Abstract
BACKGROUND:
Cafeteria diet (CAF) is a succesful tool for establishing animal obesity model. This study purposed to show immunoexpression and oxidant-antioxidant status in the various organ tissues of Wistar rats fed CAF.
METHODS:
Two groups (six rats per group) of Wistar albino rats were fed CAF and standard chow (control) diets for eight weeks. After experimental procedure, the animals were sacrificed. Intestine, stomach, liver, kidney, spleen, heart, lung, and pancreas tissues were sent to Pathology Department and inducible nitric oxide synthase (iNOS), nuclear factor- κB (NF-κB) and cannabinoid receptor-1 (CB-1) immunoexpressions were assessed. Also, plasma liver function tests and lipid levels were measured total oxidant capacity (TOC) and total antioxidant capacity (TAC) were evaluated in both plasma and liver tissue.
RESULTS:
The immunoexpressions of NF-κB, iNOS and CB-1 were higher in the experiment group for all tissues. TOC was significantly higher in the experiment group, for both plasma and liver tissue (p < 0.001 and p = 0.02). TAC was higher in the experiment group’s plasma measurement (p = 0.02), while there was no difference in the liver tissue between experiment and control groups (p = 0.11).
CONCLUSION:
CAF-induced obesity may be related to increased immunoexpression and oxidative damage in the various organs. Systemic problems should be considered and these phenomenon should be studied more comprehensively.
Introduction
Obesity has become a serious health problem as a result of high-calorie western-style nutrition and increased sedentary lifestyle in the modern world [1]. Nutrition consists of complex mechanisms. Although the nervous system such as the hypothalamus and central neurons have a critical role for apetite, many different factors such as access to food, functional status of the gastrointestinal system, and psychological factors can affect food intake [2]. The transformation of obesity into a serious public health problem has led to an increase in studies in this area to understand the pathophysiology and to find treatments [3, 4]. In addition to cardiovascular adverse effects [5], there is evidence that conditions harmful to the organism such as pro-inflammatory effects and oxidative damage are induced by obesity [6]. However, there are conflicting results in the findings. Carillon et al. [7] showed that oxidative damage in liver tissue increased in the obesity model created by giving the cafeteria diet, but pro-inflammatory marker levels such as interleukin-1 (IL-1), interleukin-6 (IL-6), nuclear factor-κB (NF-κB) did not. Gil-Cardoso et al. [8] also demonstrated an increase in gene expression of proinflammatory markers such as TNF-α and inducible nitric oxide synthase (iNOS) in the intestinal tissues of rats that developed obesity induced by the cafeteria diet, as well as increased oxidative damage. Again, recently, cannabinoid receptor-1 (CB-1), which has both anabolic and inflammatory effects, has been studied in animal models associated with obesity, and findings have been obtained about the development of obesity and its contribution to inflammation [9].
Cafeteria diet is a high-sugar, high-fat, spicy and palatable diet consisting of products such as biscuits, soft drinks, condensed milk and sausages. Cafeteria diet is seen as a successful approach to establish obesity in animal experiments [10].
In addition to diverse results in cafeteria diet-induced obesity models, studies have generally focused on a particular organ or system. No study evaluating multiple organs could be found.
The aim of this study was to investigate the expressions of pro-inflammatory parameters such as NF-κB, iNOS and CB-1 and total oxidant-antioxidant capacities in different organs in the obesity model induced by the cafeteria diet compared to the control group.
Material and method
This study was performed by Scientific and Technological Research Application, Research Center Hüseyin Aytemiz Experimental Research-Application Laboratory at Kíríkkale University, with the approval of Kíríkkale University Animal Experiments Ethics Committee (on 21.01.2021; meeting number: 2021/01 decision no: 03).
Subjects
In this study, 12 male Wistar Albino rats, 2–3 months old and weighing 250–300 g, were used. The rats were cared for in the animal experiments laboratory of Kíríkkale University Scientific and Technological Research and Application Center. The subjects were kept separately in standard wire cages at 25±3C room temperature, 55±10% humidity, and 12 hours constant light and dark cycles. Their examinations and body weight follow-ups were performed periodically.
