Abstract
Renal cell carcinoma (RCC) makes up most adult kidney cancers. The classic presentation is a spherical, well-circumscribed mass, but fewer subtypes present as more infiltrative neoplasms. Both growth patterns can affect tumor staging and prognosis. Accordingly, this review will discuss the current status of staging, grading, and pathologic prognostication in RCC. Typically, the most important prognostic parameter in RCC is tumor stage. Pathologic staging categories (i.e., pT) are influenced by tumor size (pT1-pT2) and invasion of structures including renal sinus, perinephric fat, the renal vein or segmental branches, vena cava, or adjacent organs/structures (≥pT3). This invasion is prognostically important but can prove difficult to diagnose, due to the more common nodular rather than infiltrative pattern of RCC. In clear cell RCC, more than half of tumors over 5 cm and over 90% of tumors over 7 cm invade the renal sinus soft tissue, such that true pT2 clear cell RCC is rare. However, the size to invasion relationship is less clear for other RCC subtypes, some of which become large without invading structures (e.g., papillary RCC, chromophobe RCC), and others of which are infiltrative irrespective of size (e.g., renal medullary carcinoma, FH-deficient RCC). Histologic grading in recent years has evolved to focus mostly on prominence of the nucleolus. Other emerging assessments in RCC include tumor necrosis as a prognostic parameter (predominantly for clear cell RCC), architectural grades in clear cell RCC, and specific growth patterns in papillary RCC, such as microcystic growth. Overall, this review is aimed to serve as a guideline to the staging and grading of RCC in clinical practice, as well as highlight the potential challenges and pitfalls in this crucial interface between clinical management and diagnostic pathology.
INTRODUCTION
The incidence of renal cell carcinoma (RCC) has more than doubled in the developed world over the past 50 years or so, and it is currently the 9th most common neoplasm in the United States [1]. It accounts for over 80% of neoplasms arising from the kidney, and accurate staging is crucial for clinical decision making and prognostication. The American Joint Committee on Cancer (AJCC) and Union for International Cancer Control (UICC) staging is periodically revised to reflect the prognostic categories based upon the size and anatomic extent of cancer, which have been shown to be the more crucial considerations when determining a patient’s prognosis [1–3] (Table 1). Prototypical cancer, in many organs, exhibits features like nuclear pleomorphism, irregular tumor shape, mitotic figures, infiltrative growth, desmoplastic stroma, and other such findings; however, RCC is an exception to many of these rules, often being circumscribed and round, composed of monomorphic cells, and not necessarily having brisk mitotic activity. In contrast to desmoplasia, tumors have nodular outpouching into blood vessels, typically with minimal or no stromal reaction and maintaining the endothelial layer around the intravascular tumor [4–6]. The unique findings in RCC are additionally confounded given the different characteristics of each subtype of RCC and their predilection for different manners of invasion; for example, clear cell RCC has a predilection for expansile and intravenous growth [7, 8], whereas other tumor types, like SMARCB1-deficient renal medullary carcinoma (RMC), are more directly infiltrative of the renal parenchyma. Although many RCCs are identified incidentally with imaging for other reasons (e.g., diverticulosis, staging of another tumor, etc), RCC tends to remain clinically silent (i.e., asymptomatic or associated with non-specific symptoms such as back pain) for most of its course, meaning that these tumors can grow to more advanced staged by the time they are recognized and removed from the patient. Recognizing higher stage parameters in RCC is important, not only for patient prognostication [4], but also for consideration of enrollment in clinical trials for higher-risk renal cancers.
Summary of AJCC/UICC staging of renal cell carcinoma.

TUMOR SIZE AND pT1/pT2 STAGING
The primary size of the tumor remains one of the most important prognostic factors for RCC [7, 8]. The pT stage category for pT1 versus pT2 RCC is driven by tumor size: pT1a is defined as tumor size ≤4 cm and pT1b as >4 to 7 cm. Tumors measuring >7 to 10 cm are pT2a, and those >10 cm without extrarenal extension are pT2b. This results from the well-established observation that the behavior of RCCs is directly proportional to its size [9–16]. However, there is also significant correlation between tumor size and invasion, such that the two features are difficult to fully separate. For example, the likelihood of extrarenal extension, including, but not limited to renal sinus invasion, increases dramatically with tumor size to the point that very few clear cell RCC tumors are truly organ-confined at size >7 cm. As such, a substantial number of the historical clear cell tumors reported as pT2 may have undiagnosed sinus invasion, making them in reality pT3a [17–20]. Bonsib found that only 32% of clear cell RCCs over 4 cm and only 3% of tumors over 7 cm were limited to the kidney, in a cohort carefully examined grossly and microscopically by the same genitourinary pathologist [18]. However, the correlation of size with invasion is weaker for other RCC types, like chromophobe or papillary RCC (Fig. 1A), for which tumors sometimes reach a large size without invading the sinus [18, 21].
