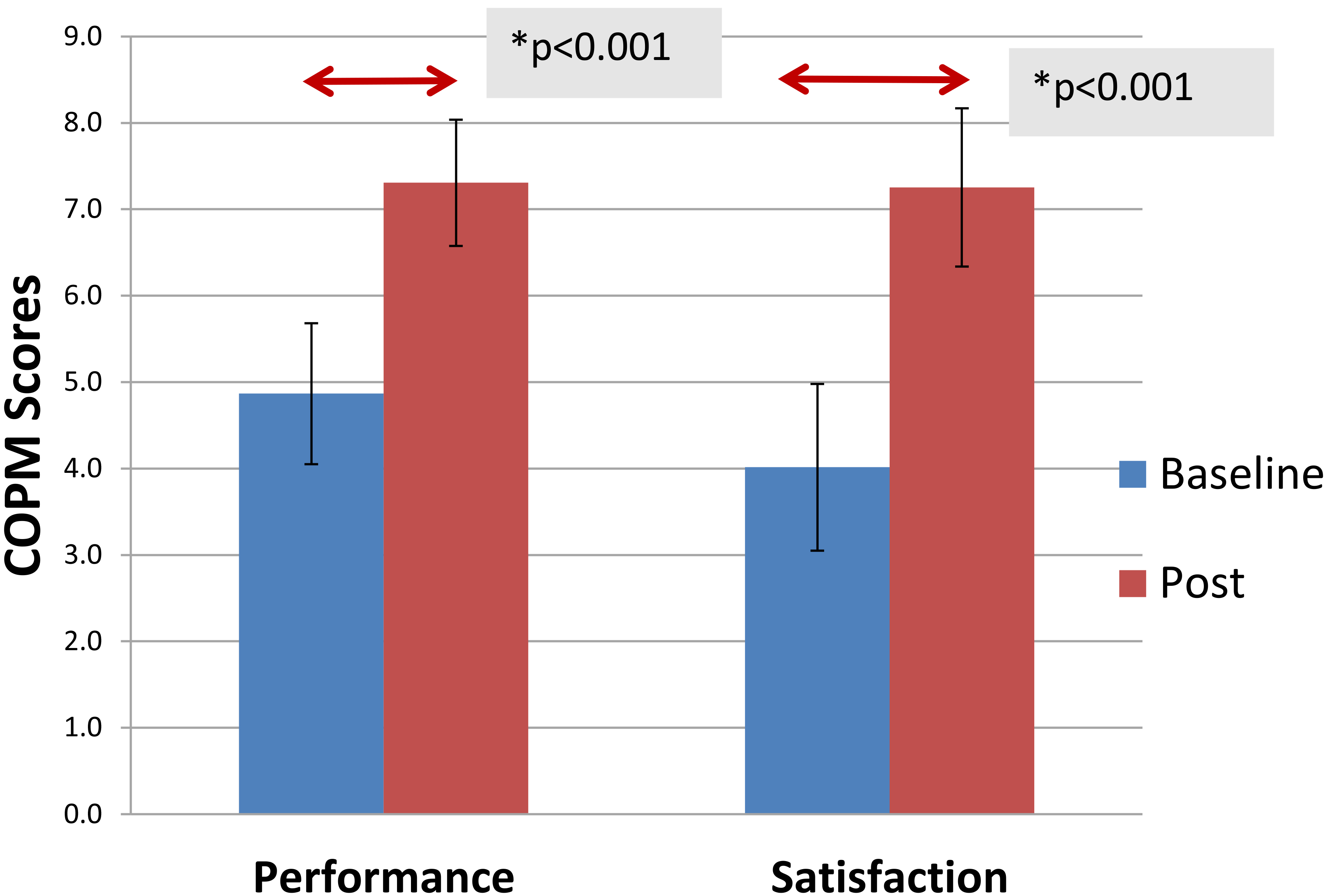
Reported falls and trips at Baseline and Post. 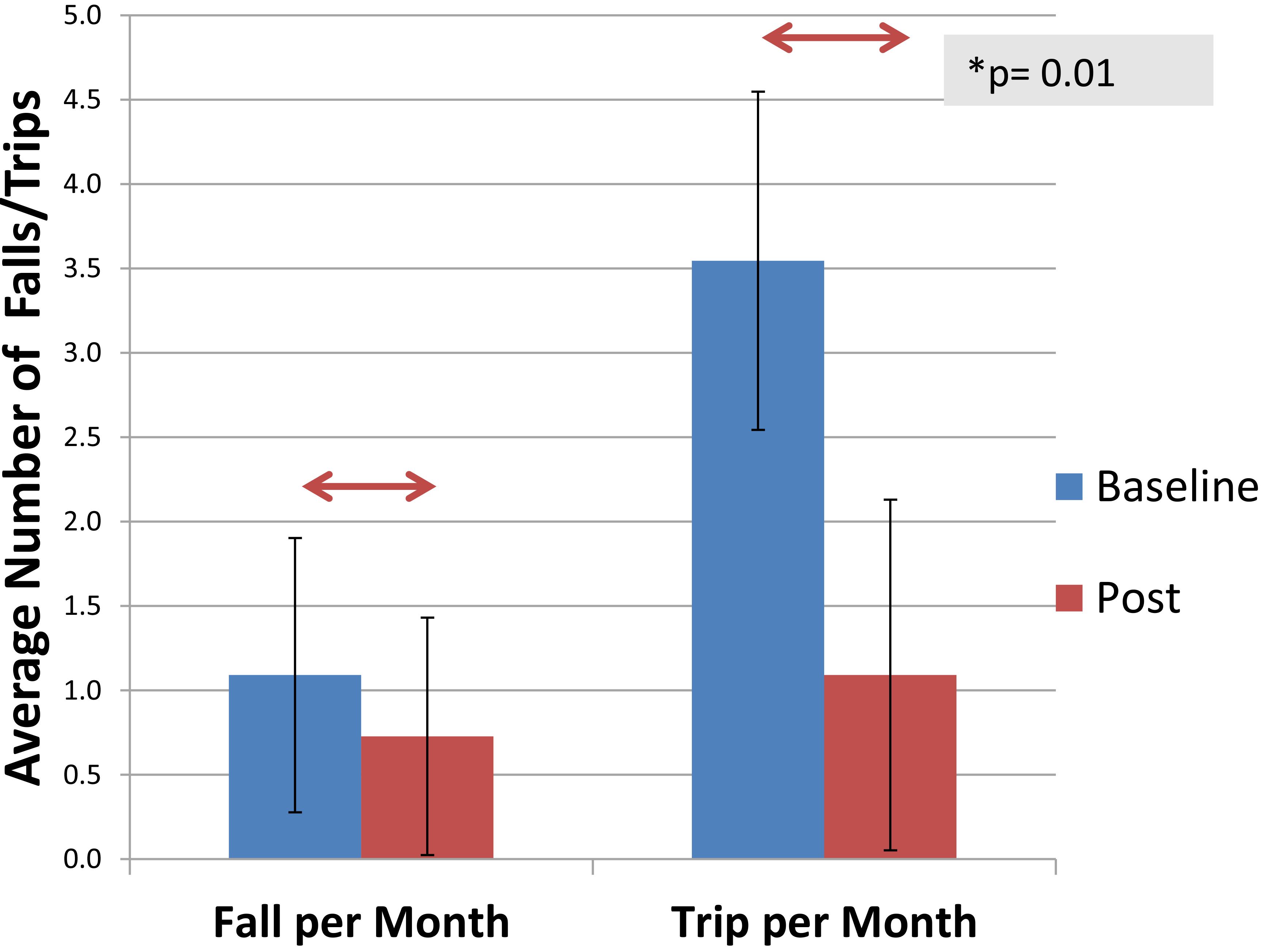
Community-based walking activity data downloaded from the SAM were available for
10 participants. SAM scores were distributed normally and assessed with the paired t test.
No significant differences were found in average daily total step count (
Baseline and post values for step watch values (
Results provided as Mean (SD)
Results from satisfaction questionnaire
The number of falls and trips were not distributed normally and were evaluated
with the Wilcoxon signed rank test. There was no significant difference in falls reported at
baseline and post with a median difference of
Eleven caregivers and three children completed the satisfaction questionnaire.
Average score for caregiver report (
This is the first study to evaluate the effects of FES neuroprosthesis use on participation and level of community-based walking activity in children with hemiplegic CP. This study demonstrated that daily use of an FES neuroprosthesis may improve satisfaction and performance with self-selected participation goals and decrease the frequency of trips. Further, satisfaction was high with all that completed the satisfaction questionnaire (11 parents, 3 children) wishing to continue to use the device at the end of the study.
This study did not demonstrate improvements in level of community-based walking
activity as measured by the SAM device which is consistent with other intervention studies
in children with CP [42, 43]. In a pre-post intervention study of children with CP (ages
