Abstract
BACKGROUND:
Reference guidelines for neonatal conjugated hyperbilirubinemia (cholestasis) management use a uniform approach regardless of gestational age (GA). We hypothesize that the clinical pattern of neonatal cholestasis is tightly related to GA. The aim of this study was to describe the effects of GA on neonatal cholestasis.
METHODS:
A retrospective 4-year cohort study in a 70-bed neonatal care unit. Neonates with conjugated bilirubin≥34.2μmol/L (2 mg/dL) were identified. The incidence, clinical characteristics, etiology, treatment, and prognosis were compared between infants <32 and≥32 weeks GA.
RESULTS:
Overall incidence of cholestasis was 4% (125/3402). It was >5 times higher and the mean duration was >1.5 times longer in neonates <32 weeks GA (10% versus 1.8%, p <0.01 and 49 versus 31 days, p <0.01, respectively). The onset of cholestasis was later in neonates <32 weeks (22 versus 10 days of life, p <0.001). This later onset of cholestasis was associated with parenteral nutrition, whereas the earlier onset was associated with other causes. Treatment using fish oil lipids was more frequently administrated to infants <32 weeks GA, whereas Ursodeoxycholic acid was administrated more frequently in≥32 weeks GA. Cholestasis resolved during hospitalization in 73% of <32 versus 38% in≥32 weeks GA infants (p <0.01).
CONCLUSIONS:
The incidence, clinical presentation, etiology, treatment, and clinical evolution of neonatal cholestasis were all significantly affected by GA. Our results support the use of a GA-oriented approach for the management of neonatal cholestasis.
Keywords
Abbreviations
Canadian Neonatal Network,
Gestational age,
Inter-quartile range,
Intrauterine growth restriction,
Parenteral nutrition,
Standard deviation,
Score of neonatal acute physiology II,
Ursodeoxycholic acid.
Introduction
Neonatal cholestasis is a frequently encountered disorder in neonatal care units. It is caused by defects in bile formation or flow, that are closely related to liver maturity [1]. It results in the accumulation of biliary substances in the blood and conjugated hyperbilirubinemia [2]. The development of neonatal cholestasis can result from a wide variety of insults and medical conditions that are relatively frequent in neonatal care units [1], including prematurity and perinatal stress. When parenteral nutrition (PN) is administered for more than 14 days, up to one in three newborns develops cholestasis [3]. Appropriate and early diagnosis of cholestasis is essential in neonatal care as its timing is closely related to clinical outcomes [4]. In some circumstances, if the cause of cholestasis is not properly diagnosed and treated, it can lead to progressive liver disease, fibrosis, cirrhosis, or even death [5].
Approaches to the evaluation of an infant with conjugated hyperbilirubinemia in reference guidelines and textbooks do not take into consideration the impact of gestational age (GA). Thus, the same approach is uniformly used for all newborns [6]. While it is well documented in the literature that the incidence of neonatal cholestasis is linked to the degree of prematurity [7], the effects of GA on the etiology, clinical presentation, management, and evolution of this pathology were not reported.
Supported by clinical observation, our hypothesis was that GA is a key element affecting the incidence, clinical presentation, etiology, treatment, and evolution of neonatal cholestasis. The goal of this study was to describe the clinical profile and management of cholestasis based on GA stratification in a retrospective large cohort in one of the largest mother-and-child centers in Canada over a 4-year period.
Patients and methods
Study design
A retrospective cohort study of neonates admitted to our 70-bed quaternary neonatal care center in Montreal, Canada, between January 1st, 2014 and December 31st, 2017, was conducted. Routine follow-up at our neurodevelopmental clinic and the division of gastroenterology and hepatology till the age of 5 years permitted the description of the long-term evolution of cholestasis in these patients (January 2019 to December 2022).
Participants
Neonates with a confirmed conjugated bilirubin value≥34.2μmol/L (2 mg/dL) were identified using laboratory records. This value is the conventional cholestasis definition used in neonatal settings [8, 9]. In our unit, any result of conjugated bilirubin value≥34.2μmol/L (2 mg/dL) is controlled within a week. All infants with a confirmed conjugated bilirubin value≥34.2μmol/L (2 mg/dL) on the control test result were diagnosed with neonatal cholestasis. Exceeding this cut-off on two consecutive occasions avoids the inclusion of false positive patients due to laboratory errors or a very transient increase in conjugated bilirubin [10].
