Abstract
Background:
Non-clear cell renal cell carcinoma (nccRCC) is a diverse group of cancers that occurs in approximately 25% of patients with renal cell carcinoma. In the advanced/metastatic setting, survival in all nccRCC subtypes is considered poor, due to the inherent aggressiveness of these cancers, and a lack of effective systemic treatment options. Clinical trials of immune/targeted agents have predominantly focused on patients with ccRCC. There is no globally accepted standard of care for nccRCC; however, recently clinical trials have been initiated in this population.
Objective:
To perform a targeted literature review of published original observational studies reporting common real-world clinical outcomes (real-world overall response rate [rwORR], real-world progression free survival [rwPFS], real-world overall survival [rwOS]) in previously treatment naïve patients with advanced/metastatic nccRCC.
Methods:
A targeted search of MEDLINE and EMBASE was conducted per PRISMA guidelines to identify observational studies in previously treatment naïve patients with advanced/metastatic nccRCC. Publications with adequate information since 2010 and from select conferences since 2020 were considered.
Results:
27 studies across 29 publications were identified. Sample sizes ranged from 7-1,573 across these studies with differences in nccRCC subtypes included and treatments received. Real-world ORR ranged from 0–37%, median rwPFS from 2–17 months, and median rwOS from 3–30 months, across 19, 17, and 24 studies, respectively. These outcomes also varied with receipt/type of treatment and demographic/clinical subgroups with outcomes tending to be worse in patients with papillary RCC compared to chromophobe RCC.
Conclusions:
Clinical outcomes varied, as patient populations, eligible histologies, treatments and methods were heterogeneous.
Keywords
INTRODUCTION
An estimated 431,288 new cases of kidney cancer were projected worldwide during 2020, with the highest age-standardized incidence rates (per 100,000) reported in Europe (9.5) and North America (12.2) [1]. The majority of kidney cancers are renal cell carcinoma (RCC) and approximately 75% of patients with RCC have clear cell histology with the remainder being non-clear cell RCC (nccRCC) [2]. Factors that have been established as important prognostic determinants of 5-year survival in RCC are tumor stage, grade, local extent of the tumor, presence of regional nodal metastases, and evidence of metastatic disease at presentation [3]. In the advanced/metastatic setting, survival in all subtypes of nccRCC is considered poor, due to the inherent aggressiveness of these cancers, and a lack of effective systemic treatment options [4].
Historically, clinical trials of immune and targeted agents have predominantly focused on patients with clear cell histology. Therefore, and due to limited data in nccRCC patients, the role of various approved agents in the treatment of nccRCC is poorly defined and there is currently no globally accepted standard of care for these patients, and clinical trial participation remains the preferred treatment [3]. A summary of recent prospective clinical trials was described by Brown and colleagues [5]. The purpose of this targeted literature review was to summarize recent publications of observational studies in patients with advanced nccRCC that remain treatment naïve or receive an initial therapy for advanced disease.
MATERIALS AND METHODS
Search strategy
A targeted literature review was conducted in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [6]. A meta-analysis of the results was out of scope for this study. A search was conducted on the EMBASE and MEDLINE databases by one co-author (S.C.) to identify relevant studies published between January 1 2010 through March 7 2022. A separate manual search of abstracts from the 2022 American Society of Clinical Oncology Genitourinary Symposium (ASCO GU) was conducted using the conference platform on March 14 2022 and the abstracts from the 2020-2021 annual conferences for the American Society of Clinical Oncology (ASCO), the European Society for Medical Oncology (ESMO), and ASCO GU were searched on the EMBASE and MEDLINE databases on March 15 2022. Recent years of conferences were selected to capture new research that may not have yet been published in manuscript form. Keywords used for all database searches include “Kidney Cancer” OR “Renal Cell Carcinoma” AND ("Non-clear cell” OR “Papillary” OR “Chromophobe” OR “Translocation”) AND ("Advanced” OR “Metastatic"). The search was conducted by one author (S.C.) in three stages. In the first stage, duplicate references were removed. In the second stage, titles and abstracts from all unique references selected were screened. In the final stage, the full-text of the remaining publications were screened. For conference abstracts, only the first two phases were utilized to select relevant publications. The other two co-authors (C.L. and M.S.) reviewed the final list of included references to ensure inclusion of eligible studies based on their knowledge of the field.