Experimental protocol
All rats were fed with adaptation food during the 4-week adaptation and preparation period. After the adaptation process was completed, the rats were randomized into two groups with six rats in each group: the study group (cafeteria diet group) (n = 6), and the control group (n = 6). The animals were fed ad libitum, and the food was renewed daily. Control chow diet ® (Kalecik, Kíríkkale, Turkey) was given to the control group. The control diet’s energy content was 2800 kcal/kg while the nutrient distribution was 13% fat, 23% protein and carbohydrate 71%, respectively. The study group was fed with cafeteria diet and water. Data related to cafeteria diet were collected as previously described in the study of Akyol et al [11]. Briefly, the highly energetic and palatable human foods include biscuits, potato and corn crisps, milk chocolate, chocolate bar, kashar cheese, jelly candy, chocolate cake and peanuts. Four of these foods were given in a cup on the cage floor daily in excess quantities. In order to maintain variety two of these foods were replaced with new ones daily. Hence, rats did not have the same foods for more than two consecutive days at a time. Cafeteria diet consisted of control chow diet (% 6 fat, % 71 carbohydrate, % 23 protein, energy 2800 kkal/kg, Kalecik, Kíríkkale, Turkey) with a random selection of highly energetic and palatable human foods. Body weights of all rats were recorded weekly with the same device.
Collection of blood samples and preparing for analysis
At the end of the 8th week, all subjects were properly sacrificed by high-dose anesthetic agents use. The fasting period was set as 12 hours before the operation. The rats were placed on their back and fixed to the operating table from their four extremities. Approximately 5 ml of blood was collected by intracardiac route into heparin tubes. Then, the rats were sacrificed with high dose anesthetic drug injection. The blood taken into heparin tubes was centrifuged at 3000 rpm for 10 min at 4C and plasma was separated. Separated plasma samples were then transferred to eppendorf tubes and stored at –80C until analysis. The livers were removed and taken in 10% formaldehyde solution.
Tissues including intestine, stomach, liver, kidney, spleen, lung, heart and pancreas were macroscopically evaluated and fixed in neutral buffered 10% formalin (pH: 7.2–7.4) for histopathological examination. Also, part of the liver tissues of the rats were separated for TAC-TOC analysis.
Biochemical analysis
Plasma aspartate amino transferase (AST) (EC 2.6.1.1), alkaline fosfatase (ALP) (EC. 3.1.3.1) (Ads, Turkey), alanine amino transferase (ALT) (TECO, USA) (EC 2.6.1.2) activities, albumin, total protein, total cholesterol, triglyceride (Ads, Turkey)levels were determined by spectrophotometer (Shimadzu UV 1700) with commercial kits. The measurements were performed at the Biochemistry Laboratory of the Faculty of Veterinary Medicine, Kirikkale University.
Analysis of total oxidant and antioxidant capacities in the liver tissue and plasma
Tissue (liver) samples were taken after blood was taken from the hearts of the animals at the end of the experiment. Tissue samples were purified from their blood (passing through distilled water and 0.9% NaCl), wrapped in aluminum foil and stored at –80C until analysis.
The liver (0.5–1 g) tissue samples were placed in the Falcon tube and the pH 7.4 buffer solution (9 ml of buffer for 1 g tissue) was added and then homogenized in ice for 1.5 minutes. The tubes were centrifuged at 3000 rpm for 30 minutes, then the supernatant was separated and stored at –80C until analysis.
Total oxidant capacity (TOC) (Erel, 2005) and total antioxidant capacity (TAC) (Erel, 2004) levels in supernatant and plasma were determined by measuring in a microplate reader (Thermo Scientific trademark Multiscan Go) with commercial test kits (Rel Assay Diagnostics, Gaziantep, Turkey).
