Abstract
Background:
Diagnosis of pharyngeal dysphagia in patients with Parkinson’s disease is often difficult as reliable screening methods are lacking so far and clinical examination fails to adequately assess the pharyngeal phase of swallowing.
Objective:
To identify clinical predictors indicating the presence of pharyngeal dysphagia in patients at risk.
Methods:
We examined pharyngeal dysphagia in a large cohort of patients with Parkinson’s disease (n = 200) divided in three clinical subtypes (tremor-dominant (TD), mainly bradykinetic (BK) and early postural instability and gait difficulty PIGD)) by using flexible endoscopic evaluation of swallowing. ANOVA-multivariance analysis and following t-tests as well as binary logistic regression analysis were performed to detect group differences and to identify clinical predictors for dysphagia.
Results:
Statistically significant differences were found in the dysphagic group: age, male gender, disease duration, stage of the disease, Levodopa equivalent dose and higher scores on the Unified Parkinson’s disease rating scale III and II, item 7. The PIGD subtype was affected more frequently than the TD and BK subtype. In a logistic regression model higher age (>63.5 years p < 0.05) and Levodopa equivalent dose (>475 mg, p < 0.01) were identified to be independent predictors for the presence of pharyngeal dysphagia.
Conclusion:
Particularly patients with an age > 63.5 years and a daily Levodopa equivalent dose >475 mg show an increased risk for pharyngeal dysphagia. These findings may partly be influenced by presbyphagia but are likely to represent disease progression. The PIGD subtype seems to be a risk factor due to more pronounced dyscoordination of oropharyngeal muscle movements.
INTRODUCTION
Dysphagia is a common symptom in Parkinson’s disease (PD) which typically intensifies during disease duration [1]. In later stages of the disease, severe dysphagia leads to complications in medication intake, dehydration, malnutrition and aspiration pneumonia but critical swallowing dysfunction is often already present in earlier disease stages [2, 3]. One of its main problems in the clinical setting consists in insufficient methods for an early detection of pharyngeal dysphagia in affected PD patients. Only 20 to 40% of these patients are aware of their swallowing impairment and less than 10% report symptoms spontaneously to their physicians or speech and language therapists (SLTs), respectively [4, 5]. Whereas oral dysphagia is able to be detected by clinical examination properly, the presence of pharyngeal dysphagia is more difficult to evaluate, as clinical examination, swallowing questionnaires and water-test-screenings do not provide clear results in regard to sensitivity and specificity [6–8]. However, severe dysphagic impairment like residues or penetration/aspiration events usually occur in the pharyngeal phase of swallowing [1]. Only more elaborate instrumental tools like flexible endoscopic evaluation of swallowing (FEES) or videofluoroscopy (VFSS) can reliably detect pharyngeal dysphagia, but these tools are not available in all occasions and therefore cannot be used as screening methods in all PD patients, either [1].
Hence, clinical predictors indicating a pharyngeal swallowing impairment are needed to identify patients at risk. In former studies, predictors like Hoehn & Yahr stadium 4 or 5, relevant loss of weight, sialorrhea, disease duration, high age, male gender or the presence of dementia had been suggested to indicate the presence of PD-related dysphagia [7,9–13, 7,9–13]. However, in most of these studies the presence of dysphagia was only assessed by subjective questionnaires or simple rating scores like the NMS-quest or the UPDRS II, item 7 [9, 10]. Only in few studies, instrumental tools, such as FEES and VFSS, have been applied [3, 11–13]. In most cases, these studies focused on detection of severe forms of oropharyngeal dysphagia defined by relevant penetration-/aspiration (P/A) events [7, 11]. A systematic study on predictors for impaired pharyngeal swallowing parameters apart from penetration and aspiration, i.e. residue, pharyngeal hyposensibility or mistimed swallowing reflex, has not been performed so far. An identification of robust predictors for pharyngeal dysphagia in earlier stages of PD would be important for clinical practice allowing an earlier treatment before onset of severe aspiration events.
Therefore, the aim of our study was (i) to characterize the occurrence and patterns of pharyngeal dysphagia in a large cohort of PD patients in all stages of the disease, (ii) to identify possible differences between clinical PD subtypes and (iii) to find robust clinical predictors for the presence of pharyngeal dysphagia in PD patients.
