Abstract
Background:
Dietary restriction (DR) is a widely used experimental intervention in aging research due to its consistent ability to extend lifespan in most species tested. DR is an all-encompassing term describing interventions that restrict some aspect of nutrition - from calorie amount to calorie type to timing of food intake - and yet share common functional endpoints including extended longevity, but also improvements in healthspan, or the time spent in good health, as well as metabolic fitness and stress resistance. Recent studies highlight the preponderance of sexual dimorphisms in the response to DR and argue for the importance of inclusion of both sexes in preclinical research.
OBJECTIVE:
We set out to perform a comprehensive assessment of documented health and lifespan outcomes of interventional DR studies in mice that display sexual dimorphism.
METHODS:
A systematic literature search was conducted according to the PRISMA statement to identify mouse DR studies in which both sexes were included using PubMed. The specific DR interventions examined included calorie restriction (CR), intermittent fasting (IF), protein restriction (PR) and methionine restriction (MetR), with experimental endpoints focused on lifespan and healthspan.
RESULTS:
Sexual dimorphism in the lifespan and healthspan effects of various DR regimens is a common finding in mice, with the magnitude and direction of dimorphic responses influenced by the specific dietary intervention as well as the strain of mouse used in the study.
CONCLUSIONS:
Despite the fact that preclinical lifespan and healthspan analyses in mice reveal sexual dimorphism in the response to DR, there is still a large gap in our understanding of how sex affects dietary outcomes. More preclinical research comparing both sexes in the same study with better attention to reporting metrics during peer review and in easily searchable text including title and abstract is required to further our understanding of the impact of sex on health and lifespan in response to DR in rodent studies.
Keywords
Introduction
The morbidity-mortality paradox, in which females tend to have worse health than males and yet live longer [1, 2], is an example of sexual dimorphism, the condition in which two sexes of the same species exhibit different characteristics unrelated to their sexual organs. Differences in size, fat metabolism and expression of drug metabolising enzymes are further examples of sexual dimorphism with potentially profound implications for biology and medicine. Nonetheless, the preponderance of both preclinical and clinical studies using only one sex has prevented a detailed accounting of which characteristics display sexual dimorphism, as well as an understanding of molecular mechanisms underlying these potentially important differences. Historically, a number of factors have precluded the use females in particular from research, including perceived increased variability amongst females due to effects of cycling sex hormones, as well as added cost of studies involving both sexes. It is important to note, that when we talk about sexual dimorphism in the context of animal studies, we consider sex a biological variable, defined genetically by XX or XY chromosomes [3].
Almost 30 years ago, the NIH recognized that excluding women from clinical research was “bad for women and bad for science” [4], and established the Office of Research on Women’s Health to address the issue in 1990. In 2016, the NIH mandated policies requiring applicants to include sex as a biological variable (SABV) in all preclinical studies, including those with primary-derived cells, or else to provide strong scientific justification for the use of only one sex based on rigorously defined exceptions that do not include cost considerations [4]. To further illustrate the importance of SABV, a cross-sectional study of C57BL/6Nia mice recently demonstrated that a number of parameters that showed an age-dependent decline in males were preserved in older female mice [5]. After controlling for multiple comparisons, lower percent body fat was associated with premature death but only among females; no health measures were significantly associated with premature death in males. This was true even for measures that differed among age groups [5].
In the context of aging research, several large-scale rodent studies [6–9] provide strong evidence of sexual dimorphism in lifespan and healthspan responses to one of the most heavily investigated anti-aging interventions, dietary restriction (DR). Defined as reduced food intake without malnutrition, DR describes a range of interventions that broadly impact the hallmarks of aging through pleiotropic mechanisms (for recent reviews, see [10–16]), resulting in extended longevity and improvement in markers of healthspan in most species tested to date.
Here we systematically review what is known about sexual dimorphisms in the lifespan and healthspan outcomes of dietary restriction interventions specifically in mice. To this end, we queried original research articles in the PubMed database describing experimental research measuring lifespan and/or healthspan outcomes in both male and female mice subject to various dietary restriction interventions, including calorie restriction (CR), intermittent fasting (IF), protein restriction (PR) and methionine restriction (MetR).
