Abstract
BACKGROUND:
An increasing amount of research has supported the positive role of repetitive transcranial magnetic stimulation(rTMS) in improving functional impairments in Parkinson’s patients.
PURPOSE:
To explore the effectiveness of using rTMS n and Balance Apparatus training to improve the balance function of Parkinson’s patients.
METHODS:
Patients were divided into 3 groups of 20 patients in each. All patients received routine rehabilitation training. The rTMS group received low-frequency rTMS therapy, while the balance apparatus group incorporated balance apparatus exercises. The observation group received both interventions. The three groups of patients were assessed at baseline and after 8 weeks of treatment using the Unified Parkinson’s Disease Rating Scale (UPDRS) motor section, Berg balance scale (BBS), Time up and go test (TUGT), and Activities-Specific Balance Confidence (ABC) scales. Additionally, all participants were tested for movement ellipse area, and Average Trace Error (ATE) using the balance apparatus.
RESULTS:
The observation group achieved better treatment results. The UPDRS score was better than that of the balance apparatus group after treatment, and the ABC score was superior to other two group. Moreover, the observation group outperformed the other two group in terms of motion length, and open-eye elliptical area, with statistical significance.
CONCLUSION
The combined application of TMS and Balance Apparatus training effectively improves enhances balance function, and boosts self-confidence in balance perception among Parkinson’s patients.
Keywords
Introduction
Parkinson’s disease (PD) is a clinically common progressive neurodegenerative disorder characterized by pathological changes involving the degeneration of dopaminergic neurons in the substantia nigra pars compacta of the brain. This leads to a reduction in dopaminergic innervation in the striatum and results in motor symptoms such as bradykinesia, muscle rigidity, and resting tremors in patients. These motor symptoms are associated with impaired balance due to disturbances in postural control. Currently, the primary treatment approach for PD is still centered around dopaminergic replacement therapy, with rehabilitation therapy playing a complementary role [1, 2]. However, the efficacy of dopaminergic medications tends to decrease as the disease progresses, and in the later stages, approximately 80% of PD patients develop severe motor impairments, including a decline in balance function [3]. Therefore, in the advanced stages of PD, rehabilitation training gradually becomes an essential therapeutic tool for slowing disease progression and improving patients’ daily living abilities [4].
For every individual, good balance function is essential for performing various complex tasks in different environments. Balance function requires continuous dynamic regulation involving interactions between the brainstem, spinal cord networks, and cognitive and sensory feedback. These three components form a closed-loop system, and any abnormality in any of these nodes can affect the entire balance regulation system [5, 6]. In PD patients, balance impairments are characterized by increased muscle tone in the trunk, difficulties in shifting the center of gravity, difficulties in initiating walking, and a hesitant gait, among other symptoms. Currently, there has been substantial research into the mechanisms underlying balance impairments in PD [7]. One of the well-established reasons is the dysfunction of the dopaminergic system. In Parkinson’s patients, the dysfunction of the dopaminergic system accumulates in the basal ganglia, leading to a decline in the ability to perform rhythmic automatic movements like standing and walking, ultimately affecting balance [7]. However, there is also evidence suggesting that Parkinson’s patients with balance impairments may respond poorly to dopaminergic medications [8]. Some studies have indicated that alterations in gamma-aminobutyric acid (GABA) and glutamatergic neurotransmission may be related to certain axial symptoms in PD [9]. Axial symptoms encompass various aspects, including balance, posture, speech, swallowing, and more, suggesting that non-dopaminergic lesions in Parkinson’s patients can also impact balance function [10]. In recent years, many scholars have explored various aspects such as central sensory-motor dysfunction, distal muscle control impairment, and cognitive function impairment [11]. The central sensory-motor function system consists of three parts: information input, central integration, and motor output. Any problems at one node can lead to abnormalities in the entire system. Research has shown that Parkinson’s patients exhibit damage to the vestibular pathway, characterized primarily by subjective vertical perception abnormalities. When there is a significant difference between the subjective vertical line and the objective vertical line, it can lead to imbalance [12]. There is also research indicating that Parkinson’s patients often have dysfunction in the medulla-midbrain-vestibular connection system, and the degree of damage is proportional to the severity of the disease [13]. This connection system affects the integration of somatosensory information in patients, thereby impacting their dynamic balance control. Some scholars believe that Parkinson’s patients have prolonged increased muscle tone in their limbs and trunk, leading to muscle contractures around various joints and a decrease in joint mobility, which ultimately weakens their postural control abilities [14]. Rehabilitation training is a comprehensive approach that can use various physical methods to target non-dopaminergic lesions and ultimately improve the balance function of patients.