MATERIALS AND METHODS
Patients
We performed a detailed video analysis of FEES examinations from our database. A cohort of 200 randomly selected PD patients (137 male, 63 female) with clinically diagnosed PD following the British Brain Bank diagnostic criteria [14] who were examined at the department of neurology at the university hospital of Muenster between 2012 and 2017 were included. Overall, 1051 FEES examinations have been done on PD patients during this time period. In accordance to our local dysphagia guidelines, all PD patients received a clinical dysphagia assessment by a trained speech-language pathologist. In those with suspected pharyngeal dysphagia or in unclear cases, a FEES was conducted by a speech-language pathologist together with a trained neurologist. 73 patients were examined at our medical ward, 127 in our outpatient clinic. Clinical data of all examined patients had been collected as well (see below). Regarding to their clinical feature, patients were divided in three clinical subtypes: patients with mainly bradykinesia (BK), tremor-dominant patients (TD) and patients with early postural instability and gait difficulty (PIGD) [15–17]. Each examination had been performed in context of a routine clinical and endoscopic dysphagia examination in clinical “on”-state by a group of 4–6 well experienced examiners.
Exclusion criteria were the presence of other neurological diseases or other medical conditions causing dysphagia. Patients suffering from atypical or secondary Parkinsonian syndromes were excluded. Age, gender, disease duration, Hoehn & Yahr (H & Y) stage, Levodopa equivalent dose and Unified Parkinson’s Disease Rating Scale (UPDRS) were documented in every patient. Furthermore, we used the UPDRS II, question 7 (0–4 point scale: 0 = normal; 1 = rare choking; 2 = occasional choking, 3 = cost adaption necessary, 4 = tube feeding necessary) to evaluate the subjective presence of dysphagia. Levodopa equivalent dose was determined after an established schema [18]. Safinamide with its anti-glutamatergic and MAO B-inhibitory effect was rated equivalent to amantadine [19]. Data acquisition and analysis were approved by the local ethics committee. Informed consent was not required as the data analysis was based on pre-collected data for clinical reasons.
Flexible endoscopic evaluation of swallowing (FEES)
FEES is considered to be the gold standard for evaluation of oropharyngeal dysphagia in PD1 and was performed in accordance to well-established protocols [20]. In brief, all patients received at least three boli of puree consistency (3×8 ml), blue-dyed liquids (3×5 ml) and soft solid food (white bread, size: 3×3×0.5 cm3), consecutively. For the examination, a flexible rhinolaryngoscope (Olympus ENF-P4) was used. Pharyngeal dysphagia was defined by the presence of delayed/mistimed swallowing reflex (no swallowing reflex was regularly triggered for at least 3 seconds after the bolus had reached the valleculae) [21, 22], pharyngeal hypesthesia, pharyngeal residue, and/or pharyngeal bradykinesia. A specific endoscopic dysphagia score was applied in a slightly modified form that has previously been validated for the use in Parkinsonian syndromes: 0 = no relevant dysphagia, 1 = mild dysphagia (any pharyngeal swallowing abnormalities, but no penetration/aspiration events), 2 = moderate dysphagia (penetration/aspiration events with one consistency), 3 = severe dysphagia (penetration/aspiration events with two or more consistencies) [23, 24]. Pharyngeal residues were defined to be relevant if they filled the pharyngeal cavaties, in particular the valleculae and/or pyriformis for at least 50%. A delayed swallowing reflex was defined as stated above (response latency >3 seconds after bolus reached the valleculae). Following suggestions of Langmore (2007) for FEES in neurodegenerative disorders, we did not score the maximum value of each FEES variable, but the “most typical” value after several swallows to exclude incidental, clinically less important findings [25]. The pattern was defined to be pathological if it was present at least in 3 swallows of one consistency. If only 3 swallows were available, the pathological pattern had to be present in the majority of swallows (at least 2 out of 3). The patients were divided in two groups: Group 1: FEES-score >0 = presence of pharyngeal dysphagia; Group 2: FEES-score 0 = no relevant dysphagia.
