Abstract
Following severe brain injuries, a subset of patients may remain in an altered state of consciousness; most of these patients require artificial feeding. Currently, a functional oral phase and the presence of exclusive oral feeding may constitute signs of consciousness. Additionally, the presence of pharyngo-laryngeal secretions, saliva aspiration, cough reflex and tracheostomy are related to the level of consciousness. However, the link between swallowing and consciousness is yet to be fully understood. The primary aim of this review is to establish a comprehensive overview of the relationship between an individual’s conscious behaviour and swallowing (reflexive and voluntary). Previous studies of brain activation during volitional and non-volitional swallowing tasks in healthy subjects are also reviewed. We demonstrate that the areas activated by voluntary swallowing tasks (primary sensorimotor, cingulate, insula, premotor, supplementary motor, cerebellum, and operculum) are not specific to deglutitive function but are shared with other motor tasks and brain networks involved in consciousness. This review also outlines suitable assessment and treatment methods for dysphagic patients with disorders of consciousness. Finally, we propose that markers of swallowing could contribute to the development of novel diagnostic guidelines for patients with disorders of consciousness.
Introduction
Over the last decades, the number of patients who have survived severe acquired brain injury has significantly increased due to improvements in intensive care medicine and life-sustaining treatments. Acquired brain injury as a result of traumatic, vascular, anoxic or metabolic origin can impact cognitive (Skandsen et al., 2010), language (Aubinet et al., 2022), motor (Thibaut et al., 2015), sphincter (Foxx-Orenstein et al., 2003) and even feeding functions (Brady et al., 2006; Mandaville et al., 2014). Following a period of coma, some of these patients may develop prolonged disorders of consciousness (DoC) and remain in these conditions for months, years, or even decades.
Patients with DoC may progress through different states of altered consciousness ranging from the vegetative state/unresponsive wakefulness syndrome (UWS) characterized by the recovery of eye opening without any behavioural signs of consciousness (i.e., only reflexive movements) (Laureys et al., 2010) to the minimally conscious state (MCS) defined by discernible but inconsistent behavioural evidence of consciousness (Giacino et al., 2002). Additionally, patients in MCS are subcategorized into MCS minus (MCS-) or MCS plus (MCS+) depending on signs of preserved language processing (Bruno et al., 2011). There is also a new subcategory called non-behavioural MCS or MCS*. Patients in MCS* are not responsive at bedside but retain brain activity compatible with a diagnosis of MCS (Gosseries et al., 2014; Thibaut et al., 2021). Finally, patients may emerge from the MCS when they recover functional communication and/or functional use of objects (Giacino et al., 2002). Consciousness exists along a continuum and these categories should be considered cautiously. Indeed, there is a spectrum of awareness between complete unconsciousness and consciousness, and the absence of purposeful behaviour does not necessarily imply the absence of consciousness (Fischer & Truog, 2017).
The Coma Recovery Scale –Revised (CRS-R) is a behavioural test used to assess the level of consciousness of patients with severe brain injury and is currently the most recommended diagnostic tool (Giacino et al., 2004; Seel et al., 2010). The scale is composed of six categories (auditory, visual, motor, oromotor/verbal, communication, arousal), and specific items denote the diagnosis of MCS (e.g., response to command, visual pursuit, localisation to noxious stimulation). To reduce misdiagnosis, at least five assessments within a period of two weeks should be administered (Wannez et al., 2017). The Simplified Evaluation of CONsciousness Disorders (SECONDs) similarly assesses level of consciousness but can be administered more rapidly (Aubinet et al., 2021; Sanz et al., 2021). In addition to the CRS-R or the SECONDs criteria used to distinguish the different levels of consciousness, other possible signs of consciousness have been explored such as resistance to eye opening, spontaneous eye blinking rate, auditory localisation, habituation of auditory startle reflex, olfactory sniffing, efficacy of swallowing/oral feeding, leg crossing, facial expressions to noxious stimulation, and subtle motor behaviours (Mat et al., 2022). The CRS-R and other potential signs of consciousness are summarised in Figure 1.
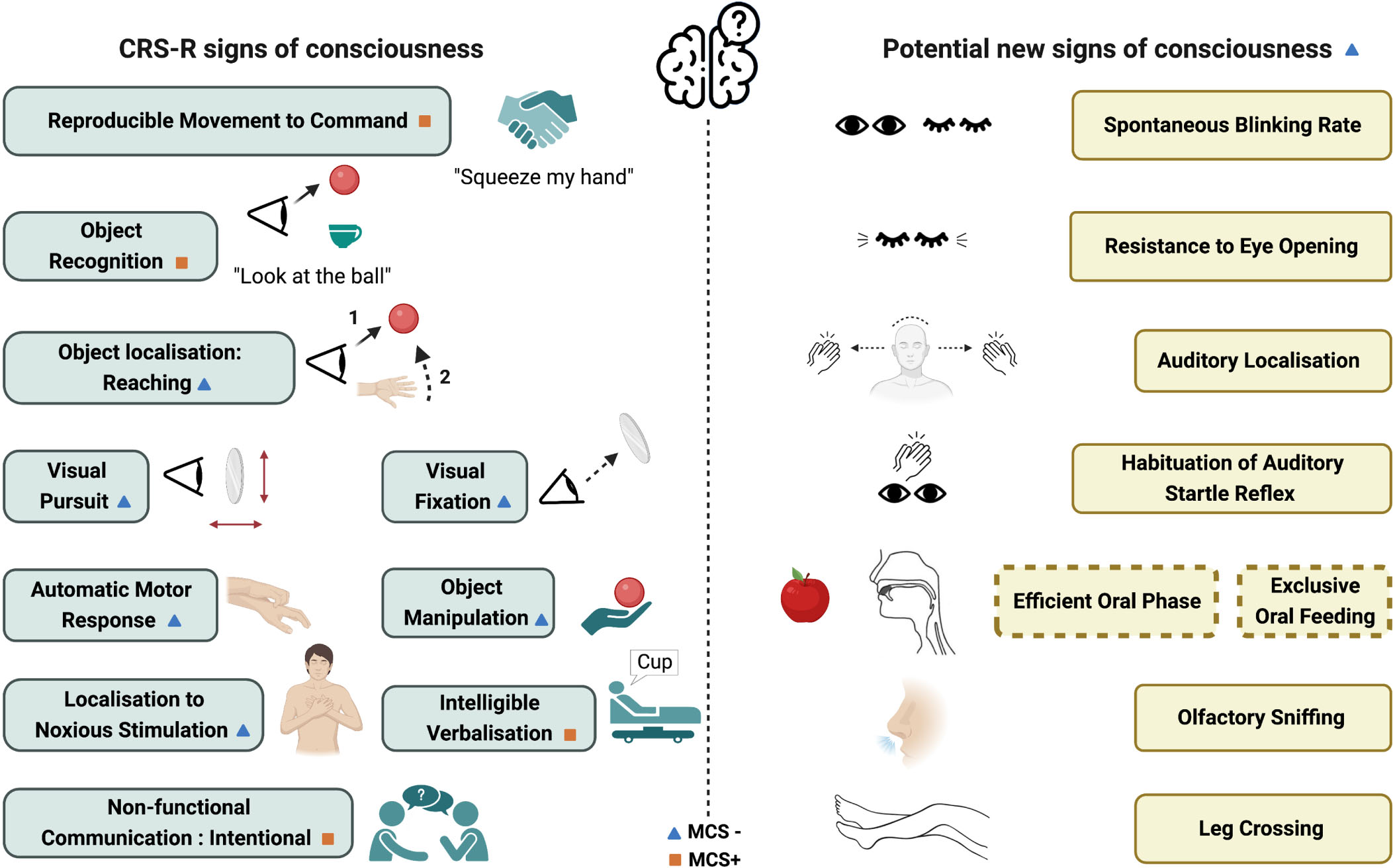
Behavioural signs of consciousness based on the Coma Recovery Scale-Revised and on recent studies (Giacino et al., 2002; Bruno et al., 2011; Mat et al., 2022). Orange square denotes a diagnosis of MCS+ and blue triangle denotes at least a diagnosis of MCS-.
