Abstract
BACKGROUND:
Improving neonatal abstinence syndrome (NAS) management is an important concern, and objective measures of its physiologic impact remain elusive. We sought to determine whether near-infrared spectroscopy (NIRS)-derived tissue oxygenation (rSO2) and fractional tissue oxygen extraction (FTOE) demonstrated physiologically plausible changes correlating with standard NAS scoring.
METHODS:
Thirty subjects (mean 39 weeks’ GA and 3 127 g BW) underwent cerebral and peripheral muscle NIRS monitoring on Days of Life (DOL) Three, Five, and Seven. We examined correlations between NAS scores and FTOE and assessed the impact of non-pharmacologic swaddling and cuddling.
RESULTS:
No statistically significant correlations between NAS scores and FTOE were observed; however, plausible trends were demonstrated between NAS scores and cerebral measurements. Buprenorphine-exposed babies (57%) showed significantly lower FTOE when swaddled (DOL7).
CONCLUSIONS:
Tissue oxygenation monitoring demonstrates potential to provide objective, clinically relevant physiologic information on infants at risk for NAS. Further study is required to determine whether NIRS-derived measures could assist in individualizing NAS care.
Keywords
Introduction
Given the ongoing nationwide opioid epidemic, neonatal abstinence syndrome (NAS) has become a major issue in newborn care. Sanlorenzo et al. reported an incidence of NAS in the United States of 5.63 per 1 000 hospital births in 2012, a nearly five-fold increase from 2000 [1]. The number of infants requiring special care nursery or neonatal intensive care unit (NICU) treatment for NAS increased approximately five-fold during this period [1, 2]. Between 2009 and 2012, cumulative hospital charges attributed to NAS climbed from $732 million to $1.5 billion [3].
NAS causes multisystem disturbances affecting postnatal function and requiring prolonged and expensive hospital stays [4]. Finding better ways to manage this increasingly common condition has become a critical concern. Current management is based on highly subjective NAS scoring systems, such as the modified Finnegan scoring paradigm [5]. An objective, reliable, and trendable estimate of NAS severity in neonates would represent an important advance in management.
Near-infrared spectroscopy (NIRS) is a noninvasive technique to evaluate and trend tissue oxygen saturation at the bedside. Similar to pulse oximetry (SpO2), which reports global anticipated oxygen input to tissues via arterialized vessels, NIRS evaluates regional tissue oxygen saturation (rSO2 = oxygenated hemoglobin/total hemoglobin) arising from all hemoglobin sources within a tissue. NIRS represents a predominantly post-capillary tissue oxygenation measure specific to the anatomic region being studied. It has been used to study cerebral and peripheral oxygen utilization. By simultaneously measuring SpO2 and rSO2, one can derive a trendable balance between oxygen supply (SpO2) and individual tissue oxygen consumption (rSO2) [6]. Commonly, SpO2 and rSO2 are used to calculate regional fractional tissue oxygen extraction (FTOE): FTOE = (SpO2 –rSO2)/SpO2 [7–9].
In a recent review, Andersen et al. (2017) described the use of NIRS in the assessment of tissue “oxygen adequacy” given that states of oxygen deprivation, with associated increases in local tissue oxygen extraction, may now be noninvasively observed and trended at the bedside [8]. It is plausible that NAS results in heightened tissue oxygen utilization such that increased withdrawal symptoms are associated with higher tissue oxygen extraction. Thus, NIRS-based oxygenation data may provide an objective and trendable assessment of NAS severity. We therefore sought to determine whether NIRS-derived cerebral and peripheral muscle rSO2 and FTOE demonstrate physiologically plausible changes in tissue oxygenation behavior that correlate with standard NAS scoring measures. We hypothesized that, among neonates at high risk for NAS, those with elevated modified Finnegan NAS scores (≥eight) would demonstrate ten percent lower cerebral rSO2, indicative of increased tissue oxygen utilization, compared to similarly at-risk neonates without elevated NAS scores. We secondarily hypothesized that a direct correlation would be observed between NAS scores and cerebral and/or peripheral muscle FTOE. We finally hypothesized that nonpharmacological measures, specifically swaddling and cuddling, would be associated with a ten percent increase in rSO2 with a proportional decrease in organ-specific FTOE. This study is an early step towards better individualized care for babies at risk for NAS.
