Abstract
Background:
Cognitive impairment is a clinical feature of multiple system atrophy (MSA). However, the prevalence and factors influencing the prevalence of cognitive impairment and dementia in MSA patients remain unclear.
Objective:
We aim to provide an estimate of the prevalence of cognitive impairment and dementia in patients with MSA and to evaluate the possible effect of demographic, clinical and methodological factors on the prevalence.
Methods:
We systematically searched the PubMed, Embase, and Web of science databases to identify studies that report the prevalence of cognitive impairment or dementia in MSA published up to February 2022. We computed the estimates of the pooled prevalence using random-effects models. Heterogeneity was investigated by subgroup analyses and meta-regression. Differences between MSA patients with and without cognitive impairment in demographic and clinical features were explored.
Results:
A total of 23 studies comprising 2064 MSA patients were included in meta-analysis. The pooled prevalence of cognitive impairment in MSA patients was 37% (95% CI: 29% –45%), the prevalence of dementia was 11% (95% CI: 7% –15%). The subgroup analyses showed the prevalence of dementia in pathologically-confirmed MSA was 7% (95% CI: 0% –12%), in clinically diagnosed MSA was 14% (95% CI: 10% –18%). Cognitive impairment in MSA patients was associated with older age, lower education, longer disease duration and more severe motor symptoms.
Conclusion:
Cognitive impairment is a common non-motor symptom in MSA. Dementia can develop in a few patients with MSA as well, but usually in the late stage.
INTRODUCTION
Cognitive impairment is a non-motor symptom in multiple system atrophy (MSA) [1–3], which mainly affects executive function and verbal fluency [4], greatly diminishes patient’s quality of life. It is believed that cognitive impairment is uncommon in MSA or is a symptom of mid to advanced stage of MSA, unlike in Parkinson’s disease (PD) which has been reported in the prodromal stage [5]. Dementia was a non-supporting feature of MSA according to the consensus diagnostic criteria in 1998 and 2008 [6, 7]. However, some studies have shown that up to 17% of neuropathologically confirmed MSA patients would develop dementia, and their pathological features were frontal lobe atrophy and white matter lesions [8]. There is still substantial uncertainty regarding the prevalence of dementia and cognitive impairment in MSA.
The prevalence of dementia and cognitive impairment in MSA varied greatly across studies, with dementia ranging from 2% to 26% [9, 10], cognitive impairment ranging from 17% to 62% [3, 11]. The large discrepancy among the studies may be partly explained by several methodological factors, such as different phenotypes of MSA, cognitive assessment scales and sample size. Demographic characteristics like age, gender, and years of education may also be associated with the heterogeneity of prevalence of cognitive impairment in MSA, but this association has not always reached statistically significant levels [12]. Besides, the prevalence of dementia in pathologically confirmed MSA was always lower than in clinically diagnosed MSA, which still need further investigation. Thus, an adequate investigation of the prevalence and clinical characteristics of cognitive impairment and dementia in the MSA patients is required.
The first objective of the present study was to conduct a systematic review and meta-analysis of the prevalence of cognitive impairment and dementia in MSA patients, as well as to explore heterogeneity across prevalence estimates. The second objective was to evaluate the possible influence of demographic characteristics, clinical characteristics, and methodological factors on the prevalence rate.
METHODS
This system review and meta-analysis was conducted in adherence with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines [13, 14]. The protocol for this study was registered in advance on PROSPERO (registration number: CRD42022309511).
Search strategy and selection criteria
Literature searches for eligible studies were performed up to February 2022 in PubMed, Web of Science and Embase using the following MeSH (Medical Subject Heading) terms and keyword variations: (“multiple system atrophy” OR “MSA” OR “olivopontocerebellar atrophy” OR “OPCA” OR “striatonigral degeneration” OR “Shy-Drager syndrome” OR “MSA”) AND (“cognitive impairment” OR “MCI” OR “cognitive deficit” OR “cognitive dysfunction” OR “cognitive decline” OR “Mild Cognitive Impairment” OR “cognitive” OR “cognition” OR “memory” OR “frontal-executive” OR “dementia”). Afterwards, the reference lists of the included articles were searched for additional eligible publications.