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows, Version 22.0. Armonk, NY: IBM Corp. First, a descriptive analysis of the study parameters was performed. Before statistical testing, each continuous variable was tested for normal distribution (“Kolmogorov-Smirnov-test”). Independent t-tests were used to compare the continuous variables (age, disease duration, UPDRS III, Levodopa equivalent dose) of the two study groups (Group 1: FEES-score >0 = presence of pharyngeal dysphagia; Group 2: FEES-score = 0 = no relevant dysphagia). Chi-square-tests were applied to compare the two groups in terms of gender, clinical subtypes, H & Y stage, and UPDRS II, item 7. An ANOVA multivariance analysis and follow-up t-tests were performed to compare the clinical subtypes of PD on continuous measures (age, disease duration, UPDRS III, Levodopa equivalent dose). Chi-square-tests were used to compare the clinical subtypes of PD in terms of gender, H & Y stage, and the UPDRS II, item 7. Bonferroni corrections for multiple testing were applied using SPSS framework. The mean difference was significant at the level of 0.05 for the cumulated values.
A binary logistic regression was performed to identify predictors for the presence of pharyngeal dysphagia. The regression model included five clinically relevant predictors: age, stage, disease duration, Levodopa equivalent dose and UPDRS III. The parameters were tested using a backward stepwise procedure (likelihood ratio). To obtain cut-off values to predict the presence of pharyngeal dysphagia, ROC (Receiver Operator Characteristic) curves were calculated for the parameters remaining in the final regression model. For parameters showing a significant area under the ROC curve, the cut-off value per parameter was defined as the value with the highest Youden-Index ((sensitivity + specificity) –1).
RESULTS
The mean age of the 200 included PD patients was 68.2±9.6 years, the mean H & Y stage 2.8±0.8 (I = 9 [4.5%], II = 63 [31.5%], III = 73 [36.5%], IV = 51 [25.5%], V = 4 [2%]) and the mean disease duration 7.6±5 years. In a first step, we compared clinical data of PD patients showing pharyngeal dysphagia in FEES examination (n = 120; mean FEES-score 1.34±0.59) to patients without any signs of pharyngeal dysphagia in FEES examination (n = 80; FEES-score = 0). Table 1 summarizes the main patient characteristics including p-values after group comparison.
Differences between dysphagic and non-dysphagic PD patients
PD, Parkinson’s disease; H & Y, Hoehn & Yahr; TD, tremor-dominant type; BK, mainly bradykinetic type; PIGD, early postural instability and gait difficulty type; UPDRS, Unified Parkinson’s disease rating scale. *statistically significant at a p-level <0.05.
Following independent t-test, we found statistically significant differences between the dysphagic and the non-dysphagic group with older age (t(198) = –2.5, p < 0.05), longer disease duration (t(198) = –2.3, p < 0.05), higher Levodopa equivalent dose (t(198) = –4.4, p < 0.05), and higher scores for UPDRS III (t(198) = –3.1, p < 0.05) in the dysphagic group. The differentiation between the presence of pharyngeal dysphagia (FEES-score >0: dysphagia, FEES-score = 0: no dysphagia) in comparison to gender difference (χ2(1) = 5.9; p < 0.05), H & Y stage (χ2 (7) = 21.7, p < 0.05), and presence of subjective dysphagia (UPDRS II, item 7 : 0 = no dysphagia, >0 = dysphagia; χ2(4) = 18, p < 0.05) using chi-square comparison showed statistically significant differences with more severe affection of male gender and subjective affection in the dysphagic group as well. No significant group differentiation was found comparing patients with and without dysphagia in PD subtypes.
Performing a PD subtype analysis (Group 1: TD type; Group 2: BK type, Group 3: PIGD type), multivariance ANOVA analysis and follow-up tests showed statistically significant differences for age (p < 0.05) and Levodopa equivalent dose (p < 0.05) between the BK and the PIGD group as well as for H & Y stage (TD vs. PIGD: p < 0.05; BK vs. PIGD: p < 0.05), UPDRS II, item 7 (p < 0.001) and UPDRS III (TD vs. PIGD: p < 0.001; BK vs. PIDG: p < 0.05) between the TD and PIGD and BK and PIGD group with higher values for the PIGD subtype in each case. Neither differences were found for disease duration and gender nor between the BK and TD group, in general.
