Abstract
BACKGROUND:
Kidney cancer exerts significant disease burden in the United States and possesses a rapidly evolving treatment landscape. The expansion of novel systemic treatment approaches and the use of immunotherapy has been accompanied by increased costs over time. However, the cost-effectiveness of immunotherapy in renal cell carcinoma (RCC) has not been fully assessed. The current study presents a systematic review of cost-effectiveness studies of immunotherapy-based treatment in the context of RCC.
METHODS:
A literature search utilizing PubMed, Embase, Web of Science, and the Cochrane Library was undertaken to find articles related to the cost-effectiveness of immunotherapy treatment in renal cell carcinoma (RCC). The inclusion criteria for articles were as follows: English, published between 1983 and 2020 and evaluated cost-effectiveness in any of the currently approved immunotherapies for RCC. Exclusion criteria included being a review article, commentary or editorial, as well as possessing no specific cost-effectiveness evaluation or analysis relevant to the current review.
RESULTS:
The current review identified 23 studies, published between 2008 and 2020, across 9 different countries. The studies identified tended to focus on patients with locally advanced or metastatic RCC and examined the cost-effectiveness of immunotherapy across various lines of treatment (first-line treatment (n = 13), second-line treatment (n = 8), and first-line and beyond (n = 2). Eight studies examined the use of interferon-alpha (IFN-alpha), with some reports supporting the cost-effectiveness of these agents and an equal number of studies demonstrating the opposite, with sunitinib often demonstrating superior cost bases. The majority, fourteen studies, included the use of novel immune checkpoint inhibitors (nivolumab, ipilimumab, pembrolizumab), half of which found that checkpoint inhibitors were more cost-effective when compared to oral systemic therapies (sunitinib, everolimus, axitinib, pazopanib, and cabozantinib).
DISCUSSION:
Novel immune checkpoint inhibitors constituted the most frequently examined agents and were likely to be deemed cost-effective as compared to other treatments; although this often required higher willingness-to-pay (WTP) thresholds or healthcare systems that possessed more cost-constraints. These observations have clinical and health system applicability, with the ability to potentially reduce the cost of treatment for locally advanced or metastatic RCC.
BACKGROUND
Kidney cancer is a major disease burden in the United States. In 2021 alone, it is estimated that 76,080 new cases and 13,780 cancer-related deaths will occur, the majority of which will be classified as renal cell carcinoma (RCC) [1]. While the incidence of kidney cancer has increased at a rate of 0.5% per year among males, deaths are trending down overall due to the development of advanced diagnostics that enable earlier detection of disease as well as novel therapeutics. Localized disease is typically treated with resection for curative intent [2], however metastatic disease is considered incurable and typically requires lifelong systemic therapy.
RCC has been a pioneer in early utilization of immunotherapy approaches due to poor response to chemotherapy-based approaches. Therefore, utilization of immunotherapy based approaches such interferon-alpha (IFN-alpha) or interleukin-2 (IL-2) were the standard of care until canonical phase III clinical trials demonstrated activity of targeted oral therapeutics, known as tyrosine kinase inhibitors (TKIs) [3]. Today, novel immune checkpoint inhibitors, such as programmed cell death protein 1 (PD-1), programmed death ligand 1 (D-L1), and cytotoxic T lymphocyte associated protein 4 (CTLA-4) inhibitors are employed in the frontline setting across all risk categories alone or in combination with TKIs for patients with metastatic disease [4, 5].
The rapid expansion of available therapies has been accompanied by an increase in treatment cost over time, especially in patients with more advanced disease [6]. Due to the availability of multiple systemic treatment options, understanding the costs associated with treatment would be beneficial in order to minimize patient financial toxicity. Effective therapies are rarely compared head to head and choosing a particular treatment can be difficult for patients, physicians, and health systems [7].
The cost-effectiveness of immunotherapy approaches across tumor types has been evaluated extensively. However, analyzing cost-effectiveness of immunotherapy treatment in advanced RCC is useful to guide health systems on preferred treatment options. Cost-effectiveness, usually measured in incremental cost-effectiveness ratio (ICER = ratio of cost difference vs. clinical efficacy for two compared therapies), is often calculated using data from phase III studies and real world cohort data [8]. Using this calculation, a particular country’s health system might decide to offer certain treatments based on their willingness to pay (WTP) threshold. The current paper presents the results of a systematic review of cost-effectiveness studies of immunotherapy in the context of RCC.