Severe dysphagia (i.e., swallowing difficulties) is almost universally present in patients with DoC and can lead to major functional consequences and comorbidities such as a dependence on tracheostomy, pulmonary discomfort, congestion or infection, dehydration, and malnutrition (Mélotte et al., 2021). In all hospitalised patients, dysphagia is a bad prognosis indicator and influences the length of the hospital stay (Altman et al., 2010). A large proportion of patients with DoC also develop oral apraxia (i.e., impairment of nonspeech volitional movement), which may interfere with the swallowing assessment (Zhang et al., 2021). Assessment and swallowing therapy in patients with DoC are therefore essential aspects of their daily management. Moreover, as recently suggested by Mat and colleagues (2022), swallowing ability should be considered in discriminating patients’ level of consciousness. Indeed, Mélotte and collaborators (2018, 2021) suggested links between swallowing components and consciousness levels.
The primary aim of this review is to establish a comprehensive understanding of the relationship between an individual’s conscious or volitional behaviour and the swallowing activity. The enhanced comprehension of this relationship holds the potential to contribute to the development of novel diagnostic guidelines for patients with DoC that include considerations of swallowing components in their criteria. This will be achieved first, by identifying which components of swallowing can be characterised as conscious through an analysis of the phases of swallowing and by examining which swallowing tasks require conscious processing. Secondly, we will shed light on brain activations that occur during swallowing by reviewing neuroimaging data acquired from healthy subjects (i.e., voluntary vs. non-voluntary tasks). Finally, we will provide an overview of the assessment tools and therapeutic methods that can be used, to our knowledge, for the management of dysphagia in patients with DoC.
Can swallowing components be considered “conscious”?
Distinguishing conscious from unconscious behaviours is historically based on the principle of differentiating reflexive from volitional behaviours (Giacino et al., 2002). However, as described by Fischer et al. (2015), there are no generalisable empirical characteristics that reliably distinguish between reflexive and conscious behaviours. As suggested by Mélotte et al. (2022), analysing the characteristics of the different phases of swallowing may help distinguish reflexive from volitional behaviours.
Swallowing is classically divided into 3 phases: oral, pharyngeal, and oesophageal. The oral phase includes the motor actions of opening the mouth appropriately (depending on what we ingest), lip prehension, lingual manipulation, mastication (in case of solid food) and lingual propulsion. Like any other motor activity, the oral phase includes some automatic processes that allow us to eat without thinking about what we are doing, but it is also the only phase that can be interrupted, modified, and consciously controlled (Mélotte et al., 2022). Consciously controlling this phase can also influence its length and efficacy (Furuya et al., 2014). Moreover, no empirical data has described UWS patients as having an efficient oral phase (mouth opening, lip prehension, lingual propulsion) (Mélotte et al., 2021; 2018) suggesting that the presence of an efficient oral phase may constitute a sign of consciousness. For these reasons, the oral phase may be considered voluntary/conscious (Fig. 2A).
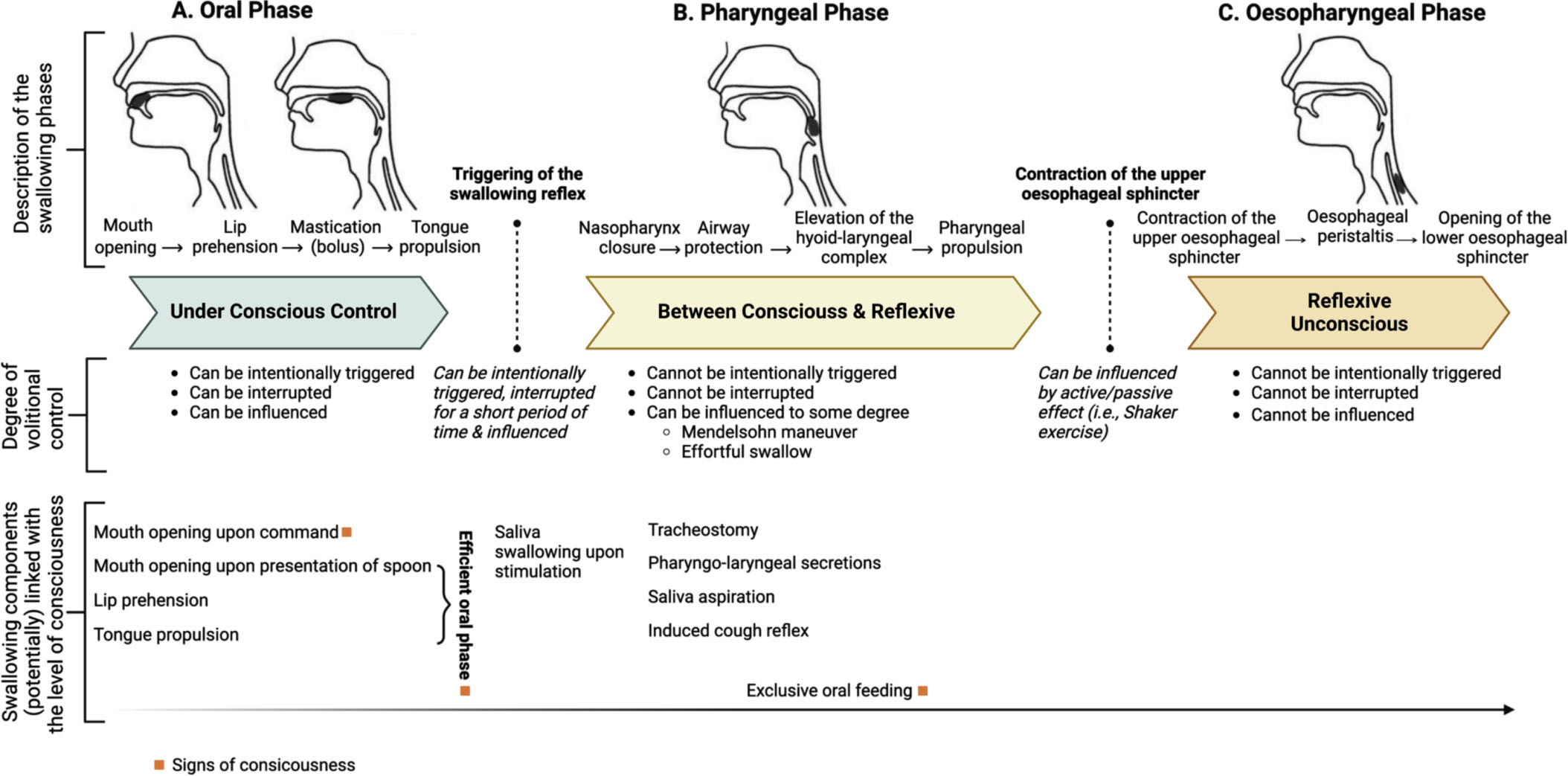
Characteristics of the oral (A), pharyngeal (B) and oesophageal phases (C) regarding their conscious or unconscious nature. Orange square denotes a sign of consciousness. Adapted with permission from Mélotte et al. (2022).