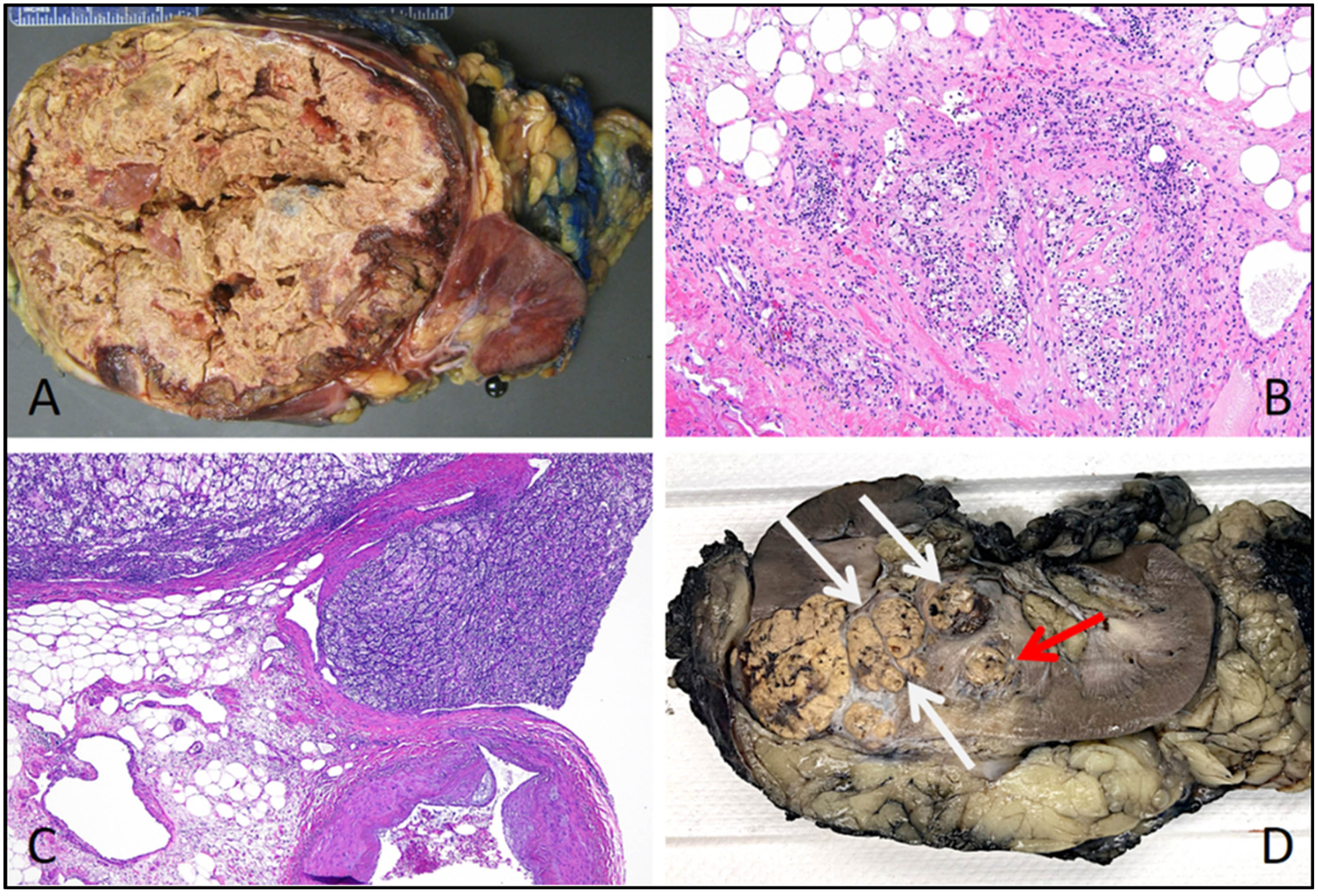
A) This papillary renal cell carcinoma is very large and effaces much of the kidney; however, it remains round and well-circumscribed without obvious infiltration/extension into perinephric or hilar soft tissues. B) Renal sinus invasion in this clear cell renal cell carcinoma manifests as tumor cells directly intermingling with fat. C) This renal cell carcinoma nodule extends into a large vein branch in the renal sinus. D) In this clear cell renal cell carcinoma, the primary tumor is at the far left; there is also extensive vein involvement heading toward the main renal vein (white arrows) and a nodule of retrograde venous invasion (red arrow) appearing as a discontinuous nodule due to backward spread along other vein branches.