4–12 years), Christy [42] assessed the effect of a
3 week intensive therapy program on walking activity. In a randomized crossover trial of
children with CP (ages 3–6 years) Bjornson compared walking activity in an AFO to non AFO
condition [43]. Both studies utilized five days
of SAM data for analysis. While the current activity dataset is more limited due to device
availability (three days of data monitoring instead of five), the baseline average daily
step count in our sample (4678, SD 1772) is similar to Bjornson [43] (4660, SD 1421) but greater than Christy [42] (2575, SD 1766). While both prior studies [42, 43] included children
GMFCS Levels I-III, Christy [42] included a
majority of children at GMFCS Level III who walk with an assistive device which likely
explains their lower average number of daily steps. In an earlier study by Bjornson [5], step activity in 81 youth with CP GMFCS Levels
I-III (ages 10–13) was compared to that of typical children. The average daily step count in
the current study was also similar to the 81 youth with CP in that study (4222 SD 2319). A
study by Stevens [6] compared 4 days of step
activity data in a younger group (4
Children in the current study spent less time active (22.6%) than the groups studied by Bjornson [43] (AFO on (49%) and AFO off (46%)), Bjornson [5] (40.2%) and Christy [42] (34.7%), but similar time active to Stevens [6] older group (19.3%). Some of these difference may be due to age as Bjornson [43] included ages 3–6 years. However age cannot explain the differences entirely as the children studied by Christy [42] were 4–12 years, and Bjornson [5] included children 10–13 years.