Analyzing FEES examinations in detail, a FEES score >0 was found in 60% of PD patients. A relevant penetration or aspiration (score > 1 of the penetration/aspiration scale) was present in 15%, delayed swallowing reflex in 38.4% and residues in combination with pharyngeal hypaesthesia in 87.5% of the dysphagic patients. In PIGD type delayed swallowing reflex was found significantly more often than in BK and TK groups (50% of all PIGD type patients). There were significantly lower values in penetration/aspiration events at the BK in comparison to the PIGD type as well (4% vs. 27%), but no differences in comparison to the TD type or in comparison of BK and TD type, respectively. Comparing the different food consistencies more in detail, P/A-events were found to be most present for liquids followed by soft and solid food in all three subtypes. No statistically significant differences were found in the three subtypes regarding residues (see Table 2).
FEES findings of the three PD subtypes
TD, tremor-dominant type; BK, mainly bradykinetic type; PIGD, early postural instability and gait difficulty type. *statistically significant at a p-level <0.05.
In a further step, we performed binary logistic regression analysis to identify independent predictors for the presence of FEES proven pharyngeal dysphagia. The logistic regression model was statistically significant, (χ2(2) = 15.9, p < 0.001). The model explained 10.3% (Nagelkerke R2) of the variance in presence of pharyngeal dysphagia and correctly classified 85.8% of cases. Increasing age and Levodopa equivalent dose were found to be independent predictors for the presence of pharyngeal dysphagia. In a next step, ROC curve analyses were performed for both significant predictors. The area under the curve (AUC) was significant for both parameters, (age: p < 0.05, AUC 0.589 (CI: 0.508–0.670); Levodopa equivalent dose: p < 0.01, 0.631 (CI: 0.551–0.710), although the effect was small.
The Youden Indices indicated that an age above 63.5 years and a Levodopa equivalent dose of more than 475 mg per day are predictors for the presence of pharyngeal dysphagia (see Fig. 1).
