Abstract
Inflammation and immune dysregulation have been linked to the pathogenesis and progression of Parkinson’s disease (PD), and represent an attractive target for therapeutic intervention, given the potential for repurposing of existing anti-inflammatory and immunomodulatory agents. Despite the fact that initial studies of drugs with secondary anti-inflammatory effects did not yield positive results, agents specifically targeting immune and inflammatory pathways may hold more promise. This article will briefly review the evidence base for targeting the immune system and neuroinflammation in PD, and discuss in detail the recently completed and currently active trials of primary anti-inflammatory/immunomodulatory drugs in PD.
Plain Language Summary
Parkinson’s disease is caused by a loss of dopamine-producing nerve cells in the brain. Recent research has suggested that activation of the immune system, leading to inflammation in the brain and body, can contribute to this loss. Current medications that are used to treat Parkinson’s disease only help with the symptoms, and do not slow down the damage to nerve cells in the brain. New treatments, aiming to reduce inflammation and thereby slow disease progression, are under investigation in a number of clinical trials which are reviewed in this article. These treatments include medications that have been used in other diseases, as well as new drugs designed to target inflammation and immune activation in PD. Some of these early studies have had encouraging results but further larger trials are needed to determine whether medications targeting inflammation will have benefit for people with PD.
INTRODUCTION
In spite of effective symptomatic therapies which improve many of the motor symptoms of PD, the neurodegenerative pathology inexorably progresses, such that two thirds of patients will develop significant postural instability and nearly half will develop dementia within the first 10 years, 1 with no treatments available to slow this process. A wealth of research has suggested that sustained neuroinflammation, driven by both the innate and adaptive immune system, can precede and perpetuate neuronal degeneration in PD, making it a potential disease modifying therapeutic target.2–4
McGeer et al. initially proposed that sustained neuroinflammation is linked to the development and progression of PD in 1988, demonstrating an increase in reactive HLA-DR+ microglia in the substantia nigra at post-mortem, which correlated with neurodegeneration.
5
Subsequent
Microglia constitute the first line of the brain’s immune defense, responding to cell damage and pathogen-derived stimuli. Activation leads to the clearance of cellular debris and toxic proteins, subsequently leading to tissue repair. 10 Chronic activation of microglia leads to sustained secretion of pro-inflammatory cytokines, chemokines and neurotoxic factors, creating an uncontrolled inflammatory environment which is proposed to contribute to neurodegeneration in PD. Neuronal death, with associated release of misfolded alpha synuclein and other toxic species may lead to a vicious cycle of microglial activation andneurodegeneration. 11