Histopathological examination
Tissue samples were trimmed, and also processed routinely in automatic tissue processor (Leica, TP1020, Germany). Then, the tissue samples were embedded in paraffin wax (Thermo Shandon, EG 1150, Germany). The paraffin blocks were cut in 5μm thickness at rotary microtome (Shandon, AS325). Microscopic evaluation were performed by an experienced pathologist. The pathologist was blinded to the study. The sections were stained according to haematoxylin-eosin staining procedure and evaluated under a light digital microscope (Olmypus BX51, Japan) and findings were illustrated by camera attachment (Olympus DP5). For scoring of histopathological findings in all tissues, degenerated and necrotic cells were counted totally at 10 High Power Fields (10 HPFs).
Immunohistochemical methods for nuclear factor kappa b (NF-κB), inducible nitric oxide synthethase (iNOS) and cannabinoid receptor1 (CB1)
Nuclear Factor Kappa b (NF-κB) were used for differentiation in altered immune response and for evalation negative effect of CAF diet over cell survival and proliferation. iNOS were used for showing high cellular oxidative stress by CAF diet in all organs. Cannabinoid receptor (CB1) were used for showing power of cellular resistance capacity during high oxidative stress and cellular catastrophe. The tissue samples prepared from the paraffin blocks were sectioned at 5μm thickness on positive charged slides. For showing reactive oxidative substance by iNOS, ROS-related degenerative and necrotic activity in cells by NF-κB, and also Cannabinoid system-associated cellular protection by CB1, an immunoperoxidase technique was utilized. Among the immunoperoxidase tecnique, avidin biotin complex peroxidase (ABC-P) method were followed up accordingly to the instruction manual of kit (Novocastra kit, RE7120K, Leica). Firstly, the tissue sections were deparaffinized in xylene and rehydrated in degraded ethanol series into distilled water. Then, for revealing antigenic determinants in tissues, the sections were placed in citrate buffer-EDTA (pH 6.0, Bioptica, Italy). The tissue sections were kept in a microwave oven at 750 Watt for 25 minutes. Afterthat, endogenous peroxidase activity were released for prevention false positivities. Sections were kept in 3% hydrogen peroxide (H2O2)-methanol solution for 20 minutes. Non-specific protein activity in tissue was prevented using blocking serum (Novocastra- RE7110, Leica). Incubation with polyclonal rabbit iNOS antibody (Abcam,ab3523), polyclonal rabbit NF-Kb antibody (Abcam,ab209799), Rabbit Polyclonal Cannabinoid CB1 antibody, (NovusBio, NLS32) were completed in incubator (Nüve, EN055) at 37C for 60minutes. Biotinylated- and Horse Radish Peroxidase-labelled antibodies (Novocastra kit, RE7120K, Leica) were dripped on sections. Incubation period were waited in incubator at in incubator (Nüve,EN055) at 37C for 45 minutes. Up to end of this process, the tissue sections were always rinsed twice for 5 minutes using Phosphate Buffered Saline (PBS) after the ending of each phase, except for the protein blocking phase. For the negative control sections, PBS was used instead of primary antibody. As a positive control, mice tissues were used for showing cross-reaction. 3,3’-Diaminobenzidine (DAB, Novocastra) as chromogen and Gill’s hematoxylin was used as counterstaining. The glass slides were mounted by coverslip using a non-aqueous mounting medium (Entellan, Merck).
Statistical analysis
Data processing was performed with SPSS 25.0 version (SPPS Inc, Chicago, Illinois USA). The normality of all data was assessed by Shapiro-Wilk test. Independent samples t-test was used to analyse the differences between the two groups. All data were expressed as means±S.E.M (standard errors to the means). For histopathological examinations, the mean and standard error values were obtained by counting 10 fields in x40 objective (Microsoft Excel programme, 2016). Differences between groups and immunoexpressions were evaluated using the Two-way ANOVA method and confirmed by post-hoc Bonfferoni test (GraphPad 5.0, USA). The level of significance was set at p < 0.05.
