Abstract
BACKGROUND:
Urinary tract infections (UTIs) and antibiotic overexposure are common neonatal problems. Recently, evidence has emerged that a standardized approach to neonatal UTI can reduce unnecessary diagnosis without complications. This quality improvement project aimed to achieve those goals in our neonatal intensive care unit (NICU).
METHODS:
A UTI diagnosis guideline was adapted from the literature with the goal of maximizing the proportion of diagnosed UTIs that conform to accepted diagnostic criteria: >10,000 CFU/mL of one organism with pyuria, or >50,000 CFU/mL of one organism regardless of pyuria. The guideline was implemented in a level III NICU. Adherence, results, and complications were monitored for 12 months.
RESULTS:
Guideline adherence after implementation was favorable, as evidenced by increased adoption of urinalysis with microscopy. There was a non-significant increase in diagnostic adherence to criteria, 87% to 93%, and non-significant decrease in the rate of UTI diagnosis, 39% to 36%. Complications were not significantly changed. Most UTIs were due to gram-negative rods, especially
CONCLUSIONS:
A guideline for diagnosing UTIs can safely increase uniformity. In contrast to previously published results, no significant changes were seen in the rate of UTI or the proportion of diagnoses conforming to the criteria for UTI. It is likely that guideline effects depend on the established practices of NICU providers. Additionally, a consistent pattern was seen in which organisms were more likely to be judged the source of true UTIs or contaminated cultures, meriting further study.
Introduction
Urinary tract infections (UTIs) are the most common bacterial infection of neonates. The estimated prevalence varies by study, with a range of 4.6% to 13.6% cited in a recent review [1]. Extremely low birthweight infants are at particularly high risk [2]. Neonatal UTIs are a source of significant morbidity and potentially even mortality. Reported rates of concomitant bacteremia vary from 5% to 30% or higher; young age and vesicoureteral reflux are risk factors for bacteremia [3]. Delay in treatment of UTI may increase the risk of renal scarring [4], and hypertension looms as a long-term concern. On the other hand, there is evidence that unwarranted antibiotic exposure in premature infants increases the risk of serious conditions including late-onset sepsis, necrotizing enterocolitis and death. Concerns about the risks of antibiotic overuse may increase even further as the impact of microbiome disruption on chronic disease is investigated [5]. The appeal of tools to diagnose UTIs sensitively and specifically is obvious.
Some variability in estimates of UTI rates can be attributed to using different criteria for diagnosis. The most recent American Academy of Pediatrics (AAP) clinical practice guideline for the diagnosis of UTI in febrile infants and young children recommends a cutoff of 50,000 colony-forming units per milliliter (CFU/mL) of a single organism in a culture obtained by transurethral catheterization, but also cites an emerging opinion that a cutoff of 10,000 CFU/mL may be appropriate in the setting of supporting information such as symptoms of infection and the presence of pyuria on urinalysis (UA) [6]. A lower CFU/mL threshold for samples obtained by suprapubic aspiration is also discussed. This guideline specifically concerns febrile patients 2–24 months old, circumstances that are not likely to apply in most UTIs diagnosed in the NICU, and its parameters are not universally applied in that environment. Uncertainty about appropriate diagnostic criteria and concern about the morbidity that could arise from untreated UTI leads some providers to treat more aggressively than the AAP guidelines would suggest [7, 8].
Historically, UA has been considered to be of limited utility in diagnosing neonatal UTI. Recently, studies have given cause for more optimism about the predictive utility of pyuria in neonates, with a sensitivity of pyuria for predicting UTI at 87% or higher and comparable specificity [9, 10]. A recent meta-analysis of reports of UTI in jaundiced infants found that definitions of UTI varied widely, with only a minority of studies requiring pyuria to diagnose UTI. The authors of that analysis reported that the overall reported UTI prevalence would drop by more than half if a requirement for pyuria were consistently applied to the definition of UTI [11].
In our NICU, prior to this project, there was no practice guideline for UTI diagnosis and cultures were interpreted by individual providers based on their judgement. UA was sent with only a minority of cultures and thus pyuria was usually not used to diagnose UTI. This quality improvement project aimed to decrease provider variability in diagnosis of UTI and to increase appropriateness of antibiotic use by aligning UTI diagnoses with commonly used criteria,
Methods
Setting
The project took place at a 54 bed Level III NICU, the highest level defined by the Illinois Department of Public Health. The unit admits approximately 500–600 infants annually, including both those inborn and those transferred from referring hospitals. Patients include extremely premature infants and infants with congenital anomalies requiring surgery.