Transcranial magnetic stimulation (TMS) is a non-invasive and painless neuroregulation technique that relies on Faraday’s electromagnetic induction principle. It applies pulsed currents to different areas of the cerebral cortex to alter the action potentials of nerve cells, thereby affecting cell metabolism and neural electrical activity [15]. Currently, TMS is widely used in the diagnosis, assessment, and treatment of various neurological disorders. In recent years, an increasing amount of research has confirmed the positive and effective role of TMS in improving functional impairments in Parkinson’s patients. Traditional balance training has been proven to improve the balance function of Parkinson’s patients [16]. Balance Apparatuss add visual, auditory, and proprioceptive stimuli to traditional training and incorporate therapy into games, making training more enjoyable and advantageous compared to conventional balance training [17]. This study aims to explore the effectiveness of using TMS and Balance Apparatus training together to improve the balance function of Parkinson’s patients. The goal is to further enhance the balance function of PD patients and provide reference for clinical rehabilitation prescriptions.
Materials and methods
Participants
Patients included in this study should meet the following inclusion criteria and exclusion criteria. Inclusion criteria: Participants exhibited core symptoms and signs of PD and met the diagnostic criteria for PD as outlined in the “Chinese Diagnostic Criteria for PD (2016 edition)” established by the Neurology Branch of the Chinese Medical Association [18]; Participants were clinically diagnosed through our department’s outpatient services; Hoehn-Yahr staging 1 to 4 levels; Participants aged under 75 years old and abled to cooperate with assessments and treatment; Participants should have no cognitive impairment or communication barriers, as evidenced by a Mini-Mental State Examination (MMSE) score of > 17 points. Exclusion Criteria: Patients with secondary Parkinson’s syndrome caused by central nervous system diseases such as cerebrovascular disease, traumatic brain injury, or intracranial lesions were excluded; Patients with concurrent musculoskeletal disorders and neurological diseases affecting standing balance were excluded; Patients with severe cardiovascular or pulmonary diseases who cannot cooperate with assessment and treatment were excluded. Additionally, individuals with severe mental disorders or currently undergoing treatment with psychotropic drugs were also excluded; A history of epilepsy or a family history of epilepsy; A history of epilepsy or a family history of epilepsy; Individuals with cardiac pacemakers, cochlear implants, or a history of cranial bone repair surgery were excluded from the study.
Research design
A total of 60 eligible patients who were admitted to our department between October 2019 and March 2023 and meet the specified criteria would be included in the study.
These patients would be sequentially numbered sequentially according to their order of admission or hospitalization, ranging from 1 to 60, prior to enrollment. This numbering system would help ensure the organization and tracking of participants. Subsequently, randomization using Minitab software would be performed by a blinded staff member, whereby participants are evenly divided into three groups: observational group, consisting of 1 to 20 participants; the magnetic stimulation group, consisting of 21 to 40 participants; Balance the equipment group, consisting of 41 to 60 participants. This randomization process aimed to ensure the equitable distribution of participants across the different treatment groups. Among these groups, one participant in the magnetic stimulation group dropped out due to reasons related to the pandemic, resulting in a total of 19 participants completing the treatment. Statistical analysis of the enrolled participants’ gender, age, disease duration, Hoehn-Yahr classification, and UPDRS-III scores (please refer to Table 1 for details) showed no statistically significant differences (P > 0.05), indicating comparability among the groups. All patients provided informed consent for participation in the clinical study, and the research received approval from our hospital’s Medical Ethics Committee (Approval Number: [2019012]). All patients followed medical advice and regularly took dopamine agonists and other anti-Parkinson’s medications. They also attended regular neurology outpatient clinics for follow-up assessments and medication guidance [19]. All patients underwent routined rehabilitation training. The magnetic stimulation group received low-frequency repetitive rTMS therapy in addition to the routine training. The balance training group incorporated the use of a Balance Apparatus into their training alongside the routine training. The combined group received both low-frequency rTMS and Balance Apparatus training in conjunction with their routine rehabilitation. Each patient received the specified treatment once daily, six times a week, for a total of 8 weeks. Refer to Fig. 1 for details.
