Abstract
Background:
Orthostatic hypotension (OH) in Parkinson’s disease (PD) is frequent and associated with impairments in quality of life and reduced activities of daily living. Abdominal binders (AB) and compression stockings (CS) have been shown to be effective non-pharmacological treatment options.
Objective:
Here, we investigate the effect of AB versus CS on physical activity using a digital mobility outcome (sit to stand [STS] frequency) collected in the usual environment as a primary endpoint.
Methods:
We enrolled 16 PD patients with at least moderate symptomatic OH. In a randomized, single-blinded, controlled, crossover design, participants were assessed without OH treatment over 1 week (baseline), then were given AB or CS for 1 week and subsequently switched to the other treatment arm. The primary outcome was the number of real-life STS movements per hour as assessed with a lower back sensor. Secondary outcomes included real-life STS duration, mean/systolic/diastolic blood pressure drop (BPD), orthostatic hypotension questionnaire (OHQ), PD quality of life (PDQ-39), autonomic symptoms (SCOPA-AUT), non-motor symptoms (NMSS), MDS-UPDRS, and activities of daily living (ADL/iADL).
Results:
Real-life STS frequency on CS was 4.4±4.1 per hour compared with 3.6±2.2 on AB and 3.6±1.8 without treatment (p = 1.0). Concerning the secondary outcomes, NMSS showed significant improvement with CS and AB. OHQ and SCOPA-AUT improved significantly with AB but not CS, and mean BPD drop worsened with CS but not AB. Mean STS duration, PDQ-39, MDS-UPDRS, ADL, and iADL did not significantly change.
Conclusion:
Both AB and CS therapies do not lead to a significant change of physical activity in PD patients with at least moderate symptomatic OH. Secondary results speak for an effect of both therapies concerning non-motor symptoms, with superiority of AB therapy over CS therapy.
Keywords
INTRODUCTION
The overall prevalence of orthostatic hypotension (OH) in patients with Parkinson’s disease (PD) is estimated to be between 30% and 52% [1, 2]. The pathophysiologic hallmarks comprise neurodegeneration with Lewy body pathology in sympathetic ganglia, hypothalamus, Locus coeruleus, rostral mesencephalon, and postganglionic sympathetic cardiac denervation leading to reduced vasoconstriction and cardiac sympathetic stimulation [3]. OH is defined as a sustained reduction of systolic blood pressure of at least 20 mm Hg or diastolic blood pressure of 10 mm Hg within 3 min of standing or head-up tilt to at least 60° on a tilt table [4], leading to venous abdominal and femoral accumulation of intravasal blood and, consequently, to transient brain hypoperfusion [5]. Based on literature, it is reasonable to assume that OH not only leads to a change in mobility behavior in general, but also to a change in sit-to-stand (STS) behavior. First, on a performance level, OH is associated with decreased mobility, more frequent falls, and decreased activities of daily living in PD patients OH [1, 7]. Moreover, the extend of the blood pressure drops (BPD) is negatively associated with the ambulatory capacity measure, which is a surrogate of mobility performance and associated with Five Times Sit-to-Stand, Timed Up-and-Go, and Berg Balance Scale performances, as well as with gait speed [8]. These results argue in favor of a correlation between (severity of) OH and number of STS movements in daily life. Second, in a geriatric cohort with OH, the extent of systolic BPD was negatively associated with chair stand test performance [9], suggesting a close correlation between OH and speed/dynamic aspects of the STS maneuver. It is thus tempting to speculate that OH leads to reduced STS performance also in patients with PD, and, consequently, to decreased STS frequency in the usual environment (although we cannot rule out with absolute certainty that the relationship between STS and OH could also be the reverse, i.e., that reduced STS exacerbates OH, e.g., through increased venous stasis). STS frequency has also been shown to correlate positively with the number of steps per day [10] and negatively with frailty scores [11], suggesting that STS frequency can serve as an (OH-associated) marker of physical activity. This assumption may be— at least indirectly— supported by a recent meta-analysis that found an association between OH and reduced physical functioning in 16 out of 42 studies [12]. Since OH has great impact on quality of life and FDA and EMA require data on OH as part of clinical trials, appropriate studies are urgently needed.
