Abstract
Background:
Repetitive transcranial magnetic stimulation (rTMS) showed potentially beneficial effects for the treatment of post-traumatic stress disorder (PTSD). Low-frequency (LF) rTMS decreases neuronal excitability and may have better safety compared to high-frequency (HF) rTMS. However, there lacks meta-analysis specifically focusing on LF rTMS.
Objectives:
To specifically explore the efficacy and safety of LF rTMS for treating PTSD.
Methods:
Databases including PubMed, EMBASE, MEDLINE, and Web of Science were systematically searched from inception to October 17, 2023. Both randomized controlled trials (RCTs) and open trials of LF rTMS on PTSD were included, and we additionally included RCTs comparing HF rTMS and sham treatment on PTSD. First, we qualitatively summarized parameters of LF rTMS treatment; then, we extracted data from the LF rTMS treatment subgroups of these studies to examine its effect size and potential influencing factors; third, we compared the effect sizes among LF rTMS, HF rTMS and sham treatment through network meta-analysis of RCTs.
Results:
In all, 15 studies with a sample size of 542 participants were included. The overall effect size for LF rTMS as a treatment for PTSD was found as Hedges’ g = 1.02 (95% CI (0.56, 1.47)). Meta-regression analysis did not reveal any influencing factors. Network meta-analysis showed that compared to sham treatment, only HF rTMS on the right dorsolateral prefrontal cortex (DLPFC) demonstrated a significant advantage in ameliorating PTSD symptoms, while LF rTMS on the right DLPFC showed a trend toward advantage, but the difference was not significant.
Conclusion:
The current literature shows LF rTMS has effect in treating PTSD caused by various traumatic events. However, present limited number of RCT studies only showed LF rTMS to have a trend of advantage compared to sham treatment in treating PTSD caused by external traumatic events. In the future, more RCTs are needed to be made to confirm the efficacy of LF rTMS. Additionally, studies are required to elucidate the underlying mechanism in order to further improve its efficacy in different traumatic populations.
PROSPERO registration number:
CRD42023470169.
Introduction
Post-traumatic stress disorder (PTSD) is a common psychiatric disorder that develops after a traumatic event, which may be external factors (e.g., natural disasters, violence, and combat) or internal factors (e.g., critical illnesses like stroke attack, myocardial infarction, and cancer). 1 The lifetime incidence of exposure to traumatic events that may cause PTSD is estimated to be over 50% in the general population, and the incidence of PTSD is estimated to be ranging from 3 to 7%. 2 For certain populations such as veterans, the prevalence may increase up to 15%. 3
Psychotherapies 4 and pharmacological therapies 5 for PTSD vary, although efficacious but not always effective. As an alternative or supplementary method, repetitive transcranial magnetic stimulation (rTMS) shows potentially beneficial effects. 6 rTMS has the advantages of noninvasiveness and good safety. Its side effect is at a rate of 5% or more, and the symptoms are generally mild and diminish rapidly 7 . High-frequency (HF) rTMS can increase the excitement of neurons, while low-frequency (LF) rTMS decreases the excitement. The right or left dorsolateral prefrontal cortex (DLPFC) is the common stimulation target. 8 Compared to HF rTMS, LF rTMS may have better safety, since it has an antiepileptic effect while the prior may induce seizures. 9 Additionally, LF rTMS of the right DLPFC ranked first in acceptability among all treatment protocols. 8 Considering PTSD may often be combined with brain lesions such as traumatic brain injury, 10 stroke11,12 and brain tumor, 13 which have a high risk of causing epilepsy, LF rTMS may have its unique role in treating these specific types of PTSD. Meta-analysis studies published in recent years only included literature published up to 2020,6,8,14,15 and most studies did not focus on a specific stimulation target or protocol. Hence, the objective of the present work was to make an updated review focusing on the efficacy and safety of LF rTMS in treating PTSD and its comparisons with HF rTMS and sham treatment.