Results
While the mean body weights of rats in both groups were similar at baseline, the body weights of CAF-fed rats were significantly higher than controls at the end of week 8. AST and ALT levels in the study group (CAF-treated rats) were significantly higher than in the control group at the end of the experiment. Also, albumin level was lower and total cholesterol was higher in the experiment group (Table 1).
Weights and laboratory results after experiment
Weights and laboratory results after experiment
Macroscopically, liver and kidneys of experimental groups (coded by H) was mottled in appearance. Other organs were hyperemic. In contrast to this group, all tissues of control group (coded by K) were normal in appearance except some hyperemic areas in intestine.
Histopathological findings
The histopathological findings of the organs are displayed in Table 2. In histopathological examination of experimental group, intestine and stomach epithels were desquamated or degenerated /necrotic in appearance in many fields. Vessels of propria mucosa and submucosa were hyperemic in this group. The tissues of control group were generally normal in appearance. Degenerated and necrotic hepatocytes covered in almost all zones of liver and tubules epitheliums covered cortical parts of kidney, although there were only a few degenerative and areas in those organs in control group. The degenerative changes belonged to paranchyme-vacuoler degeneration including roughly, confined clear vacuoles and blurred nuclei in hepatocytes and tubule epithels. In addition to this, fatty changes were also present in liver of experimental group. However, there were no conspicuous degeneration and fatty changes in the liver tissue of control group. In the spleen, some lymphoid follicles were hyperplastic in both groups. In the heart tissues, hyalin degeneration were present in dark pinkish cytoplam and blurred nuclei in myocardiocytes of