Abstract
For decades the greatest goal of Parkinson’s disease (PD) research has often been distilled to the discovery of treatments that prevent the disease or its progression. However, until recently only the latter has been realistically pursued through randomized clinical trials of candidate disease-modifying therapy (DMT) conducted on individuals after they received traditional clinical diagnosis of PD (i.e., tertiary prevention trials). Now, in light of major advances in our understanding of the prodromal stages of PD, as well as its genetics and biomarkers, the first secondary prevention trials for PD are beginning. In this review, we take stock of DMT trials to date, summarize the breakthroughs that allow the identification of cohorts at high risk of developing a traditional diagnosis of PD, and describe key design elements of secondary prevention trials and how they depend on the prodromal stage being targeted. These elements address whom to enroll, what interventions to test, and how to measure secondary prevention (i.e., slowed progression during the prodromal stages of PD). Although these design strategies, along with the biological definition, subtype classification, and staging of the disease are evolving, all are driven by continued progress in the underlying science and integrated by a broad motivated community of stakeholders. While considerable methodological challenges remain, opportunities to move clinical trials of DMT to earlier points in the disease process than ever before have begun to unfold, and the prospects for PD prevention are nowtangible.
Keywords
Now is the time to start conducting clinical trials for the prevention of Parkinson’s disease (PD), a chronic multisystem progressive neurodegenerative condition. The rising prevalence of the disease with its substantial societal and economic burden1–5 adds to the urgency of pursuing its prevention. Although efforts to date have focused on modifying the disease course after its diagnosis, recent advances in our understanding of the very early stages of PD (i.e., preclinical and prodromal clinical) along with the establishment of at-risk cohorts, and the discovery of potential early diagnostic biomarkers for PD, have made it feasible to design and implement trials for PD prevention (which in this review refers to the prevention of traditionally diagnosed PD unless otherwise specified). In fact, several clinical protocols for trials of candidate PD preventatives are in the pipeline now, with a couple of them actively recruiting participants.6–8