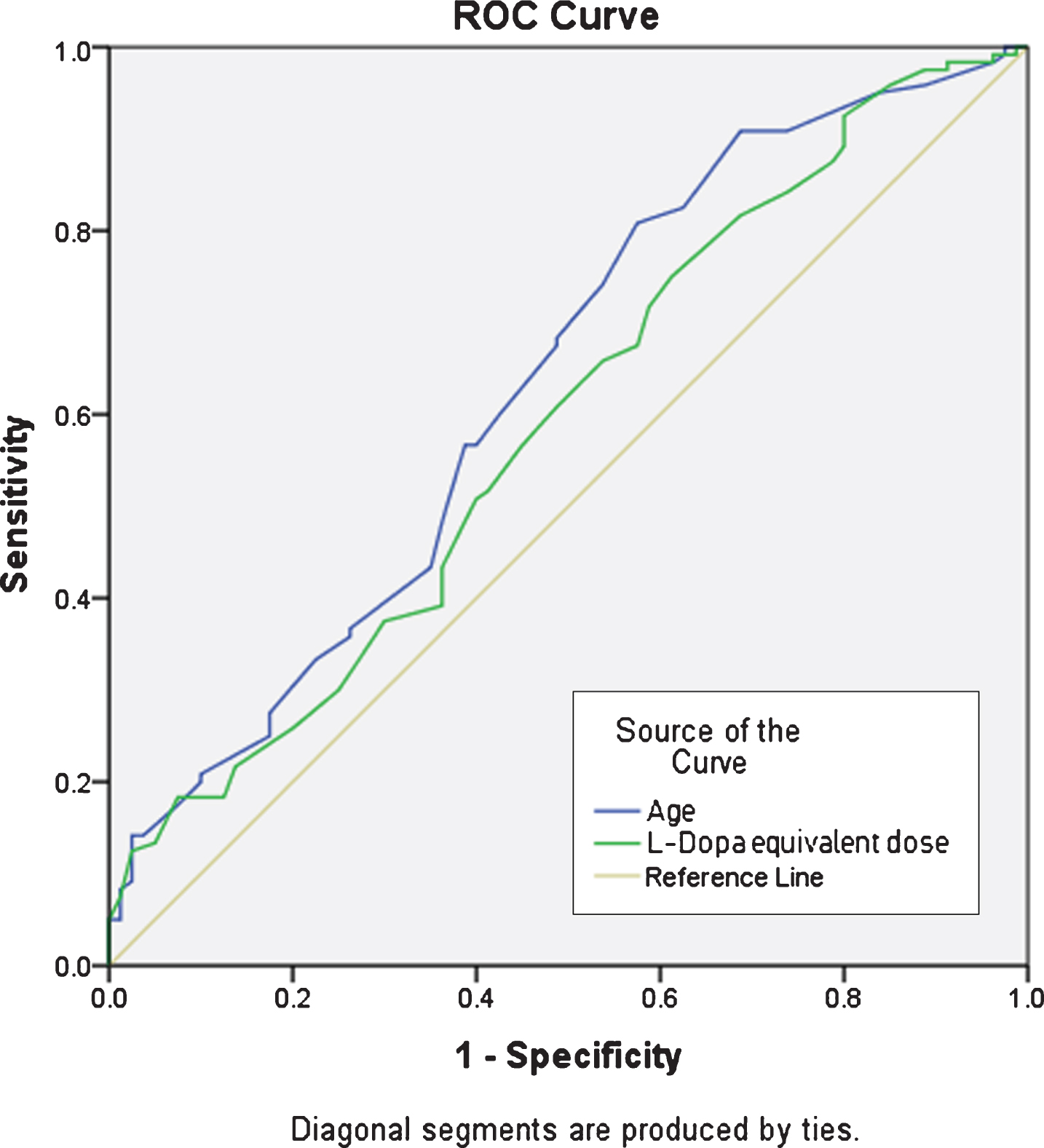
ROC curve analyses for significant predictors age (p < 0.05) and L-Dopa equivalent dose (p < 0.01).
DISCUSSION
In the present study a large cohort of PD patients with different clinical subtypes and in all stages of the disease was assessed in order to describe clinical features which could indicate the presence of pharyngeal dysphagia. Our study analysis showed (i) significant higher values for age, disease duration, H & Y-stage, Levodopa equivalent dose as well as increased scores for UPDRS III and UPDRS II, item 7 in the dysphagic in comparison to the non-dysphagic group. Besides, an increased risk for the presence of pharyngeal dysphagia was shown for male gender. Comparing different clinical subtypes of PD, we found (ii) a higher affection of the PIGD in comparison to the BK and/or the TD subtype with decreased values for age, Levodopa equivalent dose, stadium, UPRDS II, item 7 and UPDRS III. Furthermore, specific FEES findings like delayed swallowing reflex and penetration/aspiration events occurred more often in the PIGD subtype. Finally, in a logistic regression model (iii) higher age (>63.5 y) and Levodopa equivalent dose (>475 mg) were identified to be independent predictors for pharyngeal dysphagia in the examined patients.
Oropharyngeal dysphagia is known to be a frequent and clinically relevant phenomenon in PD. According to recent meta-analyses its prevalence ranges from 11% up to 87% which might be due to differences in measurement tools and heterogenous PD patient populations varying between one-third and three quarters of PD patients in the majority of studies [6, 26]. The prevalence of PD-related dysphagia in our study was only 60% as we focused on the pharyngeal phase of swallowing. If symptoms of oral phase impairment had been included in our study analysis, the prevalence of PD-related dysphagia would have been increased, however. Furthermore, in comparison to other studies dealing with this topic we applied a much stricter definition of relevant pharyngeal dysphagia (see Methods section) [3].