Subjects and methods
Study design
This proof-of-concept, prospective, single-center observational cohort study was approved by the Institutional Review Board Human Subjects Committee of Stony Brook University as a minimal risk study. Written informed consent was obtained for all participants. Cerebral and peripheral muscle NIRS monitoring was performed to identify cerebral and peripheral muscle rSO2 changes taking place with swaddling and holding infants at high risk for NAS. All care decisions were solely made by Well-Baby Nursery attending physicians according to unit-specific standard Well-Baby Nursery guidelines. These included length of observation in the Well-Baby Nursery, feeding goals, and timing of and dosing for initiation of morphine, if applicable.
Patient population/sample size
All subjects were admitted to the Well-Baby Nursery at Stony Brook Children’s. Inclusion criteria encompassed newborns with single or multiple in-utero exposures (any length) to opiates (heroin, methadone, buprenorphine, and/or other opiates); newborns with a current medical plan including NAS scoring; and mother who was 18 years of age or older. Of note, newborns with additional exposures such as cocaine and marijuana were not excluded. Exclusion criteria included medical condition(s) requiring transfer to the NICU and major congenital anomalies.
Power analyses were conducted based on previously reported less than ten percent cerebral NIRS variability in this patient population during the first postnatal week [6]. In addition, a modified Finnegan NAS score of eight was selected as a cut-off for severe NAS symptomatology as initiation of treatment is often considered beyond this threshold [10]. On sample size calculations, considering three monitoring periods per subject, a total of 30 subjects was determined to be sufficient to assess for correlations between modified Finnegan scores and cerebral rSO2. Per our hypothesis, we projected a≥ten percent lower cerebral rSO2 for patients with modified Finnegan scores≥eight compared to those with lower scores with 80% power and a two-tailed alpha of 0.05. Of note, this did not require further subdivision of subjects. Rather, it only required grouping of individual subjects’ NAS scores into≥eight versus lower scores. Based on a review of previous Well-Baby Nursery babies’ scores, we determined that there would be a broad range of NAS scores available for us to analyze. Stony Brook Children’s sees approximately 110 infants at risk of NAS per year. In 2016, for example, 107 infants’ medical plans in the Well-Baby Nursery included NAS scoring. Given these numbers, which have been rising over the past several years, our study sample of 30 subjects was well-suited for our study timeframe.
Eligible subjects were identified through daily screening of the Well-Baby Nursery admission census for history of in-utero drug exposure.
Procedures
All subjects underwent three NIRS data collection periods (Postnatal Days Three, Five, and Seven). Each period consisted of 30 minutes of NIRS monitoring. These time frames were chosen based on their likelihood of demonstrating the onset of NAS-related symptoms. Per Nursery guidelines at the conception of our study, infants on an NAS monitoring protocol typically remained in the hospital for seven days. In the event that a subject was discharged before this point, Day Three and Day Five data alone were utilized. 30-minute NIRS monitoring intervals occurred for the half hour prior to scheduled feedings. Modified Finnegan NAS scores were derived utilizing a broad range of patient information and documented per routine by Well-Baby Nursery nurses after the feed that followed each monitoring period. Well-Baby Nursery physicians, nurse practitioners, and bedside nurses were blinded to NIRS data collection. Within each 30-minute NIRS monitoring interval, 20 minutes took place with the infant in the crib. For the final ten minutes, the infant was swaddled and held by the infant’s mother, if present, or else by the first author in order to minimize excess variables. For the same reason, the first author consented each mother, performed all data collection for each baby, and swaddled each baby. Blinded NIRS data collected during the study were for research purposes only and were not used in any manner for clinical decision-making.