Inclusion was based on the development of cholestasis while hospitalized in our department during the study period. Patients who developed cholestasis after the neonatal period (>28 days after a term birth or the equivalent of corrected age) were excluded.
Cholestasis management protocol
The protocol for cholestasis management, implemented in our unit since 2008, included an early conjugated bilirubin measurement during the first 48 hours of life, and a weekly monitoring of biochemical markers (conjugated and total bilirubin, ALT, GGT, and alkaline phosphatase in addition to albumin as needed) in all infants on PN or with abnormal conjugated bilirubin. Oral fat-soluble vitamin supplementation (A, D, E, K) was provided to cholestatic infants not receiving PN.
Other treatments including oral ursodeoxycholic acid (UDCA), intravenous mixed oil lipid emulsion (SMOFlipid®) and fish oil lipid emulsion (Omegaven®), were administered at the discretion of the attending physician.
Data collection
For cholestatic infants, the data collected included demographic and clinical information, complications of prematurity, nutrition (enteral and/or parenteral), UDCA usage, investigations for etiology of cholestasis and its complications, bilirubin evolution up to 4 months after the end of the episode and peak levels of liver function tests during cholestasis.
In order to compare cholestatic infants to non-cholestatic infants admitted to our Division of Neonatology during the same period, we used the hospital’s Canadian Neonatal Network (CNN) database. This national database maintains standardized neonatal data of 29 hospitals and 17 universities across Canada collected through a medical chart review and/or prospective data collection in real-time [11]. To be able to adjust for baseline illness, the Score of Neonatal Acute Physiology II (SNAPII) was collected. The higher the SNAPII score is, the higher the illness severity and mortality risk are [12].
Statistical methods
Data were analyzed using SPSS Statistics 25 (SPSS, Inc., an IBM Company, Chicago, IL). The continuous variables achieving normality were presented as mean and standard deviation (SD), the variables not achieving normality were presented as median and inter-quartile range (IQR), and the proportions were presented as percentages. Normality was tested by the Kolmogorov-Smirnov and the Shapiro-Wilk tests. T-test was used for continuous data achieving normality, Mann-Whitney U test for continuous data not achieving normality, and Chi-square for proportions. Univariate and multivariate logistic regressions were used as appropriate. Statistical significance was set at p <0.05.
Ethical considerations
The Research Ethics Board of the institution’s research center approved this study (project identification number 2019-2258).
Results
Setting
Our 70-bed neonatal care center is part of a mother-and-child quaternary care academic center. During the study period, the Division of Neonatology recorded a mean of 827 (SD 42) admissions per year, of which 67% are inborn, 32% are outborn and transferred by the hospital’s own transport team from other centers, and 1% are readmissions. Of all admissions, 9% were extremely preterm infants (<28 weeks of GA), 13% were very preterm infants (≥28 and <32 weeks GA), 34% were moderate to late preterm infants (≥32 and <37 weeks GA), and 44% were term infants (≥37 weeks GA).
Overall incidence
Over the 4-year period, 125 patients met inclusion criteria and showed an equal sex distribution. The overall incidence of cholestasis was 3.7% of all admissions. GA distribution within the cohort made it possible to visualize two subpopulations (Fig. 1). Based on the World Health Organization classification of preterm births, stratification was made on 32 weeks of GA [13]. Using linear trend estimation, a significant increase in the incidence of cholestasis was found in lower GA of infants admitted to the neonatal unit (p <0.001; Fig. 1).