Methods
The protocol for this review was registered with PROSPERO (international database of prospectively registered systematic reviews): CRD42023470169. We adhered to PRISMA guidelines (Supplemental Materials). 16 Two independent authors systematically searched databases, including PubMed, EMBASE, MEDLINE and Web of Science to locate published studies up to October 17, 2023, using the keywords (“Repetitive Transcranial Magnetic Stimulation” OR “rTMS” OR “Repetitive TMS”) AND (“Posttraumatic stress disorder” OR “PTSD” OR “Post-traumatic stress disorder”). Additionally, we manually screened published reviews and meta-analyses for relevant studies. The full search strategy is detailed in the Supplemental Materials.
Two researchers independently assessed studies for eligibility. Inclusion criteria were as follows: (1) primary investigation using LF rTMS as a treatment for patients diagnosed with PTSD according to standard operationalized diagnostic criteria, or primary randomized controlled trial (RCT) studies comparing HF rTMS and sham treatment; (2) published in English language. Exclusion criteria were as follows: (1) studies published as conference abstracts without full text or book chapters and dissertations; (2) rTMS that was not conducted in a clinical setting under supervision from a clinician; (3) studies that did not use questionnaires or clinician assessments based on symptom severity but used physiological markers.
Selected studies were further reviewed by two authors to extract the relevant information and assess the quality of each article independently. Disagreements were resolved with a third author. First, all articles included were qualitatively reviewed. Data extracted included demographic data, treatment parameters (i.e., stimulation target, %MT, frequency, coil type, total number of pulses, and number of treatment sessions), outcome measures, assessment time, and complications. Second, we extracted data from the active treatment groups in all included studies that received LF rTMS targeting on DLPFC, in order to examine the effectiveness of LF rTMS. Third, we compared the effect sizes among LF rTMS, HF rTMS and sham treatment by extracting data from RCT studies. For studies that had multiple outcome measures, we combined those outcomes to obtain a single treatment effect size. When only figures were presented, data was extracted using Webplotdigitizer 4.0 (https://apps.automeris.io/wpd/).
As this was a structured review and meta-analysis of studies that had previously received ethics committee approval, no additional ethics approval was required.
The Cochrane Risk of Bias Assessment Tool (ROB 2.0) 17 was used to evaluate the risk of bias by one of the authors. This tool explored bias sources across five dimensions: randomization process, deviations from intended interventions, missing outcome data, measurement of the outcome, and selection of the reported result. Then an overall result was generated and the risk was categorized as high risk, some concerns, or low risk.
Data was analyzed with Stata/SE 17.0 statistical software (StataCorp, College Station, TX, USA). A random-effects model was used to analyze the effect sizes (Hedges’g for correcting for possible bias of small sample sizes 18 ) given the clinical and methodological diversity among included trials. Heterogeneity among the studies was assessed using the I2 statistic. The potential reasons for heterogeneity (total pulse and intensity) were explored by network meta-regression using R gemtc package. In addition, we performed subgroup analysis of significant sources of heterogeneity. Publication bias was assessed by Egger’s test and its visual inspection of symmetry was also presented by a funnel plot. Meta-regression analyses were used to probe the association between potential influencing factors and effect sizes. Network meta-analysis was used to compare the differences in effect sizes among LF rTMS, HF rTMS and sham treatment. R software netmeta package (version 3.6.3) was used to perform a network meta-analysis combining direct and indirect comparisons in the Frequentist model.19,20 SMD and 95% CI were calculated for all pairwise comparisons, and relevant results were presented in a forest plot. An α level of 0.05 was used to determine statistical significance.