We included the studies meeting the following criteria: (1) participants were diagnosed with MSA pathologically or clinically based on the consensus criteria published in 1999 or 2008; (2) prevalence of cognitive impairment or dementia was reported; (3) cognitive impairment or dementia was assessed by specific validated scales or the standard battery of neuropsychological tests; (4) samples were independent; (5) the types of studies are cohort studies, cross-sectional studies and case-control studies that reported the prevalence of cognitive impairment or dementia in MSA; (6) was published in peer-reviewed journals in English. The studies were excluded if they: (1) were review articles, case reports, editorial letters or conference proceedings without full report publication; (2) included parkinsonian syndromes without formal MSA diagnoses; (3) defined cognitive impairment or dementia according to a non-objective or non-standardized manner; (4) small sample size (n < 20). In case of studies based on the same sample had varying numbers of patients included, only the study with higher number of participants was included. Two researchers (YSC, FFL) independently screened the abstracts and full articles for inclusion according to these criteria. When any discrepancies existed between the included articles, this was resolved in a consensus meeting and referred to a third researcher (TF) when required.
Data extraction
Two researchers (YSC, FFL) extracted the following information independently from each study: study characteristics (authors, publication year, country, type of research, major exclusion criteria of the total sample), demographic and clinical characteristic of included studies (sample size, gender, age, age at onset, disease duration, education, Unified Multiple System Atrophy Rating Scale-III (UMSARS-III), the prevalence of cognitive impairment or dementia in MSA patients, diagnostic criteria of MSA, and cognitive assessment scales). In cases of disagreement, two researchers (YSC, SSC) consulted a third researcher (TF) to get a unified result.
Quality assessment
Quality assessment was performed using the Newcastle–Ottawa-Scale (NOS), which was widely used to evaluate observational studies. The total scores of NOS ranged from 0 to 9 for cohort studies (low quality = 0–4 points; moderate quality = 5–7 points; high quality = 8-9 points), and the total scores ranged from 0 to 6 for cross-sectional studies (low quality = 0–2 points; moderate quality = 3-4 points; high quality = 5-6 points). The NOS was suitable for using in a systematic review [15]. Two reviewers (YSC, SSC) scored each study, discrepancies in scores were resolved by consensus with a third reviewer (TF).
Statistical analysis
All statistical analyses were performed using R Version 4.1.0. The primary result of our study was the prevalence of cognitive impairment and dementia in patients with MSA. The meta-analysis of prevalence was conducted using a random-effects model. The pooled prevalence was calculated with 95% confidence interval (CI). As the secondary result, we explored the differences between MSA patients with and without cognitive impairment in demographic and clinical features. For continuous variables, we computed standardized mean differences (SMDs) with 95% CIs using Hedges’ correction. We defined a positive SMD as indicating MSA patients with cognitive impairment had higher scores than patients without cognitive impairment. For dichotomous outcomes, we employed a fixed-effect model using the Mantel-Haenszel method to estimate the summary risk if the heterogeneity was low to moderate (I2 <50%). Otherwise, we applied a random-effects model using the DerSimonian-Laird method. Heterogeneity between studies was assessed using the I2 statistic and Q test. A P value of less than 0.05 was taken to indicate statistical significance. I2 statistic was used to categorise the magnitude of heterogeneity as follows: 0% to 24% : low heterogeneity; 25% to 49% : moderate heterogeneity; 50% to 74% : substantial heterogeneity; and 75% to 100% : considerable heterogeneity. To investigate the possible reasons for heterogeneity in the prevalence of cognitive impairment, we explored heterogeneity across a series of predefined subgroups of interest: different diagnostic criteria for MSA and different cognitive assessment scales. Subgroup differences were assessed using the χ2 test. Meta-regression models (including age, different diagnostic criteria for MSA, and disease duration) were also fitted to further explore heterogeneity across study findings. We performed sensitivity analyses to evaluate the robustness of the results by computing how the overall effect size would change by removing 1 study at a time. Possible publication bias was investigated via funnel plot, roughly symmetrical funnel plots indicating low risk. Egger’s regression method was used to tests asymmetry of the funnel plot, a p < 0.05 was considered statistically significant. Then we performed a trim and fill method to address the problem of publication bias.
RESULTS
Results of the search
A total of 5,735 articles were collated using our search strategy (536 articles in PubMed, 2,285 articles in Embase, and 2,932 articles in Web of science). After removing 935 duplicated articles, 4,818 articles were remained. Then, 1,367 articles were excluded because they were case reports, books, review articles, or conference abstracts. Based on titles and abstracts, we further excluded 3,354 articles as they did not include MSA patients with cognitive impairment or dementia. Last, full-length original articles of the remaining 106 studies were assessed, we excluded 4 studies that did not use the consensus criteria published in 1999 or 2008, 78 studies did not report the prevalence of cognitive impairment or dementia in MSA, and 1 study had duplicate participants with others. The remaining 23 articles with a total of 2,064 participants showed a good methodological quality (cross-sectional studies scores varied from 5 to 6 points, whereas cohort studies ranged from 7 to 8 points) were included in the meta-analysis (Supplementary Table 1). The details for selection articles after full-text screening are shown in the flow chart (Fig. 1).