The nucleotide-binding domain, leucine-rich–containing family, pyrin domain–containing-3 (NLRP3) inflammasome is a complex of proteins which forms on activation of microglia and other innate immune cells, and is a key amplifier of the inflammatory response. It has recently emerged as a potential contributor to the pathogenesis of PD, evidenced by elevated levels of NLRP3 and downstream cytokines such as IL1β in the blood in PD patients, 12 coupled with increased NLRP3 protein levels in dopaminergic neurons in the midbrain at postmortem. 13 NLRP3 inflammasome activation can be driven by the recognition and internalization of α-synuclein by microglia,14,15, 14,15 as well as by mitochondrial dysfunction, which is prominent in PD. 16 Upon inflammasome activation, pro-inflammatory cytokines IL-1β and IL-18 are released, and cell death can occur via pyroptosis. 16 The exact mechanisms of neurodegeneration due to inflammasome activation remain unknown; however, direct neurotoxicity and the seeding of protein aggregatesis proposed. 16
This pro-inflammatory environment in PD extends beyond the central nervous system. Multiple studies have documented elevated serum levels of inflammatory cytokines such as IL-6, TNF-α, IL-1β, IL-2, and IL-10 in PD, with a more inflammatory cytokine profile in early disease being associated with accelerated motor and cognitive decline.17,18, 17,18 An increase in the neutrophil to lymphocyte ratio (NLR), a marker of peripheral inflammation, can pre-date the development of PD and correlates with motor disease severity and dopamine transporter binding in the striatum.19–21 Moreover, numerous studies have revealed shifts in both innate and adaptive peripheral immune populations, with elevated levels of pro-inflammatory subsets. In the T lymphocyte population, this includes an increase in TH1 and TH17 pro-inflammatory effector cells, 22 and a lack of the CD8 lymphocyte immunosenescence that is normally seen with aging.23,24, 23,24 T cell subsets have been shown to correlate with motor severity25–27 and notably these effector T cells recognize alpha synuclein in PD, 28 and are present years before disease onset. 29 Studies have also demonstrated that there are fewer anti-inflammatory T-regulatory cells in PD,22,30,31, 22,30,31 and that these cells are less able to suppress the pro-inflammatory action of effector cells. 27 A similar pattern of a skew towards pro-inflammatory subsets is seen in other cell populations in the blood, including B cells,30,32, 30,32 NK cells33,34, 33,34 andmonocytes.35,36, 35,36
The relevance of the peripheral immune system in PD pathology has been explored in numerous PD models. In an
Genetic and epidemiological studies provide added support for the role of the immune system in the pathogenesis of PD. Genome wide association studies demonstrate a link between PD risk and polymorphisms in the human leukocyte antigen locus as well as genes involved in antigen presentation and recognition, inflammation and the complement system.41,42, 41,42 Moreover, proteins implicated in genetic forms of PD, such as LRRK2, are highly expressed in cells of the innate immune system and are involved in numerous immune signaling pathways. 43 Patients with a diagnosis of an autoimmune disease, including inflammatory bowel disease, have an increased risk of developing PD, 44 and this risk is attenuated in patients on anti-TNF immunological therapy. 45 Use of non-steroidal anti-inflammatory drugs and immunosuppressive therapies has also been reported to be associated with a lower risk of PD in large epidemiological studies.46–48
Overall, there is compelling evidence that the immune system plays a pivotal role in the pathogenesis of PD (Fig. 1). Coupled with promising results from therapies targeting inflammation in pre-clinical models of disease, this suggests that modulating inflammation may be a highly promising disease modifying therapeutic approach.