Near-infrared spectroscopy (NIRS) monitoring
NIRS monitoring of rSO2 was performed using the INVOS 5100 C Cerebral/Somatic Oximeter with OxyAlert NIRSensors –Infant Model IS [Neonatal] (Medtronic, Boulder, CO). As in previous studies in infants, cerebral rSO2 was obtained using transverse forehead sensor placement [11]. Peripheral muscle rSO2 was ascertained via sensor placement along the right lateral thigh with the monitoring cord projected distally.
While cerebral NIRS monitoring is well-described in numerous neonatal studies [8, 13], we additionally performed peripheral muscle monitoring for several reasons. First, cerebral perfusion is robustly maintained by redundant vasculature, unlike more peripheral vascular beds. Peripheral muscle monitoring may thus be more likely to demonstrate changes in tissue oxygen utilization than cerebral monitoring. Secondly, peripheral muscle monitoring may be easier to maintain and better tolerated than cerebral monitoring given the sensor location. We thus planned to explore and compare oxygenation behavior between the cerebral and peripheral muscle sites as a proof-of-concept for future utility of peripheral muscle monitoring.
Data collection
Maternal data included maternal age, reported drug exposure, pregnancy complications, and toxicology results. Infant/birth data included gestational age, mode of delivery, birth weight, Apgar scores, resuscitation measures, feeding plan (breastmilk vs. formula and bottle-feeding vs. breastfeeding), infant toxicology results, and infant medication management if applicable. Clinical data included cerebral and peripheral muscle rSO2, vital signs, NAS scores, and timing of swaddling/cuddling during NIRS data collection periods.
After confirmation of cerebral and peripheral muscle rSO2 signal stability and initial reporting of cerebral and peripheral muscle saturations on the INVOS device, NIRS data collection continued throughout each monitoring period with the study member blinded to the readings. Cerebral and peripheral muscle data were captured every five seconds as per the most frequent data acquisition setting possible on the INVOS 5100 C device.
Cerebral and peripheral muscle FTOE were calculated as FTOE = (SpO2 –rSO2) / SpO2 [7–9].
Data management/statistical analysis
As noted above, given repeated modified Finnegan NAS scores≥eight are often used as a threshold to initiate pharmacological treatment, we similarly used this cutoff to assess NIRS data. Thus, subjects were divided into those ever experiencing an NAS score≥eight versus those whose scores remained < eight throughout their Well-Baby Nursery stay. NIRS data were compared between these groups in comprehensive and individual day-by-day analyses using t-tests. Between-group analyses were performed to evaluate correlations between NAS scores and both raw NIRS data (rSO2) and calculated oxygen extraction (FTOE) with t-tests.
For secondary analyses, Pearson correlations were calculated between individual NAS scores and NIRS monitoring (both raw rSO2 and calculated FTOE). Finally, the effects of swaddling and cuddling on both cerebral and peripheral muscle rSO2 and FTOE were assessed in a before-after manner for each NIRS monitoring period again using t-tests based on data distributions.
Results
Subject recruitment and enrollment occurred between October 2018 and December 2019. Thirty neonates with mean gestational age 39 weeks and mean birth weight 3 127 grams were enrolled (Fig. 1). 22 (73%) were exposed to one opiate while eight (27%) had multi-drug exposures. 17 neonates (57%) were exposed to buprenorphine, and seven (23%) received morphine for NAS treatment.