Tumor measurements have also been reported to differ between imaging and the pathologic specimen (both unfixed and fixed). In general, imaging measurements appear to be slightly larger than those of the pathologic specimen, possibly with some further reduction in size after formalin fixation. However, the magnitude of these discrepancies was as little as 1–3 mm in some studies [22–33]. When there is a potential discrepancy between clinical or imaging-based tumor size and pathologic specimen, it may be relevant to re-check the pathologic measurements. For example, reapproximating the tumor slices may reveal that the greatest dimension is perpendicular to the plane of section, or adjacent benign cysts may appear part of the tumor by imaging but not by histology.
In regard to the latter, rare tumors may be extensively cystic, with only a solid nodule localized to one area. It is not totally agreed upon whether the cyst should be included in the measurement in this situation. In an interobserver agreement study [5], most of the participating genitourinary pathologists responded that they would use the entire cyst size if the cyst lining contained neoplastic cells morphologically similar to those of the tumor. However, if the cyst appeared morphologically different, resembling a benign cortical cyst with cells dissimilar from the tumor, it was concluded that only the solid component of the neoplasm should be taken into consideration for staging.
pT3 STAGING
In general, pT3 stage is associated with tumor extension beyond the kidney parenchyma. This can occur when tumor extends peripherally into perinephric adipose tissue, centrally into hilar soft tissue or into the pelvicalyceal system, or into the renal vein/inferior vena cava.
Renal sinus invasion
The renal sinus is the central fatty compartment surrounding the hilar vascular structures and the collecting system. Invasion of this tissue, which has no significant capsular barrier from the kidney, has been considered a form of pT3a disease since the 2002 AJCC system [17]. Extensive work by Bonsib highlighted the importance of invasion of the renal sinus, especially in clear cell RCC [17–20]. It was shown that the likelihood of invasion at this site increases dramatically with increasing tumor size [18, 21]. Therefore, particular consideration should be given to larger tumors, especially clear cell RCC, sampling several tissue blocks of the sinus-tumor interface, when tumors exceed 4 or 5 cm in size [21]. In addition to direct infiltration of tumor into fat (Fig. 1B), consensus is that any lymphovascular invasion in the renal sinus can be considered renal sinus invasion, whether it be large vein nodules (Fig. 1C) or microscopic lymphovascular invasion [34]. Older staging schemes referred to “grossly-visible” vein branches or “muscle-containing” vein branches; however, these limitations are no longer used, since vein branches may be identified microscopically and may not necessarily have muscle [35]. An impressive study revisited the gross specimens from patients with originally-diagnosed pT1 RCC who later died of metastases [4]. In most settings, such a study would be impossible, since, for example, United States regulations require that residual gross specimens be kept only for a few weeks after final pathology reporting, not for many years as it would require to reproduce this study. Nevertheless, in this unique study, the authors found a substantial rate of undiagnosed renal sinus or vascular invasion in this cohort when re-examining the gross specimen, compared to a control group of patients who had pT1 tumors and did not die of metastatic RCC [4], suggesting that even subtle invasion is prognostically important.