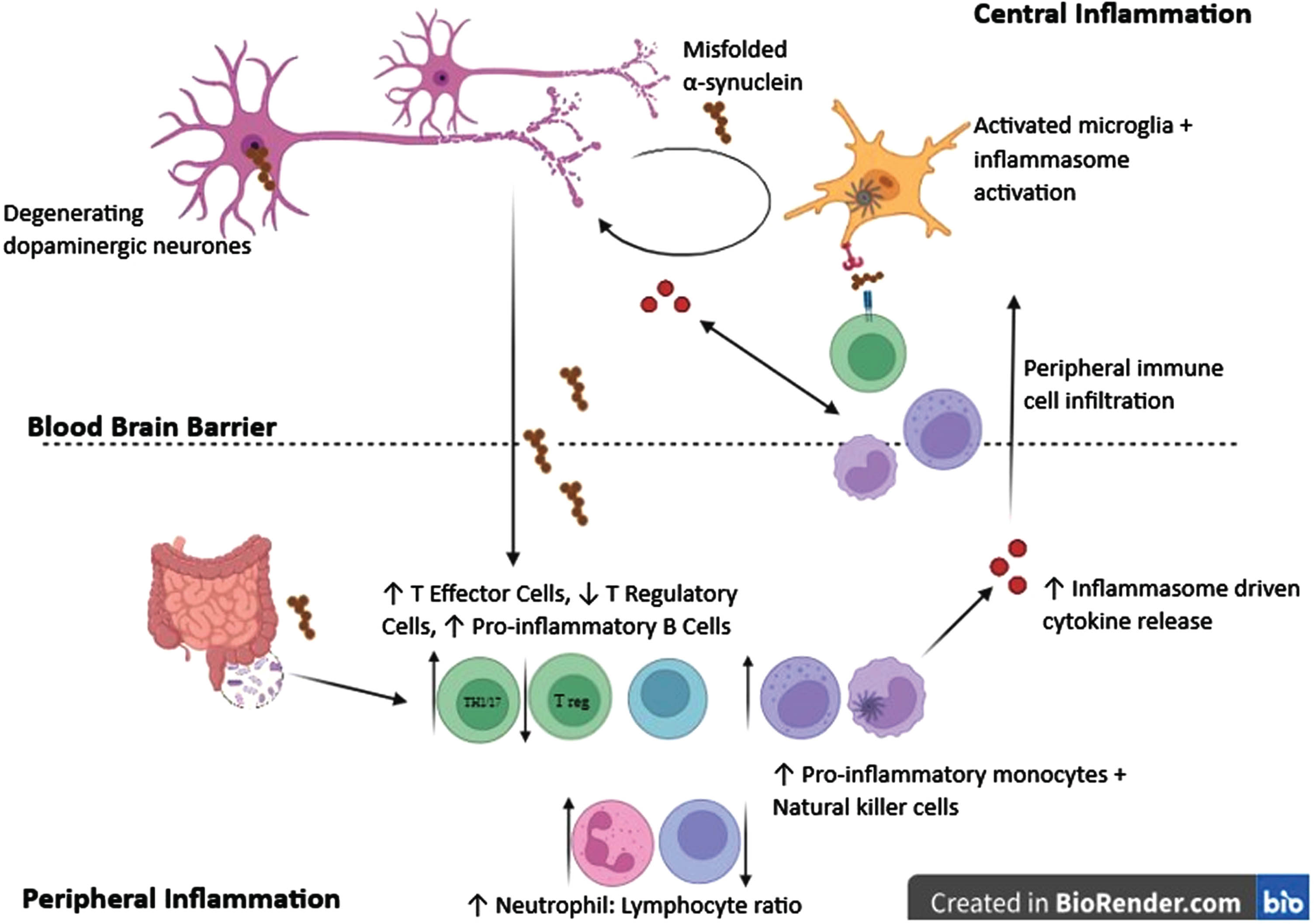
Overview of the immune system and inflammation in Parkinson’s disease (PD). Immune changes and inflammation occur both within the brain and periphery in PD. Activated microglia are found throughout the brain with evidence of NLRP3 inflammasome activation. This leads to the release of inflammatory cytokines which have a toxic effect on neurons, and release of chemokines which recruit peripheral immune cells to the central nervous system. Peripherally there is evidence of both innate and adaptive immune cell dysfunction with an increase in the neutrophil to lymphocyte ratio in the blood, and a shift towards a more inflammatory phenotype of T and B lymphocytes as well as natural killer cells and monocytes. NLRP3 inflammasome activation occurs within peripheral innate immune cells as well as centrally, and proinflammatory cytokines in peripheral blood are increased. Misfolded alpha synuclein may act as a driver of innate immune activation both peripherally and centrally, and trigger a specific adaptive immune response.
OVERVIEW OF ANTI-INFLAMMATORY/IMMUNOMODULATORY DRUGS IN PD CLINICAL TRIALS
Trials involving medications with secondary anti-inflammatory properties
Despite growing evidence for the role of the immune system in PD since the late 1980 s, historically there have been relatively few trials primarily targeting inflammatory mechanisms.
Trials using repurposed medications or supplements with secondary anti-inflammatory actions started to enter clinical trials in PD in the early 2000s.
Minocyline, a tetracycline antibiotic, was the first anti-inflammatory therapeutic agent tested in PD. It has been shown to penetrate the central nervous system and reduce microglial activation and proinflammatory cytokine production in animal models,49,50, 49,50 which is thought to be mediated through a reduction in the pro-inflammatory enzymes inducible nitric oxide synthase (iNOS) and caspase 1.50,51, 50,51 A phase 2 double blind placebo-controlled futility trial was completed in 2003 recruiting 200 participants randomized to receive either creatine, minocycline or placebo.52,53, 52,53 Futility criteria were a 30% reduction in Unified Parkinson’s disease rating scale (UPDRS) progression over the treatment period, based on data gathered from the placebo/tocopherol arm of the DATATOP (Deprenyl And Tocopherol Antioxidative Therapy Of Parkinsonism) trial. 54 Although minocycline was not rejected as futile, it was not progressed to phase 3, likely due to high discontinuation rates, safety concerns, and a lack of efficacy in slowing disease progression, as evidenced by comparable UPDRS scores to the placebo arm.52,53, 52,53
Pioglitazone, a peroxisome proliferator-activated receptor
Statins are thought to exert numerous neuroprotective actions, including reducing alpha synuclein aggregation, oxidative stress and microglial activation. 64 Nevertheless, the PD-STAT trial, a large multicenter phase 2 study recruiting 235 participants, with a treatment duration of 2 years, demonstrated no beneficial impact of simvastatin on PD progression. 65 Similarly, a 48-week trial of lovastatin in 77 PD patients, showed a trend in reduction of UPDRS part III scores which did not reach statistical significance. 66 Inflammatory biomarkers were not assessed as a part of either of these trials.
