Abstract
Papillary renal cell carcinoma (pRCC) represents the second most common renal cell carcinoma. Diagnosis of pRCC has been challenged by emergence of several novel renal tumor entities with papillary morphology. Some of these have been found to constitute independent entities with specific molecular drivers, e.g. fumarate hydratase deficient renal cell carcinoma or
EVOLUTION OF PAPILLARY RENAL CARCINOMA CLASSIFICATION
Papillary renal cell carcinoma (pRCC) represents the second most common type of renal cell carcinoma (RCC) after clear cell renal cell carcinoma (ccRCC), building up to 10-20% of renal epithelial tumors [1, 2]. They may be multifocal and bilateral, especially in the background of chronic kidney disease [1] and in patients with end stage renal disease [3]. A subset of pRCC occurs in the setting of hereditary pRCC with a germline mutation of
pRCC were already reported in the first classification attempts by Ewing 1919, Melicow 1944 and Fite 1945 (for review see [5]), but terminology and diagnosis of this tumor type with his characteristic histological growth pattern has been modified over the last decades. In many classification proposals, different renal neoplasias were grouped together, regardless of histological architecture (papillary) or cytoplasmic staining (clear tumor cells). Manzilla Jimenez et al. described specific clinical and morphological features of papillary RCC and compared them with clear cell RCC in 1976 [6]. Thoenes et al. proposed in 1986 the Mainz renal tumor classification, based on cytoplasmic staining features [7]. They separated “chromophilic” RCC with mainly papillary architecture from clear cell and chromophobe RCC and proposed that “chromophilic” RCC originate from proximal tubule. In their classification, a basophilic small cell type and an eosinophilic (granular) cell type of “chromophilic” RCC were distinguished. Cytogenetic analysis of such tumors performed by Kovacs et al. showed characteristic trisomy and tetrasomy of chromosomes 7 and 17, and loss of the Y chromosome [8]. The validation of different renal tumor categories in the Mainz classification by these cytogenetic studies led to attempts for implementation of molecular findings in renal tumor classification.
Later, the Heidelberg classification [9] and Rochester Consensus Conference in 1997 formed the basis for the 2004 World Health Organization (WHO) classification with pRCC as an entity. Delahunt and Eble described in 1997 two major patterns of pRCC: one composed of small cells with scant basophilic to amphophilic cytoplasm and thin papillae lined by one row of tumor cells with inconspicuous nucleoli, frequently showing aggregates of foamy histiocytes and scattered psammomatous calcifications (type 1 pRCC), and another composed of cells with densely eosinophilic cytoplasm, larger nuclei with prominent nucleoli and more complex papillae with stratification of tumor cells (type 2 pRCC) [10, 11]. This was supported by molecular studies, describing that type 1 pRCC showed more consistent and uniform gains in chromosome 7 (encoding the
In the following years, various new renal tumor entities with papillary growth were introduced and tumors with an unequivocal genotype-phenotype correlation were better described, e.g. fumarate hydratase-deficient RCC, mucinous tubular and spindle cell RCC (MTSCC), tubulocystic RCC and
DIFFERENTIAL DIAGNOSIS OF RENAL TUMORS WITH PAPILLARY FEATURES
Papillary features/architecture can be observed in almost all renal tumor entities, even outside the spectrum of RCC [29] (including, for example, urothelial tumors of the kidney, metanephric tumors, mixed epithelial and stromal tumors, juxtaglomerular cell tumors, among others). Also, not all pRCC show an overt papillary architecture, which can be rather tubulopapillary or even hardly apparent and replaced by solid growth [29, 30]. In other words, there is a marked difference between having a papillary architecture / growth pattern, which can be observed in multiple renal neoplasms, and between calling a renal tumor pRCC (which refers to a specific tumor entity with defined criteria, and showing gains in 7/17 and loss of Y chromosomes). Papillary adenomas are currently defined upon the basis of size (<1.5 cm) as well as morphology (WHO/ISUP grade 1-2, unencapsulated) [1].