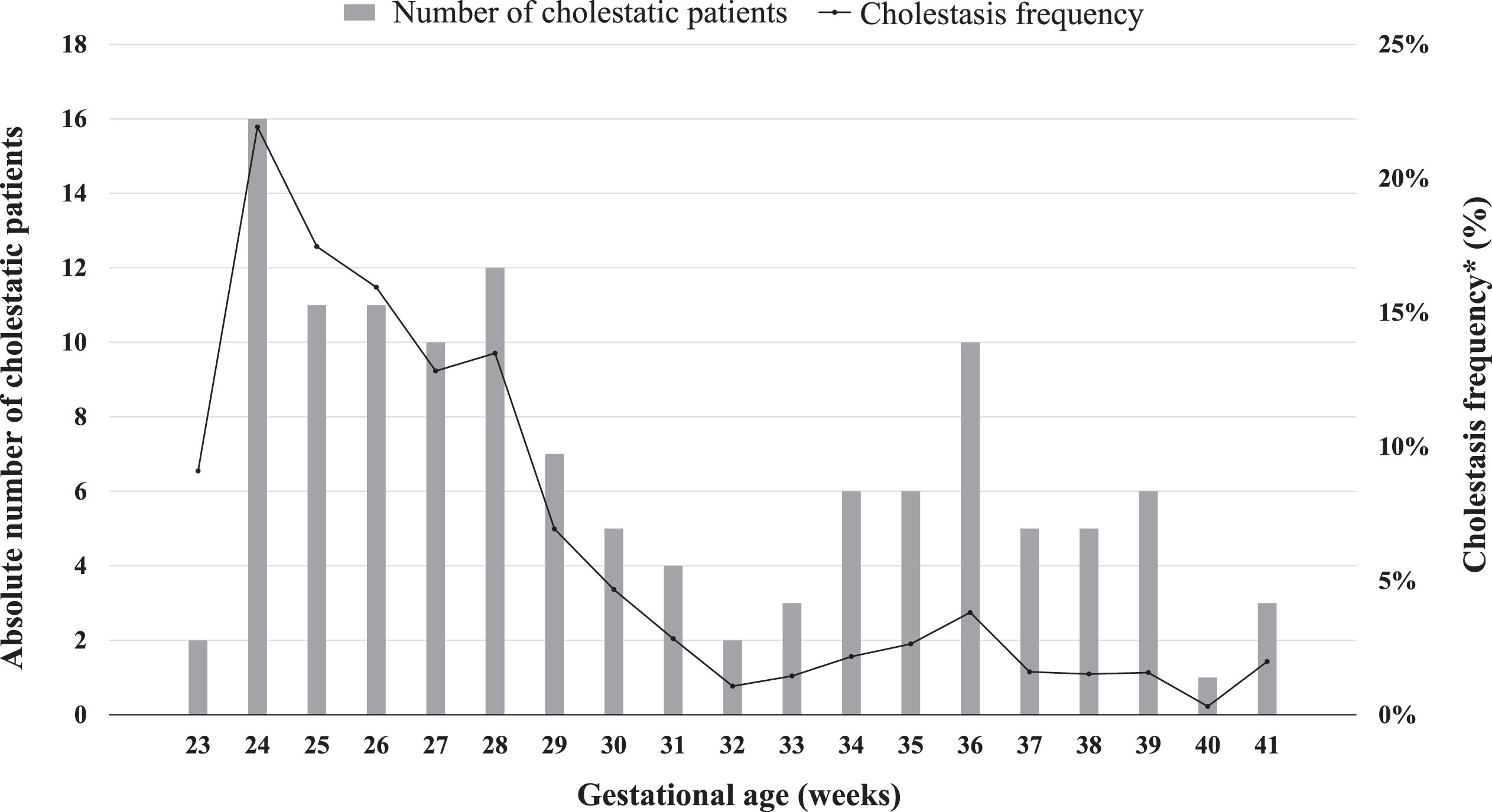
Absolute number of infants with neonatal cholestasis (total n = 125) and cholestasis frequency among GA weeks. *Cholestasis frequency among GA weeks was calculated as the number of cholestatic patients included over the number of all admissions of the same GA.
When comparing cholestatic infants admitted to the Division of Neonatology during the study period to non-cholestatic infants using the GA stratification, patients with less favorable baseline characteristics and more morbidities were at higher risk of developing cholestasis (Table 1).