Gemfibrozil (FHL-301), currently used in lipid modification, has also entered phase 2 clinical trials in PD (NCT05931484). Gemfibrozil has been found to increase the production of glial derived neurotrophic factor in astrocytes and protect dopaminergic neurons in PD models. 67 It is also thought to have anti-inflammatory effects mediated via a reduction in pro-inflammatory cytokines leading to lower production of nitrous oxide and expression of iNOS, 68 as well as reducing microglial activation and reducing release of pro-inflammatory cytokines in response to lipopolysaccharides. 69 Forest Hills Labs are running a phase 2, double blind placebo-controlled trial with a 3-week dose titration period followed by a 48 week treatment period in 32 participants with early phase PD, with change in MDS-UPDRS part III as the primary outcome measure. However, the trial does not appear to include analysis of inflammatory biomarkers. Completion is anticipated at theend of 2024.
Vinpocetine, an agent that improves cerebral blood flow and has anti-inflammatory effects through phosphodiesterase mediated pathways, was explored in a short, randomized, double-blinded study of 89 participants who received 2 weeks of treatment. 70 The active treatment arm showed reduced TLR2 and TLR4 gene expression and downstream production of the pro-inflammatory cytokine TNFα. 70 Cognitive performance also improved post treatment, although this finding is difficult to interpret in such a short study and likely due to a learning effect of repetitive testing over 2 weeks, evidenced by a similar increase in the standard therapy arm. 70 Nevertheless, the biological effect is of interest given that TLRs have been implicated in mediating an inflammatory response to aberrant alpha synuclein in PD, 71 and further exploration through a longer duration placebo-controlled trial could beconsidered. 72
Niacin, a form of vitamin B3, reduces the translocation of NFkβ, a key transcription factor in the inflammatory cascade, and the production of downstream inflammatory cytokines, in response to LPS stimulation in a cellular model of PD. 73 This is thought to be mediated through its interaction with the niacin receptor GPR109A. 73 Supplementation has minimal side effects hence this is an attractive therapeutic candidate. However, in a recent trial in 47 PD patients, no significant change in UPDRS-III scores was observed during the 6-month double-blind placebo-controlled phase. An improvement in motor function was observed in a subsequent 6-month open label phase, but a placebo response seems the likely explanation for this given the lack of benefit in the placebo-controlledphase. 74
Sulforaphane, a compound found in vegetables such as broccoli and cauliflower, has anti-oxidant, anti-inflammatory, and anti-apoptotic properties, which has sparked interest into its use as a supplement in numerous neurodegenerative disorders including PD.
75
In
Currently active trials targeting the immune system/inflammation
The growing awareness of the role of inflammation and the immune system in the etiology of PD has led to an increasing number of early phase trials of novel and repurposed drugs specifically targeting immune and inflammatory pathways. For this review, both ClinicalTrials.gov and WHO clinical trials registries were searched for all active interventional clinical trials of drugs with primary anti-inflammatory/immune modulating mechanisms in Parkinson’s disease. Eight clinical trials were identified as either active or recently completed (within the last two years) (Table 1).