Renal vein and tributary invasion
RCC has a tendency to invade vein branches as what has been described as venous tumor “thrombi”. This form of extension can spread beyond the renal vein into the inferior vena cava or even extending up to the level of the heart via the superior vena cava. Despite this conceptually extensive disease, the intravascular tumor can sometimes still be removed from the vein, since the “tumor thrombus” may be unattached to the vein wall, like a finger in a glove. Venous nodules other than in the main renal vein can exhibit a rounded contour and can be underappreciated and potentially misinterpreted as tumor multinodularity instead of vascular invasion [36]. Accordingly, we recommend that any deviation (i.e. outpouching) from the main spherical tumor mass or an oval shaped tumor projection be viewed with extreme concern for vascular involvement, considering this potentially subtle form of invasion compared to that of other non-renal cancers. Any level of venous invasion less than vena cava involvement is currently considered pT3a. However, some authors have found that involvement of the main renal vein (visible at the hilum) is more significant than segmental vein branch invasion [37].
Since RCC tends to involve large veins, nephrectomy specimens may contain a “tumor thrombus” protruding from the en face vein margin of the pathologic specimen. Current consensus is that this should not be considered a positive renal vein resection margin unless the tumor is microscopically adherent to the vein wall at the final margin [34, 36]. Documentation should be made whether or not tumor invades the renal vein wall at the resection margin, as this can affect clinical trial eligibility; however, mentioning the presence or absence of vein wall invasion at sites other than the margin is not necessary, except for discriminating pT3b from pT3c (vena cava wall invasion).
Retrograde venous invasion
A phenomenon that may mimic tumor multifocality has been referred to as “retrograde venous invasion” (Fig. 1D). In this situation, the main renal vein is occluded by tumor, and tumor begins to spread ‘backwards’ into smaller vein branches within the kidney, forming multiple nodules, “satellite” lesions, confluent multinodular masses, or smaller nodules away from the main mass. This is thought to occur in 5–8% of RCCs, mostly clear cell type [21, 38]. Overall, we recommend that the combination of a large tumor and multiple smaller tumors with similar histological features be viewed with concern for this phenomenon. In contrast, sometimes secondary smaller tumors may be of different histology than the main mass, such as a main clear cell RCC and multiple benign papillary adenomas or clear cell papillary renal cell tumors (formerly known as clear cell papillary RCC) [21, 39], which would represent true multifocality of various tumor types. Of course, multiple clear cell RCC can occur in the setting of von Hippel-Lindau disease, but outside of this setting, this is rare.
Vena cava involvement
Higher pT3 stage categories are driven by involvement of the vena cava. pT3b includes RCC with extension to the vena cava below the diaphragm and pT3c represents extension above the diaphragm or invasion of the vena cava wall below or above the diaphragm. It may not be apparent from a conventional nephrectomy specimen whether tumor is in the main renal vein or the vena cava. Therefore, correlation between pathology and operative findings is helpful. When vena cava tumor thrombectomy (i.e., pT3b tumor) is provided as a separate specimen, histologic examination should be performed to evaluate for tumor invasion into fragments of vein wall (if present) to diagnose or exclude pT3c; however, this can be challenging, since these specimens are often fragmented and unoriented.
Perinephric fat invasion
Perinephric (i.e., peripheral) fat invasion also yields a pT3a stage designation; however, some authors suggest that since the renal capsule is a more formidable barrier, this is often a later event than renal sinus invasion, occurring in addition to sinus invasion rather than as an early event [21]; of course, a peripheral versus centrally-based mass may also have an effect on the location of direct tumor extension. Perinephric invasion can be more controversial, as tumors can bulge significantly beyond the normal contour of the kidney without an infiltrative pattern. Overall, there is mostly agreement that this is insufficient to diagnose perinephric soft tissue invasion, particularly if the mass remains spherical without smaller outpouchings [34, 36]. Findings that are considered more definitive for perinephric invasion include direct infiltration of tumor cells into fat, secondary smaller nodules in the fat, or mushroom-shaped herniation of tumor into fat (Fig. 2A). Some authors suggest that there may be different prognostic implications for perinephric invasion vs sinus invasion [40]. A few scenarios in perinephric invasion remain controversial: 1) there is a lack of robust criteria to determine the difference between biopsy site artifact, in which tumor cells may be displaced into the fat by the biopsy needle, vs true perinephric invasion; And 2) other neoplasms, such as oncocytoma, may intermingle with perinephric fat, yet they are still considered benign. As such, it is less clear if perinephric involvement conveys adverse prognosis in oncocytic RCC like eosinophilic chromophobe type.