The pathophysiology underlying PD-related dysphagia has not been understood completely so far but dopaminergic as well as non-dopaminergic mechanisms may be involved in its development [1]. As swallowing function of some PD patients improves after adjusting levodopa medication, nigrostriatal dopamine deficits in the supramedullary swallowing system might lead to akinesia and rigidity of the pharyngeal muscles [24, 28]. Non-dopaminergic effects in brainstem and cortical areas caused by alpha-synuclein-pathology according to Braak staging may play an important role in the development of swallowing dysfunction in PD as well [29]. There is also growing evidence for an involvement of peripheral mechanisms suggesting an alpha-synuclein presence in peripheral motor as well as sensory nerves which innervate pharyngeal muscles with greater affections in dysphagic in comparison to non-dysphagic PD patients [30, 31]. With a prevalence of 87.5% in the dysphagic group pharyngeal residues have been by far the most common finding in FEES of pharyngeal dysphagia in our cohort which agrees with former studies [1, 32]. Pharyngeal residues in PD may result from pharyngeal bradykinesia in association with a reduced hyoid elevation as well as pharyngeal sensory deficits [13, 32–34].
Looking at clinical subtypes of PD, the subgroup analysis of our PD patients seemed to indicate a different presentation of pharyngeal dysphagia of the PIGD in contrast to the TD and BK subtype. It was previously shown that the PIGD subtype is associated with higher mortality [16]. As severe pneumonia is known to be the main cause of death in PD patients [2], a clinical relationship to the presence of severe dysphagia in comparison to the other subtypes can be supposed [16]. Furthermore, the tremor subtype is said to be associated with milder motor deterioration and striatal degeneration which could play a role in the development of dysphagia severity as well [16, 35]. Mao and colleagues recently explored the relationship between PD motor subtypes and drooling finding a higher risk in non-TD subtypes with higher daily Levodopa equivalent dose, H & Y –stage, UPDRS scores and male gender preference [36]. In our study, the prevalence of mistimed swallowing reflex was significantly increased in the PIGD subtype (50% vs. 21% and 19% of the PIGD in comparison to the TD and BK subtype) as well as the prevalence of P/A events (27% vs. 4% and 12% of the BK and TD subtype). These findings of a higher dysphagic affection of the PIGD subtype might be a result from poor coordination of the oropharyngeal muscles with rigidity and prolonged oral transit time [37].
Reviewing the literature, several clinical factors have been suggested to indicate the presence of PD-related dysphagia. Lam and colleagues found a combination of higher Hoehn & Yahr stadium, relevant loss of weight (low Body Mass Index) and difficulty of keeping food or drink in the mouth to predict severe oropharyngeal dysphagia [11]. Cereda and colleagues identified older age, male gender, a longer disease duration and the presence of dementia to be clinical predictors for PD-related dysphagia [10]. Additionally, Nienstedt and colleagues found higher age, male gender and affirmed subjective aspiration signs to be predictors at risk [7]. Especially the stronger affection of male gender, which was found in several studies (see above) is an interesting finding, as in our study population, male gender was more represented in the dysphagic group as well. Reviewing the literature, gender differences of pathophysiological features in PD patients showed a greater impairment of male gender and a higher mortality during the cause of the disease as well [38–40]. These findings might be due to higher physiological striatal dopamine levels caused by oestrogen activity leading to a lower incidence and higher age at disease onset in female gender [38]. Even in imaging studies, striatal dopamine transporter activity-related gender differences could be found in striatal subregions, especially in the antero-dorsal striatum [39]. Therefore, male gender seems to be a risk factor for the development of pharyngeal dysphagia in PD patients as well.