While there has been a lot of effort to subtype pRCC and segregate new entities, several patterns of growth and morphologies have been reported to impact prognosis across the pRCC spectrum, including solid, micropapillary, hobnail and microcystic patterns [26, 31]. WHO/ISUP grading and architecture remain strong predictors of tumor behavior and prognosis, and should always be reported [32]. Molecular features of pRCC are somewhat heterogeneous, reflecting the evolution in classifications. Besides the aforementioned typical chromosomal gains in 7/17 and
The differential diagnosis of pRCC encompasses tumors with basophilic cells, with clear cells, with eosinophilic cells and tumors with a mixture of eosinophilic and clear cells.
In the event of a very solid pattern of pRCC with basophilic cytoplasm, the differential with metanephric adenoma (or epithelial-predominant nephroblastoma) is also challenging. Metanephric adenoma is composed of very compact tubules, branching ducts and abortive glomeruli, often with psammomatous calcifications, which mimic a tubulopapillary growth of pRCC [43]. Clues for the diagnosis include the higher nuclear-to-cytoplasmic ratio, a thick scarred-like/hyalinized stroma and absence of a fibrous capsule in metanephric adenoma [44]. The differential is solved by immunohistochemistry, as metanephric adenoma shows diffuse WT1 and CD57 positivity, negative or focal CK7 staining and usually positive BRAF staining [45], given the frequent
Tubulocystic RCC is a rare tumor with a typical spongy gross appearance. Tumor cells show prominent hobnailing [61]. Tubulocystic RCC is usually an indolent tumor with a good prognosis.
PROVISIONAL/EMERGING TUMOR ENTITIES WITH PAPILLARY GROWTH
There is an emerging body of evidence, that is compelling but not sufficient for placing certain clinicopathological and genomically distinct tumors into the formal classification. The 2022 WHO Classification currently regarded some papillary neoplasms as emerging entities [80]. One is biphasic squamoid-alveolar RCC, composed of one population of larger eosinophilic cells with higher nuclear grade arranged in nests, encased by a second population of smaller cells with high nuclear-to-cytoplasmic ratio, lower nuclear grade and basophilic cytoplasm, giving a low-power appearance of alveoli [81]. A quite characteristic features is the finding of emperipolesis, with engulfment of inflammatory cells by the larger cells [82]. Staining for cyclinD1 and CD57 specifically highlights the larger cells [83, 84]. Cytogenetically, however, tumors show the typical gains in chromosomes 7/17 found in pRCC [82, 85], as well as a high prevalence of MET alterations [86], supporting a link towards pRCC. The second subtype is the biphasic hyalinizing psammomatous RCC. These tumors are composed of one population of very small cells with hyperchromatic nuclei and somewhat spindle-shaped arranged around basement membrane material admixed with a second population of larger cells forming tubules, papillae or acini [87]. EMA stains preferentially the smaller cells, while CK7 highlights the larger cells. Interestingly, these tumors have been shown to harbor mutations (with biallelic loss) in the
Papillary neoplasm with reverse polarity (PRNRP) has been increasingly reported in the literature [89, 90]. These are overall small cystic tumors, composed of thin papillae with edematous or hyalinized cores covered by a single layer of eosinophilic small cells with small low-grade nuclei (sometimes with optical clearing) pushed against the apical membrane (“reverse polarity”). Like for ccpRCT, GATA3 nuclear staining is characteristic and no worrisome features or poor outcome are reported [90]. Interestingly, the tumors have been associated with
Thyroid-like follicular RCC, which shares remarkable morphologic resemblance with follicular cancers of the thyroid gland are composed of variably sized follicles lined by small cuboidal cells. Papillary foci have been described [93], but usually a rather follicular architecture suggests the diagnosis. While most cases are indolent, aggressive behavior has been documented. Recently, these tumors have been shown to have a characteristic
Table 1 summarizes the most frequent differential diagnoses of pRCC in routine practice and some features helping assisting in diagnosis, and Fig. 1 illustrates some of the phenotypes that come in the differential of pRCC.