experimental group in contrast to being no meaningfull findings in myocardium of control group. In the lung tissues, emphysema and hyperemic capillaries were observed as well as edema in some lumina of alveol and bronchiol of a few cases in experimantal group. But, there were no distinctive findings in lung tissues of control group. In the pancreatic tissue of experimental group, there were a few of gland epitheliums including paranchyme degeneration. But, in the control group, the number of degenerated gland epitheliums were in fewer area when compared to that of the experimental group.
Mean±standart error (SE) values of degenerative and necrotic cells in all tissues of groups
Mean±standart error (SE) values of degenerative and necrotic cells in all tissues of groups
The expressions were illustrated at Table 3.
Mean±Standard error (SE) values of the immunoexpressions in groups
Mean±Standard error (SE) values of the immunoexpressions in groups
Total oxidant capacity was significantly higher in the CAF group, for both plasma and liver tissue. Total antioxidant capacity was higher in the CAF group’s plasma measurement, while there was no difference in the liver tissue between CAF and control groups. (Table 4).
Plasma and liver tissue TOC and TAOC levels
Plasma and liver tissue TOC and TAOC levels
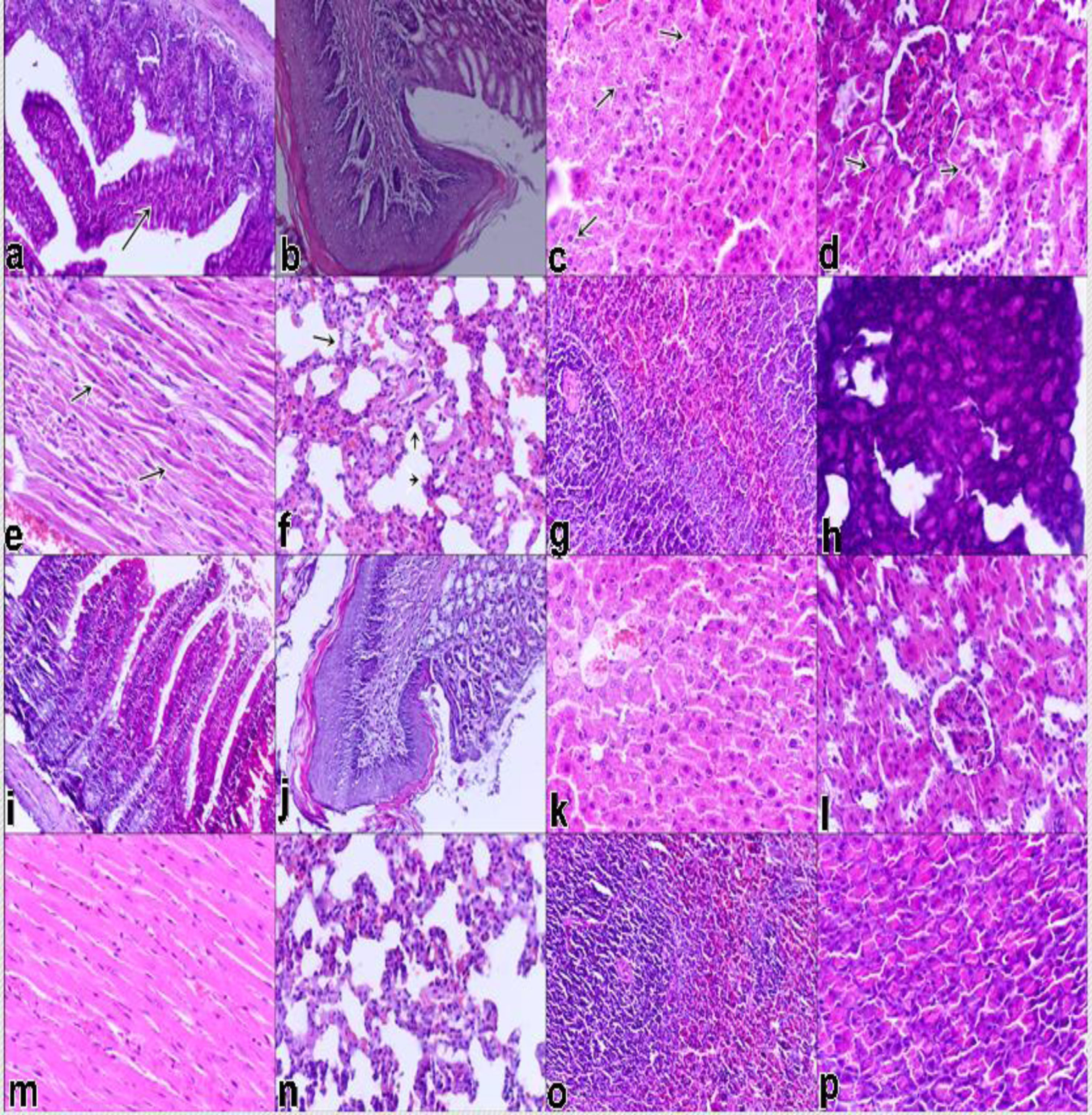
In experimental (H) group, mononuclear cell infiltration in propria mucosa of intestine (a), normal in appearence of gastric mucosa, degeneration in hepatocytes (c), degeneration in tubule epitheliums of kidney (d), hyalin degeneration in myocardiocytes (e), degeneration in alveol epithelium and emphyseme (f), hyperplastic lmyphoid follicles (g), a few degeneration gland epitheliums of pancreas (h), x100 (figure 1a and b), x400 (figure 1c to h), H&E staining. In control group, normal in appearence or no meaningfull changes, x100 (figure 1a and b), x400 (figure 1i to p), H&E staining.