Methodology
A systematic review of the literature was conducted according to the PRISMA statement [17] to identify publications reporting on mouse dietary restriction studies. PubMed was utilized as the search tool and database to screen the title, abstract and keywords of all articles (excluding reviews) using the search terms with Boolean operators as outlined in Table 1. All identified records were exported to Endnote (Endnote X9, Thomson Reuters, New York, USA), where authors removed duplicate records and irrelevant titles/abstracts and non-original research (re-analysis of previously published data, commentaries) (Supplementary Table 1). To ensure all relevant research was included, a manual review of the literature was also performed to ensure all possible research was included. The remaining potential records were then screened against the eligibility criteria as specified in Table 2, and eligible articles used as the basis for this systematic review.
Search terms used in the systematic review. Search terms including Boolean operators and permutations used in the PubMed search with standard filter for English language. 284 articles were identified for further screening
Search terms used in the systematic review. Search terms including Boolean operators and permutations used in the PubMed search with standard filter for English language. 284 articles were identified for further screening
Inclusion and exclusion criteria for the systematic review
CR, calorie restriction; DR, dietary restriction; PR, protein restriction; MetR, methionine restriction.
The definitions of terms describing the different dietary/feeding paradigms covered in this systematic review are summarized in Table 3. Lifespan/healthspan data are presented in Table 4. Assessing healthspan in mice is limited by the lack of a gold standard definition of what measure(s) constitutes an improvement in healthspan. As recently reviewed [18, 19] there are a large number of assays available in mice which measure a wide range of physiological functions in mice, many of which are altered in aging [18]. For the purpose of this review, we limited our healthspan measures to changes in body composition/body weight, measures of insulin sensitivity and glucose tolerance, incidence of tumors/neoplasia and immunology parameters as these are well established to be altered with DR.
Definition of terms describing the different feeding paradigms
Red text describes regimens involving enforced restriction of either total calories or timing of food availability; green text describes regimens consisting of diets with altered macronutrient content but fed on an ad libitum basis.
Patterns of inclusion of sex information in dietary restriction/aging publications over the past 29 years
To gain insight into the trends in use and reporting sex of experimental animals in the preclinical literature regarding lifespan/healthspan benefits of DR, we modified the search criteria specifying sex (Table 1) to highlight studies reporting the use of only males (male NOT female), only females (female NOT male), or not reporting sex in the title, abstract or keywords (NOT (female OR male)) out of the total number of studies (sex term removed). Of the total number of 2809 articles (Fig. 1A, B) returned by searching without any sex criteria, we found 1016 articles reported males only (36.2%), 402 reported females only (14.3%), 514 articles reported both males and females(18.3%), while 877 reported neither male nor female sex (31.2%).
Thus, while a PubMed search of title, abstract and keywords but lacking the full text overestimates the number of papers that fail to report the sex of experimental animals by about one half (most of which actually used one sex), this is still a surprising number of papers and indicates a failure in the reporting of data as well as in the peer review process the preceded publication and citation in PubMed.
Finally, an analysis of these trends over the past almost 30 or so years (1993–2022), including 6 full years after the 2016 NIH preclinical mandate requiring applicants to include SABV, reveals an increase in the total number of papers in the field, but little change in the percentages over time with regard to sex reporting. Taken together, this brief survey indicates that in 2022 in this subfield, there remains a majority of papers that don’t consider both sexes, and a minority that either don’t report sex at all or don’t consider it important enough to include in the text searchable by PubMed. Allowing for the inherent limitations and bias of this type of basic search as a metric, it serves to suggests an ongoing lack of inclusion of females in DR research. These findings are supported by recent work investigating the inclusion of SABV in preclinical work since the implementation of this mandate [3, 20].