So far, most PD patients with OH are treated with lower limb compression stockings (CS) and abdominal binder (AB) therapy, as a supplement to pharmacological treatment or as the stand-alone therapy. Both treatments were shown to be effective in reducing BPD and associated symptoms, such as dizziness and presyncopes. However, it remains unclear whether one of the two forms of therapy is more effective [5, 13–17]. We investigated the effect of these two treatments using a high-quality cross-over study design and— to the best of our knowledge, the first time ever in this form of trial— a digital mobility outcome (STS frequency) collected in the usual environment as a primary endpoint.
MATERIALS AND METHODS
Participants and study design
The study was registered at the German clinical trial register (DRKS00021597) and approved by the local ethics committee (D 508/17). Between 2018 and 2019, 149 PD patients were screened from the specialized Movement Disorder Center of the Department of Neurology of the University hospital Schleswig-Holstein, Campus Kiel, Germany. The screening was performed during patient’s regular clinical visits to the movement disorder outpatient clinic or inpatient treatment.
Patients were included if they met the following inclusion criteria: Diagnosis of PD according to the UK Brain Bank Criteria [18] and Hoehn & Yahr stage 1–4, symptomatic OH (OHQ symptom assessment item 1≥1 [19]), and at least moderate OH (systolic/diastolic BPD of at least 20/10 mmHg within 3 min of standing or head-up tilt to at least 60° on a tilt table) [20]. Exclusion criteria were OH due to medication side effects (diuretics, alpha adrenergics (e.g., for bladder disorder), sympatholytics, vasadilatators, antidepressants, antihypertensives; as defined by anamnestic evidence that OH symptoms occurred or worsened with start of intake of medication, cognitive deficits (Non-motor symptom scale domain attention/memory>24/36 points [21–23]), other severe neurological, psychiatric, cardiac or vascular diseases, severe diabetes mellitus, severe peripheral neuropathy as determined by examination findings, pregnancy/breast feeding, and changes of the medical therapy in the 4 weeks prior to enrollment. Judgment of disease severity with respect to exclusion criteria 1–6 was made by an experienced movement disorder specialist (SP). Pharmacological antihypotensive treatment was not an exclusion criterion.
All participants gave written informed consent. After an observation period of one week (T1-T2: baseline assessment without any OH compression therapy), participants were randomized to wear CS or AB for one week (T2-T3: treatment phase 1). Participants then crossed over to the other treatment arm, respectively (T3-T4: treatment phase 2). Drug therapy was continued unchanged through the study. Investigators were single-blinded, which means that the assessment and therapy monitoring were carried out by two different persons and the examiner did not get a chance to see the type of OH compression therapy during the assessment.
Primary, secondary, and exploratory endpoints are described in detail below. Study details are provided in Fig. 1.