Exclusion criteria
Only observational studies were considered for this summary. All editorials, reviews, non-clinically focused studies, interventional studies, and non-English language articles were excluded. To narrow the scope of the review, publications without minimum information describing the source population, histology, and treatment of each cohort of treatment-naïve / 1L treated patients with advanced nccRCC or that included patients that had previously received systemic treatment or ccRCC were excluded. In addition, outcomes for subgroups in selected publications without minimum information describing the subgroup were not extracted. The review was limited to manuscripts published between January 1 2010 through March 7 2022 and conference abstracts from ASCO (2020-2021), ESMO (2020-2021), and ASCO GU (2020–2022).
Data extraction and synthesis
From all eligible publications, including conference posters/presentations associated with the selected conference abstracts, the identifiers, design elements, population characteristics with inclusion and exclusion criteria, number of patients overall and in relevant subgroups, treatment intervention(s), clinical outcome definitions and results for overall nccRCC samples, as well as for subgroups, were extracted to a predefined data extraction table. The clinical outcomes of interest included real-world overall response rate (rwORR), real-world progression free survival (PFS), and real-world overall survival (rwOS). For publications that did not report a rwORR, but did report the number of patients with complete and partial response, as well as the total evaluated, a rwORR was calculated for that study population/subgroup. Outcomes between studies and subgroups were qualitatively compared and described. To assist with this qualitative comparison, a base definition for the clinical outcomes of interest was put forth in the protocol and publications that used different definitions were noted. For this targeted literature review, rwORR was defined as the percentage of participants who achieved either a complete response (CR) or partial response (PR). The time from the date of first dose to a) the first of documented disease progression or death due to any cause, and b) death due to any cause, were used for rwPFS and rwOS, respectively. Outcomes between studies were not combined and a meta-analysis was not conducted. All studies were organized by whether or not treatment was administered, treatment class (e.g.,
RESULTS
A total of 716 abstracts were identified by searching the EMBASE/MEDLINE databases (journals and 2020-2021 ASCO/ASCO GU/ESMO) and reviewing the complete listing of the ASCO GU 2022 RCC abstracts (Fig. 1). After excluding 3 duplicates, 713 abstracts were screened. This screening resulted in 183 full publications for review and 6 conference abstracts selected for inclusion. Of the full publications reviewed, 23 were selected for data extraction and 160 were excluded for the following primary reason: not in scope due to study population (e.g., included ccRCC) or participants were not treatment naïve (