In our study, we identified older age (>63.5 y) to be a clear and independent predictor for pharyngeal dysphagia in PD patients. An important factor explaining this observation may be the co-occurrence of presbyphagia [41]. The term “presbyphagia” describes age-related changes of swallowing including prolonged oropharyngeal phase, decreased posterior and anterior lingual movement, delayed pharyngeal triggering and initiation of the swallow reflex, smaller swallow volume, reduced hyolaryngeal movement, delayed opening of the upper esophageal sphincter, increased residual matter in the pharynx and a higher proportion of silent aspiration and penetration [41, 42]. Sarcopenia, the degenerative loss of skeletal muscle mass, strength and quality is an independent risk factor for swallowing dysfunction in older individuals [36] and leads to presbyphagia which contributes to a diminished functional reserve with a higher susceptibility for dysphagia [43]. Further aspects like a decrement in taste and smell acuity, dental problems, decrease of tissue elasticity, cervical spine alterations and hyposalivation are involved as well [37, 45]. In combination with a neurodegenerative disorder like PD, a clinically relevant age-related dysphagia (so called “secondary presbyphagia”) might occur in elderly PD patients as the ability to compensate age-related swallowing dysfunction is getting lost [44]. However, detailed signs for presbyphagia have not been evaluated in this study. A larger, prospective study with an age-matched control group would be helpful for a clearer differentiation of influences of age. However, as numerous studies have already shown, relevant PD-related dysphagia is also present in younger age and earlier stages of the disease and therefore cannot only be reduced on age-related influences [1, 6]. Nonetheless, risk increases during disease progression and is correlated to aging effects. Next to higher age, in our study a higher Levodopa equivalent dose (>475 mg) also was identified to be an independent predictor for pharyngeal dysphagia. We suggest that the Levodopa equivalent dose might be a more sensitive marker of disease progression in our study setting as compared to disease duration and H & Y - stage. This hypothesis is supported by similar findings in a large PD population study [46]. In the PD MED-study, the patient collective showed increasing Levodopa equivalent doses during the disease progression with an average Levodopa equivalent dose between 636 mg to 768 mg (depending on randomization option) after seven years. These findings are congruent to the medical dose findings in our study with an average Levodopa equivalent dose of 713 mg after an average disease duration of 7.6 years [46].
Study limitations
As our study design was based on a retrospective analysis of pre-collected data, prospective data about non-selected PD patients are not included. Nevertheless, all FEES examinations had been performed by a group of six well-experienced FEES-investigators at our medical ward or outpatient clinic under assimilable circumstances following the same established protocol. Although a slight bias for performing FEES more frequently in PD patients with subjective dysphagia symptoms cannot be excluded completely, in our clinic FEES was performed in the majority of all treated PD patients after established local guidelines (see Methods section). Therefore and because of similar data found in other studies, we feel that our results give a realistic sample about the occurrence of pharyngeal dysphagia in a large cohort of PD patients. In addition, as PD diagnosis in the examined patients only based on clinical diagnostic criteria, a future development of atypical parkinsonism in single patients cannot be completely excluded. Especially early occurrence of swallowing impairment in the PIGD subtypes could have been a hint of underlying atypical parkinsonism with similar clinical presentation in the beginning of the disease [47]. Ten steps to identify atypical parkinsonism. JNNP 2006 77 (12 : 1367–1369). Though, the mean disease duration of our PIGD patients was 9.45 y±5.98 y and no other clinical red flags for atypical parkinsonism were reported. Therefore, we feel quite sure the correct diagnosis was set in most of these patients. Concerning statistical analysis of subtype comparison, the PIGD-subtype only includes 22 patients and some reported effects were only small. However, we feel confident that our study results are relevant and worth reporting given their potential clinical implications. Though, to confirm the predictive value of our study results a further, prospective study including an age-matched control group to exclude co-existing influences like primary presbyphagia is needed.
In summary, clinical predictors may be helpful to better identify PD patients with an increased risk of pharyngeal dysphagia. Especially patients with higher age, an increased Levodopa equivalent dose or those suffering from the PIGD subtype should be considered for further instrumental examinations in order to initiate early treatment which might prevent from severe dysphagia complications.
CONFLICT OF INTEREST
Inga Claus has received honoraria from Abbvie. Paul Muhle has received honoraria from Abbvie, Fresenius Kabi, Nutricia. Judith Suttrup has no conflict of interest to report. Bendix Labeit has received honoraria from Springer Medizin Verlag GmbH. Sonja Suntrup-Krueger has received research grants from the German Research Foundation (Deutsche Forschungsgemeinschaft, DFG) and the Else Kröner-Fresenius-Stiftung. Rainer Dziewas has received honoraria from BMS, Daiichi Sankyo, Pfizer, Boehringer Ingelheim, Nutricia, Nestle, Olympus. Tobias Warnecke has received honoraria from BIAL, Abbvie, Desitin, Pfitzer and Licher, consultancies from Phagenesis and funding from Abbvie.
Footnotes
ACKNOWLEDGMENTS
This study was partly supported by Deutsche Parkinson Vereinigung e.V. (dPV) (#81). It was also supported by the Open Access Publication Fund of the University of Muenster.