The triggering of the swallowing reflex announces the start of the pharyngeal phase of swallowing. The pharyngeal phase is a complex reflex response (Steele & Miller, 2010). The swallowing reflex can be triggered voluntarily, but is usually automatic, difficult to reverse, and cannot be suppressed for long periods of time. Referring to the definition from Prochazka et al. (2000), the swallowing reflex is on the borderline between a voluntary behaviour and a reflex. At the clinical level, no data has been published yet on the frequency of swallowing in patients with DoC, but some previous studies during sleep (Sato et al., 2011), general anaesthesia (D’Angelo et al., 2014) and in post-stroke patients (Crary et al., 2014; Carnaby et al., 2019) report a significant decrease of the spontaneous swallowing frequency in these states. Mélotte et al. (2022) postulated that the frequency of triggering the swallowing reflex might be linked to the level of consciousness. However, the triggering of a swallowing reflex does not strictly require consciousness, therefore does not formally constitute a sign of consciousness.
The pharyngeal phase occurs after the triggering of the swallowing reflex and is composed of the velo-pharyngeal closure, the airway protection, the elevation of the hyoid-laryngeal complex, and the pharyngeal propulsion. These components cannot be suppressed, but can be influenced voluntarily, although at a lesser level compared to the oral phase (e.g., Mendelsohn manoeuvre or effortful swallow) (Humbert & German, 2013) (Fig. 2B). In a retrospective study on 92 patients with DoC, the efficacy of the pharyngeal phase was assessed through the presence or absence of pharyngo-laryngeal secretions, saliva aspiration and tracheostomy (Mélotte et al., 2021). More pharyngo-laryngeal secretions were observed in patients with UWS than in patients in MCS, although this was not significantly different after controlling for time since injury and etiology. The presence of a tracheostomy was also linked to the level of consciousness, with a higher proportion of patients with UWS having a tracheostomy compared to patients with MCS. The pharyngeal phase is mainly involuntary, the functional efficacy of its components may be linked to the level of consciousness. The pharyngeal phase ends when the upper oesophageal sphincter, also called the inferior pharyngeal sphincter, opens (Matsuo & Palmer, 2008).
The oesophageal phase begins with the contraction of the upper oesophageal sphincter and ends when the bolus passes through the lower oesophageal sphincter with the oesophageal peristalsis and into the stomach (Lang, 2009). This last phase cannot be voluntarily triggered or suppressed and is not reversible. However, the oesophageal phase can be influenced by the passive or active effects of the upper oesophageal sphincter. For example, the head-raising exercises described by Shaker and colleagues that strengthen the suprahyoid muscles enhance the pharynx propulsion, which improves the upper oesophageal sphincter opening (passive effect) (Tuomi et al., 2022). Recently, Winiker et al. (2022) showed that volitional modulation of the upper oesophageal sphincter resting pressure (active effect) is also possible with visual biofeedback training in healthy adults. Beyond that, no study described the possibility of voluntarily controlled oesophageal propulsion or lower oesophageal sphincter opening. For these reasons, the oesophageal phase can be considered reflexive and unconscious. Mélotte et al. (2022) go further by postulating that the opening of the upper oesophageal sphincter constitutes the border between somatic reflexes (pharyngeal phase) and autonomic reflexes (oesophageal phase). Finally, the links between components of the oesophageal phase and the level of consciousness has never been analysed (Fig. 2C). The conscious or unconscious nature of the swallowing phases and the potential links between swallowing components and consciousness are depicted in Figure 2.
To address the concept of reflexive and conscious behaviours, it is also possible to compare different types of swallowing tasks. Ertekin et al. (2001, 2011) made the distinction between reflexive, spontaneous and voluntary swallowing tasks. Recently, Mélotte et al. (2022) analysed the different types of swallowing tasks described in the literature and divided them into two categories: volitional swallowing tasks (VOST) and non-volitional swallowing tasks (NVOST). NVOST include reflexive swallowing tasks and spontaneous swallowing tasks. Reflexive swallowing tasks trigger the swallowing reflex by an external stimulus directly in the pharyngeal area (tactile or with the injection of a bolus). In reflexive swallowing tasks, the oral phase is almost “bypassed”, except for tongue movements occurring in any swallowing process (Mélotte et al., 2022). In spontaneous swallowing tasks, swallowing is assessed through swallowing of saliva, water, or food swallows, without specific external request (visual, auditory, or tactile cue). On the other hand, VOST refers to tasks implying swallowing after a specific request. With reference to Fischer’s work (Fischer & Truog, 2013), VOST imply interactive capacity defined by “the ability to receive communicated information and the intentional generation of a coherent response”. Moreover, Mélotte et al. (2022) also made a distinction between nutritive and non-nutritive swallows because of the potentially different brain activations and different physiological mechanisms involved. These different swallowing tasks are summarised in Table 1.
Description of the different types of swallowing tasks
Description of the different types of swallowing tasks
Adapted from Mélotte et al. (2022).
