Abstract
BACKGROUND:
Diet and sleep represent key behavioral risk factors for major non-communicable diseases. Dietary factors may modulate systemic inflammation and potentially affect the human brain functionality, finally affecting the sleep-wake cycle. In this context, the Dietary Inflammatory Index (DII) has been studied as a tool to investigate the dietary inflammatory potential and its relationship with a variety of health conditions.
OBJECTIVE:
The aim of this study was to perform a systematic revision of observational studies on the inflammatory potential of the diet assessed through the DII and sleep features.
METHODS:
A systematic search of observational studies on DII and sleep features was conducted on Pubmed and EMBASE electronic databases. The Meta-analyses Of Observational Studies in Epidemiology (MOOSE) guidelines were used to plan the methodology. Studies identified through the search were selected according to inclusion/exclusion criteria.
RESULTS:
The systematic search and review led to the selection of 13 studies. Most studies including the general population reported an association between higher inflammatory potential of the diet and worse sleep quality and suboptimal sleep duration. Some studies also reported an association with daytime sleepiness and sleep apnea. However, some studies including younger individuals (i.e., college students or young employees) reported null findings. Four studies conducted in individuals with a variety of health conditions also showed some relation between DII and sleep features, although results were not fully consistent.
CONCLUSIONS:
Data published so far suggests an association between DII and sleep features. However, lack of randomized clinical trials and better designed observational studies, imply that further research on this matter is warranted to understand whether a causal relation could explain current findings.
Keywords
Introduction
Over the last decades, accumulating research has shown that lifestyle factors, such as dietary habits and sleep behaviors are important risk factors for chronic diseases globally [1, 2]. Both inadequate diet and sleep quality have been related with higher likelihood of suffering from cardiometabolic disorders [3, 4] and certain cancers [5, 6]. Moreover, sleep disturbances are often associated with neurodegenerative diseases and only recently being considered as potential prodromal symptoms for such conditions to be considered in older adults [7]. In fact, an increasing body of scientific literature provides evidence for a relation between diet and sleep features with cognitive health and Parkison’s disease patients [8, 9]. The essentiality of these physiological behaviors for the human body and the chronicity to their exposure represent the key rationale for their importance in influencing a number of biological systems and pathways, including cardio-metabolic and the central nervous ones [10, 11].
The relation between diet and sleep has been hypothesized and demonstrated over the last decades. While a large body of scientific evidence is dedicated to the influence of sleep duration and quality on dietary features, such as total calorie/nutrient intake or overall diet quality [12, 13], the exploration of the inverse relation has only recently gained interest [14]. Dietary factors may in fact affect neuroendocrine regulation in the brain and may play some regulatory role on the sleep-wake cycle through such para-physiological mechanisms [15]. Moreover, dietary factors may influence the overall inflammatory state of the human body, which in turn lead to neuroinflammation and alteration of the physiological activities of the brain [16]. A large and consistent evidence from the scientific literature shows a direct effects of certain dietary factors on biomarkers of inflammation and provides the rationale describing, at least in part, the numerous mechanisms explaining the effects of diet on the immune system [17]. Diet is related with sleep most likely through a bidirectional relationship, although the exact pathways are still under investigation and yet to be more clearly elucidated.
The dietary inflammatory index (DII) is a tool demonstrated to be able to capture the inflammatory potential of dietary habits by taking into account a multitude of parameters including foods, nutrients, and non-nutrient factors [18]. The use of DII has been widely validated in the general population and related to the risk of various chronic diseases, including cardio-metabolic conditions and certain cancers [19]. Although current evidence suggests a relation between dietary factors potentially known to affect inflammatory pathways and most common non-communicable diseases, only a limited number of studies tested the association of DII scores with sleep features, while no systematic evaluation of the evidence has been published so far. Thus, the scope of this study was to perform a systematic revision of observational studies investigating the relation between the inflammatory potential of the diet assessed through the DII and sleep features.