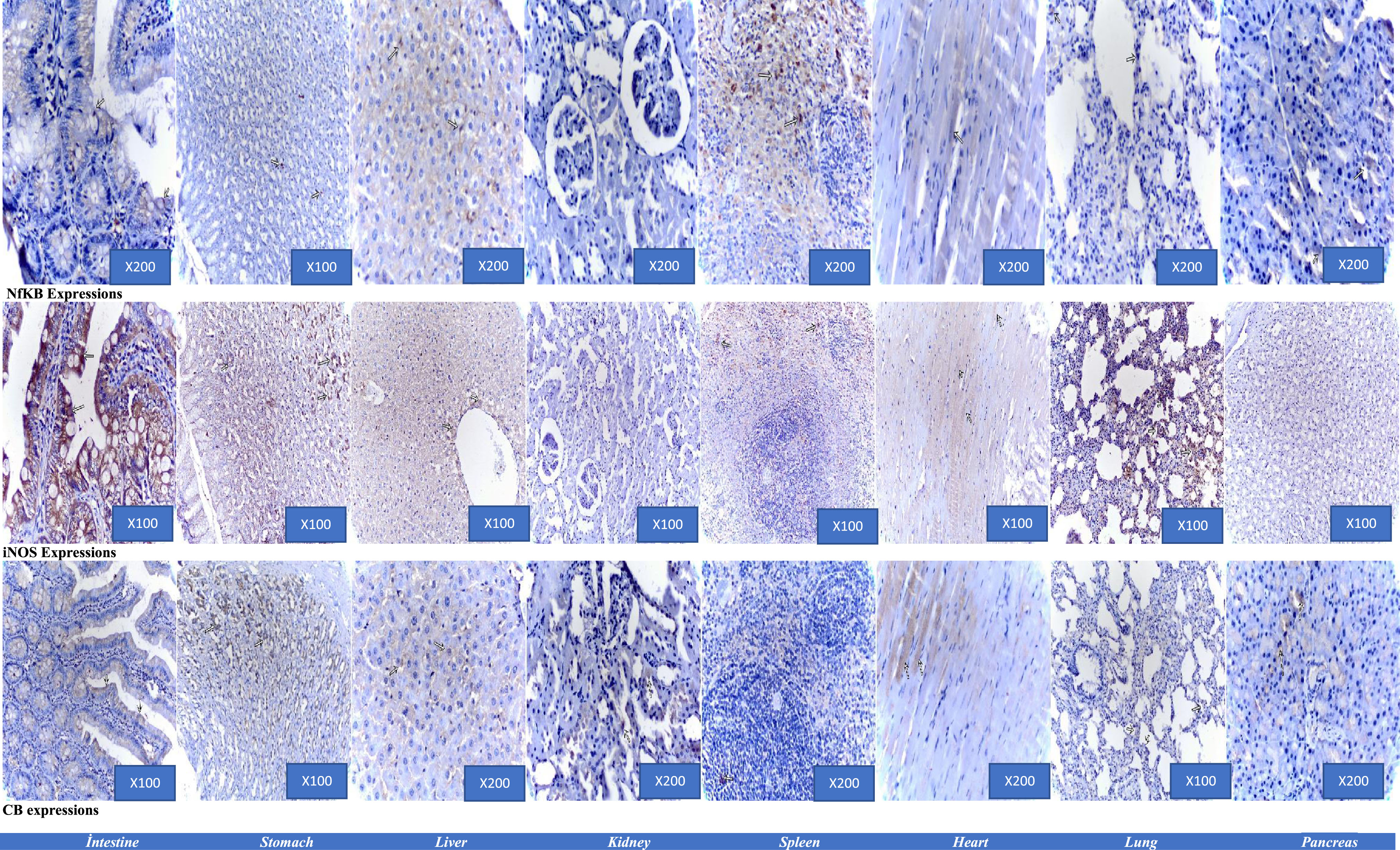
The expressions showing positivities (arrows), ABC-P.
The present study revealed that cafeteria diet induced obesity can lead to increased immunoexpression in the various tissues and increased plasma oxidative stress. In plasma, Increases in the TAC levels would be a compensatory nature responding to the deteriorating oxidant/antioxidant balance in the liver. In liver tissue, the CAF group had increased levels of plasma TOC, a marker of oxidative stress and an indicative of lipid peroxidation of ROS in the cell membrane. Reduced TAC level in the present study can be due to the increased lipid peroxidation.