Differential diagnosis of papillary renal cell carcinoma.

Abbreviations: FISH – fluorescence
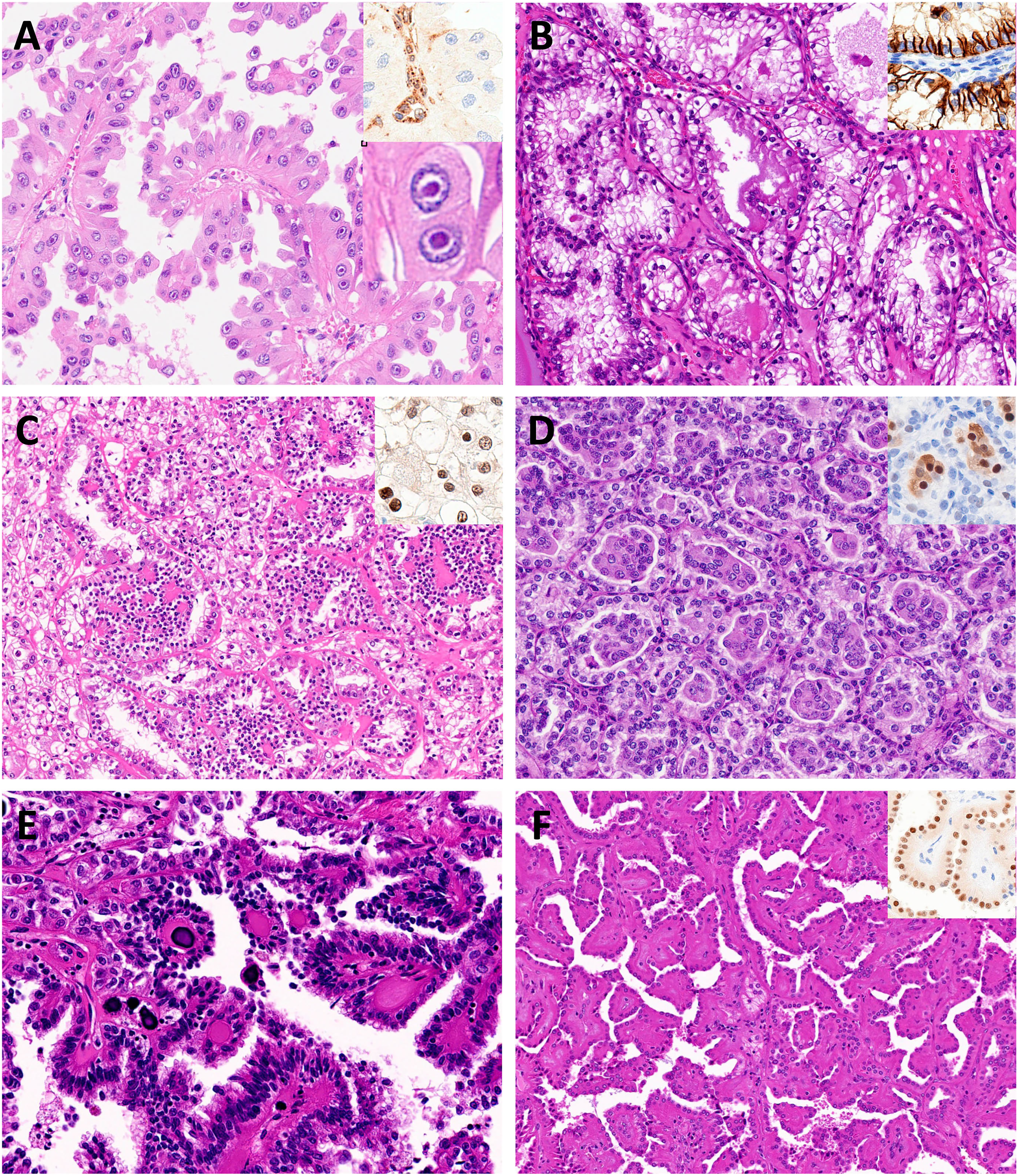
The examples of renal neoplasia with papillary architecture. A – FH-deficient RCC, showing papillary structures covered with cells with abundant eosinophilic cytoplasm and at least focal eosinophilic macronucleoli, often shows perinucleolar haloes (inset). B – Clear cell papillary renal cell tumor, showing tubulopapullary architecture lined by cuboidal cells with clear cytoplasm. The low-grade nuclei often exhibit linear arrangement apart from the basement membrane. The tumor cells show cup-like staining for CAIX with no luminal border staining (inset). C – TFEB-rearranged RCC, showing a biphasic pattern comprising nests of larger and smaller tumor cells surrounding basement membrane material. The tumors show diffuse TFEB nuclear positivity (inset). D – Biphasic squamoid alveolar RCC, showing a biphasic pattern with squamoid cells with abundant eosinophilic cytoplasm in the center of the alveolar structures. Squamoid cells are positive for cyclinD1 (inset). E – Biphasic hyalinizing psammomatous RCC, showing biphasic pattern with tubulopapillary growth of larger cells and also showing smaller cells, with psammomatous calcifications and having a glomeruloid pattern. F – Papillary renal neoplasm with reverse polarity with thinly branching papillae lined by eosinophilic cells with low-grade nuclei aligned at the apex. The tumors show diffuse GATA3 nuclear positivity (inset).
FUTURE PERSPECTIVES
In conclusion, the spectrum of pRCC has changed in recent years, with many entities being identified. The 2022 WHO renal tumor classification has introduced the concept of molecularly defined RCC entities and abolished the separation of type 1 and type 2 pRCC. Several new renal tumor entities with papillary growth are emerging. Therefore, it is expected that the spectrum of pRCC is becoming increasingly narrow (Fig. 2). Continuing revisiting tumors with papillary features and gathering data on particular patterns, combined with accurate genotype-phenotype and clinical correlation, will contribute to precision medicine and better patient risk stratification and selection for specific therapies [98].