Materials and methods
Systematic search and study selection
The design, analysis and reporting of this study followed the Meta-analyses Of Observational Studies in Epidemiology (MOOSE) guidelines [20]. A systematic search of published studies examining the link between DII and sleep features was performed using Pubmed and EMBASE from their inception to July 2022, with no restrictions. The search strategy was performed using the following keywords: (dietary inflammatory index OR diet inflammatory index OR dietary inflammatory potential OR anti-inflammatory diet OR pro-inflammatory diet OR DII) AND (sleep OR insomnia OR sleepiness). Moreover, the reference list of retrieved papers and relevant reviews were searched manually for any additional studies not previously identified.
Inclusion/exclusion criteria
The included studies met the following criteria: (i) observational study design, limited to prospective cohorts, cross-sectional and case-control studies; (ii) studies reporting on DII as exposure; iii) studies exploring sleep features as primary outcome; and iv) studies providing a statistical calculation/estimation of the size effect. Studies reporting on: (i) children or adolescent population (<18 years old), (ii) pregnant women, and (iii) patients with end-stage degenerative diseases were excluded.
Study selection
The whole procedure including the systematic search and study selection was performed by two authors (J.G. and G.G.). Any occurring discrepancy was examined by both authors and solved by agreeing consensus.
Data extraction
The main characteristics of all the eligible studies were extracted using a standardized electronic form. The following information was extracted: i) first author name and year of publication; ii) study design; iii) study cohort name and country; iv) population characteristics; v) number, gender and age of participants; vi) type of exposure and its main characteristics; vii) investigated outcome and its assessment method; viii) main results.
Results
Study selection process and study characteristics