METHODS
Data sources and searches
In order to obtain the most comprehensive results, a literature search utilizing PubMed, Embase, Web of Science, and the Cochrane Library was conducted to find articles related to the cost-effectiveness of therapies in renal cell carcinoma (RCC). Both keywords and index terms (MeSH and Emtree) to develop these searches. The concept of RCC was combined with concepts of selected drugs and surveillance, along with that of cost-effectiveness. The full search strategies for each database are included as a supplement. Besides the database searches, the gray literature was searched by reviewing conference abstracts downloaded from Embase, and by hand-searching the literature of our most relevant articles for additional references. Among all identified manuscripts, only studies employing immunotherapy in the review were included.
Study selection criteria & data extraction
The inclusion criteria for articles were as follows: English, published between 1983 and 2020, evaluated cost-effectiveness in any of the currently approved immunotherapies for RCC. Exclusion criteria included being a review article, commentary or editorial, as well as possessing no specific cost-effectiveness evaluation or analysis relevant to the current review. Three phases of review were undertaken, including title, abstract and then full-text review of identified articles. At each stage, a dual consensus was reached for each inclusion and exclusion, with five reviewers (EP, AB, DK, SZ, FW) independently reviewing and assessing articles. Based on inclusion and exclusion criteria and dual review consensus, a total of 23 articles met criteria for data extraction and review.
RESULTS
As presented in Fig. 1 and detailed in the subsequent tables, the current review included 23 studies published between 2008 and 2020. Studies reviewed aimed to evaluate the cost and cost-effectiveness of various immunotherapies in the treatment of advanced or metastatic RCC. The studies were performed in 9 different countries (Italy, Canada, United Kingdom, Singapore, United States, China, Sweden, Israel, & Spain) and were sponsored by government grants, national organizations, and private companies. Although not a specific inclusion criterion, the studies identified in this review focused on patients with mRCC or advanced RCC and examined the cost-effectiveness of immunotherapy across various lines of treatment. Included studies utilized four different methodological approaches in their analysis of the cost and cost-effectiveness of RCC treatment. Sensitivity analyses were not always performed, but when utilized, included probabilistic, deterministic, univariate/one-way sensitivity analyses. The study horizons ranged from 11 months to patients’ lifetime but was not always specified in the included papers.
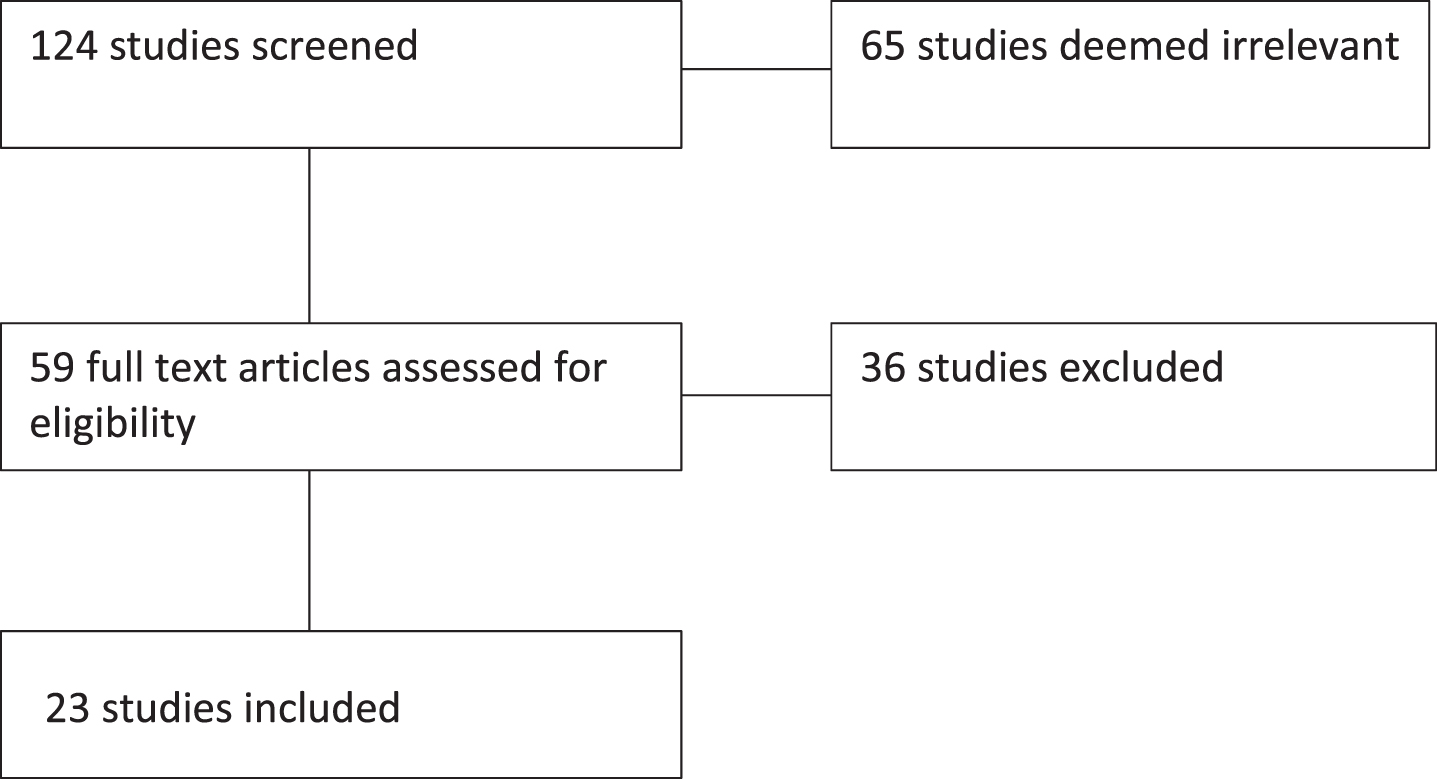
PRISMA Flow chart.
Study Characteristics