Swallowing tasks may be challenging in patients with DoC because of their multiple cognitive and motor impairments. If we only consider the level of consciousness, we can expect that patients in MCS+ may be able to perform volitional tasks as they can respond to verbal or visual commands, whereas patients in UWS or MCS- may be unable. Non-nutritive tasks and nutritive tasks can be assessed regardless of the level of consciousness. However, nutritive tasks should only be proposed to patients who demonstrate a certain level of swallowing ability, which can be determined through a detailed swallowing assessment. Some of these tasks have been used in neuroimaging studies to further investigate swallowing capacities.
To expand on the distinction between VOST and NVOST in light of neuroimaging data, we analysed previous studies exploring cerebral areas activated during VOST and/or NVOST in healthy adult subjects that both used positron emission tomography (PET) and/or functional magnetic resonance imaging (fMRI). Among the 39 identified studies (see Appendix 1 for description of studies and results), the majority (n = 36) focused on VOST tasks. VOST studies used both nutritive (water, barium, capsule) and non-nutritive tasks (saliva), and some studies compared the induced brain activity with that induced by other tasks, such as motor imaging of swallowing, speaking out loud, tongue movement or finger tapping. Regarding brain location, most of the studies (n = 30) focused on whole brain analysis, whereas four targeted regions of interest such as the insula, primary sensorimotor cortex, cerebellum, and pons (Lowell et al., 2012; Malandraki et al., 2011; Mihai et al., 2014; Toogood et al., 2017) and one focused specifically on the brainstem (Komisaruk et al., 2002).
Numerous cortical and subcortical areas are found to be involved in the control of VOST tasks (Fig. 3). Based on the 36 studies using VOST tasks, the main brain regions (identified in at least 50% of the studies) that are activated are the primary motor cortex (n = 33), insula (n = 31), primary somatosensory cortex (n = 29), anterior cingulate cortex (n = 23) (right > left), premotor cortex and supplementary motor area (n = 22), and cerebellum (n = 19) (left > right). Regions identified in at least 25% of studies are the superior temporal gyrus (n = 16), inferior parietal lobule (n = 15), thalamus (n = 14), posterior cingulate cortex (n = 13), putamen (n = 13) (left > right), inferior frontal gyrus (n = 13), frontal operculum (n = 11), precuneus (n = 10), superior frontal gyrus (n = 10), cuneus/lingual gyrus (n = 9), middle frontal gyrus (n = 9), middle cingulate gyrus (n = 9) and middle temporal gyrus (n = 9).
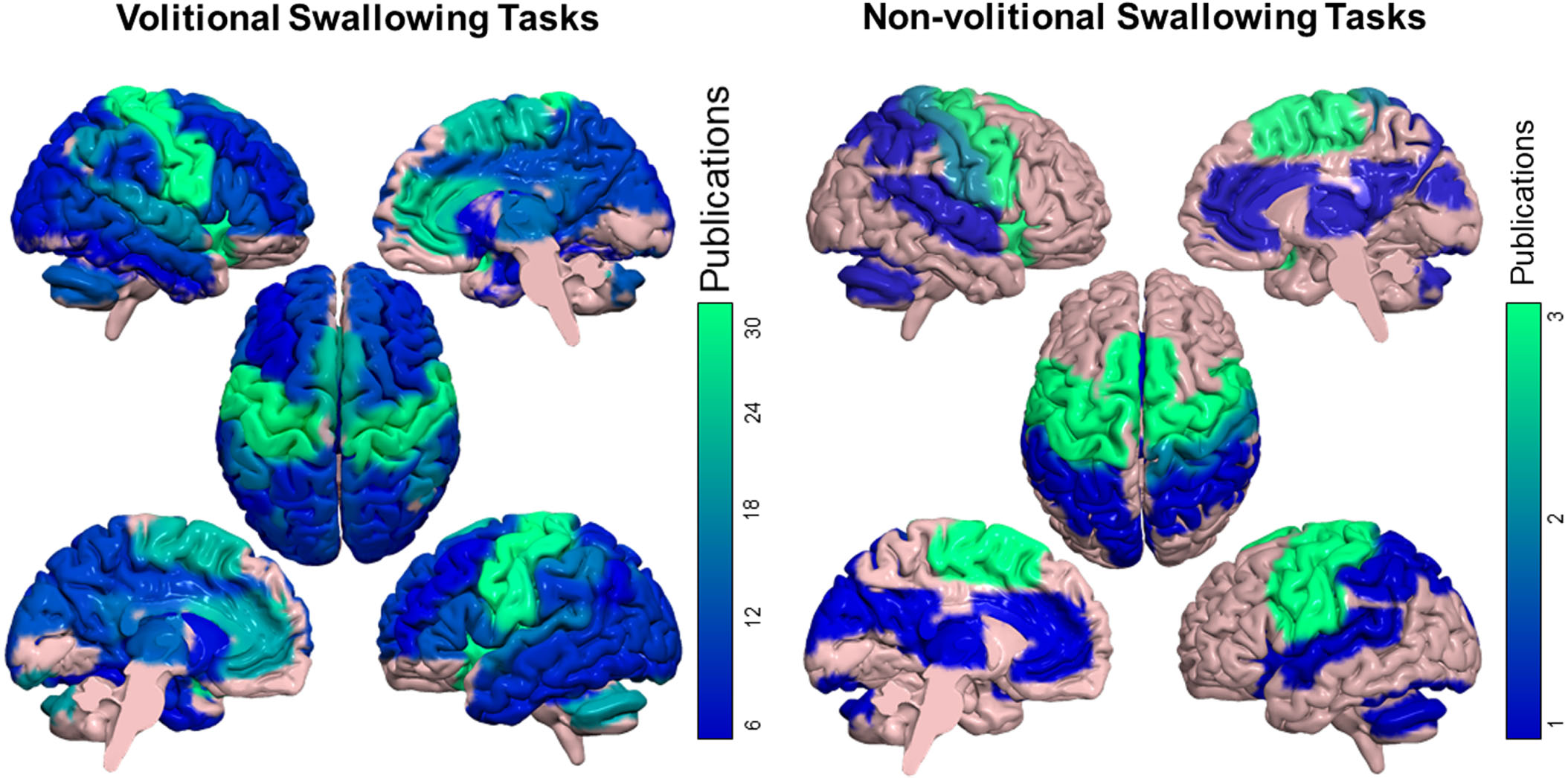
A qualitative analysis showing the brain areas activated in volitional swallowing tasks (VOST) and non-volitional swallowing tasks (NVOST) in healthy individuals, based on the brain areas and Brodmann areas mentioned in the studies. Each brain area identified was linked to brain areas in the AAL Atlas (Rolls et al., 2015). Colours refer to the number of studies that mentioned the specific area. For studies using VOST, brain areas mentioned in at least 3 studies are illustrated. Due to the low number of studies in NVOST (n = 3), all study-related brain areas are shown in the figure.