General information of 3 groups of patients
General information of 3 groups of patients
rTMS: Repetitive transcranial magnetic stimulation.
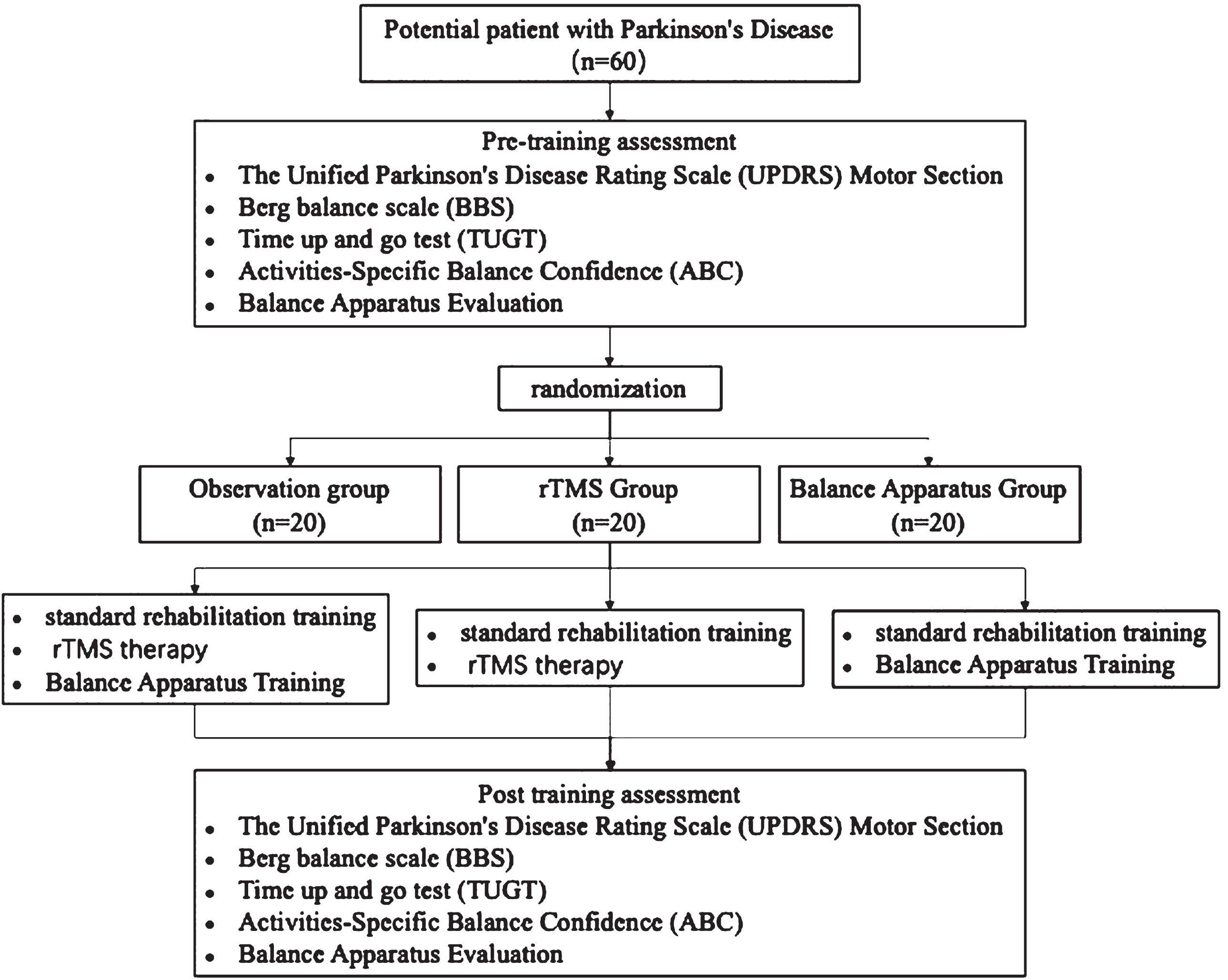
The flow chart of the study.