In assessing the ratio of medium level to low level activity, children in the present study demonstrated a ratio of 0.51. This is greater than findings of both Bjornson [5] (0.33), and Stevens [6] older group (0.27), suggesting that the children in our study spent relatively more time in medium level activity. Last, the children in our study spent less time in high activity (2%) than children in both the Bjornson [5] (5.6 %) and Stevens [6] (4.5 %) studies. These differences in % time active, ratio of medium to low activity and % time at high activity may be due to geographical variations in children’s physical activity. Children in the current study are from Ohio, while Bjornson’s studies [5, 43] included children from Washington, Christy [42] from Alabama, and Stevens [6] from Tennessee. We are not aware of any studies that compare physical activity levels in children with CP across regions in the US. However geographic differences in children’s activity level have been reported in a nationally representative sample of typical children [44]. Specifically Sisson [44] reported more children in Washington participate in greater than or equal to 3 days a week of physical activity compared to children in Ohio or Tennessee. Further information regarding physical activity levels for children with CP across regions of the US would be useful.
Results from this study are in agreement with other studies reporting the COPM
as responsive to meaningful changes due to therapy interventions [36, 42, 45]. In the current study, the mean improvement in
performance and satisfaction was clinically meaningful
Similar to the adult literature using this satisfaction questionnaire [27, 39], a majority of our participants were satisfied and all wanted to continue to use the FES neuroprosthesis after the intervention period. When considering interventions, therapists should choose options that reflect the motivations and interests of the individual [47]. The oldest participant in our study (16 years) preferred the neuroprosthesis to her AFO because she could easily turn it off and drive without removing the device. She was unable to drive while wearing her AFO. Wingstrand [48] reports AFO wear decreases with age which is consistent with the clinical experience of these investigators. It has been our experience that some children, especially adolescents, may be more motivated to wear a neuroprosthesis over an AFO due to its novelty and perceived technological benefits.
Reducing falls and trips is a possible benefit of FES neuroprosthesis intervention [16]. Similar to Pool [28], our study reports a decrease in the number of trips but unlike Pool, we did not report a decrease in falls. While the current study relied on recall of the participant and caregiver Pool used a five point ordinal scale ranging from “I never fall over” to “I fall over every day”. This scale may have improved the ability to detect change in reported falls and trips. Prospective data collection on falls and trips prior to intervention should be used in future studies to minimize recall bias.
Limitations of this study include the lack of repeated baseline measures, use of a small sample of convenience, the absence of a comparison group and the potential for intervention bias as the caregivers may have anticipated improvement in trips/falls compared to their recollection of prior trips or falls. Additional weaknesses were in reliance on the caregiver’s diary of device use, and limited activity monitoring due to the reduced availability of SAM devices. Future studies may benefit from improved body sensor technology to track neuroprosthesis wear time rather than relying on caregiver diaries of device wear.
Conclusion
The purpose of this pilot study was to investigate the effects of FES neuroprosthesis on performance and satisfaction with participation goals, level of community-based walking activity and to monitor safety and satisfaction with its use in children with hemiplegia. Findings suggest that use of an FES neuroprosthesis resulted in positive effects on self-selected participation goals and self-reported trips. Further, satisfaction was high when using the device with a high level of support that included therapy visits and communication with therapists trained in using the device. To our knowledge this is the first study to measure participation and community-based walking activity after neuroprosthesis use. Future research should examine the effects of FES neuroprosthesis on participation and community-based walking activity in a larger sample of individuals with hemiplegic CP with varying baseline function, across geographical regions and compare FES neuroprosthesis to AFO. Validated measures of satisfaction would be important to identify factors contributing to long term use of the device.
Footnotes
Acknowledgments
This study was approved by the primary investigator’s Institutional Review Board #2012-4682. This study was funded by a grant from the Pedal with Pete Foundation. We gratefully acknowledge the children and parents who participated, and Bioness Inc. for donating the NESSL300 units for use in this study.
Conflict of interest
The authors report no conflicts of interest.