The systematic search of the literature led to identification of 360 potential reports, 175 of which were excluded after revision of the title and 165 after abstract, leaving 20 articles for full-text evaluation. Based on the full-text screening, 7 articles were excluded for either not reporting on the exposure of interest, or the outcome of interest or not reporting on the relation between exposure and outcome of interest or reported results on an intervention study, leaving 13 articles meeting eligibility criteria [21–33]. The study selection process is shown in Fig. 1.
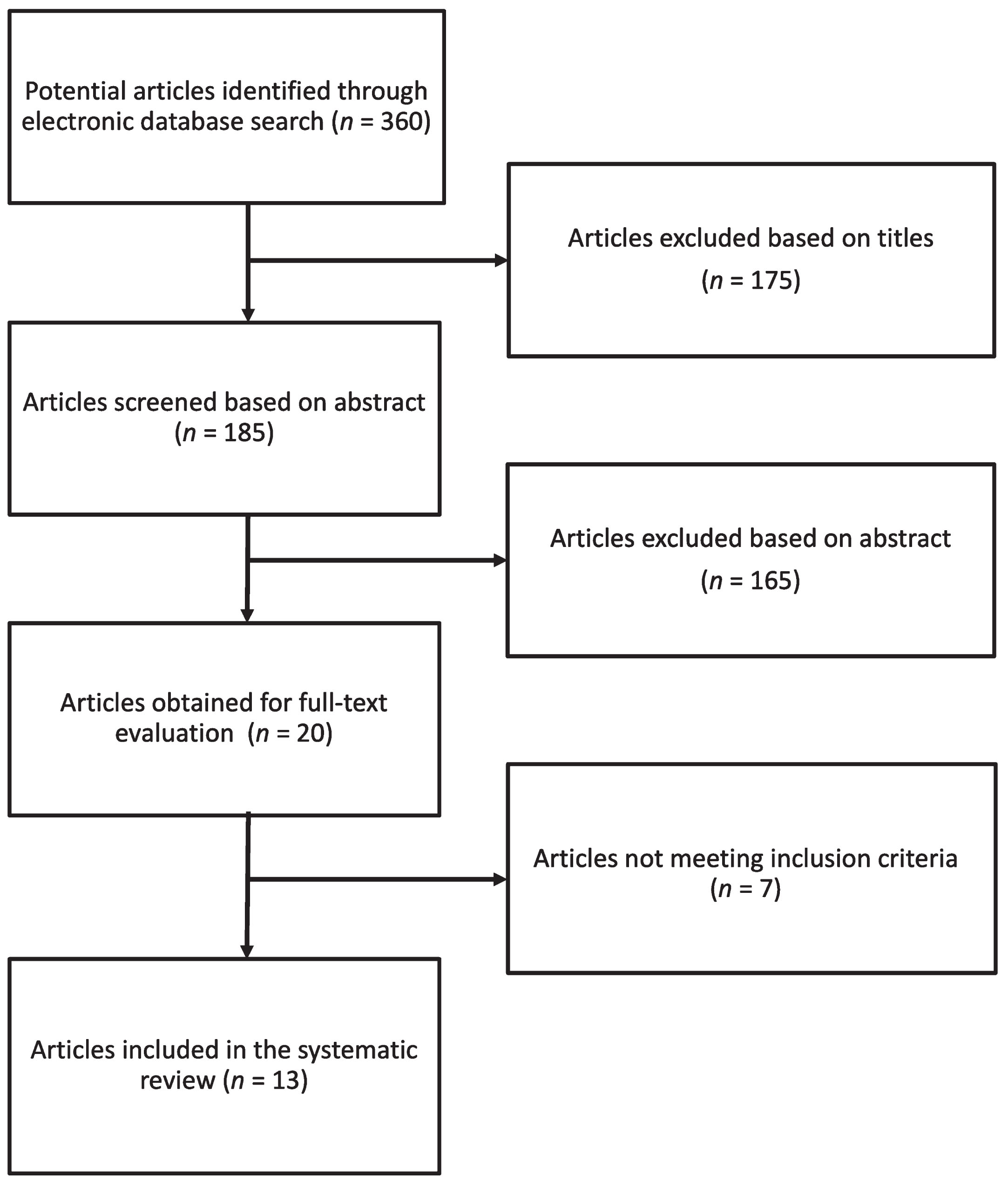
Flow chart of study selection.
A summary of the descriptive characteristics of the studies selected from the systematic search are provided in Table 1. All studies had a cross-sectional design with exception of one longitudinal evaluation of changes in parameters analyzed over time [32]. Three studies only included females while ten articles reported on both sexes. Concerning the continents where the participants of the studies were located, 3 studies included individuals from European countries, 5 from America, 4 from Asian countries, and one from Australia. Ten studies have been conducted on the general population (out of which, 2 specifically on students and 2 on employees) and 4 studies on individuals with health conditions. Dietary data was generally assessed through food frequency questionnaires (FFQs). Concerning sleep parameters, most studies evaluated them through subjective tools, including the Pittsburgh sleep quality index (PSQI) and the Epworth Sleepiness Scale (ESS) for daytime sleeplessness.
The main characteristics of the studies included in the systematic review reporting on the relation between dietary inflammatory index and sleep parameters
Abbreviations: BCOPS (Buffalo Cardio-Metabolic Occupational Police Stress); E-DII (Energy-adjusted Dietary Inflammatory Index); ESS (Epworth Sleepiness Scale); F (female); M (male); MEAL (Mediterranean healthy Eating, Aging, and Lifestyle); NHANES (National Health and Nutrition Examination Survey); NWAHS (North West Adelaide Health Study); OPDC (Oxford Parkinson’s Disease Discovery Cohort); OR (odds ratio); OSA (obstructive sleep apnea); PSQI (Pittsburgh Sleep Quality Index); STOP-BANG (Snoring, Tired, Observed apnea, Pressure, Body mass index, Age, Neck, Gender); y (years).