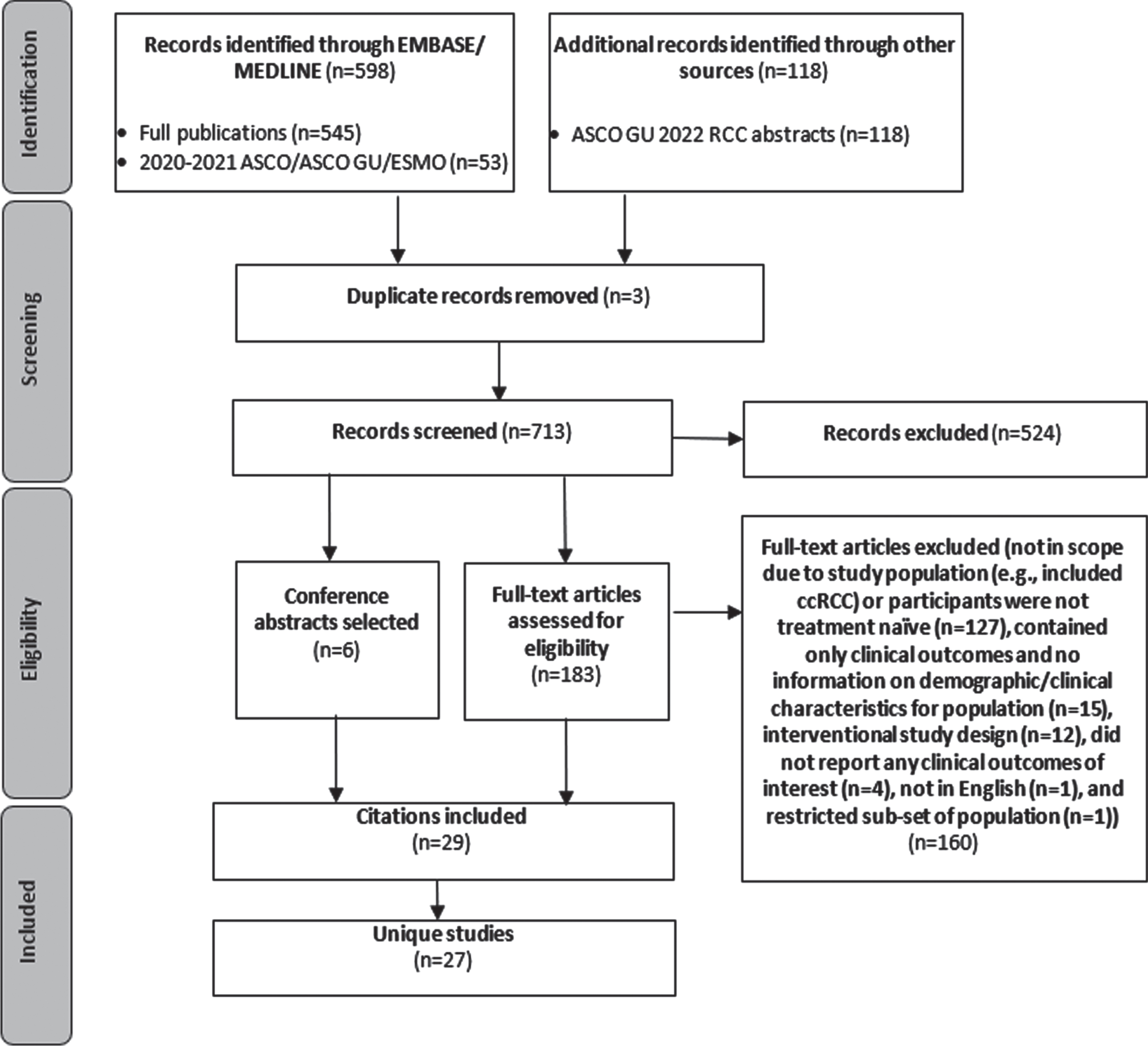
Inclusion and exclusion for targeted review of observational studies with outcomes for advanced non-clear cell renal cell carcinoma.
Real-world ORR, PFS, and OS in observational studies of nccRCC
‡Outcome definition differed from base definition. *Calculated based on data reported in publication (ORR = CR+PR). Abbreviations: ATE: atezolizumab; AXI: axitinib; BEV: bevacizumab; CB: clinical benefit; CI: confidence interval; CN: cytoreductive nephrectomy; CR: complete response; DC: disease control; DOR: duration of response; EVE: everolimus; ICI: immune checkpoint inhibitor; IFN: interferon-α; IL2: Interleukin-2; IPI: ipilimumab; IQR: interquartile range; ITT: intention to treat; mTORi: mammalian target of rapamycin inhibitors; NE: not evaluable (per publication); NIV: nivolumab; NR: Not reported; PAZ: pazopanib; PEM: pembrolizumab; rwORR: real-world overall response rate; rwOS: real-world overall survival; rwPFS: real-world progression free survival; SOR: sorafenib; SUN: sunitinib; TEM: temsirolimus; TKI: tyrosine kinase inhibitors.
Across the identified observational studies, sample size ranged from 7 to 1,573 patients and all but one [13] utilized a secondary data study design. When reported, the most common method for evaluating tumor response was RECIST (17/28). The clinical outcomes of interest are reported for each publication in Table 1 and the distribution of these clinical outcomes across the identified publications are reported in Table 2. Differences were noted in the start of follow-up for both rwOS and rwPFS with some studies using diagnosis rather than start of treatment as the index date for these clinical outcomes. Results that utilized diagnosis or deviated from the base definition are denoted in Table 1. Overall, rwORR ranged from 0–37% (Fig. 2), median rwPFS from 2–17 months (Fig. 3), and median rwOS from 3–30 months (Fig. 4), across 19, 17, and 24 studies, respectively (Table 2). The majority of studies (14/19) reported a rwORR of 25% or less, most publications (12/17) reported a median rwPFS of 8 months or less, and for many (21/24) the median rwOS was 25 months or less. When limited to studies that used outcome definitions that aligned with the base definition of the clinical outcomes as described in the prior ‘Data extraction and synthesis’ section, the minimum in the range of median rwOS increased from 0 to 8 months and the maximum was unchanged. Different definitions for rwORR were not reported in the selected publications and the range for median rwPFS was not impacted when limiting to publications using a definition similar to the protocol definition. Some studies also reported results for subgroups of study samples; for subgroups with adequate demographic and clinical information within the publication, the reported results are also included in Table 1.