In keeping with the circa 1735 adage of Benjamin Franklin that, “An ounce of prevention is worth a pound of cure”, 9 preventing PD would have an enormous impact not only on the individual but also on society at large via reduced burden on the population, healthcare, government, etc. Prevention medicine, at its simplest, promotes health and prevents disease, and can be classified as primary, secondary, or tertiary in nature, as shown in Table 1 using the example of a dental cavity.
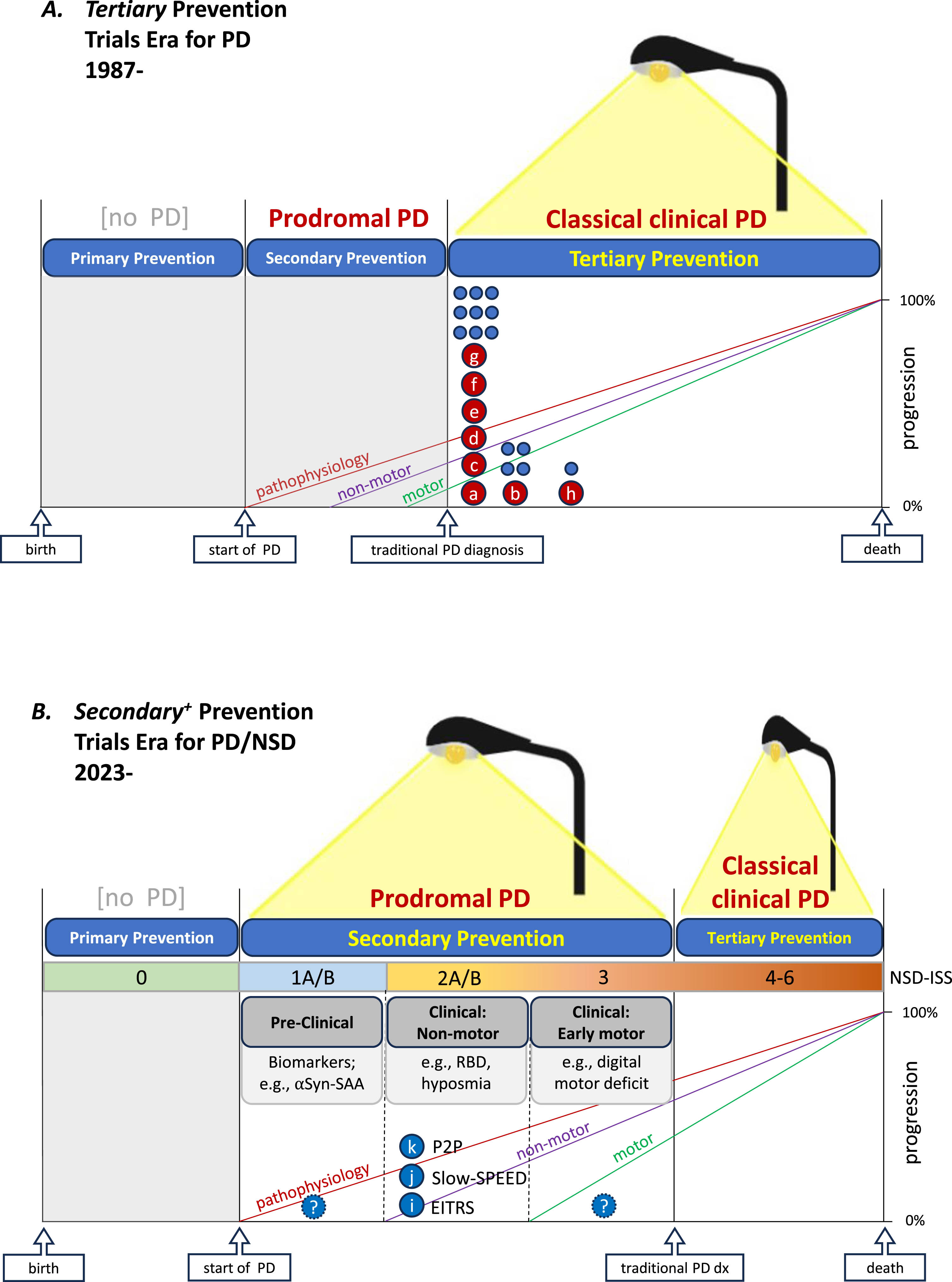
Given the advances in our understanding of PD pathophysiology, prevention medicine can also be applied to PD (Fig. 1) with: primary prevention to reduce the risk of developing PD (prodromal or traditional motor PD) in individuals without PD; secondary prevention to stop or delay the development of traditional motor PD (e.g., as defined by current clinical diagnostic criteria for PD) during the prodromal period of the disease, which defined as the time between the start of a discrete measurable pathophysiologic process (often characterized by α-synuclein-based biomarkers) and the traditionally diagnosable state of PD or related neurodegenerative synucleinopathy like dementia with Lewy bodies (DLB); and, tertiary prevention to slow the progression of classical parkinsonian features and the development of complications like motor fluctuations and cognitive impairment in individuals with established PD (i.e., after traditional clinical diagnosis).
Prevention medicine using the example of a dental cavity
PARKINSON’S DISEASE-MODIFICATION TRIALS TO DATE (TERTIARY PREVENTION)
To date, clinical trials in PD have pursued tertiary prevention through disease modification in individuals with a traditional diagnosis of PD. Since 1987 there have been numerous phase 3 tertiary prevention trials in PD (Fig. 1A). Most of these trials have enrolled participants with de novo PD (usually within 2 years of diagnosis, and not yet started on symptomatic dopaminergic therapy). Enrolling these participants as early as possible after diagnosis is in keeping with the broader principle that the earlier one intervenes with a disease-modifying therapy (DMT) in the pathophysiology and neurodegeneration of PD the more likely it will be effective. Intervening too late in the disease process, as proverbially presaged and discouraged by John Heywood through his 1546-published English aphorism, “Don’t shut the barn door after the horse has bolted” (as adapted from Old English), could be one reason why no tertiary prevention of PD has been established to date. Intervening with a DMT in participants with prodromal PD (Fig. 1B) may be more fruitful.