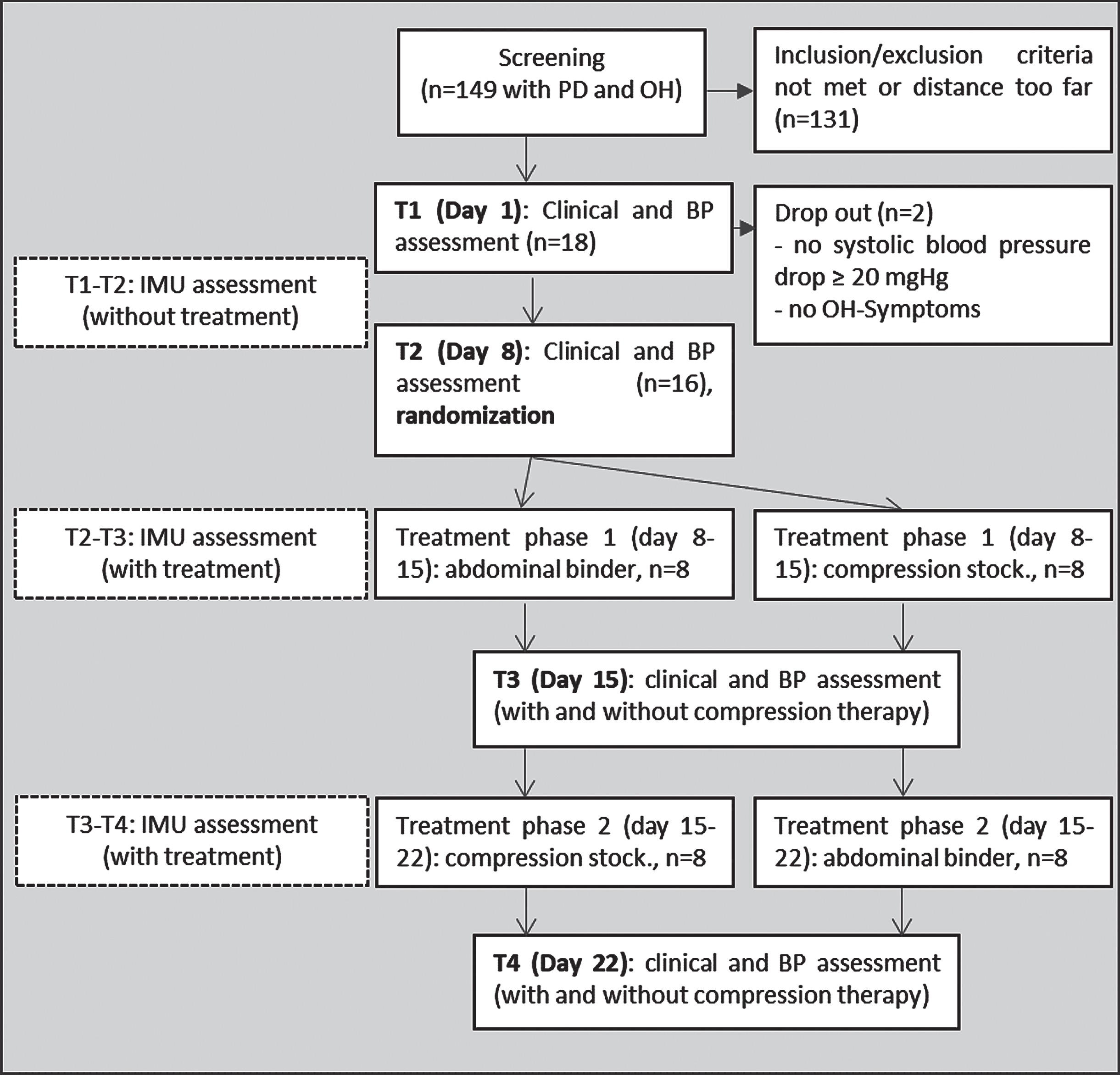
Study flow chart. Compression stock., compression stockings; IMU, inertial measurement unit; OH, orthostatic hypotension; PD, Parkinson’s disease.
CS were individually adjusted and reached to the thighs or to the hips, in some cases made-to-measure production was necessary. Compression class 3 was selected, building up a pressure of 34 to 46 mmHg [13, 17]. Some study participants needed a dressing aid. The three-stripes Abdo Syncro (Syncro-med GmbH, Linz, Austria) was used as AB, building up an abdominal wall pressure of 20±2 mmHg [15]. Compression therapy was applied from the morning after getting up and taken off in the evening at bedtime. This was monitored via a patient diary, in which participants noted at what times AB/CS was worn and which donning or wearing problems occurred.
Primary, secondary, and exploratory endpoints
Primary endpoint
The primary endpoint was real-life STS frequency, which was defined as the number of STS movements per hour during the respective observation period. This parameter was assessed 3 times (without OH treatment, during treatment phase 1 and during treatment phase 2) over an observation period of 7 days, respectively (Fig. 1).