Results
The results of the literature search are shown in Figure 1. Finally, 15 studies with a sample size of 542 participants were included and reviewed. Among these studies, 13 were studies on LF rTMS (Table 1), and the rest two were RCTs comparing HF rTMS and sham treatment. All participants were adults receiving rTMS for PTSD. Three subgroups included treatment protocols of LF rTMS plus exposure procedure. Among the 13 studies on LF rTMS, the targets of LF rTMS stimulation were right DLPFC (n = 11), left DLPFC (n = 1), or bilateral motor cortex (n = 1). Coil types were eight-shaped (n = 11), angular-shaped (n = 1) or circular shaped (n = 1). The pulse intensity ranged from 70% to 120%, and the number of pulses ranged from 1000 to 86,400 (except one study 21 utilized only 30 pulses). Trauma types included both external factors (accidents/assault/combat) and internal factors (stroke attack), and the population included both civilian and military personnel. Outcome measures in these studies included CAPS (n = 7), CAPS-5 (n = 1), IES (n = 3), IES-R (n = 1), PCL (n = 4), PCL-5 (n = 2), Modified PTSD Symptom Scale (n = 1), and treatment outcome PTSD Scale (n = 1).

Flow diagram of literature search.
Summary of studies on LF rTMS.
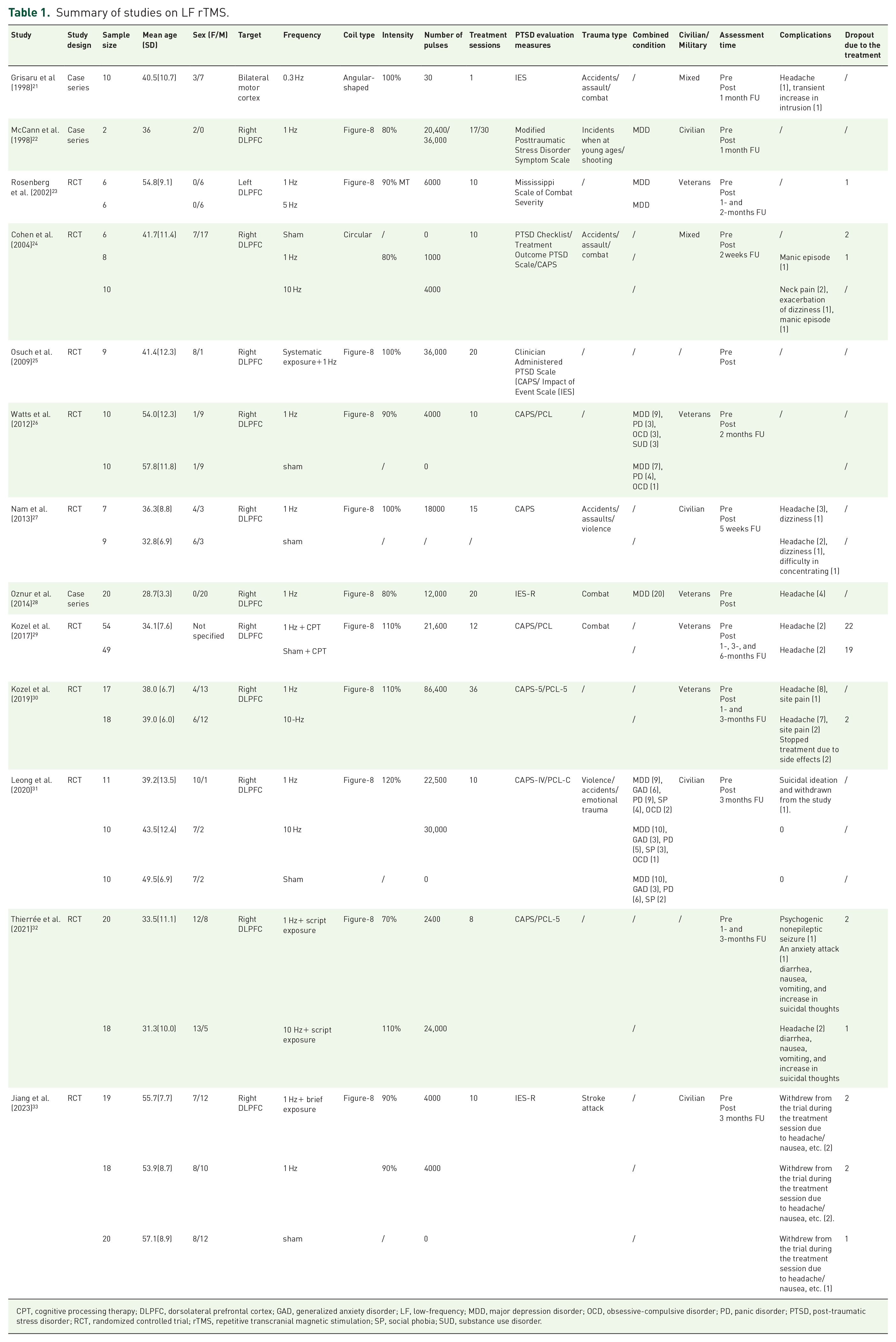
CPT, cognitive processing therapy; DLPFC, dorsolateral prefrontal cortex; GAD, generalized anxiety disorder; LF, low-frequency; MDD, major depression disorder; OCD, obsessive-compulsive disorder; PD, panic disorder; PTSD, post-traumatic stress disorder; RCT, randomized controlled trial; rTMS, repetitive transcranial magnetic stimulation; SP, social phobia; SUD, substance use disorder.