Clinical trials of immunosuppressive/anti-inflammatory drugs in PD which are either active or recently completed
A phase 2 trial of azathioprine commenced recruitment in 2021 and is the first to repurpose a peripherally-acting immunosuppressant drug for PD. It is a randomized, double-blind, placebo-controlled trial of 60 patients with early PD, receiving treatment for one year, with a primary outcome measure of the gait-axial subscore of the MDS-UPDRS. 77 The results of this trial are expected in 2024 and remain highly anticipated as the drug itself does not cross the blood brain barrier. Hence if the results of this trial are positive, it will support the role of interplay between the peripheral and central immune system contributing to the progression of PD. The trial incorporates proof of mechanism outcome measures including immunophenotyping in the blood and cerebrospinal fluid and TSPO PET/MR imaging to evaluate brain inflammation. 77
Sargramostim, a human recombinant granulocyte-macrophage colony-stimulating factor (GMCSF) that can induce T-regulatory immune cell responses, is currently undergoing extended phase 1 testing. Initial results with 8 weeks of treatment in 20 patients with PD demonstrated target engagement in terms of increasing the numbers and suppressive function of peripheral T regulatory cells, and there was a modest reduction in UPDRS part III scores in the sargramostim-treated versus the placebo arm. 78 Unfortunately, there were numerous side effects at the 6 μg/kg/day dose, with daily subcutaneous injections leading to significant injection site reactions. 78 Ongoing multi-year trials with fewer participants receiving lower treatment doses (3 μg/kg on five days per week) are underway in a bid to improve its tolerability in the PD population, however its efficacy at this dose needs furtherexploration.79,80, 79,80
With concerns over the tolerability of Sargramostim, a novel, longer acting GMCSF, PDM608, is under development in a phase 1 safety, tolerability and dose finding study in healthy volunteers (NCT05950906). Following a single administration in MPTP mouse models, PDM608 has shown an anti-inflammatory effect with a reduction in activated microglia and increased T-regulatory cell numbers and function for 10 days. 81 If successful in humans, a single subcutaneous injection could be given 1-2 weekly, alleviating the need for daily injections, hopefully mitigating some of the adverse events seen in the Sargramostim trialsto date.
Montelukast, a cysteinyl leukotriene receptor type 1 (CysLT1) antagonist, currently licensed as an oral therapy for asthma, has shown efficacy in animal models of PD.82,83, 82,83 In a phase II open label trial in 15 patients, the medication was well tolerated with no significant safety concerns despite the high doses used (four times the licensed dose in asthma), with a significant improvement in MDS-UPDRS part I–III scores post treatment (EUCTR2020-000148-76-SE). Intelgenx has recently won approval from the Swedish Medicines Agency for a new formulation of montelukast, which is a thin film applied orally which can be absorbed directly and without water, and will be trialed in a large phase 2, multi-center, double-blind, randomized, placebo- controlled trial (EudraCT number 2023-504278-39-00) in PD. 84 This has a 50% higher bioavailability, bypasses first pass metabolism, thereby lowering potential toxic effects. 84 The new trial aims to recruit 90 participants who will be randomized to receive montelukast or placebo for a duration of 18 months.
With inflammation now implicated in a broad range of diverse diseases, research is focusing on identifying and modifying novel therapeutic targets. To date, two novel anti-inflammatory agents have completed trials in PD. NE3107 is an orally administered brain penetrant molecule that binds to and inhibits extracellular signal-regulated kinase (ERK) mediated pathways in macrophages, leading to a decrease in the downstream inflammatory cascade. 85 In a phase 2 double-blind placebo-controlled trial in 46 participants over 28 days, treatment with NE3107 resulted in a clinically meaningful benefit in PD with a 3-point reduction in UPDRS part III scores and a reduction in OFF time in comparison to placebo. 86 AKST4290 is a small molecule antagonist of CCR3, a chemokine receptor expressed on leucocytes, as well as on microglia and astrocytes in neuroinflammatory conditions, 87 which plays a role in T cell migration into the brain. 88 In a randomized placebo-controlled phase 2 trial with 12 weeks of oral treatment in 110 participants with PD, there was no evidence of a beneficial change in the primary outcome measure of motor progression (MDS-UPDRS Part III) (NCT04369430). 89
The NLRP3 inflammasome, an integral component of the innate immune system, has attracted much interest in recent years, leading to the development of multiple novel selective inhibitory molecules. Some of these molecules can cross the blood-brain barrier, with the potential for translation to PD clinical trials. Nodthera have recently announced completion of their phase 1b/2a trial on NT-0796, an orally administered brain penetrant NLRP3 inhibitor. 90 Full details of the trial protocol and results are not yet available, but a press release has suggested that the novel molecule had an impact on peripheral and central cytokine production, lowering levels of IL-1β, IL-6, CCL2, CXCL1, and CXCL8 in up to 10 PD patients following 28 days of treatment, with minimal adverse events. 90 A phase 1b, randomized placebo-controlled trial investigating selnoflast, another orally administered NLRP3 inflammasome inhibitor, is underway. The investigators are aiming to recruit 72 participants with early PD, treating participants for 28 days with either selnoflast or placebo, with primary outcome measures related to safety and tolerability and secondary outcomes assessing pharmacokinetics and target engagement using [ 18 F]-DPA-714 TSPO PET. Results are expected in 2025 (NCT05924243).