Range of rwORR, rwPFS, and rwOS in observational studies of nccRCC overall and by initial therapy class and histological subtype
Abbreviations: ICI: immune checkpoint inhibitor; mTORi: mammalian target of rapamycin inhibitors; NR: Not reported; rwORR: real-world overall response rate; rwOS: real-world overall survival; rwPFS: real-world progression free survival; TKI: tyrosine kinase inhibitors.
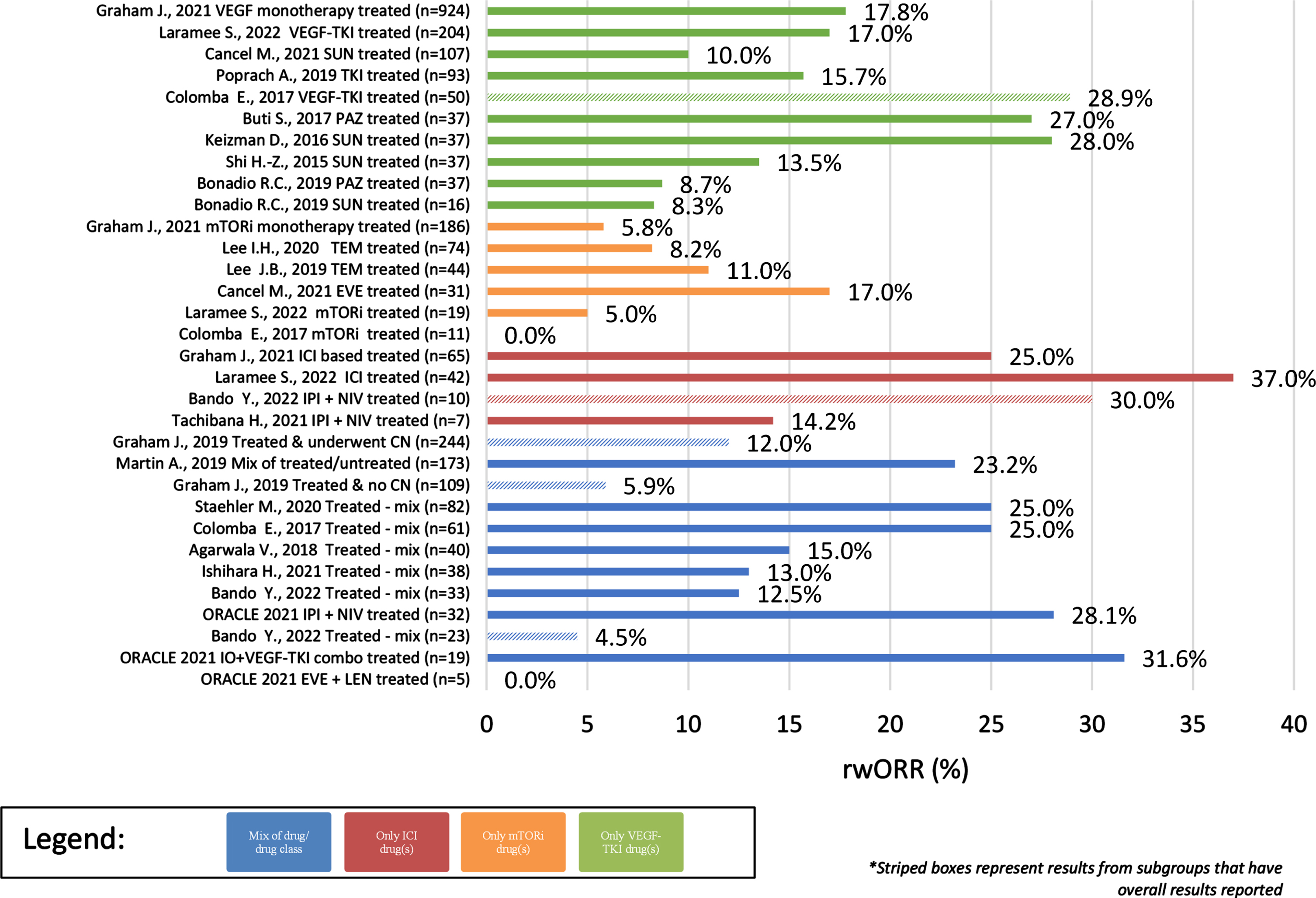
rwORR of Observational studies (overall and subgroups) by treatment/sample size.