These findings demonstrate that VOST primarily activates sensory and motor brain areas involved in planning, control, and execution, as well as brain regions implicated in volitional processes, such as the insula and anterior cingulate cortex. Furthermore, VOST activates numerous other brain areas, which accounts for the variability in swallowing function observed in individuals with various brain lesions. Additionally, VOST also share activation with brain areas involved in consciousness networks including the posterior cingulate cortex, inferior parietal cortices, precuneus and superior frontal gyrus (Broyd et al., 2009; Heine et al., 2012).
Only three studies explored NVOST tasks. Two of them induced spontaneous saliva swallowing while one induced reflex swallowing by introducing a predetermined threshold volume in the pharynx. The small number of studies exploring NVOST tasks makes it difficult to compare them with VOST tasks. However, we observed that the activated regions were not specific to NVOST, as they were all similarly activated in VOST tasks. Some brain areas (i.e., precuneus, inferior, middle, and superior frontal cortex, frontal operculum, putamen and middle cingulate cortex) were activated during VOST tasks, but not NVOST tasks. Moreover, NVOST (i.e., reflexive swallowing) is characterized by greater cortical activation in the left hemisphere, whereas VOST shows greater volume activation in the right hemisphere (Kern et al., 2001).
Paine et al. (2011) compared their results obtained with spontaneous saliva swallowing (NVOST) with the results of another study (Malandraki et al., 2009), which used voluntary water swallowing (VOST). They showed that regions activated in both tasks were almost identical: regions related to motor control, sensory input and somatosensory integration. However, the authors reported that the significant activations from the spontaneous swallowing study were much more localised in motor control areas. In addition, Humbert et al. (2009) compared brain activity, as measured by fMRI, during the swallowing of three types of boluses (saliva, water, and barium), and found a higher cerebral activity during the swallowing of saliva condition. Saliva swallowing may elicit larger responses due to the increasing effort required, despite minimal sensory stimulation in the oropharynx compared to the other conditions (water and barium). Furthermore, swallowing tasks and specific oral (e.g., jaw clenching, tongue movements) or pharyngeal (i.e., throat clearing) tasks share areas of activation. In fact, the main brain areas activated during isolated oral or pharyngeal tasks (i.e., sensorimotor cortex, insula, anterior cingulate cortex, putamen, cuneus, precuneus) were also activated with swallowing.
Some authors have also highlighted age differences in brain cortical activation during swallowing. They found that older adults displayed larger neural activation particularly in the prefrontal cortex and middle temporal gyrus compared to younger adults, suggesting that swallowing may require greater neural control in this population (Humbert et al., 2009; Moon et al., 2016).
Finally, to overcome some limitations of fMRI and PET –notably the need of a supine position (fMRI) or radiation (PET) –Gallois and collaborators (2022) investigated in a meta-analysis the efficacy of alternatives that could be used during an “ecological swallowing task”. This task refers to any food intake situation in a usual/natural position (seated or standing up) with no invasive evaluation method. Three techniques can be used in ecological swallowing task: electroencephalography (EEG), magnetoencephalography (MEG) and functional near infra-red spectroscopy (fNIRS). These techniques cannot however be used during mealtime, are limited in terms of spatial exploration (fNIRS) and require multiple trials (EEG and MEG). The authors suggest exploring the use of both EEG/MEG (neuronal signal) and fNIRS (hemodynamic signal). Moreover, fNIRS could have promising therapeutic applications using visual neurofeedback during swallowing.
As previously mentioned, assessing swallowing in patients with severe brain injury is challenging. In fact, in the absence of functional communication, response to command or severe spasticity limits considerably the possibility to perform a “classic” swallowing bedside assessment. To remedy the lack of appropriate bedside tools to assess swallowing in patients with DoC, Mélotte et al. (2021) developed the Swallowing Assessment in Disorders of Consciousness (SWADOC). This tool includes 48 qualitative items and 8 quantitative items. Qualitative items invite clinicians to pay attention to the presence or absence of a wide range of components related to swallowing. By their discrete nature, quantitative items allow clinicians to monitor the evolution of a patient, compare between patients, and to assess the effectiveness of a specific management program. For each quantitative item, a patient’s abilities are rated on a four-level scale ranging from 0 to 3. Four items are linked to the oral phase and four to the pharyngeal phase. This tool is in the process of validation in 104 patients with DoC, but a preliminary version is already available in French and English (Mélotte et al., 2021; Regnier et al., 2023). However, even if this tool contributes towards a better understanding of the swallowing profiles of patients, it does not replace an objective swallowing assessment with a Fiberoptic Endoscopic Evaluation of Swallowing (FEES) or Video Fluoroscopic Swallowing Study (VFSS) when we consider feeding a patient orally. Indeed, the prevalence of silent aspiration is high in patients with severe brain injury (Terré & Mearin, 2007); relying only on external signs of dysphagia (e.g., cough, voice changing) to determine if a patient can receive oral feeding may risk bronchoaspiration or denutrition.
Administering a detailed bedside assessment of swallowing is the first step to better understand the clinical swallowing profile of a patient. As a complement, in patients with DoC, FEES and VFSS administered by an ear, nose and throat specialist (ENT) and/or a radiologist can objectively describe pharyngo-laryngeal sensitivity, laryngeal mobility, presence or absence of pharyngo-laryngeal secretions and saliva aspiration. Collaboration with ENT specialists can also guide clinicians during the process of tracheostomy weaning. A systematic review aiming to assess the feasibility and safety of FEES in patients with DoC showed that FEES can be used in this population and has the potential to facilitate advancement in the patients’ oral intake (Checklin et al., 2022). Finally, whilst noting that objective swallowing assessments are essential to determine the possibility to feed a patient orally. Not all patients are able to perform a functional swallowing test. Four criteria are necessary to examine before considering doing a FEES or a VFSS: (1) semi-seated position for a minimum of 15 min; (2) mouth opening; (3) at least minimal tongue propulsion; and (4) swallowing reflex present spontaneously or elicited by stimulation in the pharyngo-laryngeal area (Mélotte et al., 2022).
To further explore swallowing in patients with DoC, it will be interesting in the future to find other ways that do not require the patient’s participation. Recently, some authors explored the possibility of using spontaneous swallowing frequency to determine the severity of dysphagia. Spontaneous swallowing frequency has been studied in various populations of patients: post-stroke (Crary et al., 2014); Parkinson’s disease (Pehlivan et al., 1996); head and neck cancer (Carnaby et al., 2021); cerebral palsy (Crary et al., 2022) and elderly population (Murray et al., 1996; Tanaka et al., 2013). Measuring the spontaneous swallowing frequency seems to be a simple and non-invasive technique that does not require active participation and can be administered in patients with DoC. Currently, several methods of recording the swallowing frequency are used, including the use of a microphone (sound analysis), an accelerometer (vibration analysis) or a surface electromyography. In due course, reliable measures of spontaneous swallowing frequency may allow for additional information about saliva management, quantitative measure of the patient’s progress and a way to appraise the effect of a therapy.