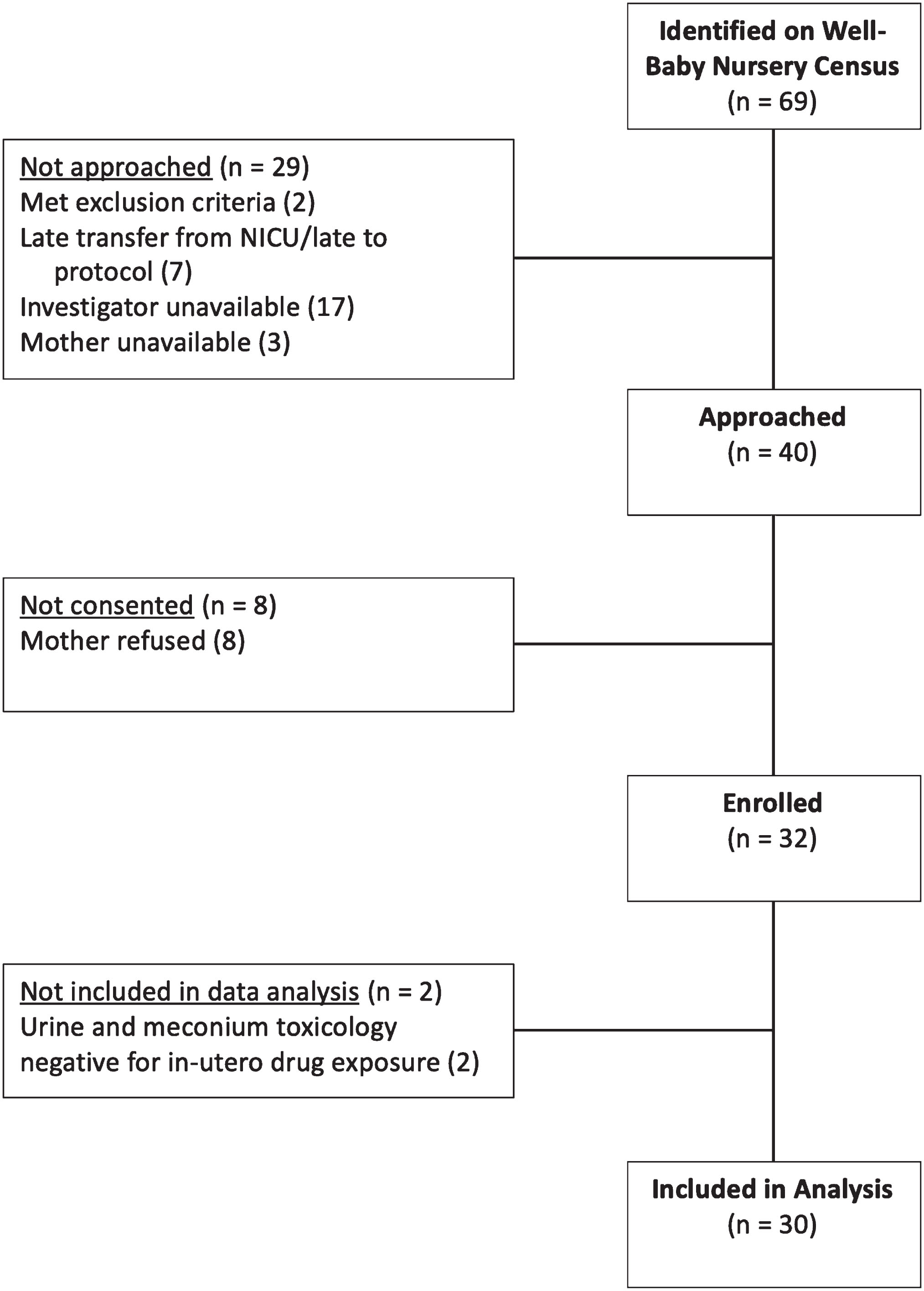
Subject recruitment and enrollment.
Correlations between NAS scores and cerebral FTOE are presented in Table 1 and Fig. 2. The correlations between NAS scores and peripheral muscle FTOE are presented in Table 1. Neither cerebral nor peripheral muscle FTOE demonstrated significant correlation with modified Finnegan scores.