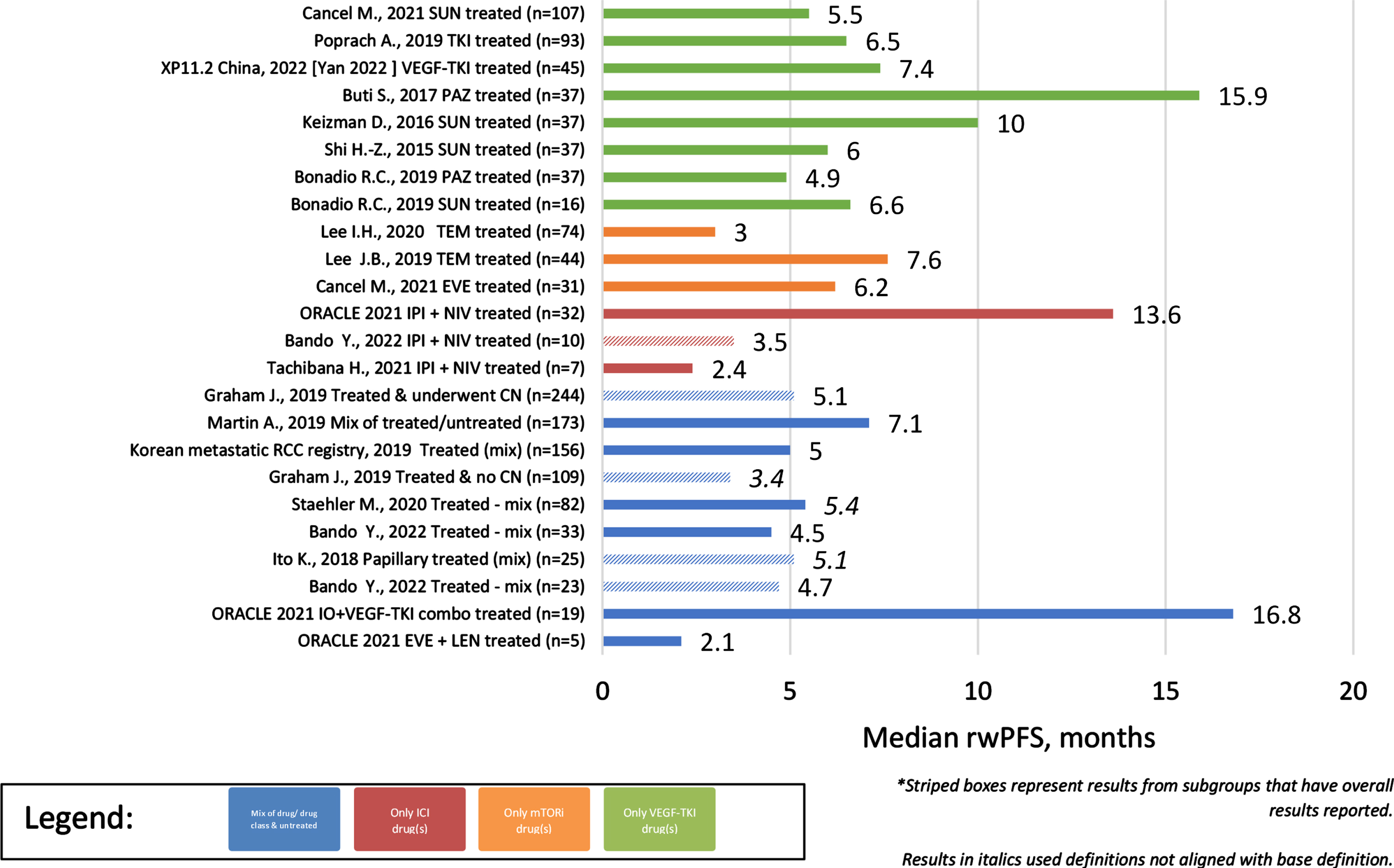
Median rwPFS of Observational studies (overall and subgroups) by treatment/sample size.