A total of 11 studies of LF rTMS (nine RCTs and two open trials) included 11 subgroups of participants who received active treatment, reflecting a sample size of 176 participants were included in the meta-analyses for the effect sizes of LF rTMS (one study 23 showing only combined effects of LF rTMS and HF rTMS and one study targeting motor cortex were excluded). The effect sizes with associated 95% confidence intervals for the included studies were explored (Figure 2(A)). In the 10 studies of PTSD caused by external traumas, LF rTMS showed a significant reduction of scores of PTSD outcome measures with large effect size (Hedges’ g = 1.02, 95% CI (0.56, 1.47), p < 0.001). Overall treatment effects of the majority of studies (n = 8) were evenly distributed between g = 0.08 and g = 1.96. Cochran’s Q statistics (Q = 34.94, p < 0.001, I2 = 74.2%) indicated heterogeneity suggesting unexplained variance due to differences among studies. The funnel plot showed no publication bias (Figure 2(B)), which was confirmed by a non-significant Egger’s test (p = 0.11). The study by Jiang et al. 33 including individuals of PTSD caused by stroke showed a slightly larger effect (Hedges’ g = 1.94, 95% CI (1.13, 2.74)).

(A) Standardized effect sizes of LF rTMS treatment on PTSD outcome measure scores with a random-effects model and (B) Funnel plot (with pseudo 95% confidence limits) of included studies.
By meta-regression analyses, potential influencing factors on effect sizes were investigated (coil type, total treatment pulses, number of treatment sessions, motor threshold, combined treatment with exposure, research design, and publication year), but none showed significant differences (Table 2).
Meta-regression results of potential influencing factors on effect sizes of LF rTMS.

LF, low-frequency; RCT, randomized controlled trial; rTMS, Repetitive transcranial magnetic stimulation.
The LF rTMS was relatively safe and no serious treatment-related side effects were reported. Side effects of rTMS were generally mild, including headache, site pain, and dizziness. However, dropouts occasionally happen for uncomfortableness besides other treatment-unrelated reasons (Table 1).
Nine RCTs with sufficient data, including a sample size of 383 participants, were included in the network meta-analyses for comparing effect sizes among LF rTMS, HF rTMS (right DLPFC or left DLPFC) and sham treatment. Three studies had three treatment arms and six studies had two treatment arms. Network relationship diagram for effect sizes of different treatments is shown in Figure 3(A). Results indicated that compared to sham treatment, only HF rTMS to the right DLPFC (d = 0.70, 95% CI (0.14, 1.27)) demonstrated a significant advantage in improving PTSD symptoms, while there was no significant difference in comparisons between other treatment modalities (LF rTMS to the right DLPFC showed a trend toward benefit compared to sham). Forest plot was presented in Figure 3(B). The results of our network meta-regression show that neither total pulse (shared beta: 0.49, 95% CI (−0.53, 1.57)) nor intensity (shared beta: 0.49, 95% CI (−0.53, 1.57)) significantly affects our research results. The results of our network meta-regression indicate that total pulse did not significantly affect our findings (shared beta: 0.49, 95% CI (−0.53, 1.57)). It is worth noting that intensity significantly affected our findings (shared beta: 0.71, 95% CI (0.02, 1.41); estimates at the centering value: intensity = 100) and served as a major source of heterogeneity. Only when intensity ⩽ 100 (Figure 3(C)), HF rTMS to the right DLPFC demonstrated a significant advantage in improving PTSD symptoms compared to sham treatment, but not when intensity >100 (Figure 3(D)).