The routine rehabilitation training program was not specifically designed for the patients’ balance function but was based on assessment results. It included specific aspects such as trunk and limb strength training, active and passive joint mobility exercises, relaxation training, breathing exercises, functional training like turning, sitting up, standing, turning around, and walking, as well as hand exercises like gripping and pinching. The duration of this program was approximately 30 to 40 minutes.
Low-frequency repetitive transcranial magnetic stimulation
Low-frequency rTMS was administered using the Magneuro60-type transcranial magnetic stimulator from Nanjing on the supplementary motor area (SMA) [20]. The stimulation coil used a standard 9 cm circular coil with a maximum intensity of 1.5 Tesla. Stimulation intensity was set at 110% of the motor threshold, and the stimulation frequency was 0.5 Hz. Each treatment session consisted of a total of 1800 continuous pulses, lasting approximately 30 minutes [15].
Balance apparatus training
The training was conducted using the Pro-Kin Balance Testing and Training System (Teenobody S.r.l. PK252), an Italian product. All patients underwent static balance testing before treatment, and based on the test results, different modules and games were selected for training. These modules and games included the following categories: Static balance function training: Under the guidance of the therapist, patients stood on a locked balance board and trained by shifting their weight in different directions and maintaining stability. The training included completing various games integrated into the device that challenged their balance in different ways.;Dynamic balance function training: With the assistance of the therapist, patients stood on a movable balance board and, while maintaining dynamic balance, controlled markers on a display screen to move them according to the requirements of various games integrated into the Balance Apparatus. The balance training adhered to the following principles: progressing from supported training to unsupported training, transitioning from open-eye training to closed-eye training, and moving from static balance training to dynamic balance training. Each treatment session lasted approximately 30 minutes.
Evaluation method
The three groups of patients were assessed at baseline and after 8 weeks of treatment using the following evaluations. All assessments were conducted in the same environment by the same therapist who was unaware of the patient group assignments. Additionally, any adverse reactions such as seizures, dizziness, pain, nausea, fatigue, etc., that occurred during treatment were observed and recorded. The specific assessment methods are as follows.
The Unified Parkinson’s Disease Rating Scale (UPDRS) motor section
The UPDRS scale consists of three parts: neurological and behavioral status, activities of daily living, and motor function examination. In this study, only the third part was used to assess the extent of motor dysfunction in PD patients. This section primarily evaluates speech, facial expression, resting tremor, action or postural tremor of the hands, rigidity, finger tapping, hand movements, pronation-supination movements, leg agility, arising from a chair, posture, gait, postural stability, and body bradykinesia. It is scored on a scale of 0 to 56 points, with higher scores indicating more severe motor dysfunction [21].
Berg balance scale (BBS)
The Berg Balance Scale (BBS) comprises 14 tasks, each scored on a scale of 0 to 4, with a total score of 56. It is used to assess abilities related to sitting, standing, leaning, changing positions, and single-leg standing, among others. A lower score on this scale indicates poorer balance function [22].
Time up and go test (TUGT)
Patients sit on a chair with armrests (chair height approximately 46 cm, armrest height approximately 21 cm). From the chair, patients stand up, walk to a colored, thick line positioned 3 meters away, cross the line, quickly turn around, walk back to the chair, and then turn around once more to sit down. The total time taken to complete this process is recorded, with a longer time indicating poorer balance function.
Activities-Specific Balance Confidence (ABC)
The ABC (Activities-Specific Balance Confidence) scale, introduced by Powell and Myers in 1995, is a questionnaire designed to assess an individual’s self-confidence in maintaining balance during various activities. These activities include routine tasks like walking, going up and down stairs, picking up objects, as well as social activities such as taking a taxi, boarding public transportation, or shopping. The scale consists of 16 different tasks, and patients rate their level of confidence in completing each task without losing their balance, with scores ranging from 0% to 100%. The scores for each task are then averaged to calculate an overall confidence level. Higher scores indicate a greater perceived level of confidence in one’s balance.