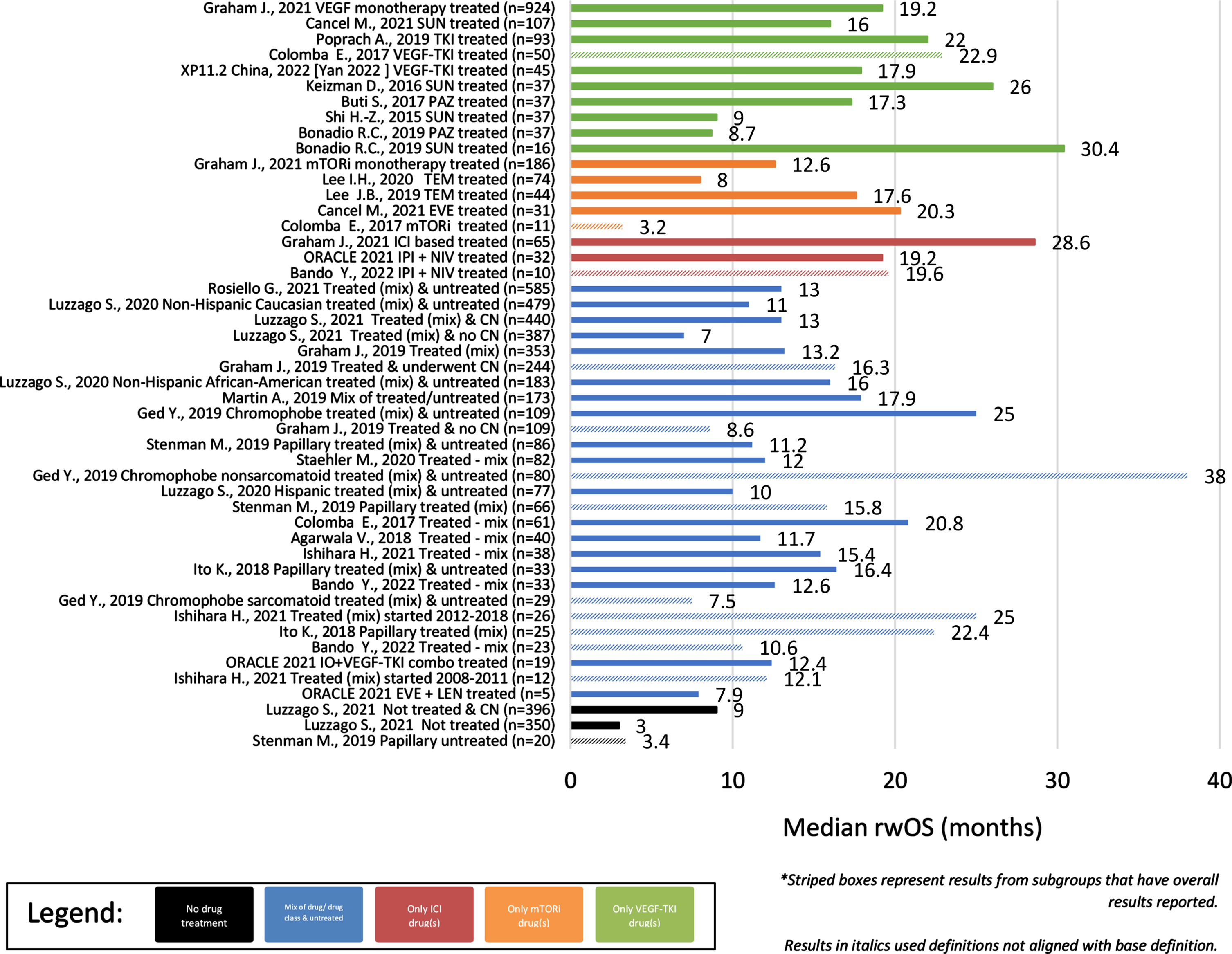
Median rwOS of Observational studies (overall and subgroups) by treatment/sample size.