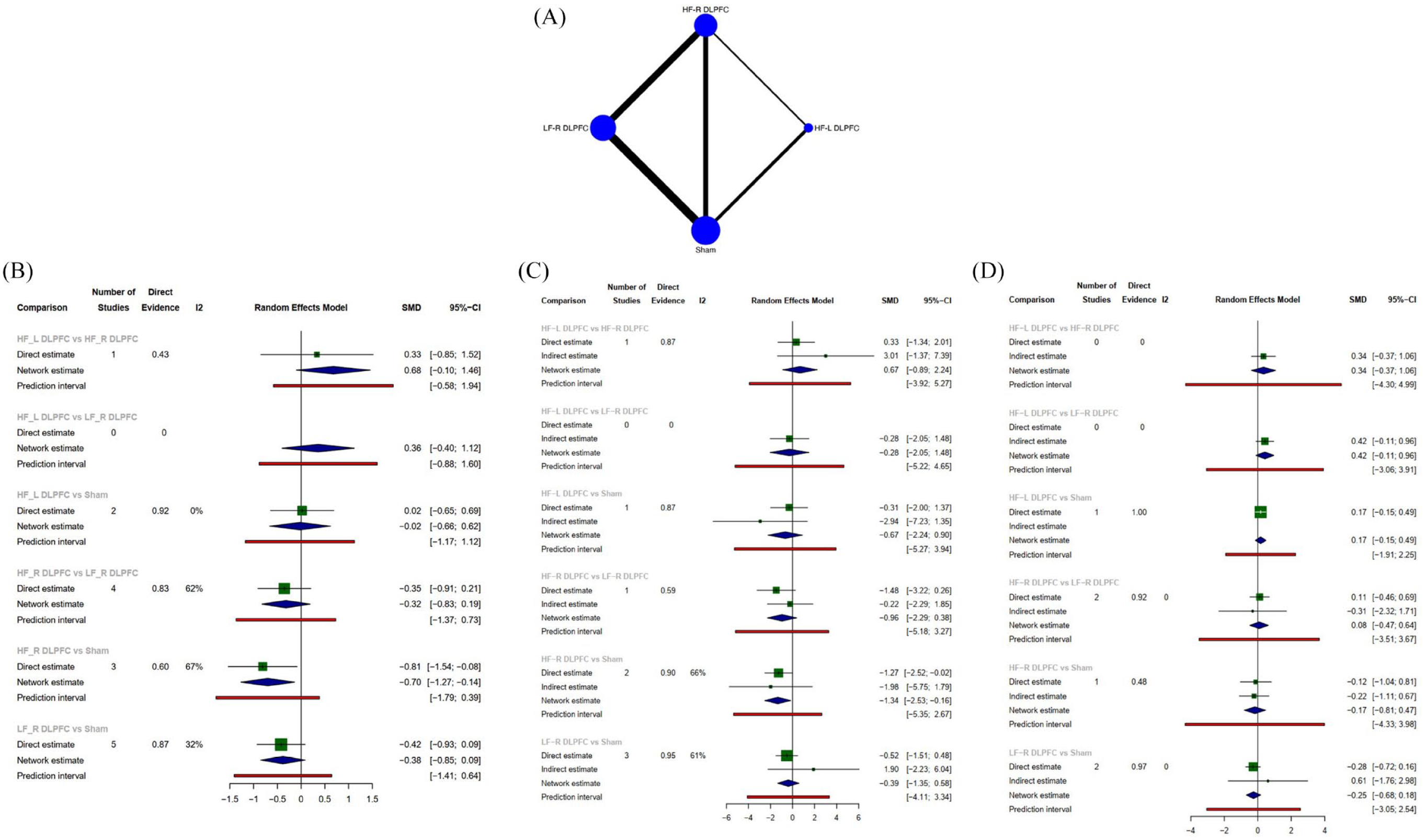
(A) Network plot, (B) forest plot of studies included in the network analyses, (C) forest plot of subgroup analysis (intensity ⩽ 100), and (D) forest plot of subgroup analysis (intensity > 100).