Although for decades there has been ample evidence that dopaminergic neuron deficits and various motor and non-motor symptoms precede the traditional clinical diagnosis of PD by years or decades, 10 the inability to clearly define at-risk populations precluded the enrollment of earlier clinical cohorts until recently. Similarly, tertiary prevention trials of candidate DMT have typically based their primary trial outcome on imperfect but validated clinical measures of motor deficit progression (as determined by clinicians and/or patients).11–18 More sensitive motor and non-motor clinical measures of disease progression before the diagnosis, not to mention biomarkers of progression that would be needed to gauge the efficacy of DMT before the diagnosis, had not yet been developed and validated. The development of these tools and strategies for the design of the first (secondary) prevention trials for PD should benefit greatly from collaborative engagement across a community of invested stakeholders comprising newly mobilized advocates from at-risk populations, 19 as well as from academic, government, industry, philanthropic, and regulatory partners committed to preventing the disease.
SECONDARY PREVENTION TRIALS
As noted above, advances in our understanding of the prodromal period of PD and the identification of prodromal features placing people at high risk for developing PD have provided an opportunity to modify the course of PD earlier, and enabled the field to design and implement secondary prevention trials now. The MDS Research Criteria for Prodromal PD were established in 2015 and updated in 2019, providing a framework to calculate an individual’s probability of being in the prodromal period of PD based on the presence of risk and prodromal PD markers. 20 Polysomnogram-proven REM sleep Behavior Disorder (RBD), a “clearly abnormal dopaminergic PET/ SPECT scan”, “subthreshold parkinsonism”, hyposmia, and neurogenic orthostatic hypotension are some of the markers among these criteria with a high likelihood ratio for prodromal PD. 20 Building on the elevated PD risk conferred by these prodromal features, PREDICT-PD, a UK-based predominantly online study used PD epidemiological risk factors (i.e., coffee consumer, NSAID use, head injury, etc.) to designate risk scores of low, medium, and high for later development of PD.21,22, 21,22 They then validated the risk score result with the presence of other high-risk prodromal PD features: RBD, hyposmia, and reduced finger tapping speed.
In addition to those clinically at risk for PD development due to the presence of motor and non-motor symptoms, some individuals are instead (or in addition) genetically at risk for PD. These individuals have genetic variants associated with a risk of developing PD later in life. The most prevalent pathogenic genetic variants are those in the LRRK2 and the GBA genes, which may confer a PD risk of 43% and 19% by 80 years of age, respectively.23,24, 23,24 Other more penetrant but less prevalent genetic variants, like several in the SNCA gene, are compelling to pursue but given their rarity, they are more difficult and less practical to target in early secondary prevention efforts.
In recent years, cohorts of clinically at-risk and genetically at-risk participants have been established. Given the high-risk nature of RBD and hyposmia, most of the cohorts to date have included participants with one or both of these features. The North American Prodromal Synucleinopathy (NAPS) Consortium for RBD has enrolled 361 participants with RBD, and these participants are undergoing longitudinal comprehensive clinical assessment. 25 The Parkinson’s Progression Markers Initiative (PPMI) is currently enrolling a “prodromal cohort” of participants who meet the following sequential criteria: age 60 years or older; presence of risk factors or clinical features related to PD: have RBD, known genetic risk variants, or “other known PD risk criteria including those based on questionnaires in PPMI Online”; hyposmia based on UPSIT testing; and a positive dopamine transporter (DAT) SPECT on visual inspection. 26 These participants undergo comprehensive clinical assessments along with the collection of biological fluids and serial neuroimaging. Participants in PPMI may be eligible to participate in the Path to Prevention (P2P) platform trial, a phase 2A, randomized, double-blind, placebo-controlled study to evaluate multiple investigational drugs in prodromal PD, 27 which will be further discussed below.