Comparison of patient characteristics, major morbidities, and mortality between cholestatic
Comparison of patient characteristics, major morbidities, and mortality between cholestatic
Factors negatively associated (p <0.001) with the development of cholestasis on univariate logistic regression were GA and birth weight. Factors positively associated (p <0.01) with its development were necrotizing enterocolitis, positive blood culture, retinopathy of prematurity (only in <32 weeks GA) and neurological injury. Data are expressed as median values (intra-quartile range, IQR) or n (percentage).
Cholestasis incidence in extremely and very preterm infants (10%, n = 78/748) was five times higher (p <0.01) than incidence in moderate to late preterm or term infants (1.8%, n = 47/2,654). Neonatal cholestasis was observed later in less mature infants as the mean age at diagnosis of cholestasis was 22 (SD 14) and 10 (SD 10) days of age in neonates <32 weeks GA and≥32 weeks GA, respectively (p <0.001). Baseline characteristics and clinical evolution were compared according to GA in Table 2.
Infants with neonatal cholestasis: population characteristics, treatments administration and clinical evolution based on GA stratification
Infants with neonatal cholestasis: population characteristics, treatments administration and clinical evolution based on GA stratification
Data are expressed as mean (standard deviation, SD), median values (intra-quartile range, IQR) or n (percentage). *All confirmed infections during the period of 2 weeks prior to cholestasis diagnosis and up to resolution. **All cholestatic preterm <32 weeks GA, and 70% of neonates≥32 weeks GA received PN. ***Peak levels of liver function tests during cholestasis. ****Cholestasis duration was only calculated for infants who had complete resolution of cholestasis during hospitalization.
Investigations performed in cholestatic infants included liver enzymes in 97% (n = 121) of patients, serum lactate in 94% (n = 117), abdominal ultrasound in 90% (n = 113), hepatic doppler in 17% (n = 21), serum cortisol in 34% (n = 43), TORCH panel test performed as follows: Toxoplasmosis was tested in 4.8% (n = 6), syphilis in 0.8% (n = 1), Rubella in 1.6% (n = 2), Cytomegalovirus in 59% (n = 74), Herpes simplex virus in 14% (n = 18), HIV in 5.6% (n = 7), urine reducing substances in 14% (n = 18), serum alpha-1-antitrypsin in 13% (n = 16), serum galactose-1-phosphate uridyltransferase in 8% (n = 10), sweat test in 5.6% (n = 7), scintigraphy or liver biopsy in 4.8% (n = 6), genetic mutation analysis for cystic fibrosis in 3.2% (n = 4), and posterior embryotoxon was looked for in 2.4% (n = 3). Investigations were performed sequentially until the etiology was reached.
To evaluate the effects of infection on the etiology or the evolution of cholestasis, we examined hemocultures and urine cultures two weeks prior to the diagnosis of cholestasis up to resolution. Eighty-nine percent (n = 111) and 62% (n = 77) of patients had at least one hemoculture and urine culture performed, respectively, during this period.
The different etiologies of neonatal cholestasis in infants <32 weeks GA and≥32 weeks GA are presented in Fig. 2.