Most studies investigated the association between the DII scores and sleep parameters in the general population. The first ever cross-sectional study has been conducted by our group on 1936 adults living in southern Italy; the study investigated the relationship between dietary habits, including the DII scores, and sleep quality assessed through the PSQI through a multivariate analyses adjusted for potential confounding factors (i.e., age, sex, marital, educational, and occupational status, smoking and alcohol drinking habits, physical activity level, and adherence to the Mediterranean diet) resulting in a direct association between higher scores of DII and lower likelihood of having adequate sleep quality [for the highest DII quantile, odds ratio (OR)=0.49, 95% CI: 0.31, 0.78] [21]. However, when investigating the specific domains of sleep quality included in the PSQI, a significant result was reported only for sleep latency (OR = 0.60, 95% CI: 0.39, 0.93) [21].
Several following studies on the general population have been conducted on individuals living in the US recruited in the National Health and Nutrition Examination Surveys (NHANES) waves 2005–2016 (n = 23,867) [27], 2005–2008 and 2015–2018 (n = 14,210) [30], 2005-2008 (N = 5594) [31]. A large investigation explored the association between DII and sleep problems assessed through direct questions concerning sleep duration [categorized according to the National Sleep Foundation’s sleep duration for young and old adults in (i) 6 hours for short duration, (ii) >6 to < 9 hours for normal (recommended) duration, and (iii) 9 hours for long duration], and sleep disturbances (assessed by asking whether they ever referred to a doctor or other health professional having trouble sleeping): the adjusted model for age, sex, ethnicity, education level, marital status, BMI, and chronic disease revealed a significant association between DII and both short and long sleep duration (for the highest DII quantile, OR = 1.40, 95% CI: 1.21, 1.61 and OR = 1.23, 95% CI: 1.03, 1.46, respectively) and sleep disturbances (OR = 1.14, 95% CI: 1.02, 1.27); interestingly, inclusion of physical activity in the model led to weakening of results concerning sleep disturbances, suggesting that lifestyle factors (diet and active life) may be clustered and both lead to worse sleep quality [27]. Another study focused on sleep apnea risk assessed using the STOP-BANG (Snoring, Tired, Observed apnea, Pressure, Body mass index, Age, Neck, Gender) questionnaire: the study showed in the multivariate model (adjusted for sex, age, race, marital status, education, income, smoking, physical activity, cardiovascular disease, cancer, diabetes, sleep duration, and alcohol consumption) that individuals with higher DII scores were more likely to have sleep apnea (OR = 1.55, 95% CI: 1.24, 1.94); in this study, the authors suggested that c-reactive protein (CRP) and BMI would have a mediating effects of the dietary inflammatory potential of the diet toward sleep apnea risk [30]. The last study including NHANES data was conducted on a subgroup of participants assessing the association between DII and sleep quality assessed via the PSQI through generalized additive model (GAM) to evaluate the non-linear relationship of the outcome and the predictors showed significant findings with a U-shape association (higher scores for both DII and PSQI indicating worse sleep quality related to higher inflammatory potential of the diet) [31].
Among other studies conducted on the general population, a report conducted on 1743 participants in the North West Adelaide Health Study (NWAHS) cohort in Australia (mean age 56.6 y) did not provide inferential analysis but only reported frequency distribution of subjective measure of sleep quality (categorized as ‘very good’, ‘fairly good’, ‘fairly bad’ and ‘very bad’) by quartiles of DII score, showing no significant differences across groups [25]. Two studies evaluated whether the quality of the diet, in terms of its inflammatory potential, may affect sleep quality among university students [24, 26]. A study conducted on a convenience sample of 379 college students at the University of Sharjah (UAE) only reporting mean DII score difference between participants with adequate and inadequate sleep quality (assessed through the PSQI and all its components) showed no significant differences aside from day dysfunction (P = 0.01) [24]. The other study was conducted on 249 female college students at Ahvaz Jundishapur University of Medical Science (Iran) reported a significant linear association in the most adjusted model (correction for age, energy intake, physical activity, and education) between DII and PSQI scores (beta = 0.30, p = 0.02) [26]. Other two studies have been conducted on employees [29, 32]. One study included 211 employees from different departments in Ahvaz Jundishapur University of Medical Sciences (Iran) showing a relation between DII and sleep duration (beta = -1.35, p < 0.001) but not with PSQI total scores [29]. The other study involved 401 active-duty police officers included in the Buffalo Cardio-Metabolic Occupational Police Stress (BCOPS) cohort followed-up from 2004 to 2019, which were assessed for objective (Micro Motion Logger Sleep WatchTM) and subjective (PSQI) sleep parameters; the study investigated various sleep features (time in bed, sleep duration, WASO, sleep latency, and sleep fragmentation index) and reported a linear association between 1-unit increase in the DII score over time, total PSQI scores (p < 0.001), and WASO by nearly 1.4 min (p = 0.07) [32].