In addition to the identification and further characterization of at-risk groups of participants, there has been ongoing research into biomarkers of risk. A recent and notable breakthrough in this area is the discovery and validation of early measures of PD pathophysiology centered on synucleinopathy, like the α-synuclein (αSyn) seed amplification assay (SAA) in cerebrospinal fluid (CSF) or phosphorylated α-synuclein detected by immunohistochemistry of the skin.28–31 The SAA has been studied in several different biological fluid samples (i.e., blood (plasma and serum), nasal secretions, and saliva) and has shown high diagnostic sensitivity and specificity in them for detecting PD.29,30,32, 29,30,32
TRIAL DESIGN ELEMENTS: PRODROMAL STAGE-DEPENDENCE
With the identification of multiple at-risk populations, candidate therapeutics, and potential outcome measures suitable for prevention trials comes the challenge of their collective selection in designing a given prevention trial. In addition to their reliance on one another, key design elements for secondary prevention trials depend on the prodromal stage targeted, and vice versa 33 —as illustrated below and in Fig. 2. The prodromal period of PD (or more broadly, of neurodegenerative synucleinopathy) can be divided into three largely sequential stages: preclinical, clinical non-motor, and clinical early motor. (See Figs, 1B and 2).

1)
a)
Another practical impediment to establishing a trial-ready cohort for secondary prevention in preclinical PD based on an αSyn biomarker is the relative invasiveness of the lumbar puncture used for the best validated αSyn biomarker at present. However, the prospects for developing a more accessible, peripheral measure for αSyn-SAA are rapidly expanding (e.g., in neuronal extracellular vesicles from plasma 32 or in αSyn immunoprecipitates from serum 34 ), and other αSyn pathobiology measures such as phosphorylated forms of αSyn in skin biopsies, 31 which have been associated with RBD35,36, 35,36 as well as PD, may provide alternative αSyn markers for cohort enrichment in preclinical prodromal PD. Stage 1 of the recently proposed neuronal synuclein disease integrated staging system (NSD-ISS), 37 which corresponds closely to the preclinical stage of prodromal PD (Fig. 1B), was inferred based on CSF αSyn-SAA data, yet this system similarly allows for alternative, adequately validated αSyn biomarkers to define active disease.37,58, 37,58 Of note, while NSD-ISS considers early PD pathophysiology to be anchored in αSyn (neuronal αSyn, more specifically and by definition), other formulations rely on mechanistically less restrictive or more heterogeneous definitions of PD allowing for synuclein-independent pathophysiology. Thus, it remains plausible that novel, αSyn-unrelated biomarkers could help define a preclinical prodromal PD cohort at high risk of PD and its prodromal features (hypothetically, for example, a mitochondrial DNA damage marker). 38
b)
Genetically at-risk cohorts targeted for secondary prevention trials may be recruited from any stage of prodromal PD, but at present most practically from its clinical stages. PD gene carriers can be efficiently screened for non-motor features (e.g., RBD or hyposmia) or early parkinsonian motor deficits via simple questionnaires and/or at-home testing (e.g., scratch and sniff tests for hyposmia, smartphone assessments for slow gait and finger movements, rest tremor, and asymmetry.) The anticipated advent of non-invasive, blood-based αSyn markers (as above) would allow for enrollment at the preclinical stage, although such screening may exclude a substantial portion of
c)
d)
Slow-SPEED (in its initial Dutch version Slow-SPEED-NL; NCT06193252 8 ), like EITRS is enrolling only RBD patients but in contrast allows those with isolated parkinsonian motor features (i.e., short of a PD/neurodegenerative disease diagnosis), with recruitment active as of January 2024. Although numerous serial motor and non-motor clinical outcome measures will be collected, the primary outcome assessment is the feasibility of increasing step count as an indicator of increased voluntary physical activity (exercise) over two years. Participants will be randomized 1 : 1 to receive guidance to gradually increase the volume and intensity of their step count-based exercise either by a modest or substantial amount relative to their personal baseline activity per instructions provided by a motivational Slow-SPEED-NL smartphone study app.