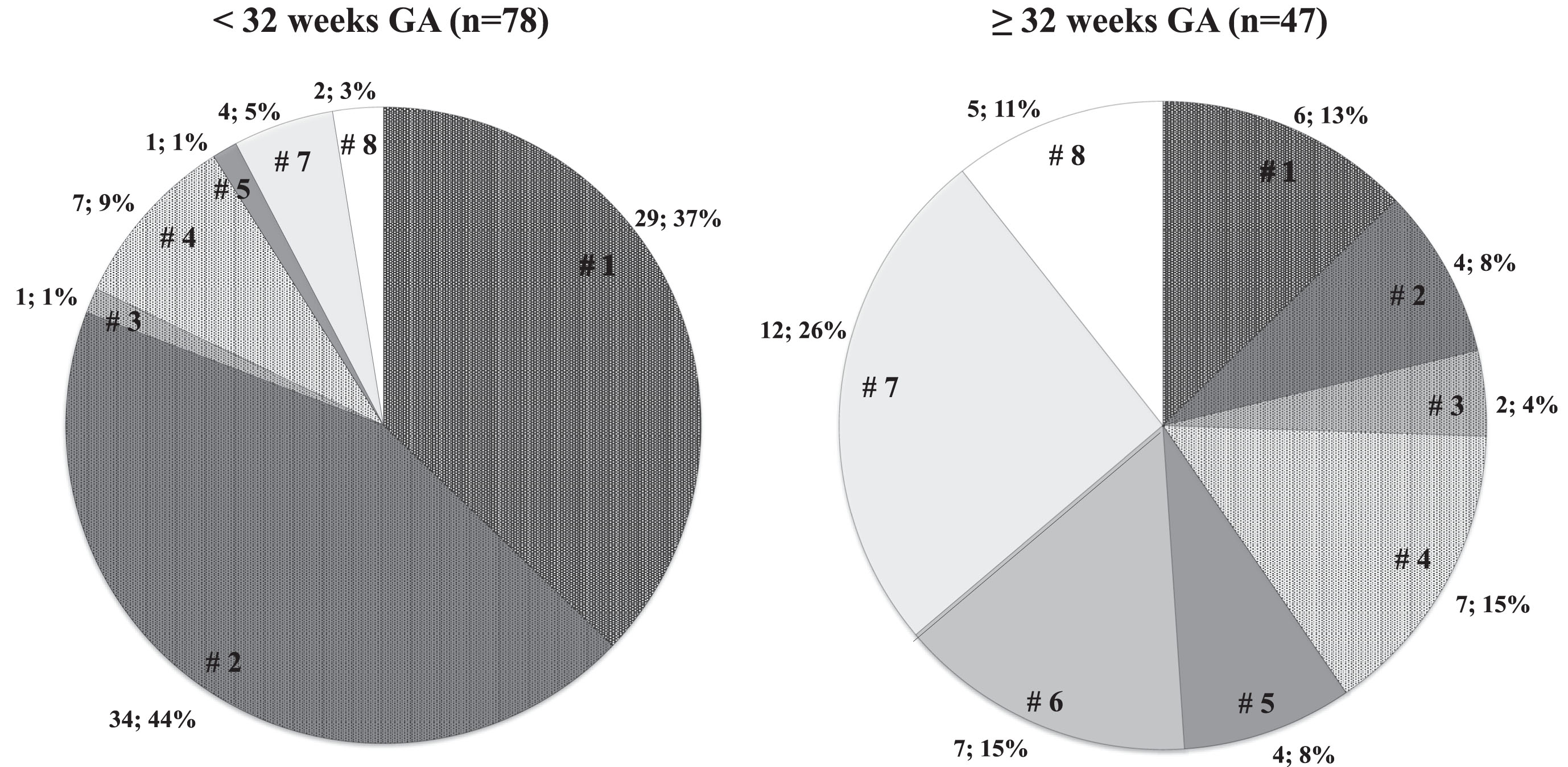
Different etiologies of neonatal cholestasis documented during initial hospital stay in the study participants according to GA (n; %). PN was considered a contributing factor to cholestasis etiology if received for 7 or more days before diagnosis. Positive culture was considered a contributing factor to cholestasis if occurred in the two weeks preceding the diagnosis. PN: parenteral nutrition considered a contributing factor, Ø PN: PN not considered a contributing factor or not applicable.
Nutritional strategy is an important part of cholestasis management. Among patients receiving PN, mixed oil lipid emulsion (SMOFlipid®) or fish oil lipid emulsion (Omegaven®) was prescribed instead of soybean oil-based emulsion (Intralipid®). Nutritional intake of cholestatic infants, the use of fish oil containing emulsions, as well as the UDCA administration and duration, and its prescription at discharge are all described in Table 2.
Gestational age effect on long term outcomes
Resolution of cholestasis was achieved when conjugated bilirubin value decreased below 34.2μmol/L (2 mg/dL). Cholestasis resolved during hospitalization (Fig. 3) in 73% (n = 57) of neonates <32 weeks GA and 38% (n = 18) of≥32 weeks GA (p <0.01). The mean duration of cholestasis, peak levels of liver function tests during cholestasis and other short-term outcomes are described in Table 2. From the multivariate linear regression, cholestasis duration was significantly associated with peak conjugated bilirubin level (β=0.32; p <0.005), duration of PN (β=0.24; p <0.05) and UDCA therapy (β=0.24; p <0.05).