The authors of this review are leading a phase 2 randomized, double-blind, placebo-controlled trial using the NLRP3 inflammasome inhibitor dapansutrile 91 , developed by Olatec Therapeutics. The trial is currently in set-up. Dapansutrile penetrates the blood-brain barrier and has neuroprotective effects in MPTP mouse models of PD. 92 Early phase trials in healthy volunteers, 93 gout, 94 and heart failure 95 have shown a good safety and tolerability profile with a treatment-induced reduction in levels of pro-inflammatory cytokines IL1β and IL6 in the blood. This trial aims to recruit 36 participants with early phase PD, with an initial randomized double-blind placebo-controlled phase of six months, followed by a 6-month open label phase. 91 The primary outcomes will be safety and tolerability, with blood and CSF inflammatory markers and [ 18 F]-DPA-714 TSPO PET imaging used to evaluate target engagement, and exploratory clinicalmeasures.
Two other early phase trials of novel NLRP3 inhibitors in PD have recently been announced. Ventyx Biosciences have completed a phase 1 trial in healthy volunteers with their novel CNS penetrant NLRP3 inflammasome inhibitor VTX3232, which was well tolerated with only mild to moderate adverse events recorded and positive results noted in whole blood IL1β stimulation assays. 96 They have announced a phase 2a trial in patients with PD, however there is no further information available about trial design yet. 96 Similarly, Zydus have completed a phase 1 trial in which 30 healthy male volunteers were recruited into a single ascending dose study, receiving a single dose of ZYIL1, and 18 healthy male volunteers were recruited into a multiple ascending dose study receiving ZYIL1 for a total of 14 days. A total of 12 adverse events were recorded over the two studies ranging in severity from mild to severe, and there was evidence of >90% reduction in IL1β and >70% reduction in IL18 release in whole blood from treated participants stimulated with LPS/ATP to activate the NLRP3 inflammasome in comparison to pre-treatment levels. 97 They have recently gained FDA approval to start a phase 2 clinical trial in PD but further details are currently unavailable. 98 NLPR3 inflammasome inhibitor studies which have recently completed, are active or planned, are summarized in Table 2.
Clinical trials of NLRP3 inflammasome inhibitors in PD
TBC, To be confirmed.
CONCLUSION
There is considerable evidence that immune activation contributes to the pathogenesis and progression of PD. Multiple tractable therapeutic targets within immune and inflammatory pathways have been identified and whilst as yet no definitively positive results have emerged from large-scale trials, several early phase studies have reported promising efficacy signals, warranting further exploration.
Immune and inflammatory mechanisms interact with multiple other complex pathological processes involved in the pathogenesis of PD, including protein aggregation, mitochondrial dysfunction, and disrupted autophagy-lysosomal mechanisms. It is likely that the contribution of these pathogenic processes varies between individuals and this may account for some of the clinical heterogeneity observed in the symptoms and rate of progression of PD. Although rarely considered in clinical trial design, both clinical and biological heterogeneity of study cohorts may have a major impact on outcomes, particularly in small scale trials. Lack of phenotypic stratification may have contributed to negative outcomes in some of the previously completed early-phase trials in this field. In future trials of immunosuppressive and anti-inflammatory therapies in PD, selecting participants based on inflammatory phenotypes using baseline biomarker analyses could potentially enhance success.
APPENDIX: SUMMARY OF IMMUNOSUPPRESSIVE/ANTI-INFLAMMATORY AGENTS UNDER ACTIVE INVESTIGATION
The eight clinical trials of immunosuppressive/anti-inflammatory drugs which are either active or recently completed are listed in Table 1 and summarized in more detail in the section below.
A clinical trial repurposing azathioprine as a potential disease modifying therapy for PD was commenced in 2021 and completed in February 2024 but results are not yet available. This proof of concept study explores the hypothesis that peripheral immune activation is a significant driver of central immune activation and disease progression through specifically targeting the peripheral immune component.