Studies on individuals with health conditions
Some studies evaluated the association of pro-inflammatory diet among individuals subjected to diverse health conditions including patients with obstructive sleep apnea [22], women with fibromyalgia [23], overweight and obese individuals [28], and patients diagnosed with Parkinson’s disease [33]. A study conducted on 296 patients with mild to severe obstructive sleep apnea tested for the relation between DII and sleep patterns analyzed by polysomnography and subjective sleep parameters (PSQI and ESS); the study showed a significant association between a higher DII score and daytime sleepiness (Q4 vs. Q1, OR = 0.47, 95% CI: 0.22, 0.99) but no relation with overall sleep quality [22]. A case-control study comprising 95 women with fibromyalgia and 98 healthy controls did not report any significant results for the relation between DII and sleep quality evaluated using PSQI for either of the groups [23]. Another study involving a total of 219 obese and overweight adult women (mean age 36.5 y) reported a linear relation between DII and PSQI scores in the fully adjusted model (beta = 0.24, p < 0.001) [28]. Finally, people diagnosed with Parkinson’s disease recruited from the Oxford Parkinson’s Disease Discovery Cohort (OPDC) were tested for disease symptoms, including daytime sleepiness assessed through the ESS: DII scores were linearly associated with ESS score in the univariate analysis, but the association was no longer significant after adjustment for various potential confounding factors [33].
Discussion
The results of this systematic review show that dietary factors potentially related to inflammation assessed through the DII are associated with sleep quality in most of the observational studies published so far. Concerning individual sleep features, sleep duration and sleep efficacy resulted as potentially most affected by such inflammatory score related to the diet. The studies were conducted on a variety of populations, thus the findings cannot be generalized and taken as conclusive. However, the consistency of results and similarity of findings across studies provide a promising basis to support the hypothesis tested in this review.
To our knowledge, clinical intervention trials exploring a direct effect of the inflammatory potential of the diet on sleep-related outcomes are substantially lacking. Only in an intervention study the authors investigated changes in DII over time following a 12-week intervention trial on diet, sleep, stress, and physical activity [34]; the study (Inflammation Management Intervention Study - IMAGINE) included a total of 95 participants (mean age 46.9 y) measured nighttime sleep parameters including sleep/wake times, sleep duration, sleep efficiency, sleep onset latency, and wake-after-sleep-onset (WASO) considered as sleep-related outcomes; the final analysis restricted to 79 participants that completed the trial showed that increase in one-point DII score change (meaning higher pro-inflammatory direction) resulted in WASO increase by 3.75±1.33 min (p < 0.01) and sleep efficiency decrease by -0.56±0.26% (p = 0.04) [34]. However, lack of randomization of the aforementioned study and the cross-sectional design of the observational studies included in the present systematic review do not allow to assess a causal relation of the dietary inflammatory potential on sleep quality parameters. Though, various mechanisms have been suggested to potentially explain such relations. Inflammation has been taken into account in various pathways to potentially affect the proper functioning of the brain and circuits assigned to the sleep-wake cycle [35]. In fact, dietary factors may induce stimulation of the microglia and induce neuroinflammation via activation of the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kB) signaling and other inflammatory pathways; the resulting effects include impaired neuronal function and, eventually, affect sleep behaviors over time [36]. Inflammatory mediators, such as tumor necrosis factor-alpha (TNF-a) and interleukin-1 beta (IL-1b) are reported to play a role in sleep initiation and promotions by affecting the circadian rhythm and hormonal regulation [37]. In fact, the inflammatory state would affect the regulation of neurotransmitters implicated in sleep patterns, such as dopamine and serotonin [38]. Several components diet components have been reported to affect low-grade systemic inflammation. Plant-derived foods, including fruits, vegetables, whole-grains, legumes, and plant-based beverages, such as wine, coffee, and tea, are rich sources of vitamins (especially the former) and phytochemicals, such as (poly)phenols, that have demonstrated important antioxidant and anti-inflammatory properties [39]. Dietary (poly)phenols are characterized