Subjects
The urine culture results for all infants admitted from January 2020 through November 2022 were considered. Cultures drawn from January 2020 through November 2021 were considered the pre-intervention baseline. The guideline was implemented on December 1, 2021, and cultures drawn thereafter were considered the post-intervention group. Characteristics of the patients in both groups are summarized in Table 1.
Characteristics of pre- and post-intervention patients in whom urine cultures were sent
Characteristics of pre- and post-intervention patients in whom urine cultures were sent
A guideline for the diagnosis of UTI was adapted from the literature [8]. The guideline was reviewed by local specialists in neonatology and infectious disease. The implemented guideline provides recommendations for interpreting urine culture results based on collection method, the number of different organisms growing, the CFU/mL of the most prevalent organism, and the presence or absence of pyuria (Fig. 1). Modifications from the previously published guideline were made for clarity and flexibility; some information was incorporated into the flowsheet itself rather than footnotes, and further details were added concerning how to manage cultures growing 10,000–50,000 CFU/mL of bacteria without pyuria. The imaging recommendations included in the previous study were not included in our institution’s guideline.
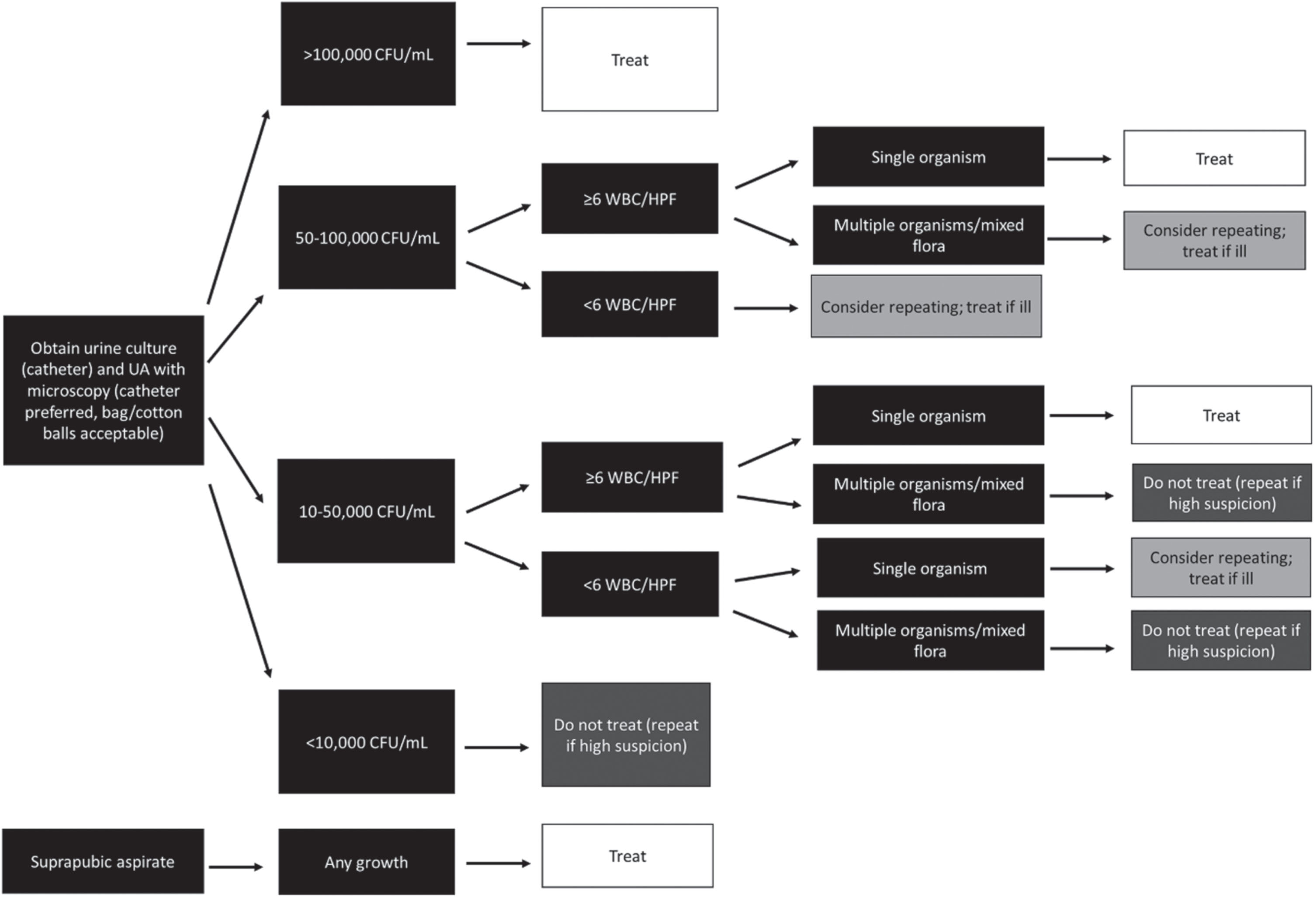
Guideline adapted for the interpretation of urine culture and UA results in the NICU. Adapted from reference [8] with modifications.