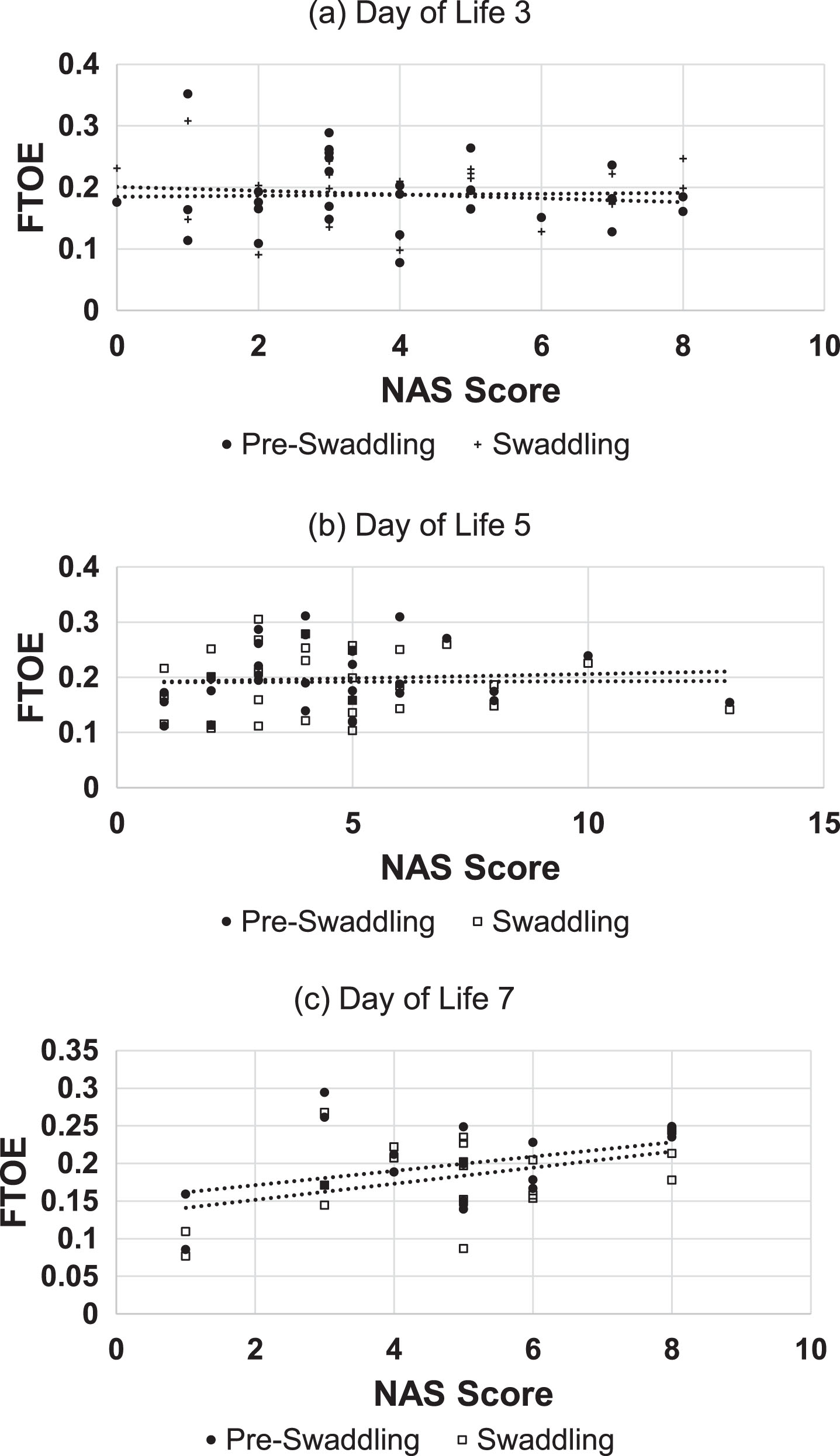
Correlations between neonatal abstinence syndrome (NAS) scores and cerebral fractional tissue oxygen extraction (FTOE) by day of life and swaddling state.
Correlations between neonatal abstinence syndrome (NAS) scores and cerebral and peripheral muscle fractional tissue oxygen extraction (FTOE)
p-value≤0.05 indicates statistical significance.
Comparisons between infants with elevated NAS scores (ever≥eight) versus never elevated NAS scores (< eight) are displayed in Table 2. There were no statistically significant effects of swaddling on cerebral or peripheral FTOE in infants of either group. When we divided the sample by exposure to buprenorphine, however, there was a statistically significant improvement in FTOE on Day Seven of life when babies exposed to buprenorphine were swaddled (p = 0.03) compared to the non-swaddled state. There were no statistically significant differences between groups or between pre-swaddling and swaddling monitoring periods when the babies were subdivided by single- vs. multi-drug exposure or when subdivided by whether morphine treatment was initiated.