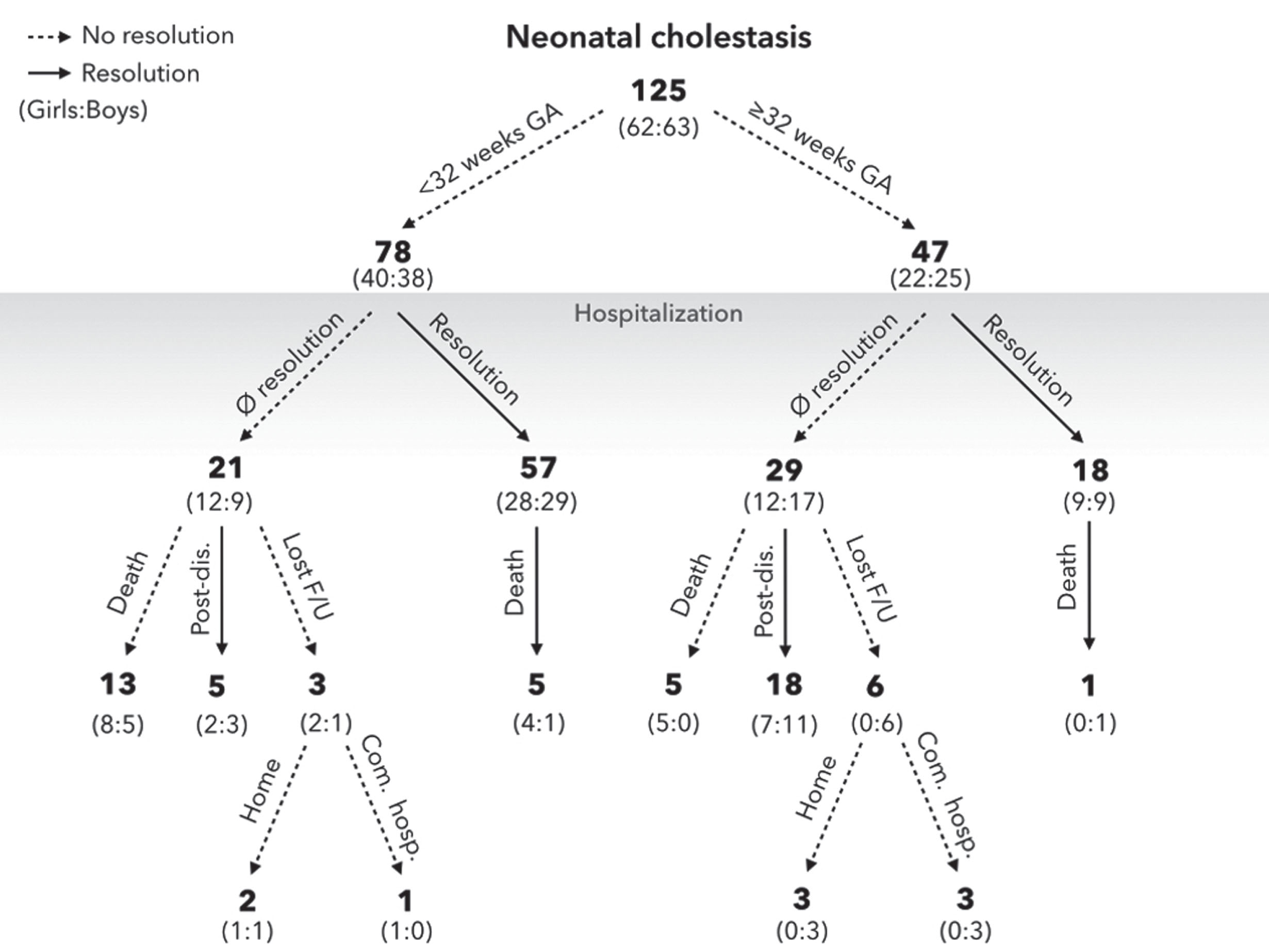
Clinical cholestasis outcome of study participants according to GA. Resolution of cholestasis was achieved when conjugated bilirubin value fell below 34.2μmol/L (2 mg/dL). Com. hosp. –Community hospital; Lost F/U –Lost to follow-up.