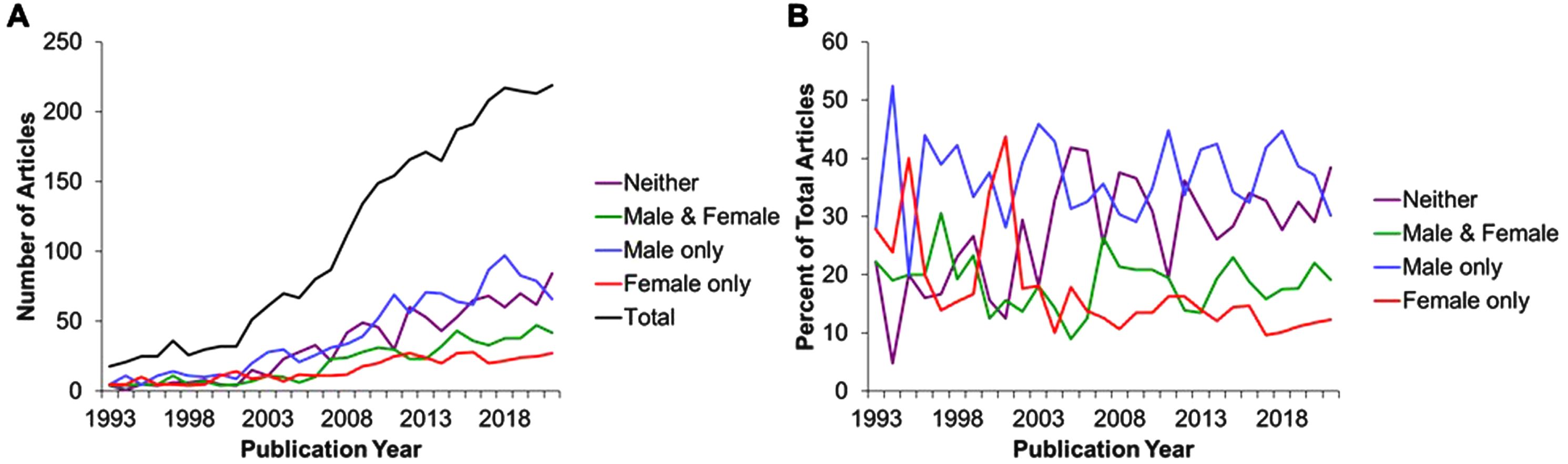
Total numbers of publications (A) expressed as a percent of total (B) over the period from 1993 through 2021 : 2809 results.
Aging is a multifactorial process resulting in a progressive breakdown in tissue homeostasis leading to a decline in physiological reserve. The consequence of this inevitably is eventual death, but often includes an increased burden of chronic disease. The underlying cellular and molecular mechanisms are complex and poorly understood, but thought to be attributable to a number so-called “hallmarks of aging” including cellular senescence, mitochondrial dysfunction, stem cell exhaustion, deregulated nutrient sensing, loss of proteostasis, epigenetic alterations, telomere attrition, genomic instability and altered intercellular communications [21]. While the underlying mechanisms of the aging process are beyond the scope of this review, we direct the reader to several excellent reviews on the topic ([21–23]).
Dietary restriction (DR) is an all-encompassing term describing interventions that restrict some aspect of nutrition, but without deficiency of essential nutrient, that broadly impacts the hallmarks of aging, resulting in extension of lifespan and improvement in markers of healthspan in most species tested to date. Such interventions include calorie restriction (CR), involving reduced food intake (usually by 20–40% relative to
The first report of the benefits of calorie restriction (CR) on lifespan in rodents can be traced back to 1935 when McCay, Crowell, and Maynard described how a severe reduction in calories after weaning retards growth and body size of albino male rats, while at the same time extending their lifespan relative to a control group given free access to food [24]. This work established the use of a dietary intervention to extend longevity and laid the groundwork to future use of this technique as a tool to interrogate the basis of the aging process itself.