For the 11 RCTs, quality assessment revealed overall low risk for five studies, some concerns for five studies, and high risk for one study (Figure 4).

Risk of bias of included RCTs.
Discussion
Although numerous studies have been made, the underlying mechanism of PTSD largely remains unknown. As traumatic events vary greatly, it has been argued by some scholars that PTSD only represents a common cluster of symptoms, rather than a disease with specific pathogenesis. 34 Typical traumatic events are external factors, like combat, accidents, sex abuse, etc. However, internal factors, that is, some acute serious illnesses, can also become traumatic events according to the criteria of DSM. 1 Among these illnesses, acute coronary syndrome (ACS), 35 stroke, 36 and cancer 37 are most commonly investigated. However, previous studies of TMS generally did not include PTSD individuals caused by illnesses and explicitly excluded those with brain lesions. 32 Only one study explored the effect of TMS in treating PTSD following illnesses, revealing favorable results of LF rTMS for the treatment of post-stroke PTSD. 33 Although brain lesions of the stroke participants in this study were minimal (with average National Institute of Health Stroke Scale scores of around 2, indicating mild symptoms caused by stroke), they may affect either the target of stimulation directly, or else may affect areas of the brain that are indirectly connected to the target. So, this study was analyzed separately as a subgroup in the present meta-analysis.
It is important to accumulate our experience in treating PTSD combined with brain lesions because PTSD is often comorbid with traumatic brain injury (TBI), 38 strokes, 39 and brain tumors. 13 For populations with both PTSD and concussion (a type of mild TBI, but without obvious brain lesions), safety of TMS has been proved by some studies, using HF rTMS targeting the left DLPFC,40,41 or LF rTMS targeting the right DLPFC, 42 or intermittent theta burst stimulation (iTBS) targeting the right DLPFC. 43 However, experience of its safety in treating PTSD populations comorbid with more severe brain lesions is lacking. On the other hand, the usage of TMS in treating neurological diseases (e.g., ischemic stroke, 44 Parkinson’s disease and other movement disorders, Alzheimer’s disease, multiple sclerosis, epilepsy, and disorders of consciousness 45 ) has been widely explored. As LF rTMS has an antiepileptic effect while HF rTMS may induce seizures, 9 it may theoretically be safer to use LF rTMS for the treatment of PTSD secondary to brain lesions (e.g., hemorrhagic stroke and brain tumor) which often causes epilepsy. Additionally, LF rTMS was found to be more acceptable than other treatment protocols for patients. 8 Compared to previous literature that included research on various types of TMS, only including research on LF rTMS can have better homogeneity for meta-analysis. For these reasons, in the first part of our work, we specifically focused on the LF rTMS. In the second part, we further included two RCTs that compared HF rTMS and sham treatment, to make direct and indirect comparisons among different treatments using network meta-analysis. Thus, this study gave a relatively comprehensive view of LF rTMS for PTSD treatment.