A) This clear cell renal cell carcinoma shows a mushroom-like nodule of tumor beyond the contour of the main mass, extending into the perinephric fat (red arrow). B) In this partial nephrectomy specimen, a tumor nodule is evident bulging from the renal parenchymal surface of the specimen (red arrow), likely representing an intravenous nodule exposed by the resection; although this may abut the pathologic margin, intraoperative correlation may demonstrate no residual tumor in the resection bed. C) This clear cell renal cell carcinoma demonstrates abrupt transition from grade 2 (bottom) to grade 3 (top), with cells having more voluminous cytoplasm and prominent nucleoli. D) Microcystic features are an emerging finding correlated with extrarenal extension and metastatic disease in papillary renal cell carcinoma. This tumor has multiple cystic spaces containing tumor cells at right and transitions into a poorly-differentiated/high grade malignancy at left.
Pelvicalyceal system invasion
Invasion of the pelvicalyceal system by RCC was added as an additional route to pT3a as of the 2016 AJCC classification guidelines [35, 41–43]. Although less commonly found, when present this novel criterion is applied in the same fashion as renal vein invasion; any nodule of tumor within the collecting system would be considered pT3a. However, questions still exist regarding bulging into (with an intact overlying urothelium) versus breaking through into the collecting system, and this remains an area of investigation.
Tumors that are inherently infiltrative
Although the classic RCC is round/spherical, with finger-like outpouchings into veins or renal sinus, rare tumor types are infiltrative, more akin to the prototypical cancer. In particular these include, but are not limited to, SMARCB1-deficient renal medullary carcinoma, a subset of fumarate hydratase (FH)-deficient RCC (often associated with hereditary leiomyomatosis and renal cell carcinoma syndrome), and collecting duct carcinoma [44–54]. These tumor types are more likely to infiltrate around and entrap normal structures like tubules and glomeruli (nephrons), or show direct infiltration into fat.
High stage in partial nephrectomy
Although invasion of structures like vein branches or renal sinus are potentially unsurprising in radical nephrectomy specimens, these findings can sometimes be also present in partial nephrectomy specimens [55–63], in which a polypoid “tumor thrombus” may protrude into a vein lumen near the hilum (Fig. 2B) or focal renal sinus tissue may be present and involved by tumor. In the case of tumor protruding out of a vein at the margin, technically the tumor may abut the margin; however, it may intraoperatively appear that there is no further tumor in the patient. Therefore, correlation between pathology and intraoperative findings may be helpful in these patients. Our approach is to note that tumor is present in the vein lumen at the margin in this scenario, to convey that it does not appear to be represent actual transection of the main tumor.
“Invasion” in benign tumors
Although renal sinus, perinephric fat, and vein branch involvement are considered high-stage findings in RCC, these findings have been reported in benign tumors, in which they do not appear to change the benign behavior of the neoplasm. As benign tumors are not staged, the designation of pT3 is not used in these circumstances. In oncocytoma, cells intermingling with the perinephric fat are thought to have no significance [5, 64–67]. Less literature exists for oncocytoma with renal sinus involvement; however, this also has not been described to affect prognosis [64]. Oncocytomas have also been reported to involve the renal vein or branches, and again no impact on behavior has been noted [68, 69]. This can be problematic, as oncocytoma and chromophobe RCC are known to overlap. As such, a tumor in this spectrum with vascular involvement could be either a benign tumor or a pT3a renal cancer. Although less common, benign renal angiomyolipoma has rarely been reported to have a renal vein “tumor thrombus” with no effect on prognosis; however, in this setting an epithelioid and/or malignant perivascular epithelioid tumor (PEComa), another member in the AML family, should be excluded [70–72]. Lastly, a recent study has shown that clear cell (tubulo)papillary renal tumors can extend into the renal vein or adipose tissue with no apparent effect on prognosis [73].
LYMPHATIC INVASION
Though lymphatic invasion is not a staging parameter in the AJCC or UICC staging systems [35], it is recommended that it be reported [74]. However, various studies have used different definitions for lymphatic invasion that confound definitive consideration as a prognostic parameter [75–78]. For example, some authors have included intratumoral lymphatic invasion, which many would consider almost impossible to assess given the extreme vascularity of RCC, given that almost all tumor cells are invested by capillaries. Conversely, others have included renal sinus lymphatic invasion in these studies, which by current convention would be considered renal sinus invasion/pT3a [34]. We would consider non-pT3a lymphatic invasion to represent microscopic foci of intravascular tumor, typically at the leading edge of the mass but within the kidney, in spaces that are unequivocally below the size threshold to be considered a vein branch. It is important to remember that some renal vein branches have inconspicuous smooth muscle in their walls despite being grossly visible/dissectible, so larger intravascular foci with an accompanying artery may be considered vein branch invasion (pT3a).