For this purpose, an inertial measurement unit (IMU) with 3D acceleration and 3D gyroscope sensors worn at the lower back was used (RehaGait®, Hasomed, Magdeburg, Germany). Participants were instructed to put the sensor on when they arose in the morning and take them off again when they went to bed. Data were extracted and analyzed using a validated algorithm [24, 25]. The algorithm detects STS movements based on the vertical acceleration of the lower back in the global frame. The duration of the STS movements was estimated based on a fitted sigmoid model [24].
Secondary endpoints
The PDQ-39 summary index (0–100, higher scores indicate decreased health-related quality of life), the orthostatic hypotension questionnaire (OHQ [19]; 0–100, higher scores indicate impaired functioning), The Activities/instrumental Activities of Daily Living (ADL/iADL) questionnaire, the Movement Disorder Society revised version of the Unified Parkinson’s disease rating scale (MDS-UPDRS, parts I–IV), the non-motor symptoms scale in Parkinson’s disease (NMSS; 0–235; higher scores indicate reduced functioning) and the Scales for Outcomes in Parkinson’s Disease –Autonomic dysfunction (Scopa-AUT; 0–75, higher scores indicate abnormal autonomic symptoms), the mean real-life STS duration (in seconds; we hypothesized that OH is associated with reduced STS velocity, e.g. due to dizziness or cautious performance) and the BPD were used as secondary endpoints.
BPD, measured by tilt table examination (Aristo®, Wesseling BV, Zwanenburg, The Netherlands), is relevant for assessing the therapeutic effect of compression therapy for OH [5, 20]. Examinations were done at T1-T4 (see also Fig. 1). At T3 and T4, they were carried out first with wearing the respective compression device, and then at least 15 min after taking off the device. Thus, a total of 6 tilting table examinations were performed on each study participant. The examinations were performed at the same time of day for each participant (in order to minimize the influence of daytime variations), using the modified Italian tilting table protocol [13]: a 5-min supine rest phase was followed by a passive 60° tilt for 20 min BPD was calculated by subtracting (mean [MAP], systolic and diastolic) arterial pressure values at 5 min supine rest from 3 min tilt (Δ 3rd min tilt: mean/systolic/diastolic). The particular BPDs were designated as follows: Baseline value at time T1: BL 1, repeat baseline determination at time T2: BL 2, at time points T3 and T4: after one-week therapy with compression stockings (CS+) and after 15-min washout (CS-), respectively, and after one-week therapy with abdominal bandages (AB+) and after 15-min washout (AB-).
Exploratory endpoints
Real-life movement intensity (m/s2), real-life turning frequency (turns/h) and real-life turning duration (all assessed during the 7-day assessment periods using the IMU on the lower back) were included as exploratory endpoints. Real life movement intensity was used as a means to indirectly assess metabolic energy expenditure [26]. It has been shown to be closely correlated with physical activity as measured with a triaxial accelerometer at the low back in sport students [27]. Real life turning frequency and mean turning duration may also be surrogate marker for physical activity, and more sensitive to mobility disability than gait assessments alone [28, 29]: In a study of 160 older community-dwelling participants, reduced turning frequency and longer turning duration were associated with less time walking and were identified as prognostic factors for more frequent falls [29].
Further exploratory endpoints comprised the clinician/patient global impression severity scales (CGI-S, PGI-S; 1–8; higher scores indicate impaired functioning), number of syncopes/severe dizziness/falls (patient diary), Beck’s Depression Inventory-II (BDI-II; 0–63, higher scores indicate worse depressive symptoms), the Levodopa Equivalent Daily Dosage (LEDD) 4 weeks prior to enrolment and obesity (body mass index, kg/m2). An ECG was performed in all patients at study inclusion.
Statistical analysis
Normal distribution of data was controlled using Shapiro-Wilk’s test. Group comparisons were performed using repeated measures ANOVA and post hoc testing. p-values were adjusted by Bonferroni correction. Violations in the assumptions required for an ANOVA were controlled and if needed Greenhouse-Geisser sphericity correction was applied. Omega squared (ω2) effect size calculation was used to provide an unbiased effect size measure.