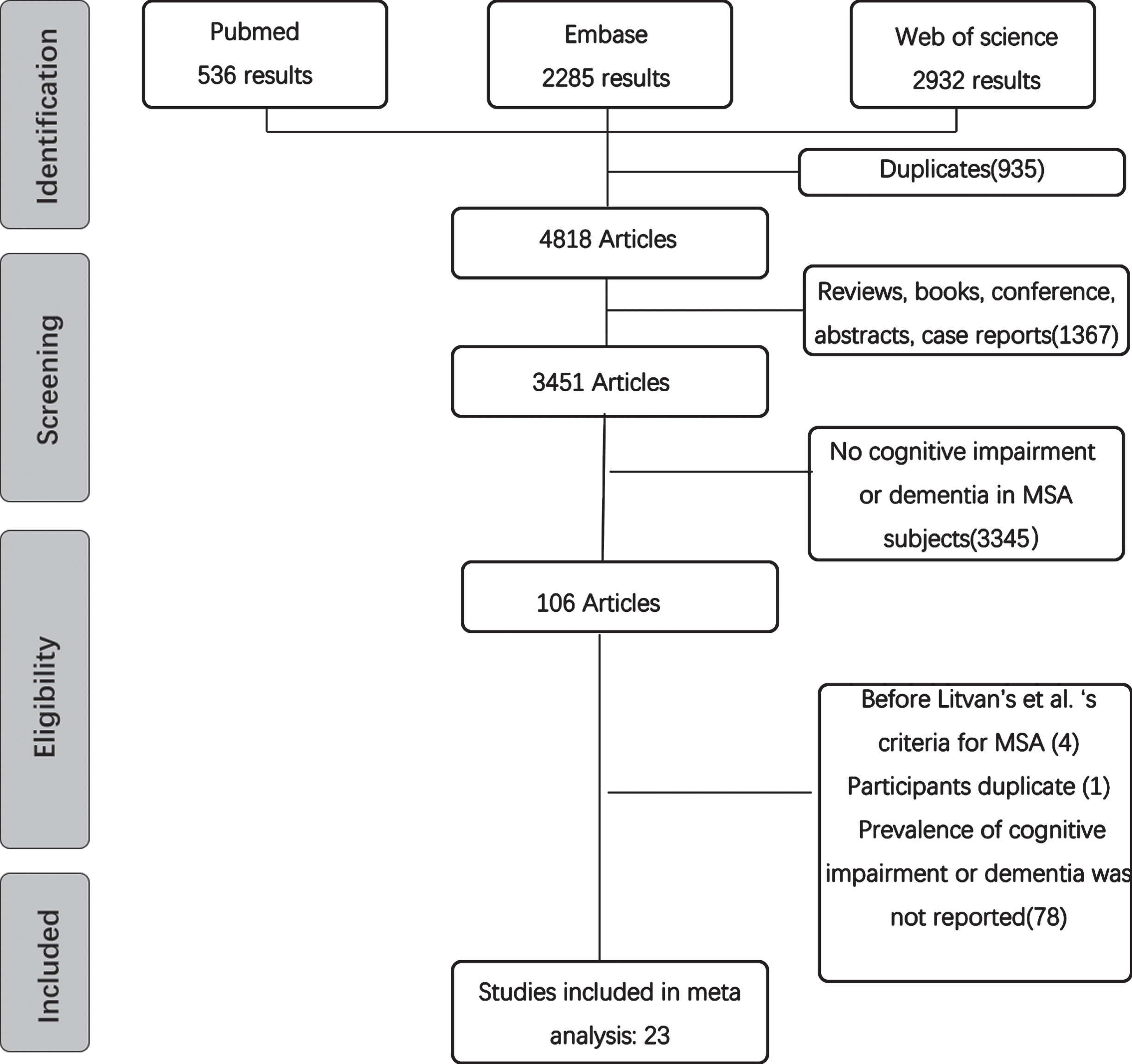
Study selection flow chart, performed according to the PRISMA 2020 guidelines.