We were able to determine the overall resolution of surviving patients who complied with the planned outpatient follow-up with the division of gastroenterology and hepatology. Thus, 95% (n = 57/60) of surviving neonates <32 weeks GA and 85% (n = 35/41) of≥32 weeks GA resolved cholestasis during hospitalization or after discharge; the remaining 3 and 6 patients respectively, were lost to follow-up. Cholestasis was not clinically suspected to be a cause of death in any of this cohort’s patients. However, 3 autopsies showed liver pathological changes in relation to cholestasis. Among deceased infants (n = 24), 6 parents consented for autopsy. Of these, only 3 autopsies showed liver pathological changes in relation to cholestasis as follows: hepatic fibrosis (n = 1; late preterm of 333/7 weeks GA with heart failure that received PN for 20 days); hepatic necrosis, grade of 2/4, centrilobular, involving zones 2/3 of the hepatic lobule (n = 1; extremely preterm infant of 26 weeks GA that received PN for 27 days and died from multiple organ failure and necrotizing enterocolitis); micronodular biliary cirrhosis (n = 1; late preterm of 341/7 weeks with porto-systemic shunt).
The goal of our study was to test the hypothesis that GA has a significant impact on the incidence, etiology, treatment, and outcome of neonatal cholestasis. In accordance with this hypothesis, the study shows that neonatal cholestasis was associated with two different clinical profiles depending on whether the newborn is an extremely or very preterm (<32 weeks GA) or a moderate/late preterm or term neonate (≥32 weeks GA). Intrauterine growth restriction (IUGR) is a well-known risk factor that increases the risk for cholestasis as demonstrated by Teng et al. [30]. However, will IUGR status modify the presentation, outcomes and management of neonatal cholestasis in each GA group is another important question. In our cohort, we had only 35 infants from 78 infants <32 weeks GA and 17 infants from 47 infants≥32 weeks GA with IUGR. With these limited numbers, our sample size could not allow answering this question.
Using this GA-based stratification demonstrated how closely cholestasis is related to GA. Extremely or very preterm neonates had a 5 times higher incidence of cholestasis compared to moderate/late preterm and term infants. The incidence of cholestasis in infants admitted to the neonatal care unit was around 100 times higher than the reported incidence in normal term infants, which is 1 : 2,500 [4]. This higher incidence is closely linked to the multiple complications of prematurity and the liver’s immaturity of preterm infants, which makes it particularly sensitive to hepatic and systemic damage [1]. As an example, lipopolysaccharides, released during infections, will produce high levels of cytokines that will downregulate bile acid transporters in the hepatocytes and induce cholestasis [14]. PN was involved in the etiology of cholestasis in 91% of patients with GA <32 weeks and 40% of patients with GA ≥32 weeks (p <0.01, Fig. 2). PN is considered the most hepatotoxic agent frequently administered in the neonatal intensive care unit [15] and its prolonged administration is known to induce many complications, including neonatal cholestasis [16]. Age at onset and diagnosis of cholestasis is linked to GA. Neonates <32 weeks GA had a later onset of cholestasis which is mostly associated with PN, whereas an earlier onset among neonates ≥32 weeks GA is associated with more prevalent genetic, innate errors of metabolism and other causes of cholestasis.
Identifying the etiology of cholestasis can present many challenges. In well-appearing newborns, it is important to investigate and treat biliary atresia [15] in its early stage. In this cohort, the incidence of biliary atresia was 1 : 3,402 while the reported Canadian incidence was 1 : 19,065 [17]. In ill-appearing newborns, it is important to first find and treat the acute cause of the disease [18]. Standard algorithmic approach to the evaluation of neonates with conjugated hyperbilirubinemia does not take into consideration the impact of GA [7, 19–21]. In our cohort, in infants <32 weeks GA, PN was the only etiology of neonatal cholestasis of 37% versus 13% of infants≥32 weeks GA (p <0.01). Other etiologies of cholestasis (not involving PN) were only in 9% of infants <32 weeks GA versus 60% of infants≥32 weeks GA (p <0.01). It is also important to note that infection was highly associated with cholestasis in infants <32 weeks GA compared to infants≥32 weeks GA (45% versus 16%, p <0.01). Based on our results, using the same approach in the evaluation of a well-appearing preterm infant on PN as in a well-appearing full term infant could result in performing many unnecessary investigations. As etiology strikingly varies between the two groups of our GA classification, cholestasis treatments and evolution will be closely linked to GA and possibly the etiology of cholestasis. However, the small number of patients in each specific etiology category does not allow us to present our results by etiology (Annex Tables 1 and 2).