P2P proposes to enroll a more clinically heterogeneous prodromal cohort accepting RBD alone, hyposmia alone, or both as the requisite non-motor anchor (while allowing early motor features short of a PD/neurodegenerative disease diagnosis) beginning in early 2025. Conversely, it will more narrowly define the cohort biologically, with eligibility requiring αSyn-SAA positivity and a DAT deficit on neuroimaging (per most recently posted protocol information), 27 consistent with Stage 2B or higher by the NSD-ISS. 42
The requirement of a DAT deficit to enroll in P2P rather than allowing αSyn-SAA+/DAT- (formally a Scan Without Evidence of Dopaminergic Deficit (SWEDD) consistent with NSD-ISS Stage 2A) may reflect a difficult decision in defining a prodromal cohort. While excluding SWEDDs altogether increases the prospects for further DAT deficit during the follow-up period, there may be a favorable intermediate option to include DAT deficits that approach but fall below the current quantitative threshold that conservatively avoids false positives. A less stringent cutoff may facilitate recruitment while enhancing prospects for demonstrating efficacy with an earlier stage population, especially if a change in DAT signal was a primary outcome in this phase 2 study. That could better inform a decision of whether to advance to a clinically focused phase 3 prevention trial. Such nuanced design decisions may depend on a growing knowledge base on the dynamic range of imaging and other biomarkers across prodromal stages, as well as the specific intervention and outcomes, under study, and program or sponsor considerations.
e)
1. Risk of excluding populations commonly underrepresented in research: Identification of at-risk participants requires additional financial and technological resources, the lack of which may preclude prevention trial participation in many low and middle-income countries, and among disadvantaged groups within wealthier nations. Investigators, funding bodies, regulators, and sponsors should work to enhance local and global representation as lack of diversity in trials affects the generalizability and accuracy of their results along with increasing inequity and injustice. Thus, extra attention, effort, and cost will be required to ensure the representation among participants.43–45 The Global Parkinson’s Genetics Program (GP2), an initiative of the Aligning Science Across Parkinson’s (ASAP), is an excellent example of investing in a global research community, inclusive of underrepresented groups in PD genetics. 46 In addition, global representation in prevention trials may be aided by innovative, decentralized, and accessible methods such as a smartphone-based exercise app used both for intervention implementation and outcome assessment. This approach employs relatively low-cost technology that is widely available even in many developing countries, and its ongoing validation may be scalable up for international, multi-lingual cohorts of individuals at high risk of or living with PD. 47
Similarly, prevention trials should ensure proportional representation of women, a group who are also generally underrepresented in manifest PD.48,49, 48,49 In at-risk individuals with iRBD, the ratio of male:female is comparable to that in manifest PD, in which a higher male prevalence is well established. 50 Thus, strategies will be needed to recruit and enroll a greater percentage of women with iRBD than typically enrolled in PD studies. Unlike idiopathic disease, PD due to a pathogenic variant of the LRRK2 gene shows parity of prevalence between sexes, thus setting the goal at 1 : 1 for male:female enrollment in LRRK2 PD prevention trials. 51
2. Risk of causing psychosocial harm: Understandably there is concern that these prevention studies in at-risk individuals may cause psychosocial harm to participants via learning about their elevated risk of PD (“risk disclosure”) or the intervention that they may receive during the trial. Risk disclosure of any type requires careful execution by an experienced professional. However, many at-risk individuals for PD feel empowered and are motivated to participate in clinical research. 19 Furthermore, research on risk disclosure in a related neurodegenerative disorder, Alzheimer’s disease has not shown increased psychosocial harm.52–54 Current prevention studies, like Slow-SPEED-NL and P2P are recruiting adults who have undergone genetic testing for PD pathogenic variants or who were previously diagnosed with iRBD by their physician who may explain their increased PD risk before being recruited into the study. Interested individuals will also generally be engaged adults who can give informed consent to participate in research of their choosing.