Statistical analysis was performed using JASP Team (version 0.14, University of Amsterdam, Netherlands). p values < 0.05 were considered statistically significant.
Data availability
Data not provided in the article because of space limitations may be shared (anonymized) at the request of any qualified investigator for purposes of replicating procedures and results.
RESULTS
Study population and baseline values
Eighteen PD patients with symptomatic OH were included into the study. After T1, two patients were excluded (one had a systolic BPD below 20 mmHg in the tilting table test and one reported no OH-associated symptoms, i.e., OHQ item 1 = 0 points). Of the remaining 16 participants, 14 were male. Mean disease duration was 10.3±5.8 years (±SD), mean MDS-UPDRS motor score (med. on) was 40±11 points). Mean LEDD was 713±486 mg/day and the medication was continued unchanged during the study (Table 1). Five patients had a history of syncopes, five took antihypertensive medication and four took antihypotensive medication. Study participants showed a moderate impairment of non-motor symptoms (OHQ: 31±15; Scopa-AUT: 18±7; NMSS: 62±30).
Patient characteristics at baseline (T1)
Patient characteristics at baseline (T1)
Data are given as mean (±SD). BMI, body mass index; LEDD, Levodopa equivalent dosage per day; NMSS, Non-Motor Symptom Scale; OHQ, Orthostatic Hypotension Questionnaire; Scopa-AUT, Scales for outcomes in Parkinson’s disease-autonomic dysfunction; UPDRS, MDS-Unified Parkinson Disease Rating scale.
Following 7 days of home monitoring, the 16 patients were re-assessed (T2). There were no significant changes in secondary and explorative outcomes compared to T1 (see Table 2; p values not shown). All 16 patients were randomized and successfully completed the crossover phase. For one participant, the analysis of sensor data for the “CS” condition failed due to technical problems. Two participants found the wearing of compression stockings annoying and constricting, and one reported a feeling of “fullness” after eating when wearing abdominal binder. Despite this impairment, compression therapy was not discontinued by any of the participants.