Citric acid cough reflex testing is another promising technique for patients with DoC. This test consists in administering a solution of a tussive agent via an aerosol and measuring the time between the start of the administration and the triggering of the cough reflex. This is a simple, non-invasive method of cough testing that also does not require the patient’s active participation. Some studies have shown a link between cough reflex testing and the presence of aspiration in an objective swallowing assessment (Miles et al., 2013; Wakasugi et al., 2008). Furthermore, there is also a link between the cough reflex elicited by a fiberscope in the pharyngo-laryngeal area and the level of consciousness (Mélotte et al., 2021). This technique can help clinicians to manage pharyngo-laryngeal and pulmonary congestion by the expulsion of secretions. The techniques described above allow clinicians to obtain quantitative measures that can contribute to monitor the effectiveness of a speech-therapy treatment.
Nutritional status and management in patients with DoC
When assessing swallowing in patients with DoC, therapists should also focus on their nutritional status, which is understudied. Currently, there are different tools that assess the type and method of feeding independently. Going forwards, it would be useful to simultaneously gather information on both aspects, especially in cases of partial or complete oral feeding. The Food Intake Oral Scale (Kunieda et al., 2013) can be used to document the degree of oral intake daily. It is classified on a 10-level scale with three categories: no oral intake, oral intake with alternative nutrition, and oral intake alone. For patients who receive oral feeding, the International Dysphagia Diet Standardisation (IDDSI) is recommended (Cichero et al., 2017) to document the type of feeding.
Although the association between the level of consciousness and the type of feeding has been reported in patients with severe brain injury (Brady et al., 2006; Terré & Mearin, 2007; Kjaersgaard et al., 2015; Bremare et al., 2016), few studies have documented the type of feeding in patients with DoC (Mélotte, 2018; Mélotte et al., 2021; Ippoliti et al., 2023). In a retrospective cohort study of 92 patients with DoC (26 patients with UWS and 66 patients in MCS), no significant association was found between the type of feeding (exclusive enteral nutrition or not) and the consciousness diagnosis (Mélotte et al., 2021). Eighty-eight percent of those with UWS and 73% of those with MCS received exclusive enteral feeding. Additionally, none of the patients in UWS received full oral feeding, and only a small proportion of the patients in MCS (7%) could safely resume full oral feeding with easy-to-swallow food.
In another retrospective cohort study of 9 patients with prolonged DoC and 11 patients who regained consciousness, primarily due to traumatic brain injury, all individuals were found severely underweight with a Body Mass Index below 15. These patients exhibited a higher prevalence of complications during the rehabilitation phase when compared to patients who were adequately nourished. Furthermore, ten of the patients who were undernourished required an additional year after the brain injury to reach the functional goal of becoming practically independent (Dénes, 2004). In contrast, in another more recent retrospective cohort study on 80 patients with DoC (Ippoliti et al., 2023), almost all patients were well nourished, with only 9% at risk of under-nutrition based on a Body Mass Index of < 18.5 kg/m2 and based on a difference between daily calorie needs and real intake of calories and proteins. Patients with severe spastic muscle overactivity had a lower body mass index. Additionally, a negative association was observed between lower limbs spasticity and body mass index, suggesting that patients with spastic muscle overactivity may require additional calorie intake. Interestingly, patients with the highest body mass index in that study received less calories, compared to those with a lowest body mass index.
Swallowing therapies in patients with DoC
The overall management of patients with DoC is complex, with its remit surpassing standard care practices. It requires a constant ethical reflection between the clinical teams and among family members. Care practices must be adapted according to the patients’ level of consciousness, prognosis, sign of discomfort and pain. The comprehensive management of dysphagia involves a team of specialists from various disciplines who work together to provide a thorough evaluation and treatment plan for the patient. The medical and paramedical team generally includes a speech-language pathologist, otolaryngologist, gastroenterologist, radiologist, neurologist, pulmonologist, pharmacist, dietitian, occupational therapist, and physical therapist (Logemann, 1994).
Swallowing management is an essential part of speech therapy, which also encompasses voice, breath, orofacial tonicity and sensitivity, communication, oral hygiene, and tracheostomy weaning (Roberts & Greenwood, 2019). Additionally, it should also be considered in the wider context of “awakening” stimulation. The general goals of the treatment are to limit sensory deprivation, improve respiratory comfort by limiting aspiration of secretions and saliva, allow therapeutic feeding (i.e., giving a small amount of food and/or thickened liquid to the patient to stimulate the oral and pharyngeal phases of swallowing) (Jakobsen et al., 2019), encourage a good mobility of the different oro-pharyngeal structures, improve orofacial comfort (diminished eventual spasticity and hypertonicity that can conduct to bite wounds), and support a good oral hygiene (Fig. 4A). Besides that, specific goals must be determined and readjusted continuously according to the swallowing profile and potential improvements of the patients (Roberts & Greenwood, 2019; Nusser-Müller-Busch & Lehmann, 2021).