The adaptive immune response has been strongly implicated in PD, with changes in both pro-inflammatory and regulatory lymphocyte subsets. In particular, a reduction in T regulatory cells has been reported with an impaired ability to suppress the proliferation of T effectors cells.27,31,103, 27,31,103 Increasing T regulatory cells through treatment with sargramostim mitigates inflammation, microgliosis and neuronal death in MPTP mice models of disease.104,105, 104,105
In 2016, a phase 1 randomized double blind, placebo-controlled trial of sargramostim (NCT01882010) was completed. Twenty PD patients were randomized in a 1:1 ratio to receive sargramostim at a dose of 6 μg/kg/day administered subcutaneously or placebo for 56 days. 78 The primary outcomes were safety and tolerability, and the trial was not powered to assess clinical efficacy. Adverse events were reported in all participants in the treatment arm, with the most common being injection site reactions, abnormal blood results and bone pain. 78 There was evidence of the anticipated effect on immune populations, with an increase in numbers of T regulatory cells and functional assays showing a restoration in their ability to suppress T effector cells. 78
Phase 1 testing of sargramostim has recently resumed but at a lower dose due to concerns regarding the tolerability of the drug at the initial doses used. Two related trials from the same academic team are currently underway.
A Phase 1, Open-label Study to Validate Treat-ment-induced Biomarkers Following Sargramostim Treatment in Parkinson’s Disease (Trial 2)
In models of CNS disease, including MPTP induced mouse models of PD, CysLT1 expression is increased. 108 Inhibition of this pathway with montelukast can attenuate dopaminergic neuronal loss, reduce microglial activation and reduce the production of pro-inflammatory cytokines TNFα and IL1β in multiple animal models of PD.82,83,109, 82,83,109 Inhibition and knockout of 5-lipo-oxygenase, which is essential in the production of leukotrienes is also neuro-protective.110,111, 110,111
To investigate the potential of montelukast as a disease modifying therapy in PD, a small trial assessing safety and tolerability was completed in 2022.
ERK is a part of the mitogen activated protein kinase family that has an essential role in coordinating cellular responses to external stimuli. 113 In human PD and DLB brains, cytoplasmic inclusions of phosphorylated ERK have been found in neuronal cells of the substantia nigra, even in early disease stages. 114 ERK mediated pathways have also been implicated in mouse models of levodopa induced dyskinesias, further supporting targeting this pathway in PD. 115
In an MPTP mouse model of PD, NE3107 attenuated dopaminergic neuronal loss, preserved motor abilities on the rotarod test, and reduced expression of proinflammatory mediators such as iNOS, TNF-α, and IL-1β. 112 In a marmoset model of PD, NE3107, administered after MPTP, was neuroprotective and led to improvements in mobility in comparison to placebo. 116 This effect was increased with the co-administration of levodopa and a reduction in levodopa induced dyskinesias was observed. 116
A phase 2 trial investigating NE3107 in PD completed in January 2022.
The evidence supporting the role of CCR3/CCL11 in PD pathogenesis is variable. Serum and CSF levels of CCL11 and expression of CCR3 on peripheral eosinophils are not increased in PD in comparison to controls,119–121 but levels of CCL11 are increased in microglial cells of the substantia nigra in post-mortem PD human and MPTP mice brains. 122 Treating MPTP mice with peripheral neutralizing antibodies to eotaxin and RANTES/CCL5 (another chemokine which also binds to CCR3 with medium affinity) reduced inflammatory cytokine production, peripheral T cell infiltration into the substantia nigra and dopaminergic neuronal death leading to an improvement in motor deficits, 122 further supporting targeting CCR3 in PD.
A clinical trial assessing the efficacy and safety of AKST4290 was completed in 2021.
Although no positive pharmacodynamic results were recorded
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
BP is supported by the National Institute of Health Research, UK (NIHR). JCG was supported by the Cambridge Centre for Parkinson-Plus. CHWG is supported by the Medical Research Council, UK (MR/W029235/1) and the NIHR Cambridge Biomedical Research Centre (NIHR203312). The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care. The authors also receive grant support from Cure Parkinson’s, Parkinson’s UK, the MRC, the Rosetrees Trust, and Addenbrooke’s Charitable Trust.
CONFLICT OF INTEREST
The authors have no conflicts of interest to report.