Effects of swaddling on cerebral and peripheral muscle fractional tissue oxygen extraction (FTOE)
All data expressed as mean±SD.
Comparisons of simultaneous cerebral and peripheral muscle FTOE measurements among subjects subdivided by NAS score categorization and day of life are displayed in Table 3. There was only one statistically significant result, namely on Day of Life Five, where the average peripheral muscle FTOE in the never elevated NAS score group was significantly higher than that for the ever elevated NAS score group. Comparisons of cerebral NIRS FTOE scores and peripheral muscle FTOE scores between subjects with ever elevated NAS scores and subjects with never elevated NAS scores are shown in Table 4. Simultaneous cerebral and peripheral muscle FTOE measurements were significantly different, taking all data into account, for subjects with ever elevated NAS scores and subjects with never elevated NAS scores as well as specifically on Day of Life Three for subjects with ever elevated NAS scores and on Day of Life Five for subjects with never elevated NAS scores.
Cerebral and peripheral muscle fractional tissue oxygen extraction (FTOE) comparisons between ever-elevated neonatal abstinence syndrome (NAS) score versus never-elevated
All data expressed as mean±SD.
Comparisons between cerebral and peripheral muscle fractional tissue oxygen extraction (FTOE) for ever-elevated neonatal abstinence syndrome (NAS) score and never-elevated NAS score groups
All data expressed as mean±SD.
This study assessed the use of NIRS in the neonatal population at high risk for NAS. On a recent literature search, no studies evaluating tissue oxygenation monitoring in neonates at high risk for NAS were identified. Therefore, our study represents a pilot project to aid in determining whether non-invasive NIRS monitoring could assist clinical decision-making for this patient population.
NAS encompasses physiologic and neurobehavioral signs demonstrated by newborns exposed to opiates in utero. The pathophysiology of NAS is not completely understood, especially given the immaturity of neurologic development and processing in neonates and the intricate pharmacokinetics governing interactions among mother, placenta, and fetus [15]. NAS affects neurologic processing in multiple brain regions. Given the widespread nature of opioid receptors, NAS affects both central and autonomic neurologic function in addition to contributing to respiratory and gastrointestinal malfunction, with clinical effects on neonates’ postnatal adaptation [4]. These realities raise concern for both the short- and long-term effects of fetal opioid exposure and postnatal opioid treatment on the newborn brain [15].
In terms of management, while most literature recommends nonpharmacological care as first-line treatment, studies comparing the efficacy of behavioral interventions are lacking [15]. In some neonates, nonpharmacological management may reduce clinical symptoms and avoid the need for pharmacological therapy [4]. Medications such as morphine are added when supportive care is ineffective, high NAS scores remain, and/or opiate-associated diarrhea and/or vomiting leads to additional clinical concerns. While morphine’s effectiveness is well-established, it contributes to prolonged length of stay, as gradual weaning is required, in addition to uncertain effects on developmental outcomes [15].
Though our correlations between NAS scores and cerebral/peripheral FTOE were nonsignificant, they did plausibly trend in the correct direction for all but one category. These positive correlations were consistent with our hypothesis –higher NAS scores could potentially be associated with elevated cerebral FTOE such that enhanced withdrawal symptomatology could result in increased tissue oxygen utilization. With a larger sample size, it remains feasible that statistically significant correlations between NAS scores and cerebral FTOE would be observed.
Regarding peripheral NIRS monitoring, some correlations between NAS score and peripheral muscle FTOE were observed in the hypothesized direction while others occurred in a seemingly random fashion. These peripheral measures varied in slope and overall did not align with cerebral monitoring results. As discussed earlier, we included peripheral muscle monitoring as an exploratory measure to assess whether peripheral muscle monitoring could perhaps serve as a potentially easier-to-manage cerebral surrogate. Our data suggested that cerebral and peripheral muscle data are not equivalent. We believe this discrepancy can be explained by the body’s response to stress. Cerebral oxygenation should be a more stable signal owing to blood flow redistribution during periods of stress whereas peripheral muscle theoretically should demonstrate a more variable oxygen utilization signal. Our comparisons of simultaneously collected cerebral and peripheral muscle FTOE data furthered this claim –cerebral and peripheral muscle act differently, and one is not a surrogate for the other. Perhaps the higher reactivity of peripheral muscle makes it more susceptible to changes that have not yet impacted the behavior of cerebral oxygen extraction. Furthermore, an anecdotal lesson learned was that the peripheral muscle sensor was actually the less tolerated sensor of the two in general by the babies enrolled in the study. While the babies were generally content with the cerebral sensor across their forehead, more agitation was observed in response to right thigh sensor placement.