Assessment of sexual dimorphism in lifespan and healthspan effects of dietary restriction
AL, ad libitum; 10CR, 10% calorie restriction; 15CR, 15% calorie restriction; 20CR, 20% calorie restriction; 40CR, 40% calorie restriction; M, male; F, female; B6, C57BL/6; EM, emory morse; FI, frailty index; N/A, not applicable; LS, lifespan; BW, bodyweight; EOD, every other day; PR, protein restriction; SD, standard diet; FBG, fasting blood glucose; dietary restriction; LPD, low protein diet; DR-LPD, dietary restriction with the low protein diet (6% protein); DR-22P, dietary restriction using the 22% protein normal chow diet; LDLc, low density lipoprotein calculated; HDLc, high density lipoprotein calculated; ↓, decreased; ↑, increased; AL-HFD, ad libitum high fat diet; AL-CR-HFD, weight cycling with periods of AL HFD then HFD-calorie restriction; 20CR-HFD, high fat diet with 20% calorie restriction; 30CR-HFD, high fat diet with 30% calorie restriction; GFN, geometric framework; MF, meal feeding; SAM, S-adenosylmethionine; SAH, S-adenosylhomocysteine.
Assessment of sexual dimorphism in lifespan and healthspan effects of dietary restriction
AL, ad libitum; 10CR, 10% calorie restriction; 15CR, 15% calorie restriction; 20CR, 20% calorie restriction; 40CR, 40% calorie restriction; M, male; F, female; B6, C57BL/6; EM, emory morse; FI, frailty index; N/A, not applicable; LS, lifespan; BW, bodyweight; EOD, every other day; PR, protein restriction; SD, standard diet; FBG, fasting blood glucose; dietary restriction; LPD, low protein diet; DR-LPD, dietary restriction with the low protein diet (6% protein); DR-22P, dietary restriction using the 22% protein normal chow diet; LDLc, low density lipoprotein calculated; HDLc, high density lipoprotein calculated; ↓, decreased; ↑, increased; AL-HFD, ad libitum high fat diet; AL-CR-HFD, weight cycling with periods of AL HFD then HFD-calorie restriction; 20CR-HFD, high fat diet with 20% calorie restriction; 30CR-HFD, high fat diet with 30% calorie restriction; GFN, geometric framework; MF, meal feeding; SAM, S-adenosylmethionine; SAH, S-adenosylhomocysteine.
The first study to consider intermittent fasting as a more translatable approach also used rats of both sexes, and was the first to uncouple reduced animal size from longevity [25]. In this study, fasting for 1 day in 3 increased the life span of males and females 20% and 15%, respectively, and proved more effective than 1 in 4 or every other day fasting. Classic studies in subsequent decades on the nutritional basis [26] and physiological effects [2, 27] of DR in rats were notable in their exclusive focus on the male sex. Nonetheless, studies in which both male and female rats were included highlight important sexual dimorphisms in multiple physiological responses to DR including, but not limited to, plasma glucose and fructosamine levels, plasma triglyceride and cholesterol levels [28], and number and onset of tumors [29].
Beginning in the 1940s, researchers were studying the effects of food restriction on tumor development using mouse models. This led to the description of a number of different phenotypes modulated by DR such as body composition, insulin sensitivity and immune functions; all of which are now recognized as hallmarks of the DR response [8]. From these investigations, the mouse emerged as an important tool to study aging and age-related diseases due to many factors not limited to their physiological similarity to humans, the ease of maintaining and breeding them in the laboratory, and the availability of many inbred strains [30]. In recent years, the mouse has become the model for testing interventions for improving health and lifespan. Below we describe those studies which have an experimental design that includes both male and female mice to address modulation of lifespan and/or healthspan by DR; these studies are summarized in Table 4. It is important to note that in addition to sexual dimorphism, strain differences in the response to DR were also observed [5, 32], urging a cautionary approach to general translation of these findings.
Traditionally, the success of DR interventions against aging have been based on their ability to increase mean and/or maximal lifespan. Over the past decade, an additional emphasis has been placed upon the ability of such interventions to improve healthspan independent of their ability to increase lifespan. In humans, healthspan can be defined as the length of adult life during which a person maintains the capacity to perform all routine activities of daily living (dressing, bathing, eating, toileting, transferring) as well as instrumental activities of daily living (finances, shopping, transportation, food preparation, managing medications, using the telephone) [18]. In mice, although a comparable accepted definition is still lacking, healthspan could be defined as the period of life under conditions of ideal husbandry in which the mouse is able to move around, feed itself, and care for itself, for example with grooming [18]. To standardize quantification of these measures in mice a number of indices have been developed to measure mouse frailty [33, 34]. These tools are analogous to human frailty such as the Fried frailty index and the Rockwood deficit accumulation index. These include both observational and functional deficit assessments and have been validated against a number of pre-clinical outcomes. In one study, lifelong 40% CR significantly reduced frailty in male, but not female C57BL/6 mice when compared to their AL counterparts [35]. There was no effect of 40% CR on reducing frailty in DBA/2J mice, another inbred strain [35]. When started late in life, 6 months of MetR is sufficient to reduce frailty [36] in male C57BL/6Nia mice. Female data has not been reported. In another study, frailty index predicted mortality in female, but not male, 3x Tg Alzheimer’s mice [37]. Considering the relevance of these findings to lifespan/healthspan uncoupling, the underlying mechanisms of sexual dimorphism require further investigation.