Characteristics of included studies
A total of 23 studies were included in our meta-analysis (Table 1). Of these studies, 8 reported the prevalence of cognitive impairment, 6 reported the prevalence of dementia, 4 reported the prevalence of both cognitive impairment and dementia, and 5 reported the prevalence of cognitive impairment which excluding patients with dementia. Fifteen studies diagnosed cognitive impairment by using neuropsychological scales for assessing global cognitive abilities: 8 used Mini-Mental State Examination (MMSE) [2, 16–19], 3 used Addenbrooke’s cognitive examination-revised (ACE-R) [1, 21], 3 used Montreal cognitive assessment (MoCA) [22–24], and 1 used Mattis dementia rating scale (DRS) [25]. Two studies used the standard battery of neuropsychological tests [3, 26] followed the MDS clinical criteria for the diagnosis of mild cognitive impairment in PD (PD-MCI) [27]. For the diagnosis of dementia, 3 used diagnostic and statistical manual of mental disorders, fourth edition (DSM-IV) [8, 29], 4 used MMSE [9, 30], and 1 used both MMSE and ACE-R [18]. Two [3, 31] studies diagnosed dementia in MSA referring to the standard diagnostic criteria of PD dementia (PDD) [32].
Study characteristics
QS, quality score; M, mean; SD, standard deviation; N, total number of participants; y, years; m, months; nr, not reported; D, dementia; OND, other neurological disorder; SMD, severe motor dysfunction; PSY, psychiatric disorders; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; DRS, Mattis dementia rating scale; ACE-R, Addenbrooke’s cognitive examination-revised; DSM- IV, Diagnostic and statistical manual of mental disorders, fourth edition; PDD, MDS standard diagnostic criteria of PD dementia; PD-MCI, MDS clinical criteria for the diagnosis of mild cognitive impairment in PD.
About the diagnostic criteria for MSA, 16 studies diagnosed MSA according to the consensus criteria published in 2008. Of these studies, 7 studies included participants who met criteria for possible MSA [2, 25], and the other 9 studies only included participants who met criteria for probable MSA [1, 33]. Six studies identified patients with a pathological diagnosis of MSA [10, 30]. Two studies included clinically diagnosed MSA but did not report the diagnostic criteria [9, 31].
Prevalence of dementia in MSA