Balance apparatus evaluation
The balance testing and training were conducted using the Pro-Kin Balance Testing and Training System (Teenobody S.r.l. PK252), an Italian-made system. Patients stood on a locked balance board under the guidance of a therapist. To ensure the comparability of the tests, all patients assumed a standard standing posture based on the balance board coordinates (with the inner ankle of each foot passing through the red line on the A3A7 axis, feet close together, both heels aligned on the same axis, the left foot’s second toe pointing to A8, and the right foot’s second toe pointing to A2). After the therapist entered the general information, they selected the static stability assessment module for testing. Patients were instructed to stand with their chest up and head held high. When prompted by the therapist’s click, the patient attempted to maintain this position for 30 seconds before completing the test. All patients were required to perform this test both with their eyes open and with their eyes closed. The main observed indicators were as follows: Movement Length (mm): This indicator represents the total length of the trajectory of the body’s center of gravity movement during the test, indicating the speed and extent of center of gravity movement. A value greater than 250 mm suggests abnormalities; Movement Ellipse Area (mm2): This indicator refers to the total area enclosed by the trajectory of the body’s center of gravity movement during the test, indicating the size of the center of gravity’s amplitude. An observation value greater than 200mm2 suggests abnormalities [23]. Average Trace Error (ATE): This indicator refers to the average error rate between the monitored trajectory and the ideal trajectory when the limbs make circular continuous movements in various directions on the electronic tilt board. The formula for calculating ATE is ATE=(length of the trajectory traced by the patient’s ankle-foot control cursor - length of the ideal trajectory) / length of the ideal trajectory. A lower score indicates better balance function [24]. All patients underwent each test twice consecutively, and the observation indicators were the better.
Sample size
The sample size was established by considering the Unified Parkinson’s Disease Rating Scale (UPDRS) Motor Section as a primary outcome and was based on thefollowing assumptions: significance level (α) =0.05,(2) type 2 error (β) =0.2, and 80% test power. For sample calculation, G*Power 3.1.9 was used based on an effect size of 0.8 (a large effect size according to Cohen). The calculated sample size was 19 patients per group. Considering a 5% dropout rate, aminimum total sample of 60 patients (20 per group) was required.
Statistical methods
All data were statistically analyzed using SPSS 26.0 software. Measurement data are expressed as mean (standard deviation) and were assessed for normality using the Kolmogorov-Smirnov (K-S) test and for homogeneity of variances using Levene’s test. One-way analysis of variance (ANOVA) was used for comparisons of measurement data, followed by Bonferroni post hoc tests for pairwise comparisons. Count data were compared using the chi-squared (χ2) test, and differences were considered statistically significant at P < 0.05 (two-tailed).
Results
Comparison of UPDRS scores, BBS scores (points), TUGT (seconds), and ABC scores before and after treatment among the three groups
Before treatment, there were no statistically significant differences in UPDRS scores, BBS scores (points), TUGT (seconds), and ABC scores among the three groups (P > 0.05). After treatment, there were significant improvements in UPDRS scores, BBS scores, TUGT, and ABC scores in all three groups compared to before treatment, and these differences were statistically significant (P < 0.05). In the intergroup comparison after treatment, the observation group had significantly lower UPDRS scores than the Balance Apparatus group, and significantly higher ABC scores compared to the other two groups, with statistically significant differences (P < 0.05). The rTMS group had higher ABC scores than the Balance Apparatus group, and this difference was statistically significant (P < 0.05). Refer to Table 2 for details.
Comparison of UPDRS score, BBS score, TUGT and ABC score among three groups of patients
Comparison of UPDRS score, BBS score, TUGT and ABC score among three groups of patients
rTMS: Repetitive transcranial magnetic stimulation; UPDRS: The Unified Parkinson’s Disease Rating Scale; BBS: Berg balance scale; TUGT: Time up and go test; ABC: Activities-Specific Balance Confidence; *P < 0.05.
Before treatment, there were no statistically significant differences in the movement length, movement elliptical area, and ATE (average trace error) results between the three groups with eyes open and closed (P > 0.05). After treatment, there were significant reductions in the movement length, movement elliptical area, and ATE results with eyes open and closed in all three groups compared to before treatment, and these differences were statistically significant (P < 0.05). In the comparison between groups, the observation group had significantly lower movement length with eyes open and closed compared to the rTMS group and Balance Apparatus group. Additionally, their closed-eye movement elliptical area was significantly lower than that of the rTMS group (P < 0.05). The Balance Apparatus group had significantly lower movement length with eyes open, as well as movement elliptical area with eyes open and closed compared to the rTMS group (P < 0.05). Refer to Table 3 for details.