The implemented changes were disseminated through both medical and nursing leadership, and the guideline was posted in the NICU. The orders placed for patients with suspected UTIs were monitored by the personnel involved in this project. Quantitative analysis of the impact of the guideline on UTI diagnosis rates and complications began approximately six months after the beginning of guideline implementation in December 2021.
Electronic charts were reviewed for all infants admitted to the NICU between January 2020 and November 2022 for whom a urine culture was sent. Data collected included basic demographic information, urine culture results, associated clinical symptoms, the results of any associated UA or cultures of the blood or cerebrospinal fluid, whether a UTI was diagnosed, treatment prescribed and its duration, whether the infectious disease service was involved in management, renal ultrasound and voiding cystourethrogram results, and any associated complications during the admission.
Complications were assessed in patients with untreated, positive cultures. Short-term complications included subsequent UTI, blood or CSF infection, necrotizing enterocolitis or increase in respiratory support (initiation of noninvasive support or escalation from noninvasive to invasive support) in the 14 days after urine culture collection; these complications were not considered if there was an intervening negative urine culture or urine culture growing a different organism. Other complications tracked were the development of renal scarring, hypertension, or death before discharge.
Because cultures were sometimes repeated in quick succession during the same episode of illness, when cultures were sent within 72 hours, they were analyzed as a single episode and the appropriateness of the ultimate decision to diagnose or not diagnose a UTI was based on the results of the last culture sent, if antibiotics had not already been started by then.
Data were stored in an anonymized form in REDCap [12, 13]. This study includes descriptive data analysis, utilizing median with interquartile range (IQR) for continuous variables and percentages for dichotomous values. Statistical comparisons used the Wilcoxon rank sum test for continuous data and Pearson’s chi-squared testing with Yates’ continuity correction for dichotomous variables. The R computing environment (version 4.3.0; R Core Team, Vienna, Austria) running on Windows was employed for statistical analyses [14]. Multiple hypothesis correction was not used.
Ethical considerations
The local Institutional Review Board (IRB) reviewed the project proposal and determined it did not constitute human subjects research subject to IRB oversight. Instead, the project was managed as quality improvement and overseen by the NICU administration. Randomization and control groups were not used; instead, the changes were implemented in the entire NICU and outcome measures and complications over time were monitored closely.
Results
Patient demographics
Demographic information was compared for patients for whom urine cultures were sent in the pre- and post-guideline implementation eras. This included 104 patients in whom cultures were sent prior to guideline implementation, from January 2020 to November 2021, and 87 in whom cultures were sent after guideline implementation, from December 2021 to November 2022. A total of four patients had cultures sent during both time periods; their statistics are included for both groups. The pre- and post-guideline implementation group did not vary significantly by gestational age, birth weight, or sex (Table 1).
Outcome measures
A total of 224 cultures over 200 episodes (defined as the set of any cultures repeated within 72 hours for the same patient) were sent in the pre-guideline implementation period. 165 cultures in 153 episodes were sent in the post-guideline period. In most cases, cultures grew no bacteria or only mixed flora and were not considered further. In the pre-guideline period, of the 56 episodes in which an identifiable organism grew, 22 UTIs were diagnosed (39% of culture-positive episodes). In the post-guideline period, UTIs were diagnosed in 15 of 42 culture-positive episodes (36%). Treatment decisions made by the treating physician were retrospectively compared to the new UTI criteria detailed above. Based on results of the last culture in an episode, 50 of 56 diagnostic decisions in the pre-guideline implementation group met criteria (89%), compared to 40 of 42 decisions (95%) in the post-guideline implementation period (Fig. 2). There was no significant difference between the pre- and post-guideline periods in the rate at which UTIs were diagnosed, whether the treatment decisions accorded with UTI criteria, or the rate at which the pediatric infectious disease service was consulted in episodes involving a positive urine culture. However, shorter antibiotic treatment courses for UTIs became more common, with courses less than 10 days in duration increasing from 0% to 47% of diagnosed UTIs (Table 2).