*mRCC = metastatic renal cell carcinoma, †ICER = incremental cost-effectiveness ratio, ‡ICUR = incremental cost-utility ratio, §QALY = quality-adjusted life-year, ∥ IFN-α= interferon-alpha, ¶IL-2 = interleukin-2.
As noted, there were four methodological approached employed in the reviewed studies: (1) Cost-Effectiveness based on Clinical Trials, (2) Cost-Effectiveness based on Hypothetical Cohorts from Clinical Trials, (3) Cost per progression-free-survival and overall-survival, and (4) Base Case Economic Model and Evidence Review Group (ERG) Analysis. Our review primarily focused on quality adjusted life years (QALY), incremental cost-effectiveness ratio (ICER), and willingness-to-pay (WTP) thresholds to determine the cost-effectiveness of each therapy. Given that four studies did not include these specific measures, we used costs associated with therapy as another proxy to assess its cost-effectiveness for these studies.
Eight of the twenty-three studies included examined the use of IFN-alpha (Table 2a). Four of these studies showed IFN-alpha to be the more cost-effective option when compared to prior standards of care (IL2, temsirolimus and sunitinib). In contrast, the remaining four reported IFN-alpha to be the less cost-effective option when compared with sunitinib and pazopanib. Two studies included the use of cytokine therapy (IL-2); one of which reported cytokine therapy to be more cost-effective when compared with sunitinib or bevacizumab, while the second noted cytokine therapy to be less cost-effective as compared to IFN-alpha or sunitinib.
Summary of cost analyses for IL-2 & IFN-α
Fourteen studies included the use of novel immune checkpoint inhibitors (nivolumab, ipilimumab, pembrolizumab) (Table 2b). Seven of these studies found that checkpoint inhibitors were more cost-effective when compared to oral TKIs (sunitinib, everolimus, axitinib, pazopanib, and cabozantinib), while three studies found that they were less cost-effective when compared with cabozantinib and everolimus. Two of the fourteen studies found that the cost-effectiveness of these agents was dependent on the WTP threshold employed, though in general, if WTP was greater than $150,000, immune checkpoint inhibitors were the more cost-effective option when compared to everolimus. A single study showed that immune checkpoint inhibitors were more cost-effective in China than the US when compared to everolimus. Finally, one study reported inconclusive findings regarding the cost-effectiveness of nivolumab compared with everolimus. Among the 14 studies examining novel immune checkpoint agents, they were relatively evenly split between those examining cost in a first-line setting as compared to those in the second-line setting. Overall, the results suggested that regional differences and consequent disparities in pricing may be an important factor in determining the benefit of first- versus second-line treatment with immune checkpoint inhibitors.