One of the hallmark features of CR is the ability to delay the onset and incidence of cancer in animal models. Indeed, before McCay connected growth restriction via CR to longevity, the influence of food restriction on the growth of transplanted and spontaneous tumors was known [38]. Interestingly, there is contrasting evidence with some suggesting that the number of tumors in mice does not appear to be sex-specific [39, 40], while others do report a sexually dimorphic effect in the number and type of tumors [41, 42]. Whether this also holds true to other healthspan and ‘hallmark features of CR’ requires further investigations (Table 4).
Certainly, daily reduction in caloric intake is not the only means to achieve such beneficial outcomes. EOD feeding, ADF, IF and other fasting paradigms also demonstrate improvements in many physiological domains that overlap with CR, including lifespan extension [43, 44]. In recent years, intermittent fasting type diets have gained increased traction as they intersperse fasting with non-fasting days and potentially seem more applicable than a daily 40% CR. In this paradigm, short periods of intense energy restriction (75–100% reduced caloric intake on fasting days) followed by ‘normal’ eating on non-fasting days. A study in Growth Hormone Receptor Knock-Out (GHRKO) mice showed that IF (EOD feeding) increased lifespan in WT mice compared to control fed mice in males but not females. They also demonstrated that GHRKO mice do not respond to IF [45]. A study of late-lie IF (EOD feeding) in C57BL/6 mice (21 mo age) reported attenuation of some hallmarks of CR including improved glucose tolerance, restoration of metabolic flexibility and decreased frailty [46] in both sexes. Lifespan however was not measured. As the majority of the studies published do not include females or do not include a lifespan outcome, we exclude them from the scope of this systematic review on sexual dimorphism and refer the reader to several excellent reviews [44, 47] on this topic.
While the underlying molecular mechanisms regarding the beneficial effects of CR on improved lifespan and healthspan have been under investigation for many years, there has been less work done to disentangle the question of whether fasting time or calories is more important for the physiological benefits. Recently, fasting time has been reported to be positively associated with the effects on survival and reduced disease incidence in mice [48]. The findings in this study were recently built upon by others using a diluted AL paradigm where food is diluted with indigestible cellulose, but provided AL, leading to an approximate 30% restriction [49]. When compared with classical 30% CR and meal feeding (30% CR fed across the day in three allotments), the authors found that fasting is necessary for the CR-induced improvements in frailty and lifespan in male mice [49].
Sex differences in the methionine restriction -mediated life- and health-span improvements
One of the major challenges in translating CR to humans is quite simply that most humans would find it incredibly difficult to reduce daily caloric intake by 20–40%, especially for the significant portion of their life required to increase longevity (if this is even possible in humans). To this end, interventions allowing for DR benefits without actual food restriction represent an attractive alternative. In the early 90’s Orentriech and colleagues reported that a reduction in a single amino acid, methionine, resulted in a 30% increase in lifespan of male Fischer 344 rats [50]. Notably, rats were able to eat as much as they wanted of a diet with 0.17% w/w (compared to a control diet of 0.86% methionine). On a technical note, it is important to clarify that when we use a methionine restricted diet, it is a diet restricted in sulfur amino acids (SAA) since the non-essential sulfur amino acid cysteine is absent in MetR diets. This lack of cysteine is required for the MetR phenotypes as it has been shown that cysteine blocks the effects of MetR [51]. Interestingly, the metabolic phenotype of MetR can be obtained with diets that are deprived of other amino acids such as leucine [52], although none have yet shown the same lifespan extension. Recent work has demonstrated that diets low in isoleucine or valine recapitulate the metabolic phenotype seen with MetR [53], however their effects on lifespan were not tested in this study.