Comparison of motor length, elliptical area, and ATE results among three groups of patients
Comparison of motor length, elliptical area, and ATE results among three groups of patients
rTMS: Repetitive transcranial magnetic stimulation; ATE: Average Trace Error; *P < 0.05.
During the treatment and assessment processes, none of the patients in the three groups experienced adverse effects such as seizures, dizziness, pain, nausea, fatigue, or other discomforts.
Discussion
TMS was initially used primarily for clinical research, assisting in diagnosis, and functional localization. However, as research deepened and related technologies improved, its application has expanded to include interventions, modulation, and treatments for neurological functions. The efficacy of TMS in treating PD has been Supported by relevant studies. Its mechanism may involve regulating monoaminergic neurotransmitter levels to improve neurotransmitter imbalance [25]. TMS can also activate the midbrain striatal dopaminergic pathway to improve motor dysfunction [26]. The 2020 European rTMS treatment guidelines updated recommend high-frequency rTMS over the bilateral M1 area for treating motor symptoms in PD (Grade B recommendation) [27]. However, recent meta-analyses have suggested that low-frequency rTMS over the SMA area can also significantly improve motor symptoms in Parkinson’s patients [28]. Because this study utilizes a combination treatment and takes into account the lower incidence of adverse effects and higher safety profile associated with low-frequency rTMS, low-frequency rTMS was chosen.
Balance Apparatus has many advantages over traditional balance training, including: Balance Apparatus provides real-time feedback to patients through sensors and computer analysis, enhancing the stimulation of the central sensory-motor system to improve balance control abilities [29]. Balance Apparatus training focuses on improving patients’ dynamic balance and proprioception [30]. Balance Apparatus incorporates a wide range of games and interactive training, making it more engaging for patients. It is particularly beneficial for Parkinson’s patients who often experience impairments in distal muscle control and cognitive functions. As a comprehensive training system, Balance Apparatus enhances lower limb proprioception through visual feedback, continuously stimulating the body’s postural control system, ultimately improving body balance and stability [31].
The improvement of balance function in Parkinson’s patients requires comprehensive training that addresses both central sensory-motor neural functions and distal muscle control abilities along with proprioception. rTMS and Balance Apparatus each provide interventions from different perspectives, achieving a synergistic effect in combined therapy. In the results, although all three groups of patients showed significant improvements in UPDRS scores, BBS scores, TUGT performance, ABC scores, and Balance Apparatus test results after treatment, the observation group demonstrated certain advantages in improving the motor function of Parkinson’s patients, enhancing their confidence in balance perception, and maintaining static balance stability. Furthermore, the study results indicate that the Balance Apparatus group showed more significant improvements in motion length and motion ellipse area compared to the rTMS group. However, there were no statistically significant differences in UPDRS scores, BBS scores, and TUGT performance between the two groups. This could be because the motion length and motion ellipse area tests are primarily designed for assessing the effects of Balance Apparatus training and may not necessarily imply that Balance Apparatus training is superior to rTMS therapy in all aspects.
There are still many shortcomings in this study: Due to the limited number of cases, patients were not grouped according to the course and severity of the disease, which resulted in the research results being less specific; A high-frequency rTMS treatment was not included as a control group for comparison; The relatively short duration of the study may have resulted in less significant differences between the groups, which could have reduced the overall persuasiveness of the findings; The sample size in the study was still relatively small, and there was no long-term follow-up of the patients, which are limitations that could be addressed in future related research to further refine the findings.
Conclusion
The results of this study Supported that after 8 weeks of combined treatment with TMS and Balance Apparatus on top of conventional therapy, the observation group showed significant improvements in ABC scores, open-eye and closed-eye elliptical lengths, and open-eye elliptical area compared to their own pre-treatment scores and the post-treatment scores of the rTMS group and the Balance Apparatus group, with statistical significance (P < 0.05). The results suggest that the combined use of TMS and Balance Apparatus training can effectively improve motor symptoms in Parkinson’s patients, enhance their balance function, and boost their self-confidence in balance perception. This approach appears to have advantages over solely using TMS therapy or Balance Apparatus training.
Conflict of interest
The author declare no conflict of interest.
Footnotes
Acknowledgments
The authors have no acknowledgments.