A total of 10 studies explored the prevalence of dementia in patients with MSA, with an overall random-effects pooled prevalence of 11% (95% CI: 7% –15%), the heterogeneity was considerable (I2 = 76%) (Fig. 2A). Significant publication bias was found (p < 0.05, Supplementary Figure 1). Trim-and-fill analysis trimmed 5 studies revealing an estimated prevalence of 5.85% (95% CI = 0.61% –11.08%, I2 = 81.1%)
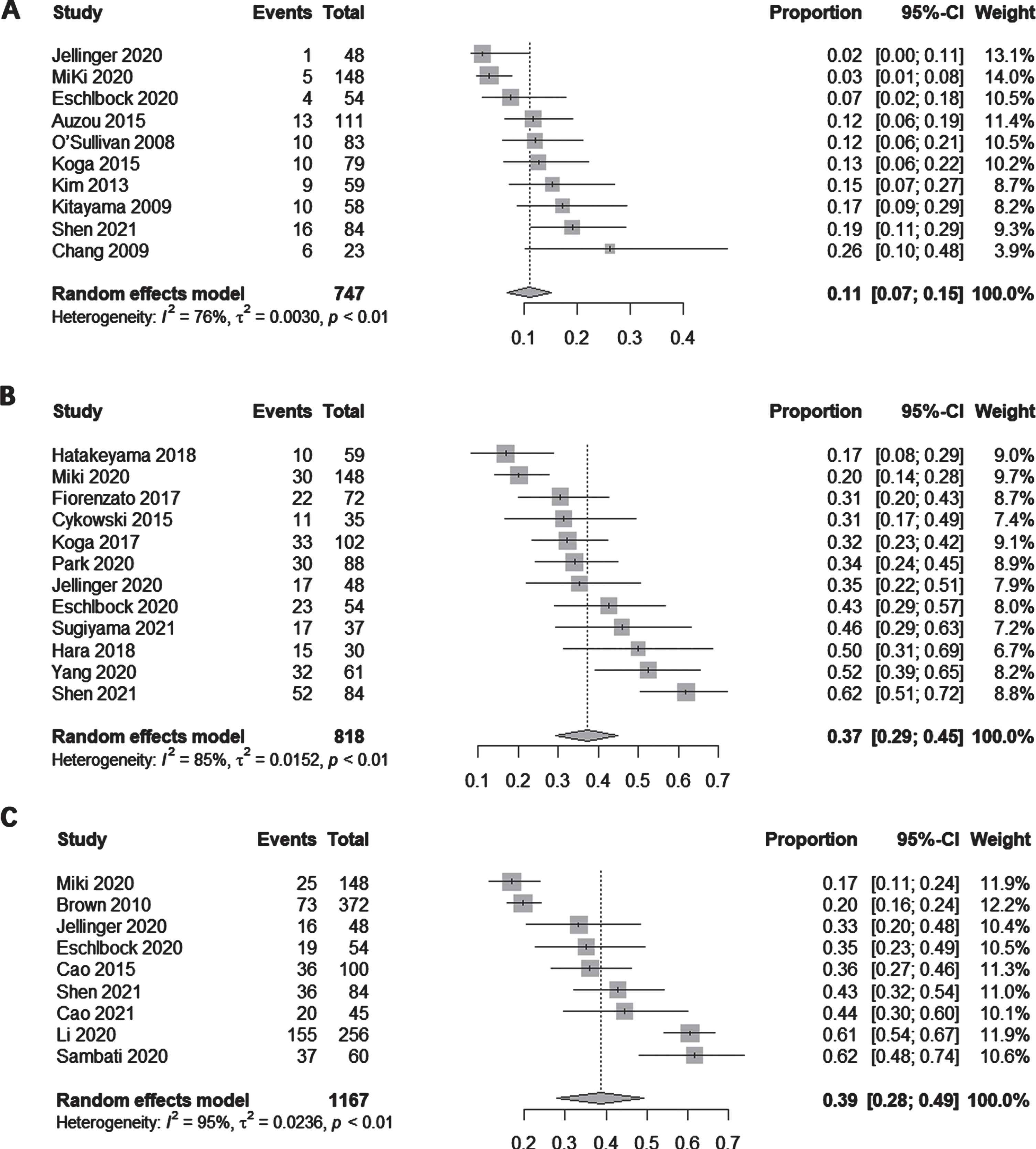
A) Forest plot for overall point prevalence of dementia in patients with MSA. B) Forest plot for overall point prevalence of cognitive impairment in patients with MSA. C) Forest plot for overall point prevalence of cognitive impairment excluding dementia in patients with MSA.
Results of the subgroup analysis suggested the prevalence of dementia in clinically diagnosed MSA was 14% (95% CI: 10% –18%, I2 = 34%). The prevalence of dementia was 7% (95% CI: 0% –12%, I2 = 73%) in the group of pathologically diagnosed MSA (Supplementary Figure 2). Statistically significant subgroup differences were detected for diagnostic criteria of MSA (p < 0.05).
Based on different cognitive assessment scales, the subgroup analysis showed the prevalence of dementia was 9% (95% CI: 0% –17%, I2 = 74%) in the group diagnosed dementia using MMSE, 14% (95% CI: 9% –19%, I2 = 0%) in the group diagnosed dementia according to DSM-4, 15% (95% CI: 0% –22%, I2 = 49%) in the group diagnosed dementia referring to the standard diagnostic criteria of PDD (Supplementary Figure 3). There were no statistically significant subgroup differences detected.
Prevalence of cognitive impairment in MSA
Twelve studies explored the prevalence of cognitive impairment in patients with MSA, yielding an overall random-effects pooled prevalence of 37% (95% CI: 29% –45%) with a high level of heterogeneity (I2 = 85%) (Fig. 2B). Significant publication bias was not found (p≥0.05) (Supplementary Figure 4).
The subgroup analysis showed that the prevalence of cognitive impairment was 34% (95% CI: 0% –44%, I2 = 75%) in the group diagnosed with probable MSA using consensus criteria published in 2008, 55% (95% CI: 0% –100%, I2 = 34%) in the group diagnosed with possible MSA, and 29% (95% CI: 0% –36%, I2 = 58%) in the group of pathologically diagnosed MSA (Supplementary Figure 5). Statistically significant subgroup differences were detected for diagnostic criteria of MSA (p < 0.05).