The initial study describing how a MetR diet (0.1–0.15%) can increase lifespan in mice was published in 2005. When female mice were fed the MetR diet from 6 weeks of age, this resulted in an increase in maximal lifespan of 9.2% (estimated from the survival curves at the time of publication) [54]. Subsequent studies have shown that this can be replicated in males (but not shown in females) when started at 12 months of age [55]. Interestingly the metabolic phenotype of MetR is dose-dependent with there being a threshold level of methionine that abrogates the beneficial effects; however this has only been reported in males and not females [56]. Given the differential response in C57BL/6 females to CR [8] it would be incredibly interesting to see if this was also true for females on MetR. Benefits of MetR that are not sexually dimorphic (at least as currently described in wildtype mice in the literature) include reductions in bodyweight, fat mass and oxidative stress coupled with improvements in insulin sensitivity as well as changes in circulating insulin, glucose, leptin, adiponectin, IGF-1 and FGF-21. It is important to note that a number of these benefits of MetR overlap with CR despite
Studies have also demonstrated the applicability of MetR as a treatment for different progeria syndromes including Hutchinson-Gilford progeria syndrome (HGPS) [57] and Cockayne syndrome [58]. Importantly, MetR was able to extend median lifespan in both male and female HGPS mice and had a lower mortality rate [57]. In Cockayne syndrome mice, MetR extends lifespan and improves healthspan parameters; although the study includes both male and female mice (personal communication), this study is limited in that the authors do not differentiate between male and female mice [58]. More importantly, the healthspan of these mice was improved significantly with MetR as evidenced by an amelioration of the loss of bone structure and lack of grooming behaviors, as well as improvements in aortic and skeletal muscle fibrosis [57]. This is interesting given that generally MetR is thought to reduce bone mass [59]. A recent study of young and old male and female mice noted that bone morphology is altered in an age and sex specific manner, with MetR mice having reduced bone mass. However after correcting for body size, MetR mice had no impairment in biomechanical properties [60]. This points to the role of sex steroids in the hormonal regulation of bone morphology in response to MetR [60]. Indeed, short term studies (up to 5 weeks) have demonstrated a sexual dimorphism in hormonal responses to MetR in young mice [61].
In 6-week-old male and female mice preconditioned with a western diet (WD) for 12 weeks before being switched to a WD deficient in methionine (or staying on WD) there is no sexual dimorphism in the physiological response to the MR diet in terms of bodyweight, food intake, insulin resistance/glucose homeostasis or energy expenditure [61]. However, sexual dimorphism was present in terms of plasma FGF21 levels with only males having increased levels despite increased levels of liver transcript in both sexes. Interestingly UCP1 expression was increased in gonadal WAT of MR fed male mice but not females. This suggests that in females, increased energy expenditure occurs via a FGF21/UCP1 independent mechanism [61]. Growth hormone has been implicated in the mechanistic response of MetR but studies have not investigated the sexual dimorphism (or lack thereof) [62] in these mice. Furthermore, there are sexual dimorphic tissue specific metabolic responses in Snell dwarf vs control mice fed a MetR diet have been observed. Hepatic hypotaurine being 3-fold higher in normal males versus females, a difference that was not seen in the Snell dwarf mice where the hypotaurine concentration in both sexes was comparable to the lower value found in normal females [63]. While the molecular mechanism underlying these sex differences is not known, it is plausible to attribute these differences at least in part to sex hormones. Indeed, estrogen removal in animals or menopause in women is associated with metabolic disturbances including hepatic triglyceride accumulation and decreased insulin sensitivity [64].