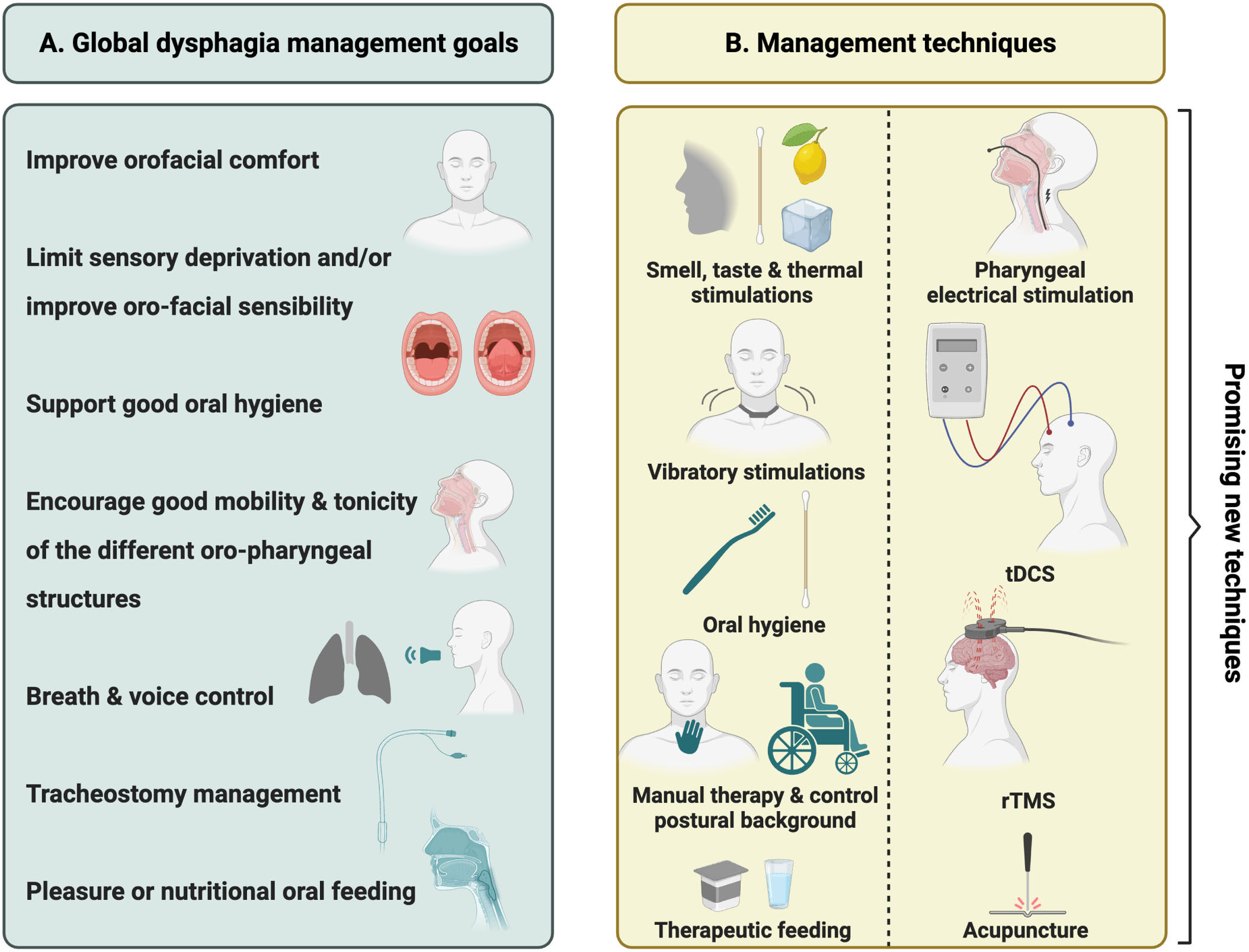
Summary of swallowing management goals (A) and therapies (B) in patients with DoC.
A range of factors, such as the specificity, intensity, and frequency of the treatment, contribute to the success of a rehabilitation therapy (Kleim & Jones, 2008). Moreover, studies focusing on motor control and learning reveal that in the context of patients with brain damages, information proposed by the therapist should be “unambiguous, concrete, clear, consistent and short, and should be repeated several times” (Mulder & Hochstenbach, 2003). Therapists should find the right balance between observation and stimulation and avoid excessive technicity that can neglect accurate observation of the patient. Therapeutic interventions must include the observation, interpretation and evaluation of all motor responses. Furthermore, stimulations of the orofacial area may feel intrusive for the patient (Nusser-Müller-Busch & Lehmann, 2021). That is why therapists must remain attentive to signs of discomfort.
To date, no study has been published about the efficacy of a specific swallowing treatment protocol for patients with DoC. Akin to assessments, classical management techniques are not applicable to patients with severe brain injuries. Currently, the most valuable clinical approach to manage dysphagia in patients with DoC is the Facial-Oral Tract Therapy (F.O.T.T.) (Nusser-Müller-Busch & Lehmann, 2021). This is an interprofessional approach based on the Bobath Concept created for patients with brain injuries. The F.O.T.T. book (Nusser-Müller-Busch & Lehmann, 2021) provides a detailed description of principles and techniques that can be applied to patients with severe brain injuries and, to some extent, to patients with DoC. The therapy is based on tactile and proprioceptive stimulations (i.e., patient touching their own face with their hands, applying firm pressure on the patient’s face, performing intraoral stimulations of targeted sites with a finger), manual facilitation techniques and control postural background (i.e., utilisation of adapted chairs, upright pelvis, relaxed head in a central position).
However, when working with patients with DoC, whose behavioural responses are often limited, therapists may encounter challenges in eliciting active participation. Some exercises recommended by the F.O.T.T. require an active participation of the patient during rehabilitation. Some of these exercises may be adapted to fit with clinical specificities of patients with DoC by proposing for instance passive and/or assisted movements of the oro-facial sphere instead of responses to commands or providing tactile support for expiration (Bicego et al., 2014). Some techniques that require no active participation have been developed and could be implemented for dysphagia management of patients with DoC. For example, the application of cold oral stimulation in 18 healthy volunteers resulted in significant variations in pharyngeal cortical excitability, indicating the existence of a sensorimotor correlation between cortical areas involved in oral and pharyngeal function (Magara et al., 2018). Besides thermal stimulations, smell and taste can also be used in clinical practice. Prum and collaborators (2022) conducted a study in 8 patients with UWS after an exposure to strong tastes and smells. Four different tastes were used: fruity (orange and banana), empyreumatic (chocolate and coffee), vegetal (tea and garlic) and spicy (mint and vanilla). To stimulate smell, substances were crushed and placed 5 cm in front of the patient’s nose for 10 minutes. To activate taste, cotton swabs were placed on the tongue for the same amount of time. Patients were trained during five sessions in one week. They demonstrated an increase of spontaneous swallowing frequency but no change in tongue or velum mobility after the fifth session.
To further treat dysphagia without requiring the active participation of the patients, Mulheren & Ludlow (2017) have described the use of vibration over the larynx in healthy volunteers. They observed an increase of spontaneous swallowing and cortical activation for swallowing during the stimulation. Similar results were found in patients with oropharyngeal dysphagia secondary to a cerebrovascular accident or following radiation treatment for head and neck cancer (Kamarunas et al., 2019). Vibratory stimulations are non-invasive and can stimulate spontaneous swallowing, however, the long-term effectiveness of this treatment has not yet been evaluated. In addition, this technique has never been proposed to patients with DoC. Acupuncture combined with rehabilitation is another potential method to treat neurogenic dysphagia. In a recently published meta-analysis aimed at assessing the efficacy of manual acupuncture in post-stroke dysphagia, it appears that the integration of this method during the rehabilitation improved swallowing efficacy and reduced the aspiration rate as well as the incidence of aspiration pneumonia (Jiang et al., 2022). Another treatment for dysphagia is the pharyngeal electrical stimulation. This stimulation is a peripheral intervention which indirectly amplifies the excitability of the motor cortex by increasing the sensori-motor input into the cortical areas responsible for pharyngeal function (Sasegbon et al., 2020). In an observational cohort study of 245 adults with neurogenic dysphagia, it was finally emphasized that pharyngeal electrical stimulation was safe and could improve dysphagia by reducing the risk of aspiration (Bath et al., 2020). This technique could eventually be implemented in patients in MCS+ since it requires a certain level of consciousness because patients must perform swallowing exercises during the stimulation. Therefore, the healthcare provider needs to assess the level of consciousness and the cognitive abilities of patients before deciding if this technique is suitable for them.