Moreover, the prevalence of cognitive impairment diagnosed using MMSE in patients with MSA was 32% (95% CI: 0.25–0.40, I2 = 79%). In the group diagnosed cognitive impairment using ACE-R the prevalence of cognitive impairment was 34% (95% CI: 0.26–0.42, I2 = 0%) (Supplementary Figure 6). Statistically significant subgroup differences were detected for different cognitive assessment scales of MSA (p < 0.05). The prevalence of cognitive impairment in MSA-cerebellar (MSA-C) was 42% (95% CI: 28% –56%, I2 = 87%), in MSA-parkinsonism (MSA-P) was 41% (95% CI: 31% –52%, I2 = 61%). Results of the subgroup analyses were summarized in Table 2.
Subgroup analysis of the prevalence of cognitive impairment and dementia in MSA patients
K, number of studies; I2, heterogeneity statistic within level; MMSE, Mini-Mental State Examination; MoCA, Montreal Cognitive Assessment; ACE-R, Addenbrooke’s cognitive examination-revised; DSM- IV, Diagnostic and statistical manual of mental disorders, fourth edition.
Nine studies reported a prevalence of cognitive impairment excluding dementia in patients with MSA, resulting in an overall random-effects pooled prevalence of 39% (95% CI: 28% –49%), the heterogeneity was considerable (I2 = 96%) (Fig. 2C). Significant publication bias was not found (p≥0.05) (Supplementary Figure 7).
Meta-regression
There was an apparent effect of diagnostic criteria of MSA (possible versus probable versus pathologically confirmed MSA, p = 0.036) and disease duration (p = 0.04) on the prevalence of cognitive impairment. There was little evidence of an effect of age (p = 0.18) on the prevalence of cognitive impairment (Supplementary Table 2).
Sensitive analysis
We performed a sensitivity analysis by excluding 1 study at a time to investigate the impact of these studies on the aggregate results. The sensitivity analysis found that the pooled prevalence of dementia (Supplementary Figure 8), cognitive impairment (Supplementary Figure 9) and cognitive impairment excluding dementia (Supplementary Figure 10) in MSA did not change substantially, indicating results of our meta-analysis was robust.
Association with demographic characteristics of MSA
Twelve studies enrolled 1,204 participants compared age of MSA patients with cognitive impairment versus MSA patients without cognitive impairment. The pooled effect size was 0.17 (95% CI: 0.05–0.30, I2 = 48.5%), indicating MSA patients with cognitive impairment were older than MSA patients without cognitive impairment (Supplementary Figure 11). Egger’s test showed there were no publication bias (p = 0.29), and sensitivity analysis showed the results was robust. Eight studies including 1,078 participants compared education years of MSA patients with and without cognitive impairment. The pooled effect size was –0.55 (95% CI: –0.83––0.44, I2 = 69.1%), indicating MSA patients with cognitive impairment had a lower level of education than MSA patients without cognitive impairment (Supplementary Figure 12). Egger’s test showed there were no publication bias (p = 0.44), and sensitivity analysis showed the results was robust. Gender was not associated with the risk of cognitive impairment in patients with MSA (0.85, 95% CI: 0.68–1.07, I2 = 29.5%) (Supplementary Figure 13), but sensitivity analysis suggested the results was not stable (Table 3).
Summary of meta-analytic results of demographic and clinical characteristics of MSA
K, number of studies; I2, heterogeneity statistic within level; UMSARS, Unified Multiple System Atrophy Rating Scale; H&Y, Hoehn-Yahr stage; OH, Orthostatic hypotension; MSA-CI, MSA with cognitive impairment; MSA-NCI, MSA without cognitive impairment.
Association with clinical features of MSA
Ten studies (N = 1,250) compared disease duration of MSA patients with and without cognitive impairment. The pooled effect size was 0.19 (95% CI: 0.06–0.31, I2 = 27.5%), indicating MSA patients with cognitive impairment had longer disease duration than MSA patients without cognitive impairment (Supplementary Figure 14). The heterogeneity across studies was low, with Egger’s test showed there were no publication bias (p = 0.22). A total of 3 studies compared UMSARS1 scores of MSA patients with or without cognitive impairment. The pooled effect size was 0.59 (95% CI: 0.37–0.82, I2 = 18.7%), indicating MSA patients with cognitive impairment had more severe motor symptomatology than MSA patients without cognitive impairment (Supplementary Figure 15), and there was no publication bias. Four studies compared H&Y stages of MSA patients with or without cognitive impairment. The pooled effect size was 0.42 (95% CI: 0.02–0.82, I2 = 78.6%), indicating MSA patients with cognitive impairment had higher scores on the Hoehn-Yahr stage (H&Y) (Supplementary Figure 16). The funnel plot was asymmetric and sensitive analysis suggested the results was not stable. The frequency of orthostatic hypotension (OH) was not associated with an increase in cognitive impairment risk (OR = 1.30, 95% CI: 0.75–2.23, I2 = 47.6%; Table 3).