pT4 STAGING
The pT4 category is defined as when the main tumor grows beyond the Gerota fascia- a thin collagenous layer of connective tissue that surrounds the kidneys and adrenal glands that joins with the posterior renal fascia to separate the kidneys from other organs. The Gerota fascia is not obvious via histologic evaluation, so direct invasion into surrounding structures (e.g., liver, bowel, etc) is consistent with pT4 disease. pT4 also includes the situation where tumor is growing directly into the ipsilateral adrenal gland [79, 80]. Thorough gross evaluation is important to give macroscopic assessment of the relationship of the tumor to the adrenal gland, as a noncontiguous nodule in the adrenal gland is instead regarded as metastasis (pM1). It should be noted that incidental benign adrenal cortical nodules and adenomas are not uncommon, and may have a similar gross appearance to RCC (yellow-orange) and a similar microscopic appearance with cells containing clear cytoplasm in a nested arrangement. Simple immunohistochemistry with PAX8 and SF1 antibodies can be used to support diagnosis of neoplasms as primary to the kidney and adrenal gland, respectively.
LYMPH NODES (N)
In current practice, lymph nodes are not routinely dissected by urologists in all RCC patients [81, 82], as it is not clear that this benefits cancer control [83, 84]. There is some evidence that extranodal extension of tumor may be more important than number of lymph nodes involved [85], so lymph node dissection is typically performed selectively [82]. In routine nephrectomy specimens, the hilar area usually contains no lymph nodes, or less commonly 1–2 [34]. Mehta et al. found metastases only in grossly recognized lymph nodes, suggesting that random microscopic samples of hilar fat for lymph node search may be unnecessary. Interestingly, non-hilar lymph nodes were also found, with a lower rate of positivity (65% hilar, 16% nonhilar) [86]. Previous staging systems had different N categories based on number of lymph nodes and size of metastases; however, the current systems for RCC have only a single pN1 category for involvement of one or more regional lymph nodes [35].
DISTANT METASTASES (M)
RCC, especially clear cell RCC, is known to have somewhat surprising metastatic behavior, sometimes with unusual metastatic sites, such as the gallbladder or pancreas. Other tumors, such as papillary RCC and translocation RCC may be more prone to spread to lymph nodes. Metastases can also occur years to decades after initial diagnosis, requiring long term follow-up. As mentioned, adrenal gland involvement that is not continuous with the primary tumor is also considered pM1.
HISTOLOGIC GRADING
In recent practice, the Fuhrman grading system has been replaced with the ISUP/WHO grading system. In this system, the focus for histologic grading is the prominence of the nucleolus, and a helpful starting point is 100× magnification (10× microscope objective). If nucleoli are prominent at this magnification, then grade 3 is assigned, whereas nucleoli prominent only at higher levels of magnification is more consistent with grade 2, and entirely inconspicuous nucleoli are grade 1. Grade 4 encompasses extreme nuclear pleomorphism, sarcomatoid and/or rhabdoid features [87–89].
Some areas of uncertainty that remain in histologic grading include variation in the degree of nucleolar prominence required to assign the higher grade. Whereas some references indicate that the nucleolus should be prominent “and eosinophilic,” most genitourinary pathologists would assign the higher grade if the nucleolus is visible but not necessary eosinophilic [90]. The presence of multinuclear tumor cells is also not well agreed upon. Giant cells with bizarre multilobate nuclei are considered grade 4; however, whether cells with more than one small round/oval nucleus automatically reflects grade 4 is debatable [91].
Grading is also not applied to all RCC subtypes. Grade is best validated in clear cell and papillary RCC, and it is debatable whether to use the ISUP/WHO system for other tumor types. For chromophobe RCC, grading as “not applicable” is recommended, since these tumors inherently have atypical nuclei, but their behavior is usually favorable [92, 93]. Other grading systems have been proposed for chromophobe RCC, incorporating features such as nuclear overlap and necrosis, but these have not gained widespread use thus far and are not required unless desired for institutional practices [94, 95]. The introduction to the 2022 WHO Classification notes that grade may not be useful for TFE3 rearranged RCC, and may be misleading for certain tumors types, such as [pure] tubulocystic carcinoma, acquired cystic kidney disease-associated RCC, eosinophilic solid and cystic RCC, and the familial RCCs such as FH-deficient RCC and succinate dehydrogenase (SDH)-deficient RCC. In these tumors, nucleoli are characteristically prominent by their nature; however, prognosis is variable and based on tumor type more than nuclear grade [96].