Soybean oil emulsions, administered first to newborns, are documented to contribute to the development of PN-associated liver disease and cholestasis [22]. These emulsions contain an unfavorable ratio of omega-6 to omega-3 fatty acids as well as phytosterols, which may have a deleterious effect on the liver [23]. Thus, the most common therapy among neonates <32 weeks GA is the change of lipid emulsion, with the administration of mixed oil lipid emulsion (SMOFlipid®) followed by fish oil emulsion (Omegaven®) if needed. Parenteral fish oil has been shown to be effective in treating parenteral nutrition-associated liver disease [23, 24]. On the other hand, for more mature (≥32 weeks GA) and feeding infants, UDCA was the most frequently used therapeutic medication.
Liver enzyme peaks during cholestasis were not significantly different between our two groups of GA classification, except for alkaline phosphatase which was significantly higher among neonates <32 weeks GA. This enzyme is nonspecific and is also used as a marker of mineral status for metabolic bone disease diagnosis, more prevalent among neonates <32 weeks GA [25].
The effects of peak conjugated bilirubin and duration of PN fit our assumptions and were positively associated with a longer episode of cholestasis. However, the positive association between UDCA therapy and a longer duration of cholestasis seems unlikely. Due to selection bias, UDCA was frequently more used in infants with higher conjugated bilirubin peaks. We have previously reported the effect of UDCA on increasing the rate of direct bilirubin decline in neonatal cholestasis [26].
The resolution rate of cholestasis during hospitalization might seem low. Having two times less resolution among neonates with a higher GA is explained by the fact that these patients were clinically stable, ready for discharge with outpatient follow-up till achieving resolution at home. After discharge, previous studies reported similar resolution rates, by 6 months among living patients, of 99% and 95% [27, 28].
Strengths and limitations
The main originality of this study is putting into evidence the effect of GA classification (<or ≥32 weeks GA) on the incidence, etiology, treatment and clinical evolution of neonatal cholestasis. It also highlights the importance to consider GA in any algorithmic approach to the evaluation and management of infants with conjugated hyperbilirubinemia.
Our cohort included cholestatic patients with different etiologies in one of the largest mother-and-child centers in Canada over a 4-year period, making this work a large descriptive profile of neonatal cholestasis. Our study sample size is larger than most published cohorts [27–30], in which the number of cholestatic infants ranged from 27 to 92.
The presence of a protocol for cholestasis management in our unit enabled regular monitoring of biochemical markers and better data quality. However, the retrospective aspect of the study made the cofounders unavoidable, so we must remain cautious in generalizing our results. Our cholestasis management protocol did not include regular measurement of serum bile salts or coagulation studies which could have been of specific interest. While our cohort can be considered as one of the biggest reported neonatal cholestasis cohorts, the numbers are still insufficient to make the presentation of treatment and outcome evolution by specific cholestasis etiology possible.
Conclusion
In our cohort, neonatal cholestasis had two different clinical profiles based on GA stratification. The incidence, etiology, treatment, and clinical evolution of neonatal cholestasis were clearly affected by whether the newborn is an extremely or very preterm neonate (<32 weeks GA) or a moderate/late preterm or term neonate (≥32 weeks GA). Our results support the use of a GA-oriented approach for the evaluation and management of newborn infants with conjugated hyperbilirubinemia.
Footnotes
Acknowledgments
The authors would like to thank Mr. Bryan Quinn for his invaluable help with the English language editing of this manuscript.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Disclosure statements
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