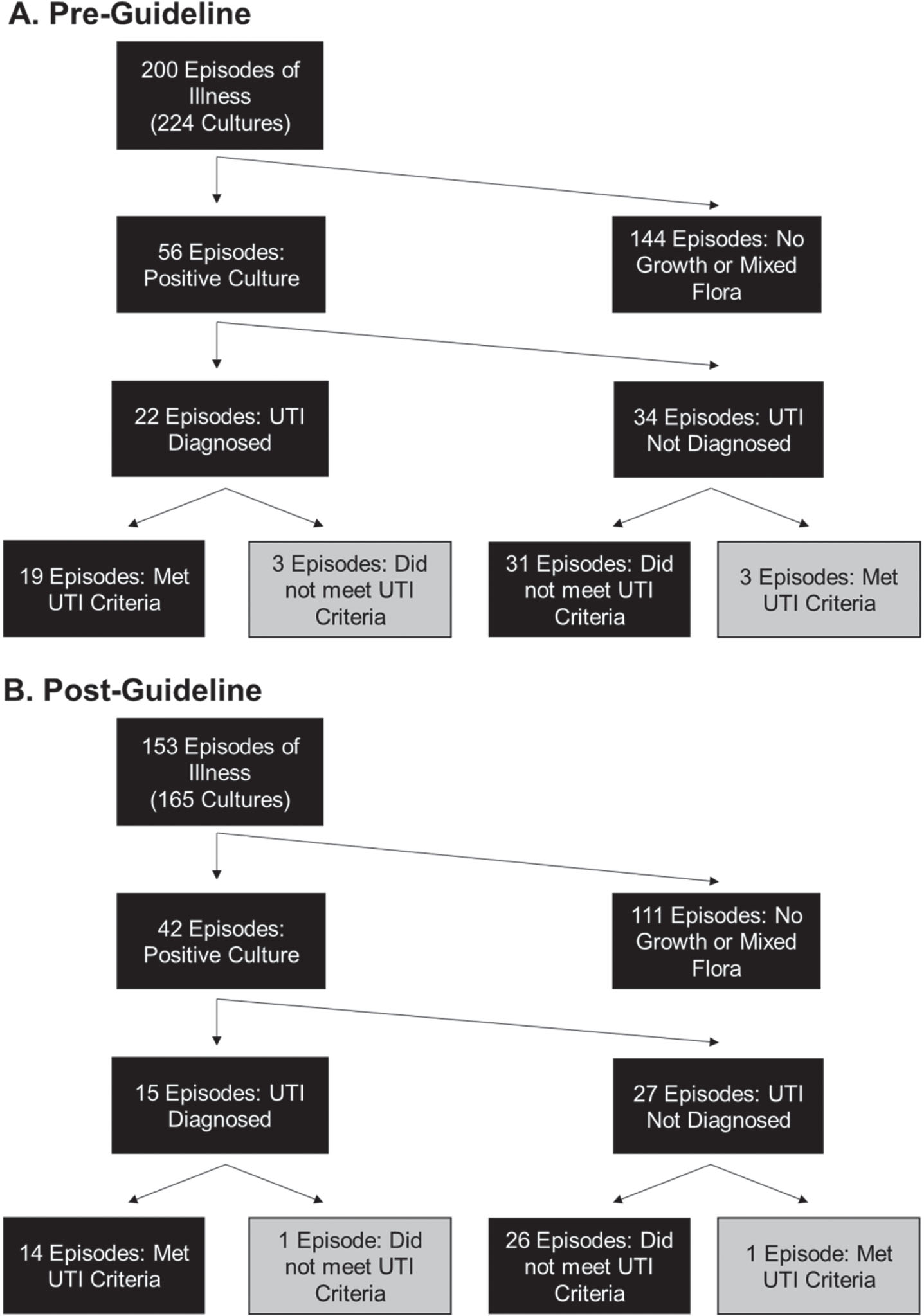
Interpretation of urine cultures before (A) and after (B) guideline implementation. The UTI criteria considered when evaluating diagnostic decisions after the fact were either >50,000 CF/mL of a single organism, or >10,000 CFU/mL in the presence of pyuria (≥6 WBC/HPF); otherwise, criteria were not met.
Summary of diagnostic and treatment decisions in patients with positive urine cultures
a: Significant by Pearson’s Chi-squared test with Yates’ continuity correction.