DISCUSSION
This meta-analysis revealed that the overall pooled prevalence of cognitive impairment in MSA was 37%, the prevalence of dementia was 11%. Subgroup analysis revealed that the prevalence of dementia was 7% in pathologically confirmed MSA, 14% in clinically diagnosed MSA. Different cognitive assessment scales contributed to the inconsistence in the prevalence of cognitive impairment as well, the prevalence of cognitive impairment measured by MMSE was lower than others. By comparing MSA patients with and without cognitive impairment, our meta-analysis highlighted the predominant patient-related characteristics associated with cognitive impairment including older age, lower education, longer disease duration and more severe motor symptoms.
Our meta-analysis showed that about 10 percent of MSA patients would develop dementia, and 7% of patients with pathologically diagnosed MSA had dementia as well. In studies using DSM-IV to diagnosis dementia, the prevalence of dementia in clinical diagnostic MSA was 14%, with no heterogeneity. Dementia was considered non-supporting feature by consensus diagnostic criteria in 1998 and 2008 [6, 7]. However, in the latest diagnostic criteria of MSA in 2022 [34], it was revised to state that dementia within 3 years of disease onset was an exclusion feature for MSA diagnosis. In line with this, the average disease duration of MSA patients with dementia ranged from 3.28 to 8.4 years in our meta-analysis, and a longitudinal study suggested time interval from MSA diagnosis to clinically dementia was estimated to be 6.4 years [29]. Thus, the diagnosis of MSA cannot be clinically excluded if the dementia develops 3 or more years after disease onset. Besides, our subgroup analysis found pathologically confirmed MSA resulted in a lower prevalence of dementia than possible or probable MSA. MSA with dementia can be difficult to differentiate from dementia with Lewy bodies (DLB), especially when DLB patients present with some atypical symptoms such as autonomic failure. Twenty-three autopsy-confirmed MSA patients (13%) were not clinically diagnosed with MSA, but instead with progressive supranuclear palsy, PD, PDD, or DLB [35], supported the results of our subgroup analysis. This indicated that dementia could also be present in patients with MSA, which was always overlooked. Thus, for levodopa-unresponsive parkinsonism with autonomic failure and dementia, the diagnosis of MSA combined with dementia should also be considered in addition to DLB.
Postmortem findings further point towards cognitive decline as a characteristic feature in some MSA patients. For example, MSA patients with cognitive impairment had a greater burden of neuronal cytoplasmic inclusions in the dentate gyrus, neocortex, and medial temporal region [17]. Neuronal cytoplasmic inclusions burden in the hippocampus and para-hippocampus was proved correlated with memory impairment in MSA [18]. Furthermore, concomitant Alzheimer disease neuropathological changes, such as cortical tau pathology, also had impact on cognitive changes in MSA [10].
In our meta-analysis, cognitive impairment occurs in about a third of patients with MSA, and the shortest mean disease duration of MSA patients with cognitive impairment was 1.88 years. According to a recently published study, cognitive impairment was a frequent and prominent early finding within the 2 years before diagnosis of MSA [36]. Thus, cognitive impairment may not only be a presentation in early stage of clinical diagnostic MSA but also in prodromal MSA. Moreover, our meta-regression analysis showed that disease duration had a statistically significant effect on the prevalence of cognitive impairment in MSA. A large prospective study demonstrates in patients with MSA surviving more than 8 years, almost 50% are reported to be cognitively impaired [29], suggesting that the prevalence of cognitive impairment in MSA would be higher if the disease duration was longer.
The subgroup analysis revealed that the prevalence of cognitive impairment in possible MSA was higher than probable and definite MSA. Cognitive impairment and autonomic failure could be both present from the earliest phase of all α-synucleinopathies, coexistence of which was a pitfall in the differential diagnosis of α-synucleinopathies [37]. DLB and PD were more likely be misdiagnosed as possible MSA [38], which might lead to a higher prevalence rate of cognitive impairment in possible MSA.
For cognitive impairment excluding dementia, the pooled prevalence was 39%, abnormally higher than cognitive impairment including dementia. However, when using ACE-R, the prevalence of cognitive impairment excluding dementia was 36%, lower than cognitive impairment including dementia. Thus, unified cognitive assessment tool with high sensitivity and specificity were important to characterize the cognitive function in MSA.