by a great variety of chemical structures underlying different biochemical properties and functions [40, 41]. Interestingly, several metabolites derived from certain flavonoids (i.e., anthocyanins), phenolic acids, and stilbenes (i.e., resveratrol) are able to cross the blood-brain barrier and directly exert antioxidant and anti-inflammatory action directly in the central nervous system tissues and cells [42]. Additionally, also macronutrients can modulate the immune system depending on various factors to be considered. Certain dietary fats, such as mono- and polyunsaturated fatty acids (MUFA and PUFA, respectively), may have anti-inflammatory effects by limiting saturated fatty acid-induced lipotoxicity, serving as precursors of anti-inflammatory prostaglandin synthesis, improving the synthesis of neurotrophic factors and ultimately limiting the production of mediators of inflammation [43]. These compounds have been shown to provide beneficial effects in the central nervous system by also increasing neuron membrane fluidity/stability and improving neuronal homeostasis positively modulating neurotransmission [44]. Conversely, saturated fatty acids may exert different effects depending on the length of the carbon tails, being short- and medium-chain saturated fats may have immunomodulatory functions providing anti-inflammatory effects [45], while excess of long-chain saturated fats lead to accumulation of lipid intermediates into cells damaging the endoplasmic reticulum and activating the stress sensors [46]. The pro-inflammatory stimulus has been shown to reach the hypothalamic neurons leading to neural activity dysfunction [47, 48]. Among carbohydrates, refined simple sugars have higher glycemic index leading to a faster insulin secretion and spike of blood glucose, which are both associated with an acute inflammatory response [49]. Moreover, refined sugars have been found to directly induce neuroinflammation in certain brain areas (i.e., hippocampus) leading to impairment of their proper functionality [50]. Finally, proteins are important for the production of mediators potentially affecting sleep quality: for instance, sources of protein rich in tryptophan and branched amino acid can modulate the metabolism of melatonin and serotonin [51]. Certain foods, such as milk and fish, also contain oligopeptides that have been reported to exert anti-inflammatory properties by inhibiting stress-induced intracellular pathways activation of the central nervous system [52].
Together with the aforementioned direct mechanisms, nutrient and non-nutrient factors may also indirectly exert pro- or anti-inflammatory effects by playing a role in influencing the gut microbiota and exert effects on the central nervous system through the so-called gut-brain axis [53]. Modifications of the gut microbiota taxonomic configurations and metabolic activities, a condition known as dysbiosis, may affect the permeability of the intestinal mucosa (“leaky gut”), which allow the translocation of lipopolysaccharides from the bacterial cell wall to the bloodstream and stimulation of the inflammatory response by activation of circulating macrophages and monocytes and increase in circulating mediators of inflammation (including the aforementioned TNF-a and IL-1) [54]. Dietary (poly)phenols are shown to be strong regulators of gut microbiota, as they serve as substrate for replication of certain strains with favorable effects for the human host [55]. Moreover, such compounds are heavily transformed into smaller metabolites by the gut microbiota with actual anti-inflammatory effects on vascular and neuronal cells [56, 57]. Among nutrients, complex carbohydrates are not digested by humans while broken down by certain gut bacteria strains and fermented in short-chain fatty acids (SCFA) (such as, acetate, propionate, and butyrate), which demonstrated anti-inflammatory activities through immune system modulation via neuronal and glial signaling [58]. The mechanisms describing the ability of long-chain saturated fats to stimulate the inflammatory response are not clear, but involvement of Toll-like receptors able to specifically recognize bacterial lipopolysaccharides is currently under investigation [59]. Also certain peptides derived by digestion from gut microbiota of different proteins (i.e., depending whether originated from animal or vegetable proteins) are associated with dysbiosis or improvement of oxidant status of the host and contribute to the inflammatory effects of protein [60]. Moreover, excess consumption of protein sources containing choline/lecithin, L-carnitine, and ergothioneine has been related to higher production of a gut-microbiota metabolite trimethylamine N-oxide (TMAO), which has been associated to an increase of pro-inflammatory activities [61].