Our results showed an overall effect size of g = 1.02, 95% CI [0.56, 1.47], indicating an overall positive treatment effect associated with LF rTMS and PTSD outcome measure scores. However, by network meta-analysis, the effect size of LF rTMS only had a trend toward benefit compared to sham treatment (d = 0.38, 95% CI (0.09, 0.85)), but the difference was not significant. This result was in line with previous meta-analysis, 6 and offered preliminary evidence for therapeutic effect of LF rTMS. As presently the number of studies included in the analyses is small, more RCT studies comparing LF rTMS and sham are needed to be made to further confirm the efficacy of LF rTMS. Harris et al. 6 pooled previous reports of RCTs and other designs, comparing effect sizes of TMS at pre and post-treatment, and revealed that HF rTMS led to a significantly stronger treatment effect than LF rTMS. However, other studies that included only RCTs by both meta-14,15 and network meta-analysis 8 did not show significant difference between low and high frequency, although high frequency always had a slightly superior effect. This result is not unexpected, because HF rTMS generally has higher stimulation doses than LF rTMS (Table 1). Our analysis also showed no difference between LF and HF rTMS, and a trend of benefit of HF compared to LF when targeting the right DLPFC (Figure 3(b)). Further studies are required to test if the effect of LF rTMS can be enhanced by increasing total stimulation doses. Although TMS exerts its effect by regulating neuronal networks, the exact mechanism is unknown. It has been proposed by some scholars that low and high-stimulation protocols targeting different brain regions may address different clusters of PTSD symptoms. 6 LF rTMS may be especially beneficial for intrusion 27 and hypervigilance. 25 However, current studies often did not report subscale scores of PTSD measures. The respective emphasis on treatment effect of low and high frequency deserves further study. Moreover, as HF rTMS often targets the left DLPFC while LF rTMS targets the right DLPFC, whether effects can be enhanced by combination therapy needs to be explored. Until now, few studies utilized bilateral targets. 21 Compared to first-line therapy like cognitive behavior therapies, TMS offers an easier way of conducting for therapists. After simple trainings, it can be applied by nurses or technicians under the supervision of psychologists. This procedure is also more standardized and has better repeatability than psychotherapies. Either used alone or combined with other therapies, TMS can be a viable treatment choice. Personalized interventions targeting specific PTSD symptoms also require further research.
DLPFC has a central role in emotion regulation. 46 Specifically, the right hemisphere’s dominant role in stress modulation has been linked to PTSD, as studies indicated more structural abnormalities existed in the right hemisphere. 47 According to our review, the majority of studies (11/13) of LF rTMS targeted the right DLPFC. It is interesting that HF rTMS of the right DLPFC also led to symptom reductions, being more beneficial than the left DLPFC,8,14 indicating there may be other underlying mechanisms (i.e., nonspecific mechanisms) behind initial stimulation or inhibition of cortical activity.
The role of intrinsic connectivity networks in PTSD has been widely studied. The default mode network (DMN), which possesses core nodes within the cingulate cortex, medial prefrontal cortex, cuneus/precuneus, and temporoparietal junction/angular gyrus, is a crucial brain network involved in contextual processing. 48 It functions as integration of information from various brain regions and creating a coherent sense of self. 49 By electroencephalographic measure, the alpha oscillatory rhythm (8–13 Hz) represents the DMN activity. In PTSD, dysregulated alpha oscillation in the DMN50,51 and disrupted connectivity among multiple DMN structures (especially the medial prefrontal cortex (PFC) 52 ) have been reported. 53 This may be the reason why individuals with PTSD react improperly to environmental stimuli. Aside from DMN, the salience network (SN) also plays a role in PTSD. The SN consists of the insula, dorsal anterior cingulate cortex, and the amygdala, is involved in environmental monitoring and avoidance behaviors, and decodes innate alarm system signals in the context of threatening stimuli.54,55 Alteration of SN connectivity was found in individuals with PTSD,53,56,57 and alpha rhythm reduction in the visual-cortex-DMN system is hypothesized to be a global index of hyperarousal associated with SN connectivity.58,59 A recent study showed that by increasing cortical alpha center frequency and synchronizing alpha oscillation through personalized rTMS (which means adjusting the stimulation strength, frequency and site based on encephalogram guidance), PTSD symptoms can be improved. 49 In this study, the stimulus frequency range was 8–13 Hz, similar to HF rTMS. So, this theory of alpha oscillation may also be an explanation for the superiority of HF rTMS protocol in treating PTSD, as it may most effectively rectify the deviated frequency to the intrinsic alpha center frequency or increase cortical oscillatory synchrony.