Summary of Cost Analyses for novel immune-checkpoint inhibitor (PD-1, PD-L1, and CTLA-4 inhibitors)
Of the 23 studies included, thirteen examined first-line therapy, eight focused on second-line therapy, and two studies examined first-line therapy and beyond. The drugs most commonly used in first line therapy included sunitinib (12 studies), IFN-alpha (8 studies), and immune checkpoint inhibitors (4 studies). Across studies, the agents most commonly used in second-line treatment included immune checkpoint inhibitors (8 studies), everolimus (8 studies), and axitinib (3 studies). Finally, agents examined in studies of first-line therapy and beyond included sunitinib, nivolumab, pazopanib, axitinib, and cabozantinib.
DISCUSSION
The current study was a systematic review of a global literature to identify studies examining the cost and cost-effectiveness of immunotherapy agents in the treatment of RCC. As newer therapeutic agents and combination regimens gain approval in this space, it is critical that the economic consequences of treatment decision-making be examined more fully. This review identified 23 studies across nine countries that met inclusion criteria and reported on the cost or cost-effectiveness of an immunotherapy agent in the treatment of RCC. These high-level findings provide insight into the relative economic advantages of these newer therapeutic agents and can help guide the refinement of treatment guidelines and shared decision making among various stakeholders in the RCC field.
The majority of studies focused on the costs associated with immune checkpoint inhibitors, including nivolumab, ipilimumab and pembrolizumab, with most suggesting that these agents were more cost-effective when compared to oral TKIs sunitinib, everolimus, axitinib, pazopanib or cabozantinib. Importantly however, the region of study and WTP threshold were critical contributors to the eventual cost-effectiveness assessment, with these agents often requiring a higher WTP threshold (e.g. [9]) or a non-US based healthcare system (e.g. [10]). As shown in Table 2b, immune checkpoint inhibitors were compared to oral TKIs and observed to demonstrate a gain in QALYs. Interestingly, these data demonstrated that regional differences in benefit of first- versus second-line treatment with immune checkpoint inhibitors may occur due to differences in pricing.
There was no clear consensus among those studies that examined the costs associated with IL-2 & IFN-α, with some reports supporting the cost-effectiveness of these agents and an equal number of studies demonstrating the opposite, with sunitinib often demonstrating superior cost bases. These results make it difficult to draw firm conclusions, and may once again suggest that the geographic location, and subsequent healthcare system, in which a study is conducted may play a role in guiding such results.
Implications for healthcare systems
Cost-effective analyses (CEA) aim to promote health equity by prioritizing distributive benefits for the entire health system [11]. Observations from this systematic review suggest that immunotherapy approaches are more cost-effective, however, may require higher WTP thresholds. While standard CEA assume interventions are independent [12, 13], this assumption may not hold valid for low-income countries that require alteration of health delivery platforms in order to dispense certain agents. This is especially relevant when an analysis includes an intravenous versus orally administered medication. Therefore, as healthcare systems determine the applicability of a CEA, understanding the interdependence of the intervention with other aspects of health care delivery will be required.
Strengths & limitations
The current study possesses several strengths, including the systematic nature of the review dual review of studies at each stage of analysis, and the global nature of the studies included in the final analysis. This review also possesses limitations that should be acknowledged; including restricting analysis to studies published in English and to those examining immunotherapy agents. It is possible that further studies exist that could provide insight into the cost-effectiveness and contribution of other aspects of RCC treatment, including surgical interventions, and thus warrants further research.
CONCLUSIONS
To our knowledge, this is the first contemporary systematic review examining literature of cost-effectiveness of immunotherapy approaches in RCC treatment, regardless of treatment setting. Immune checkpoint inhibitors constituted the most frequently examined agents and were likely to be deemed cost-effective as compared to other agents; although this often required higher WTP thresholds or healthcare systems that possessed more cost-constraints. Regional differences in pricing may lead to differences in measured benefit of use of immune checkpoint inhibitors in first- or second-line setting for advanced RCC. These observations have clinical and health system applicability, with the ability to potentially reduce cost of treatment for advanced RCC.
FUNDING
HB is funded by Prostate Cancer Foundation and Lazarex Cancer Foundation.
AUTHOR CONTRIBUTIONS
Study conception: EP, HB. Study review: EP, AB, DK, SZ, FW. Interpretation: EP, AB, DK, SZ, FW, HB. Manuscript development: EP, AB, DK, SZ, FW, HB. Manuscript editing and review: EP, AB, DK, SZ, FW, HB.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments.