Secondary and exploratory outcomes
Data are presented as mean (±SD). *p < 0.05, **p < 0.01. anot normally distributed.
Real-life STS frequency was not significantly different between the two interventions (F(1.2, 16.2)=4.5, p = 0.6, ω2 = 0.0). Baseline assessment over 1 week (T1-T2) showed 3.6±1.8 STS/h (±SD). During AB therapy, participants performed 3.6±2.2 STS/h (p against baseline = 1.0), and during CS therapy 4.4±4.1 STS/h (p against baseline = 1.0; p against AB = 1.0. Details are provided in Fig. 2 and Supplementary Table 3).
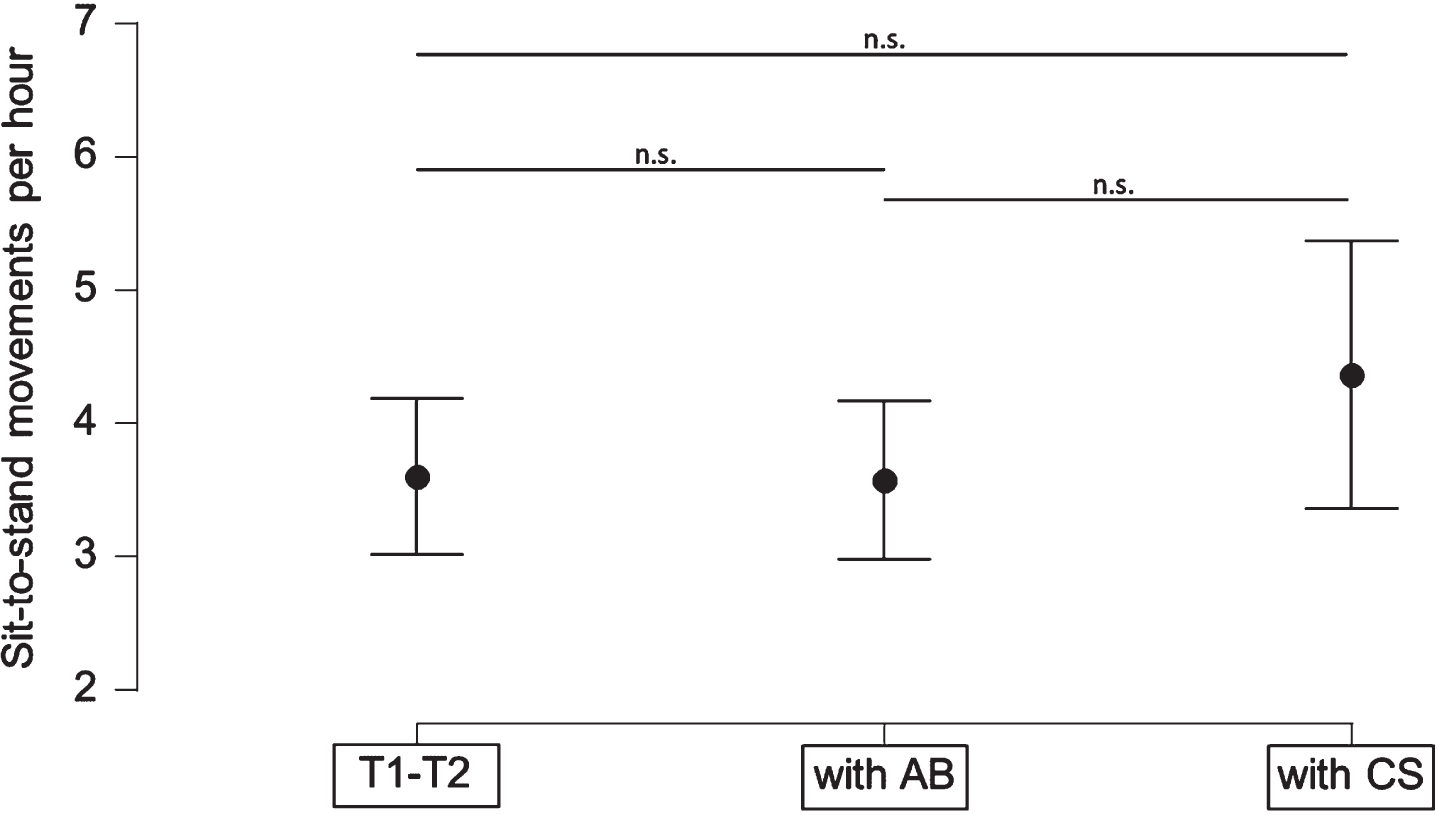
Sit-to-stand movements (STS) per hour during control, abdominal binder (AB) and compression stockings (CS) conditions. No significant difference could be observed between the three groups. (n.s., not significant. Data given as mean±SD).
A total of 1,768.9 h and 5,710 STS movements were recorded with the sensors over all three measurement weeks. Per participant, an average of 43.2±21.7 h/week were recorded. The analysis of variance showed no differences between the three conditions baseline, AB and CS treatment (p = 0.73).
T1 and T2 (control conditions) did not significantly differ concerning MAP drops. Concerning AB treatment, MAP drops were comparable between AB+ and T1 and AB+ and AB– but were higher in AB– compared to T1 (p = 0.007). Concerning CS treatment, MAP drops were comparable between CS+ and CS– but were higher in CS+ (p = 0.014) and in CS– (p = 0.005), compared to T1. Moreover, the intervention groups differed with an F(3.4,51.2)=5.2, p < 0.0022, ω2 = 0.072. The direct comparison between AB and CS showed no significant differences, especially no differences between AB+ and CS+ (details are provided in Fig. 3, Supplementary Tables 1 and 2).