While we hypothesized that subjects with elevated NAS scores would have lower rSO2 and therefore increased FTOE, we generally did not see significant FTOE differences when subjects were subdivided by whether they ever had an elevated NAS score. Interestingly, the statistically significant difference on Day of Life Five for peripheral muscle FTOE was the opposite of our expectation –given that this is the only statistically significant result, it appears most likely just due to random chance. It could also be proposed that, by Day of Life Five, the stress of withdrawal in the group of subjects having experienced elevated NAS scores caused a redistribution of blood flow to vital organs such that the peripheral muscle signal became less reactive to moment-to-moment changes in perfusion.
We additionally observed that babies exposed to buprenorphine had statistically lower cerebral FTOE scores when swaddled compared to pre-swaddling on Day of Life Seven. This may be explained by a later peak of withdrawal for babies exposed to buprenorphine compared to those exposed to other opioids [16]. Based on these findings, nonpharmacological measures such as swaddling and cuddling, or simply having parents room-in, for babies exposed to buprenorphine could be an additional area of inquiry, particularly later in these neonates’ clinical NAS observation period [17].
Our study’s particular strengths were its welcome reception by clinical staff members, our high consent rate, and the study team’s consistent approach to data collection. Given our high consent rate, we are confident that our enrolled subjects encompass a representative sample of babies undergoing NAS monitoring in our institution. Further, given the small size of the study team, variations in data collection were minimized, and we are thus confident of the reliability of our findings in this pilot project. A larger sample size is needed, however, to further study the NIRS monitoring approach in managing neonates at risk for NAS.
In terms of limitations, we acknowledge that our results are preliminary and require further study given our investigation took place at a single center with 30 subjects. While the inclusion of 30 subjects was determined after sample size calculations to assess for correlations as detailed earlier, we realize that this study design limits generalizability to the greater population and varying care practices. Furthermore, while each NIRS data collection period included time in the crib as well as time swaddled and held to draw comparisons, our study did not have a control group per se, again limiting our ability to draw conclusions on the impact of swaddling and holding on cerebral and peripheral muscle rSO2. Furthermore, while the timing of 20 minutes in the crib and 10 minutes swaddled and held was kept consistent across NIRS data collection periods and subjects, it is possible that swaddling and holding for the final 10 minutes alone may not have been long enough to fully assess the impact of swaddling and holding on rSO2. We also acknowledge that NAS severity is multifactorial, so there are other potential confounding factors that could have influenced study results and were not specifically assessed.
Tissue oxygenation monitoring demonstrates potential to provide objective and clinically useful physiologic information on infants at elevated risk for NAS. Further studies with larger sample sizes are required to best determine which subjects would benefit most from NIRS monitoring.
Footnotes
Acknowledgments
We wish to express our gratitude to the mothers of our patients for the opportunity to conduct NIRS monitoring on their newborns. Most importantly, we thank them for the chance to get to know them and hear their stories. We also wish to thank the Stony Brook Children’s Well-Baby Nursery nurses, nurse practitioners, and physicians, whose support and help with this project were crucial in its success.
Disclosure statements
Financial disclosure statement
We have no financial interests to disclose.
Human research statement
We affirm that research involving human subjects submitted to the journal was conducted in accordance with the ethical standards of all applicable national and institutional committees and the World Medical Association’s Helsinki Declaration.
Funding disclosure statement
This work was supported by an American Academy of Pediatrics Resident Research Grant (07/31/2018).