UA with microscopy, recommended by the guideline, was only intermittently used to make treatment decisions in the pre-intervention period. The UA utilization rate increased from 38% to 78% of cultures (Fig. 3B). The rate began increasing from its previous baseline in the six months prior to formal guideline implementation, while these changes were only being discussed, following the publication of reference [8]. We also attempted to track guideline adherence by assessing whether the response to each culture (treated, did not treat, or repeated) adhered to the guideline shown in Fig. 1. Bearing in mind that the guideline allows room for provider discretion, with recommendations depending on the degree of clinical suspicion, the response to most cultures both before and after guideline implementation could be considered to at least plausibly accord to guideline recommendations. Overall, this measure of guideline adherence increased from 92% to 97% (Fig. 3A). For purposes of this analysis, documentation of patient symptoms in the chart was considered to correspond to the patient illness referred to in the guideline.
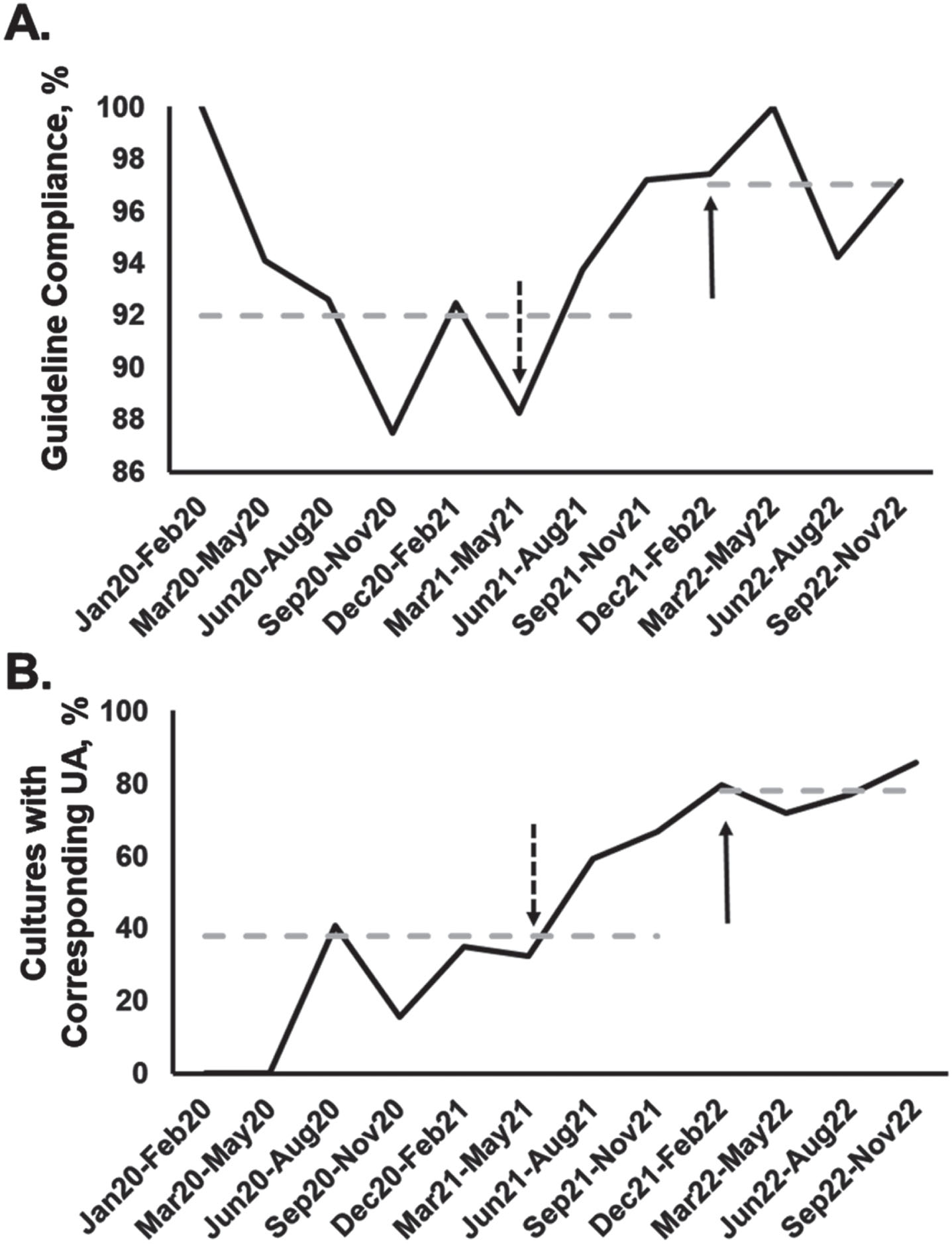
Process measures. (A) Rate at which the response to a urine culture (treating, not treating, or repeating) was compliant with the guideline shown in Fig. 1. (B) Rate at which a UA with microscopy was sent along with urine cultures. In both panels, the dashed arrow shows the point at which reference [8] was published, May 2021, and the solid arrow shows the point at which the guideline was implemented in our NICU, December 2021. Dashed horizontal lines show average rates in the pre- and post-guideline periods. Cultures are grouped in three-month intervals, except for the initial January –February 2020 interval.