In our meta-analysis, the prevalence of cognitive impairment measured by MMSE was lower than others. A meta-analysis compared diagnostic accuracy of different cognitive assessment scales found ACE-R, Consortium to Establish a Registry for Alzheimer’s Disease Battery total score (CERAD), and MoCA had similar diagnostic accuracy, while the MMSE had clearly inferior sensitivity [39]. Therefore, MMSE may not be suitable for screening cognitive impairment in MSA, whereas, the comprehensive assessment scales which evaluates memory, language, visuospatial functions, executive functions, and attention are recommended.
Our results suggested patients with MSA-P and MSA-C had the similar prevalence of cognitive impairment, in line with most previous studies [40]. However, regarding cognitive domains, differences between patients with MSA-P and MSA-C have been reported. Both MSA-P and MSA-C patients had impaired executive and visuospatial functions, but attention deficit was predominant only in MSA-C. It was also found patients with MSA-P had difficulties in immediate recall, patients with MSA-C had reduced long-term recall and recognition, which may be related with the function of cerebellum in encoding and maintenance of verbal information [41]. Neuroimaging studies revealed that cognitive dysfunction in MSA-P patients were significantly correlated with cerebral perfusion in the prefrontal cortex [41], while in the MSA-C patients the cognitive function was correlated with disruption of cerebellar-cortical circuits [40].
The comparisons between MSA patients with and without cognitive impairment showed that MSA patients with cognitive impairment was characterized by later H&Y staging, higher UMSARS scores, and longer disease duration, all of which reflect the severity of the symptoms. The relationship between motor function and cognitive impairment can be explained by a pathological change in MSA, such as widespread fronto-temporal subcortical GM, basal ganglia, and cerebellum atrophies in MSA patients; these changes may affect frontal functions such as executive function and attention [42]. Additionally, cognitive impairment in MSA patients also associated with older age and lower educational level. Higher education level had protective effects on developing dementia, which is accordance with the cognitive reserve (CR) hypothesis [43]. CR refers to the ability to tolerate the disease related pathology in the brain without developing clinical symptom, it assumed some aspects of life experience such as education protects against the onset of dementia. Gender was not associated with the risk of cognitive impairment in patients with MSA in our meta-analysis, but the result was not robust in sensitive analysis. When article of Brown et al. was excluded, the result showed female was a risk factor for cognitive impairment in MSA. Previous studies also revealed a worse global cognitive state in female patients with MSA as compared to male. And at follow up, female patients deteriorated severely than men over time in attention [12]. Therefore, further longitudinal studies are needed to explore the relationship between gender and cognition in MSA patients.
There were some limitations of the current study. First, the meta-analysis was limited by a relatively high level of heterogeneity in demographic characteristics, diagnostic methods, and measurement approaches. Thus, we included studies using validated cognitive assessment scales or the standard battery of neuropsychological tests to get a more reliable prevalence data. The subgroup analyses based on different diagnostic criteria and cognitive assessment scales were also performed to interpret heterogeneity. Second, only 4 studies reported the prevalence rate of cognitive impairment in pathological confirmed MSA patients, which indicated the scarcity of research on this area. Third, the timing of the onset of dementia in patients with MSA is unclear. Although one longitudinal study suggested time interval from MSA diagnosis to clinically dementia was estimated to be 6.4 years, the sample size of which was small [29]. More longitudinal studies are still required in the future to further explore the timing of the onset of dementia in MSA. Fourth, the most studies included were cross-section studies, which was not suitable for the derivation of causal relationships. Further longitudinal studies are needed to explore the risk factors for cognitive impairment in MSA patients. Last, the effect of factors including depression, anxiety and rapid eye movement sleep behavior disorder on cognitive impairment was not evaluated because limited studies in these aspects, and they should be further investigated in future studies.
Conclusion
Our meta-analysis revealed that cognitive impairment was not uncommon in MSA. Dementia could be developed in patients with MSA as well, but usually in the late stage. Thus, the diagnosis of MSA could not be excluded if the dementia developed 3 or more years after disease onset. Older age, lower education, longer disease duration, and more severe motor symptoms all could be risk factors for cognitive impairment in MSA. As different cognitive assessment scales contributed to the heterogeneity among primary studies, it was necessary to standardize and optimize the assessment of cognition in MSA patients in the future study.
Footnotes
ACKNOWLEDGMENTS
This work was supported by the National Natural Science Foundation of China (82071422) and Beijing Municipal Natural Science Foundation (7212031).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