2)
It is plausibly reasoned that what initiates a neurodegenerative disease can also perpetuate it – especially when neuronal degeneration is known to be a common process that underlies progression prior to and after traditional diagnosis. On the other hand, there is a strong intuitive rationale and some empirical evidence to suggest an effective DMT for (secondary) prevention may differ markedly from an effective DMT for slowing classical PD (tertiary prevention). Genetically, while some pathogenic gene variants (e.g., GBA variant L444P) can predict a higher risk of PD and a faster rate of its clinical progression, others (e.g., LRRK2 variant G2019S) show a dissociation of increased risk from a slower rate of clinical decline. 55 Thus these human data more directly support the rationale for secondary prevention trials of LRRK2 kinase inhibitors, while raising concerns over efficacy in PD trials for tertiary prevention (currently in overall encouraging phase 2 testing being conducted with clinical equipoise; NCT05348785). 56
Similarly, epidemiological studies of large, prospectively followed, initially healthy cohorts have identified several dietary, pharmacological, or lifestyle factors (e.g., caffeine consumption, ibuprofen use, glucagon-like peptide-1 (GLP-1) agonist prescription, and smoking) that predict a reproducibly reduced rate of PD diagnosis but not of PD progression after diagnosis. For example, higher caffeine consumption in unaffected individuals consistently predicts a lower risk of PD, whereas its use in people with PD is not a reliable predictor of slower progression.57,58, 57,58 Thus, even though caffeine has robust neuroprotective properties across animal models of PD, human data provide a stronger rationale to test it for PD prevention (primary or secondary) than for progression after a traditional diagnosis (tertiary). 9
a)
While low-risk and repurposed interventions may be particularly appropriate for preclinical prodromal PD (when such studies become practical), they are at present practically best suited to non-motor prodromal PD states like RBD, hyposmia, and/or other non-motor prodromal features. (See Fig. 2, row 2a.) And indeed, the two therapies currently in the first actively enrolling randomized control trials for secondary prevention are widely available with the nutritional supplement idebenone in the EITRS trial NCT04534023, 7 and titrated exercise guided via an interactive, smartphone-based app in the Slow-SPEED-NL trial NCT06193252. 8 As noted above, Slow-SPEED-NL but not EITRS allows early motor as well as non-motor prodromal features, which will aid recruitment and enrich RBD for those at greater risk of PD versus DLB, and for better or worse shifts the cohort to a slightly more advanced diseasestate.
While these interventions target general oxidative and cellular stress pathways, other repurposable agents may credibly target the products of major PD genes and thus may be considered more compelling candidate preventatives. For example, ambroxol an over-the-counter mucolytic has been found to increase CSF levels of GCase (the GBA1 gene product) in PD, 59 and is being pursued for tertiary prevention in people with GBA or idiopathic PD in a phase 3 RCT (NCT05778617). Given its long, favorable safety record 60 ambroxol may also be well suited for a secondary prevention study in GBA+at-risk individuals with a positive αSyn-SAA even before any clinical features arise, as well as in those with RBD and/or hyposmia (i.e., a non-motor prodromal population).