Alterations in macronutrient contents/ratios and impact on health and lifespan
Another method to achieve some of the beneficial effects of CR without reducing caloric intake is to alter either the ratio of protein:carbohydrate:fat (P:F:C) content in the diet (termed the geometric framework GF set of diets), or by modifying a specific component such as protein (i.e. low protein diets). Short or long-term reduced protein intake is associated with many beneficial effects including metabolic outcomes [65, 66], reduced surgical complications [67, 68] and improved lifespan [69]. The commonality in these dietary interventions is that they induce a phenotype which overlaps with CR to some extent and affect CR-related pathways.
The Geometric Framework for Nutrition (GFN) is a model that was developed to investigate how nutrients, other dietary constituents and their interactions influence physiology and health. The GFN model has been used to demonstrate how organisms across many taxa possess nutrient-specific appetites, select foods, control food intake and utilize ingested nutrients to attain their intake, growth and maintenance requirements [70]. The initial study in mice examined over 25 different diets in C57BL/6 mice found that the main determinant of lifespan is carbohydrate:protein ratio and is independent of calorie intake [69]. The authors used AL diets of normal, medium and low energy density (termed caloric dilution) to ask the question if amount and/or type of nutrient is important for health outcomes in mice [69]. It is important to note that the authors do not use CR in the traditional sense, rather energy dilution in that while the mice ingested less calories at the lower energy density foods, they eat almost twice as much food mass (available
Follow up studies to this lifespan paper have investigated the effect of P:C ratio on various outcomes and found sex dependent effects of diet on fertility [71], and skin structure [72]. Interestingly prolonged fertility correlating with increased lifespan is also a feature seen in CR mice [73]. A paper published from the same group investigated a protein titration across metabolic and cognitive outcomes compared to traditional CR (20% reduction in daily calories compared to control diet, 19% protein amounts), where they present sex as an outcome variable. This study noted sex specific differences (some variables higher in males compared to females and
Although the GFN is a relatively new tool, the idea of altering macronutrient content in the diet to improve health outcomes has been around for a number of decades. Low protein diets (LPD) have been in use since the early 1970s (in mice) as a therapeutic intervention for autoimmunity and longevity. In NZB mice fed a normal (22%) or low protein (6% from casein) diet, the LPD abrogated thymic involution, and prevented development of splenomegaly. Furthermore it was able to maintained the cell-mediated immunities, antibody-producing capacity and immune functions which are known to decline with age in these mice in a sex independent manner [76]. At 24 months of age, 9.1% of females on the LPD diet were still alive compared to 0% of mice in the other three groups [76] which is consistent with the studies by Solon-Biet et al. [69] showing low protein:carbohydrate ratio is associated with better longevity. In a subsequent study, authors compared both normal and low protein diets with and without 50% DR [77]. Mice on the LPD showed decreased bodyweight, however there was no sexual dimorphism observed [77]. Interestingly, the additive effect of 50% DR on the LPD was detrimental to lifespan in both male and female DBA2/f mice, with the effect being more pronounced in the median lifespan of male DBA2/f mice [77]. Notably, this effect was strain specific as when the same regimen was repeated in F1 offspring of the NZB×NZW F1 strain, DR had a profound sex independent effect of increasing median lifespan in both a normal and LPD of 30–42%. LPD+DR also increased maximum lifespan, although the effect was not as pronounced (7% increase in LPD-DR vs 42% increase) as the effect of a normal 22% protein diet [77]. Again, the differences were not sex dependent. The authors note that in this study no advantage was found to result from lowering the calories in addition to lowering the protein intake, and calorie restriction
Conclusions
Recent studies have illustrated the ability of sex to impact health and lifespan outcomes in mouse studies. However, with only 21.4% of studies including both sexes, it is apparent that there is still far to go. Here we present a systematic review of the literature on how sexual dimorphism may be modulated in response to different dietary restriction and feeding paradigms. These data illustrate the importance of including both sexes when considering translational approaches of these interventions to humans and highlights the potential of leveraging such differences to provide novel insights into the pathophysiology of the aging process itself.
Footnotes
Acknowledgments
This manuscript is dedicated in loving memory of Dr. Jay Mitchell. We apologize to the authors whose work is not included due to space limits.
Funding
This study was supported by the National Institute on Aging, 5R01DK090629 to J.R.M., and P01AG055369 to S.J.M.
Conflicts of interest
The authors declare no conflicts of interest.