The majority of diagnosed UTIs were diagnosed based on cultures growing >50,000 CFU/mL of bacteria (Fig. 4A). As discussed above, the rate of utilization of UA with microscopy increased significantly after guideline implementation, and this is reflected in the facts that WBCs were measured in every diagnosed UTI in the post-guideline period, and pyuria was present in most cases (Fig. 4B).
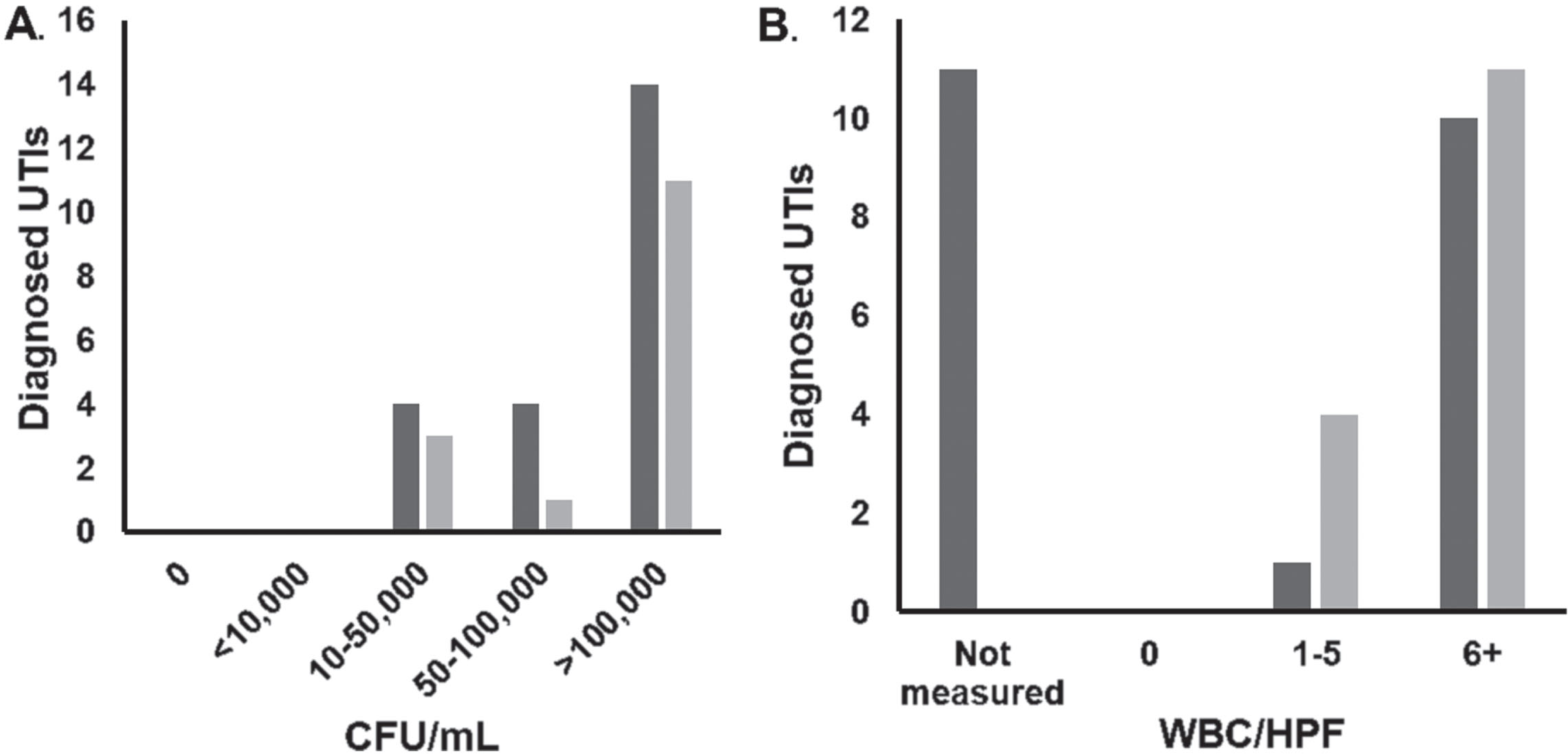
Characteristics of diagnosed UTIs in pre- (dark grey) and post- (light grey) guideline periods. (A) CFU/mL of most prevalent organism. (B) Results for WBC/HPF in UA microscopy corresponding to diagnosed UTIs.