CAF-induced obesity may lead to multi-organ damage. Zeeni et al [12] concluded that CAF was related to increased inflammation via serum IL-6 levels, and histopathologic organ damages. Another study showed that CAF can mimic the human metabolic syndrome model of liver and adipose tissue inflammation with macrophage infiltration, as well as obesity and glucose intolerance in Wistar rats [13]. Accordingly, high fat diet may induce tissue oxidative stress. Obese rats had higher oxidative damage parameters, such as malondialdehyde and protein carbonyl levels in the hepatic, renal and cardiac tissues [14]. Our data may support these results with higher liver function test results, lower albumin level and higher cholesterol levels in the CAF group.
Gil-Cardoso et al [8] studied the results of cafeteria diet on intestinal inflammation and oxidative damage. Their findings agree with the results of this study. They found that cafeteria diet-induced obesity could cause the expression of proinflammatory genes, such as tumor necrosis factor-α and iNOS. It could also raise myeloperoxidase activity and reactive oxygen species. Additionally, they showed that interstinal permability genes were expressed higher in the CAF group. Another study showed ileal dysfunction via increased plasma TNF-α levels and increased myeloperoxidase activity of ileal tissue in Wistar rats after feeding of 17-week CAF diet [15].
La Russa et al [16] conducted a study that assessed the effect of CAF induced metabolic syndrome on kidneys. They revealed that CAF had pro-oxidant effect and could deteriorate the antioxidant capacity (by reduction of plasmatic antioxidant capacity and downregulating of cytoplasmic antioxidant enzyme expression) and activate apopototic pathways such as caspase-8 and caspase-9. Another study concluded that CAF diet fed for 6-weeks increased the expression of inflammatory cytokines, such as TNF-α and IL-6 and plasma advance glycation end products. However, the plasma levels of these cytokines or kidney levels of them histologically did not change [17]. Our results do not suggest this.
There is some evidence for the effect of CAF on the central nervous system. It has been shown that Alzheimer-like models can be developed, Aβ amyloid accumulation and oxidative stress in the brain can be increased by CAF or high-fat diet. Cognitive and non-cognitive behaviours may also be affected [18–20].
There are few experiments about CAF and spleen. It was shown that CAF may cause splenocyte damage, decrease of immun response via lower production of ROS from macrophages, and decrease of specific T cell subsets [21, 22].
NF-κB is pivotal transcription factor for inflammation and oxidative stress [23, 24]. Carillon et al [7] concluded that CAF was related to oxidative stress, but they did not find any enhancement of inflammatory markers, such as IL-6, TNF-α and NF-κB. Their findings are in contrast with those of the present study, however there are some methodological differences in the study.
There are few reports about feeding and iNOS expression. A study that evaluated the effect of fermented mulberry leaves on hepatic steatosis, showed that high fat diet could lead to increase in inflammatory cytokines and mediators, such as iNOS, cyclooxigenase-2, IL-1β, IL-6, TNF-α and NF-κB [25].
CB1 receptors may have a pivotal role in the hedonic and rewarding aspects of eating behaviour, also they are widely distributed in the various tissues, especially central and peripheral nervous system. The evidences suggest CB1 receptors are critical factor in the development of metabolic abnormalities. A study showed that CB1 receptors were required for hepatic steatosis and metabolic syndrome model in mice [26]. CB1 receptors were studied as a target for weight control, improving metabolic parameters and preventin obesity with several experiments [9, 28]. But there are controversial results with peripheral receptors. A recent study concluded that chronic high fat diet might decrease the immunocontent of CB1 receptor, also motivational impairment for palatable feding [29].
Our study has several limitations. A few parameter was evaluted for immunoexpression. Other immun system parameters, such as cytokine levels could not be measured. Oxidation reactions were measured by one method. However, the evaluation of multiple organs was the major strength of this study.
In conclusion, cafeteria-diet induced obesity may increase immunoexpression and oxidative damage in the various organs. This data may be helpful for further investigations about the negative effects of obesity and cafeteria diet. Targeting these pathways may be a new insight for treatment.
Conflict of interest and funding
The authors declare there is no conflict of interest and no funding.