In addition to the use of laryngeal vibrations, pharyngeal electrical stimulation, or acupuncture, non-invasive brain stimulations techniques such as transcranial direct current stimulation (tDCS) and repetitive transcranial magnetic stimulation (rTMS) also represent other new promising approaches for dysphagia management (Sasegbon et al., 2020; Cheng et al., 2022). It has already been demonstrated that repeated left prefrontal tDCS could improve the recovery of consciousness in patients with MCS (Thibaut et al., 2014, 2017, 2019). Moreover, the use of this technique on the pharyngeal motor cortex in patients with post-stroke dysphagia has previously been shown to lead to swallowing improvements (Yang et al., 2012; Li et al., 2020). tDCS is affordable, has low side effects and can be easily implemented in a rehabilitation setting (Barra et al., 2022; Ho et al., 2022). Compared with tDCS, rTMS requires specialised equipment and trained clinicians (Rossi et al., 2009), and the device is more expensive and difficult to transport (Priori et al., 2009). When comparing both techniques, rTMS appears to induce more immediate and robust changes in cortical excitability compared to tDCS (Lefaucheur et al., 2020). The use of rTMS in patients with MCS may also improve awareness and arousal (Formica et al., 2021). In a rTMS study conducted by Park and colleagues (2017), 35 patients with post-stroke dysphagia were randomly assigned to three intervention groups: bilateral stimulation, unilateral stimulation, and sham condition. The bilateral group underwent 500 pulses of 10Hz rTMS applied at the ipsilesional followed by contralesional motor cortex. The unilateral group had the same stimulation over the ipsilesional motor cortex only, and a sham condition over the contralesional hemisphere. For the sham condition, rTMS was applied to the bilateral motor cortices. All groups had daily stimulations for two weeks. Results show that patients in the bilateral group showed significant improvements of the severity of dysphagia immediately after and three weeks after the last stimulation compared to the other two groups. rTMS applied to the bilateral motor cortices may thus complement traditional swallowing therapy. While the mechanisms by which tDCS and rTMS improve swallowing function are not fully understood, the prevailing view is that these techniques modulate the neuronal pathways involved in the control of swallowing. By targeting specific regions of the brain engaged in swallowing, these techniques may be able to enhance neural plasticity and support the recovery of swallowing function (Sasegbon et al., 2020). The techniques presented here are summarised in Figure 4B.
This narrative review outlines the characteristics, assessments, and treatments of dysphagia in patients with DoC, underlining the need for adequate assessment and targeted management. Objective assessments using instrumental investigations such as VFSS and FEES are crucial for identifying both structural abnormalities and functional disorders of the pharynx, when we consider tracheostomy weaning or oral feeding. Additionally, a novel behavioural tool, the SWADOC, provides speech-language pathologists an accurate bedside assessment of swallowing functions. Considering that patients with DoC may have an increased risk of under-nutrition, nutritional management should also be considered while evaluating patients’ swallowing function. Some therapeutic techniques that do not require active participation from the patients have also been highlighted and can be implemented in speech therapy. However, further studies are needed to investigate the efficacy and long-term effects of these techniques within this specific population.
This review raised and attempted to address three primary questions regarding i) the relationship between consciousness and swallowing, ii) the tasks that involve conscious processes, and iii) how neuroimaging can shed light on the mechanisms underlying the conscious aspect of swallowing. Based on the analysis of the phases of swallowing and empirical data on patients with DoC, it appears that swallowing components fall on a continuum from conscious to unconscious processes. An efficient oral phase may be considered as a sign of consciousness. Therefore, a patient who exclusively receives oral feeding, regardless of texture, may be considered at least minimally conscious. Components of the pharyngeal phase (presence of pharyngo-laryngeal secretions, risk of saliva aspiration, spontaneous swallowing frequency, tracheostomy) are related to the level of consciousness but do not constitute signs of consciousness. Thus, examining swallowing tasks may help differentiate between volitional and non-volitional tasks, as well as between nutritive and non-nutritive tasks. VOST tasks require interactive capacity and should be proposed to patients who respond to commands (i.e., MCS+ or higher consciousness level). Further investigations are needed to determine the conditions under which the completion of a VOST can be considered as a sign of consciousness.
Regarding neuroimaging analysis, comparing VOST to NVOST, the paucity of research does not provide sufficient comparison elements due to the limited number of studies reporting NVOST. The primary motor cortex, somatosensory cortex, postcentral gyrus, insula, premotor cortex and supplementary motor area, anterior cingulate cortex, and cerebellum (left > right) are the main areas activated during VOST. Cortical activations identified during swallowing tasks are not specific to swallowing function as they are also involved in related motor tasks. Furthermore, VOST seems to activate brain areas shared with consciousness networks, including the posterior cingulate cortex, inferior parietal cortices, precuneus and superior frontal gyrus. These findings may provide guidance for determining the optimal site for non-invasive brain stimulations techniques such as tDCS and rTMS.
Ultimately, the relationship between swallowing and consciousness exists but remains understudied, necessitating further research on this topic. Therefore, we support the further exploration of the link between swallowing process and consciousness with a view towards developing new diagnostic criteria.
Footnotes
Acknowledgments
We would like to express our gratitude to the University and University Hospital of Liège, the patients and their families, the Leon Fredericq Foundation (CNRF funds), the Belgian National Funds for Scientific Research (FRS-FNRS), the FNRS PDR project (T.0134.21), the ERA-Net FLAG-ERA JTC2021 project ModelDXConsciousness (Human Brain Project Partnering Project), the fund Generet, the King Baudouin Foundation, the Télévie Foundation, the European Space Agency (ESA) and the Belgian Federal Science Policy Office (BELSPO) in the framework of the PRODEX Programme, the Public Utility Foundation ‘Université Européenne du Travail’, “Fondazione Europea di Ricerca Biomedica”, the BIAL Foundation, the Mind Science Foundation, the European Commission, the Mind-Care foundation, the National Natural Science Foundation of China (Joint Research Project 81471100) and the European Foundation of Biomedical Research FERB Onlus, the Horizon 2020 MSCA –Research and Innovation Staff Exchange (101131344). Development of a multimodal toolbox to ensure a fast and reliable diagnosis of consciousness disorders (DOCBOX). NA is a research fellow, CA is a post-doctorate fellow, AT and OG are research associates and SL is a research director at FRS-FNRS.
Conflict of interest
The authors declare that they have no conflict of interest.