Gram-negative rods, especially
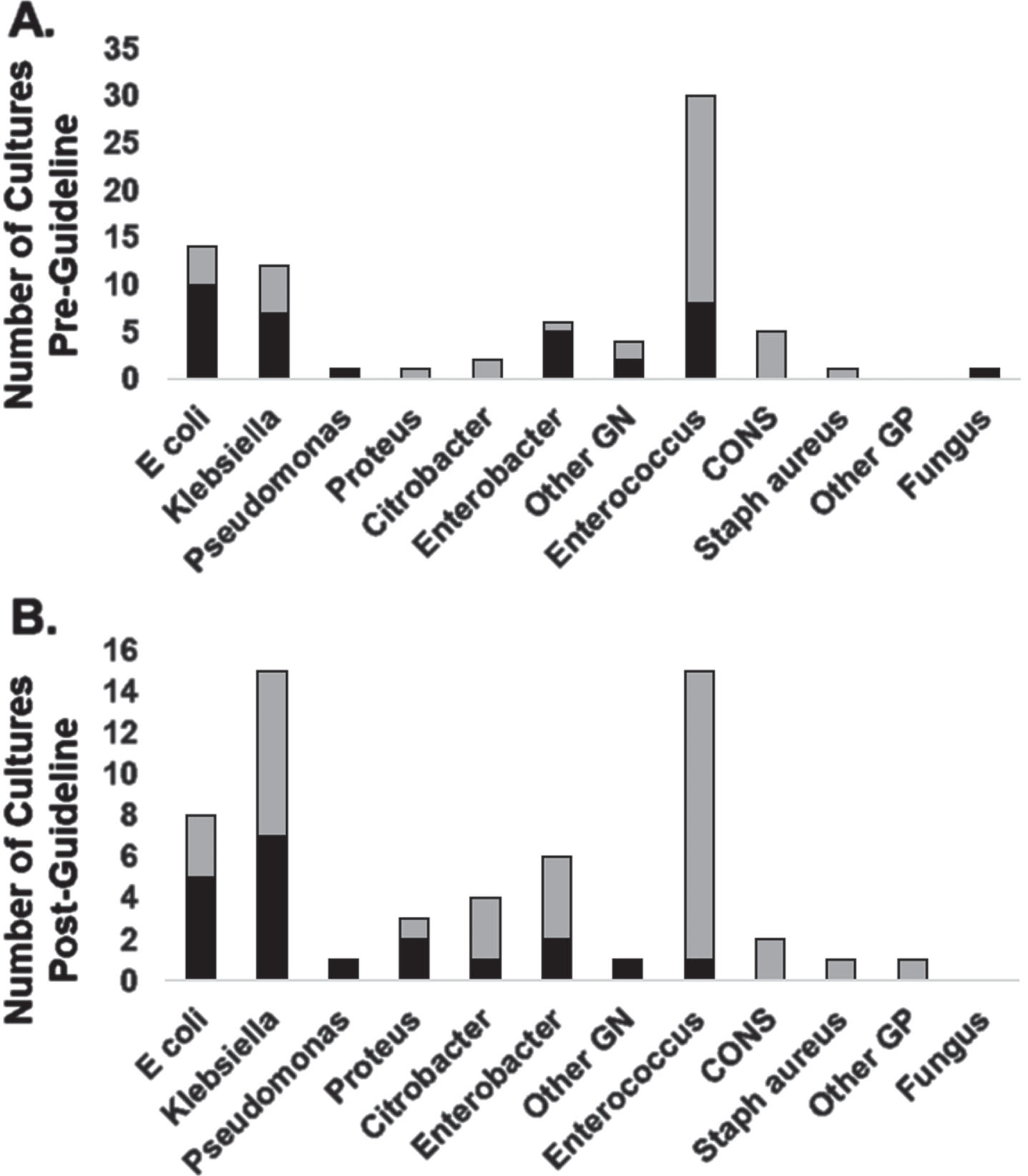
Organisms which grew from urine cultures drawn in the pre-guideline (A) and post-guideline (B) periods. Black bars represent diagnosed UTIs, and stacked light grey bars, positive cultures which were not diagnosed as UTIs. GN: Gram negative, CONS: Coagulase-negative Staphylococcus, GP: Gram positive.