PTSD individuals show impairment of executive function, which may be mostly related to the symptom cluster of negative alterations in cognition and mood. 60 DLPFC combined with parietal cortex along the intraparietal sulcus is proposed to support integration of distributed brain networks, and further supporting high-order cognitive ability including executive function. 61 Neurons that express the calcium-binding protein parvalbumin (PV) normally drive experience-dependent circuit refinement and information processing in sensory, motor and memory functions. 62 The reduction of the number of PV-positive neurons (which leads to cortical inhibitory deficit 63 ) in the PFC is the feature of various psychiatric conditions like schizophrenia and bipolar disorder. 64 GABAergic PV-positive cells have strongest ability to coordinate the actions of neural networks by rhythmic inhibition of gamma oscillation (between 30 and 90 Hz).62,65 As shown in studies on autism, both HF66,67 and LF 68 rTMS to the DLPFC could improve executive function, possibly by the mechanism of normalizing the altered gamma oscillation (i.e., increased amplitude and shortened latency). By regression analysis, different stimulation parameters like total pulses, number of treatment sessions, and pulse intensity did not influence effect sizes. This result may be due to heterogeneity of studies, but may also imply the ideal parameters are affected by more factors other than treatment doses, at least in the ranges of parameters used in previous studies.
We did not find a difference in effect sizes between single rTMS and rTMS plus exposure process. The reason may be due to the bias caused by indirect comparisons of heterogenous studies, as well as the exposure process did not follow typical procedure of exposure therapy.
The study by Harris and Reece 6 that included various types of TMS found that studies utilizing an RCT had significantly larger treatment effects than studies of other designs. This result thus addressed concerns about possible exaggerated effect sizes due to a lack of control in non-RCTs, giving support to include them in analysis. In our study, we did not find a significant difference between RCTs and other designs in effect sizes of LF rTMS. Our results may be more pertinent as our sample is more homogenous, although the difference may also be contributed to smaller sample sizes in our study.
Adverse effects occasionally occur during both rTMS treatment and sham. LF rTMS-related side effects are commonly mild. In fact, no serious side effect definitively caused by treating procedure was found. Our result proved the safety of LF rTMS.
This review has some limitations. First, the PTSD populations varied, containing military personnel and civilians. Secondly, parameters such as number and intensity of stimulations of LF rTMS were different among studies. This heterogeneity may cause bias when combining these studies to evaluate effect sizes. Since we have found that intensity was the main source of heterogeneity, we addressed this issue through subgroup analysis. Thirdly, the number of research included in the network meta-analysis is relatively small and more studies are required to confirm the efficacy of LF rTMS as compared to sham.
Conclusion
The current literature supports the efficacy of LF rTMS in treating PTSD caused by various traumatic events. However, present limited number of RCT studies only showed LF rTMS to have a trend of advantage compared to sham treatment in treating PTSD caused by external traumatic events. In the future, studies are required to elucidate the underpinning mechanisms in order to further improve its efficacy, and more widespread use of LF rTMS can be expected for different traumatic populations.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253241271870 – Supplemental material for Low-frequency repetitive transcranial magnetic stimulation for the treatment of post-traumatic stress disorder and its comparison with high-frequency stimulation: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tpp-10.1177_20451253241271870 for Low-frequency repetitive transcranial magnetic stimulation for the treatment of post-traumatic stress disorder and its comparison with high-frequency stimulation: a systematic review and meta-analysis by Che Jiang, Yong Yang, Lili Wu, Weizhi Liu and Gang Zhao in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-2-tpp-10.1177_20451253241271870 – Supplemental material for Low-frequency repetitive transcranial magnetic stimulation for the treatment of post-traumatic stress disorder and its comparison with high-frequency stimulation: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tpp-10.1177_20451253241271870 for Low-frequency repetitive transcranial magnetic stimulation for the treatment of post-traumatic stress disorder and its comparison with high-frequency stimulation: a systematic review and meta-analysis by Che Jiang, Yong Yang, Lili Wu, Weizhi Liu and Gang Zhao in Therapeutic Advances in Psychopharmacology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