The spectrum of papillary renal cell carcinoma is narrowing. Comparison of the WHO 2016 and WHO 2022 classification of papillary renal cell carcinoma.
Although several targeted therapy options are possible for patients with metastatic pRCC, many new therapeutical options have been approved based on trials including all histotypes of RCC (mainly ccRCC). Data specifically regarding pRCC is scarce [99]. In this sense, accurate histopathological characterization of renal cell tumors is important to identify specific phenotypes which may have a distinct and actionable molecular background. With the introduction of molecularly defined entities, pathologists should attempt to accompany this evolving field and acquire tools, such as dedicated NGS panels, for characterizing challenging and heterogeneous renal cancers [100]. Also, application of digital pathology algorithms for dissecting architectural and cytological features of renal cancers with papillary foci will also likely contribute to uncovering novel biomarkers to integrate with molecular data, and may be a useful tool to deconvolute the heterogeneity of renal tumors, facilitating diagnosis and predictive value of specific biomarkers [101–103].
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments.
AUTHOR CONTRIBUTIONS
Drafting of the paper, table and figure: João Lobo and Riuko Ohashi.
Supervision and final editing: Holger Moch.
All authors read and approved the final paper.
FUNDING
H.M. receives a Swiss National Science Foundation grant (No. S-87701-03-01) and R.O. receives a JSPS KAKENHI Grant Number JP22KK0273 (Fostering Joint International Research (A)).
CONFLICTS OF INTEREST
J.L., R.O. and HM. have no conflicts of interest to disclose.