No complications were identified in the pre-guideline period. Two possible complications were seen in the post-guideline period, which may be coincidental: One patient had an increase in respiratory requirements for an unknown reason after two urine cultures grew <10,000 CFU/mL of
Discussion
In this study, a standardized guideline for the diagnosis of UTI was successfully implemented without an increase in complications. After implementation, cultures were almost always interpreted in a manner consistent with the guideline. Certainly, as multiple courses of action were allowed to respond to most culture results (Fig. 1), retrospectively judging failures of adherence was difficult. Instead, the clearest evidence of the guideline impacting practice was seen in the rate at which UA was sent, which doubled in the post-guideline period. Despite this evidence of practice change, however, overall rates of UTI diagnosis did not significantly change, nor did the proportion of diagnosed UTIs that adhered to established criteria for UTI diagnosis.
These results are different from those previously obtained with a very similar guideline on which this one was based [8]. Gorski et al. saw a decrease in the rate of UTI diagnosis (from 68% of positive cultures to 28%) and an increase in those meeting the pediatric UTI definition (from 23% of diagnosed UTIs to 86%). The sample size in our study was slightly larger, with more positive urine cultures and diagnosed UTIs in both the pre- and post-guideline periods. The more important difference between the studies, however, is differing practices in the respective NICUs in the pre-guideline period. A significant portion of the UTIs seen in the baseline period of Gorski et al. were diagnosed based on a culture growing <10,000 CFU/mL of bacteria. All UTIs in our study, even before the intervention, were diagnosed with >10,000 CFU/mL of bacteria or fungus (Fig. 4). The increase in the use of UA with microscopy might have been hypothesized to reduce the rate of UTI diagnosis in cultures with low CFU/mL counts, but relatively few such cultures were seen and they were not treated aggressively. An unanticipated issue complicating analysis was that in our NICU, repeat cultures were often sent when results were difficult to interpret. A total of 24 repeat cultures were sent in the pre-guideline period and 12 in the post-guideline period. In these cases, we chose to determine the appropriateness of diagnosis based on the more recent culture results, unless the repeat culture had been done after initiation of antimicrobial therapy, to monitor response to treatment. Some cultures (three in the pre-guideline period, two in the post-guideline period) that initially did not represent clear UTIs met criteria after being repeated. Overall, a different baseline for provider aggressiveness in responding to positive urine cultures, combined with leeway in the guideline recommendations, likely accounts for different results between these studies. This is a reminder that quality improvement efforts are not necessarily generalizable and depend on institutional context.
One significant change seen over the course of this project was a trend towards shorter antibiotic courses for diagnosed UTIs. Treatment recommendations were not directly addressed on the UTI diagnosis guideline, but guideline implementation and shorter treatment courses both took place during a period of reconsideration of the local philosophy of UTI treatment. A recent review has found similar UTI recurrent risk with short (defined as ≤10 days) and long parenteral treatment courses [15]. Some of the studies considered short, entirely parenteral antibiotic courses, whereas others considered brief parenteral antibiotic courses which then transitioned to enteral antibiotics to complete a longer course.
In this study, there was no significant increase in complications and the clearest material cost, increased UA utilization, is inexpensive and likely outweighed by the changes in antibiotic prescribing practice. Overall, we found implementation of standardized guidelines for UTI diagnosis in the NICU to be safe and feasible, with evidence of sustained compliance after implementation. Only a small and non-significant effect on the rate of UTI diagnosis was seen, which likely relates to less aggressive treatment decisions in the pre-intervention baseline in our NICU. A simple change to the guideline would be to make firmer recommendations on when to repeat cultures, one effect of which would make monitoring of adherence easier. More ambitious changes could include recommendations for antibiotic duration and route, to reduce unnecessary antibiotic exposure. Similar guidelines could be used in other pediatric contexts in which patients cannot report symptoms and diagnosis may be more dependent on laboratory findings.
Lastly, the organisms growing in these urine cultures are noteworthy.
Footnotes
Acknowledgments
Dr. Ronda Oram, Dr. Jeffrey George, and Kim Vuckovich RN, MSN contributed to the modifications to the guideline and its dissemination to unit personnel.
Disclosure statements
The authors have no financial relationships or conflicts of interest relevant to this article to disclose. This project was conducted without any grant funding.