OTHER PROGNOSTIC PARAMETERS
Tumor necrosis
Tumor necrosis has also gained interest as a prognostic parameter [85, 97–99]. Currently, it is recommended to report the presence of necrosis or absence of necrosis as a prognostic indictor in RCC; however, most data are derived from studies in clear cell RCC, and it is unclear if these findings are as relevant in other RCC subtypes. The percent of tumor necrosis is not proven to be relevant clinically, especially since reproducible estimation would require a combination of gross and microscopic findings. General pathology practices would suggest that samples be typically taken either to avoid necrosis or show the junction with viable tumor, so microscopic evaluation alone would often be an underestimate. Secondly, areas thought to represent necrosis grossly are sometimes actually hemorrhage. One group proposed to combine necrosis and histologic grading into a single parameter. Grades in this scheme were as follows: grade 1 includes ISUP grade 1 and ISUP grade 2 without necrosis; grade 2 includes ISUP grade 2 with necrosis and ISUP grade 3 without necrosis; grade 3 includes ISUP grade 3 with necrosis and ISUP grade 4 without necrosis; grade 4 includes ISUP grade 4 with necrosis or sarcomatoid/rhabdoid tumors. The study found a significant difference in survival between each of the grades for clear cell RCC, but not papillary RCC or chromophobe RCC [97]. However, this system has not gained widespread use at present. There has also been interest in refining the definition of necrosis. Clear cell RCC often has zones of hyalinization or hemorrhage, which should not be considered necrosis. “Granular necrosis” has been proposed by some as a more specific definition of prognostically important tumor necrosis [100]. Additionally, it is unclear if necrosis following neoadjuvant therapy, which is now being used in a subset of clinical trial patients prior to resection, has the same prognostic significance as spontaneous necrosis.
Architectural patterns
An emerging area of prognostication in RCC is tumor grading by architectural patterns rather than nuclear features; however, this approach is currently not standard of care and is considered exploratory at this time. In clear cell RCC, groups of different architectural patterns have been proposed for clustering into architectural grades, with some low-grade patterns including tumors that resembling clear cell (tubulo)papillary tumor (i.e., cystic and/or tubular growth patterns) or having regressive features; intermediate grade patterns may have papillary, oncocytic, or inflammatory morphology; and high-grade patterns include rhabdoid, giant cell, and infiltrative morphologies [101]. For papillary RCC, recent work has suggested that “unfavorable architecture” may include solid, micropapillary, and hobnail patterns [102]. Subsequently, another group proposed refinement of this classification to focus on microcystic growth, which appears strongly associated with extrarenal extension and metastatic disease and appears more reproducible [103].
SUMMARY
RCC represents a heterogeneous group of tumors with some unique clinical and pathologic characteristics, such as invasion manifesting as rounded tumor outpouchings and extension into vascular structures with an intact endothelial cell layer. Careful attention to tumor size drives the pT1 and pT2 categories; however, one should be cautious diagnosing pT2 clear cell RCC, as tumors of this type and size overwhelmingly have renal sinus invasion, making them pT3a. In contrast, other tumor histologies may reach a large size without necessarily invading (e.g., chromophobe RCC, papillary RCC), or may be infiltrative regardless of size (e.g., medullary renal carcinoma, collecting duct carcinoma, FH-deficient RCC). Deviation from a spherical shape should raise significant concern for venous or soft tissue invasion. ISUP/WHO grading is now based on nucleolar prominence rather than nuclear size and shape, and emerging prognostic parameters include necrosis and architectural patterns.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments.
FUNDING
The authors report no funding.
AUTHOR CONTRIBUTIONS
SW: Conception, interpretation of the data, editing, and final approval. NZ: preparation of draft, analysis of data, and final approval.
CONFLICTS OF INTEREST
NZ and SW have no conflicts of interest to report.