Concerning the lack of significant findings between DII score and specific sleep features reported in some of the reviewed studies, some hypotheses can be formulated to explain the retrieved null findings. For instance, using subjective or objective (or combined) sleep quality and duration measurements may have led to different results [32]. Pre-existing conditions unrelated to sleep disorders but affecting sleep quality may also have influenced the relation between diet and sleep-related outcomes [33]. Moreover, more general conditions, such as overweight and obesity, are also well-known to affect sleep quality and may also act as mediators for its relation with dietary features [30]. Also, sleep features tend to cluster with other lifestyle factors; some behaviors affecting inflammation, such as current smoking and lack of physical activity, may be considered to potentially explain the null findings [27]. Finally, some differences may be found across age groups, as studies on younger individuals were more likely to report non-significant results [24].
The findings provided in this systematic revision of the literature should be considered taking into account certain limitations. First, the observational design of the original studies included in the review cannot imply a causal effect of the dietary inflammatory potential toward any sleep-related outcome, but only an association. Although a causal relation could not be excluded, the retrieved association may be the result of a more complex pattern of factors that may lead to chronic subclinical low-level inflammatory response (i.e., active smoking, scarce physical activity, etc.) with no actual first cause but all acting in synergy. Second, although most studies accounted for confounding factors, given their observational nature, the presence of potential unmeasured variables cannot be ruled out. Third, several studies had a relatively small sample size that may also explain the null results reported in some studies in case of insufficient statistical power. Forth, despite there is no perfect method nor instrument to retrieve dietary information, most studies estimated nutritional variables through validated FFQs or 24-h recalls, which may both suffer from some limitations (such as, under or overestimation, recall bias, etc.).
In conclusion, despite the findings presented in this study being interesting, evidence of the actual relation between the DII and sleep outcomes would benefit from studies with a more experimental approach. Thus, further intervention studies are warranted to provide evidence of a causal relationship between inflammatory-related dietary factors and sleep parameters. Whether confirmed, clinical interventions to improve diet quality toward healthier dietary patterns and reduce the DII scores could be a future suitable strategy to counteract sleep disturbances and ameliorate sleep health.
Footnotes
Acknowledgments
J.G. was supported by the co-financing of the European Union— FSE-REACT-EU, PON Research and Innovation 2014-2020 DM1062/2021; CUP: E65F21002560001.
Funding
This research received no external funding.
Conflicts of interest
The authors declare no conflicts of interest. J.G. and G.G. is an Editorial Board Member of this journal, but was not involved in the peer-review process nor had access to any information regarding its peer-review.
Author contributions
Conceptualization and methodology, J.G. and G.G.; investigation, J.G. and G.G..; writing— original draft preparation, J.G., G.L., A.M., S.C. and G.G.; writing— review and editing, J.G., G.L., R.F., F.C., S.S.C., I.E.P., A.M., S.C. and G.G.; visualization, J.G.; supervision, G.L., S.C. and G.G.; project administration, G.G. All authors have read and agreed to the published version of the manuscript.