Alternatively, PD gene-directed repurposed agents may act against neurotoxicity mediated by αSyn (encoded by SNCA), the core presumptive pathogenic pathway of PD. Albuterol, a beta-adrenergic receptor agonist commonly and chronically used for reactive airway disorders, has been found to reduce expression of αSyn, to confer neuroprotection in preclinical studies, 61 and to be linked to reduced risk of PD in epidemiological studies, though not in all of them (and with residual controversy over role of prominent candidate confounding factors).61,62, 61,62 Similarly, preclinical evidence 63 has suggested that the potential neuroprotective effects of caffeine and other more specific adenosine A2A receptor antagonists in PD may reflect attenuated αSyn neurotoxicity, further supporting its consideration as a low-risk candidate preventative in early (e.g., αSyn-SAA+ preclinical, or non-motor) prodromal PD. With additional encouraging results of GLP-1 agonism as a repurposable anti-diabetic drug class in a recent phase 2 tertiary prevention trial of lixisenatide for PD, 64 this approach may also be worthwhile pursuing for secondary prevention depending in part on anticipated upcoming results of ongoing phase 3 testing with exenatidein PD. 65
b)
In an effort to facilitate this shift, the Michael J Fox Foundation’s PPMI, which has increasingly focused on characterizing biomarkers as well as clinical features of PD’s prodromal period, has developed the P2P platform trial initiative.27,42, 27,42 P2P is inviting commercial therapeutic companies to conduct secondary prevention trials of their proprietary candidate therapeutics, and has announced its expectation that by early 2025 P2P participants with non-motor prodromal PD (with or without early motor features) will be randomized to receive the first of these as yet undisclosed treatments or placebo.
The effort to expedite industry pursuit of secondary prevention as spearheaded by PPMI/P2P will generally encourage the testing of new chemical entities (novel small molecules targeting pathogenic αSyn, GBA, LRRK2, inflammatory, etc. pathways) or biologics (e.g., αSyn-targeted antibodies, immunomodulating decoy receptors, mRNA vaccines, neurotrophic factors, etc.). The limited safety experience with these moderate-to-high risk agents warrants particular caution when administering them to a healthy population even if these participants are at substantial risk of developing a neurodegenerative synucleinopathy. (See Fig. 2, row 2b.) Thus such compounds at present are better suited to testing in the clinical phases of prodromal PD or NSD.
c)
3)
How to quantify prevention remains a major if not the greatest challenge in the design of PD prevention trials. While diagnosis of PD (or related neurodegenerative synucleinopathy) seems simplest, relying on phenoconversion can be limited practically (given the long follow-up periods required) and conceptually (given shifting definitions of PD). Preceding phenoconversion are a series of potentially informative, even if not yet definitive, biomarker and clinical metrics of PD progression (Fig. 2, rows 3a-d). Most of these are actively under investigation with their selection in early secondary prevention trials highly dependent on the prodromal stage being targeted.
a)
b)
c)
d)
e)
Nevertheless, secondary prevention RCTs adopting phenoconversion as their primary outcome have recently been planned (e.g., with 732 RBD participants to be followed over 3 years in the SAHZJU_RBD study, registered but apparently delayed; NCT05611372) 74 or initiated (e.g., with 142 participants with RBD to be followed over 5 years in the EITRS study, which is actively recruiting per NCT04534023) 7 (See Figure 2, row 3e.)
CONCLUSION
The time has come to begin conducting rigorous secondary prevention trials in prodromal PD, while pursuing improved prevention trial designs. Advances in our understanding of the prodromal period of PD along with the establishment of at-risk cohorts and the recent validation of a synucleinopathy biomarker have led the way to the design and implementation of the first trials for the (secondary) prevention of PD.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgements to report.
FUNDING
The Michael J. Fox Foundation for Parkinson’s Research Award MJFF-022854 (MAS).
This publication was supported by the Food and Drug Administration (FDA) of the U.S. Department of Health and Human Services (HHS) as part of a financial assistance award, R13FD008015 totaling $60,000 with 100 percent funded by FDA. The contents are those of the author(s) and do not necessarily represent the official views of, nor an endorsement, by FDA/HHS, or the U.S. Government.
American Academy of Neurology Clinical Research Training Scholarship (GFC).
CONFLICT OF INTEREST
The authors have no conflicts of interest to report.