In cohorts/subgroups without any systemic treatment (
Across the observational cohorts/subgroups focusing on a single drug, rwORR ranged from 8–28% in patients treated with sunitinib (
The range of rwORR in patients comprised of those treated with a single drug combination was 14–30% in patients treated with ipilimumab + nivolumab (
Clinical outcomes also differed per histological subtype (Table 2), including longer survival in patients without sarcomatoid features. In cohorts/subgroups of solely patients with papillary RCC, rwORR ranged from 6–25% (4/6), median rwPFS from 2–6 months (5/7), and median rwOS from 3–22 months (5/8). Of those unique studies/cohorts with papillary RCC that received treatment, median rwOS ranged from 9–22 months (4/7). Of those unique studies/cohorts solely comprised of patients with chromophobe RCC, rwORR ranged from 0–29% (2/3), median rwPFS was 10 months in the single study/cohort reporting it, and median rwOS ranged from 3–38 months (3/5). Overall, outcomes of patients with papillary RCC tended to be worse than patients with chromophobe RCC. Only 1 study reported results stratified by presence of sarcomatoid features, specifically median rwOS, with those without sarcomatoid features overall having longer survival than those with sarcomatoid features (38 vs 7.5 months).
DISCUSSION
While observational studies were identified in this review that focused on clinical outcomes in 1L eligible patients with advanced/metastatic nccRCC, heterogeneity in the patient populations and treatments included, as well as study design used, were found to contribute to an incomplete understanding of the clinical outcomes of the advanced/metastatic nccRCC population as a whole. This heterogeneity in study population was also noted in a recent systematic literature review that focused solely on clinical trials evaluating 1L treatment in metastatic nccRCC [5]. Overall, Brown et al. reported that the range of ORR and PFS was wide, favoring TKI and ICI-based combination regimens, which was also noted in this review. While clinical outcomes varied with patient demographics, histologic subtype and choice of treatment (e.g., ICI vs TKI, monotherapy vs combination therapy), no studies controlled for variability in population characteristics. Additionally, there were inconsistencies in defining the histologic subtypes included within nccRCC (e.g. ccRCC with sarcomatoid features) and in defining the clinical endpoints (e.g. rwPFS and rwOS). As such, interpretation of these differences is limited given the variance in the populations within/across studies and the heterogeneity of study designs. Therefore, comparisons of historical controls with future studies, such as those ongoing trials in patients with nccRCC, must be carefully selected to ensure similarity of patient characteristics and methodologies/outcome definitions.
This literature review focused solely on patients with treatment naïve advanced/metastatic nccRCC that either remained without treatment or received front line treatment in the observational setting. There have been a number of notable studies, including clinical trials, which focused on patients with advanced/metastatic nccRCC treated in this/other settings [35–40]. The KEYNOTE-427 cohort B Phase II study (NCT02853344) evaluated pembrolizumab monotherapy in untreated patients with advanced nccRCC (
Although the literature review was comprehensive and utilized a pre-specified approach, the findings may be incomplete as systematic literature review was not undertaken nor was a meta-analysis performed. Additionally, publication bias is an inherent limitation of any literature review. Selection bias may also be a limitation as some publications were excluded if the characteristics and outcomes for patients with nccRCC were not reported for this subgroup but instead were reported as part of the overall RCC population.
Overall, this literature review demonstrates that the response to available treatments for nccRCC in the real-world setting is still relatively low across the majority of observational studies. While Brown et al. (5) provided an overview of clinical outcomes for patients with nccRCC, it was limited to the interventional setting. This review expands upon what was previously reported to complete the snapshot of clinical outcomes in patients with advanced/metastatic nccRCC. These collectively emphasize the continued need for research to identify treatments that improve the prognosis of patients with advanced/metastatic nccRCC.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgements.
FUNDING
The authors report no funding.
AUTHOR CONTRIBUTIONS
SC, MS, and CL attest to substantially contributing to the conception, design or planning of the study; and/or the acquisition or analysis of the data and/or interpretation of the results; and substantially contributed to the drafting of the manuscript and/or critically reviewing or revising it for important intellectual content; and reviewed the final version of the manuscript to be submitted and am in agreement with its content and submission; and had access to all the relevant study data and related analyses, and vouch for the completeness and accuracy of the data presented.
CONFLICT OF INTEREST
SC and MS are employees of Merck & Co., Inc. SC holds personal stock in Merck & Co., Inc. and Organon & Co. MS holds personal stock in Merck & Co., Inc. In the past 36 months, CL has received: research funds to his institute from AstraZeneca, BMS, Calthera, Eisai, Eli Lilly, Exelixis, and Merck, consulting fees from Aveo, BMS, Exelixis, Eisai, Merck, Pfizer, EMD Serono, and Cardinal Health, payment or honoraria from AiCME for a CME event and from Ideology Health, Intellisphere, Medscape, and Research to Practice for educational events. CL is on the Medical Steering Committee for the Kidney Cancer Association.