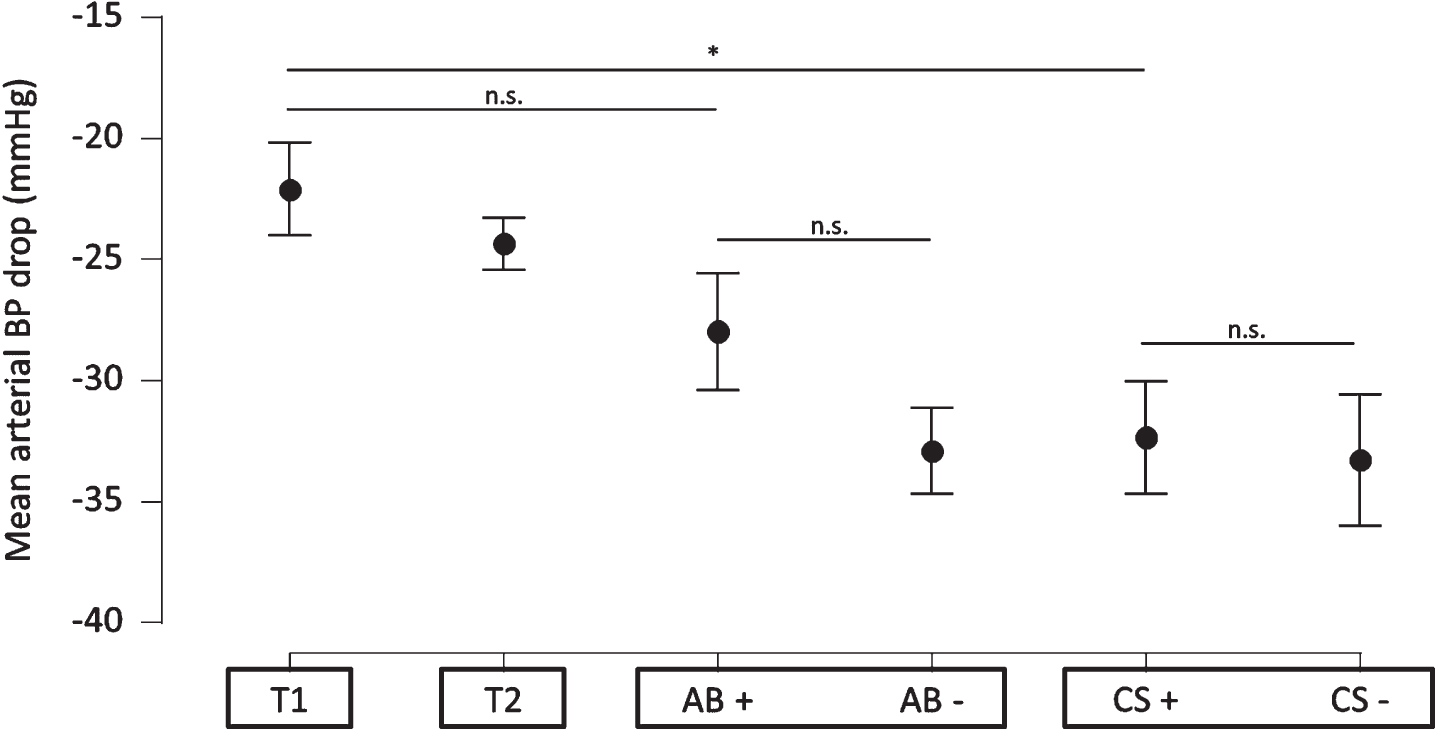
Mean arterial blood pressure (MAP) drops (calculated by subtracting 5th minute supine mean blood pressure from mean blood pressure at third minute of head-up tilt) at baseline (T1) and after abdominal binder (AB) and compression stockings (CS) therapies. T2, control assessment one week after T1. Assessments performed with the respective devices (AB+, CS+) and after 15 min washout (AB–, CS–). Data are given as mean±SE. *p<0.05, n.s., not significant.
Treatment with AB but not with CS reduced OH-associated symptoms as assessed with the OHQ (F(1.4,21.1)=5.7, p = 0.018, ω2 = 0.082, see Fig. 4 and Table 2). Post-hoc testing showed a reduction of 8 points by AB (SE±2.5, pbonf against baseline = 0.0062) and 5 point by CS therapy (SE±2.5, pbonfagainst baseline = 0.4) without significant differences in direct comparison between AB and CS treatment (Table 2).
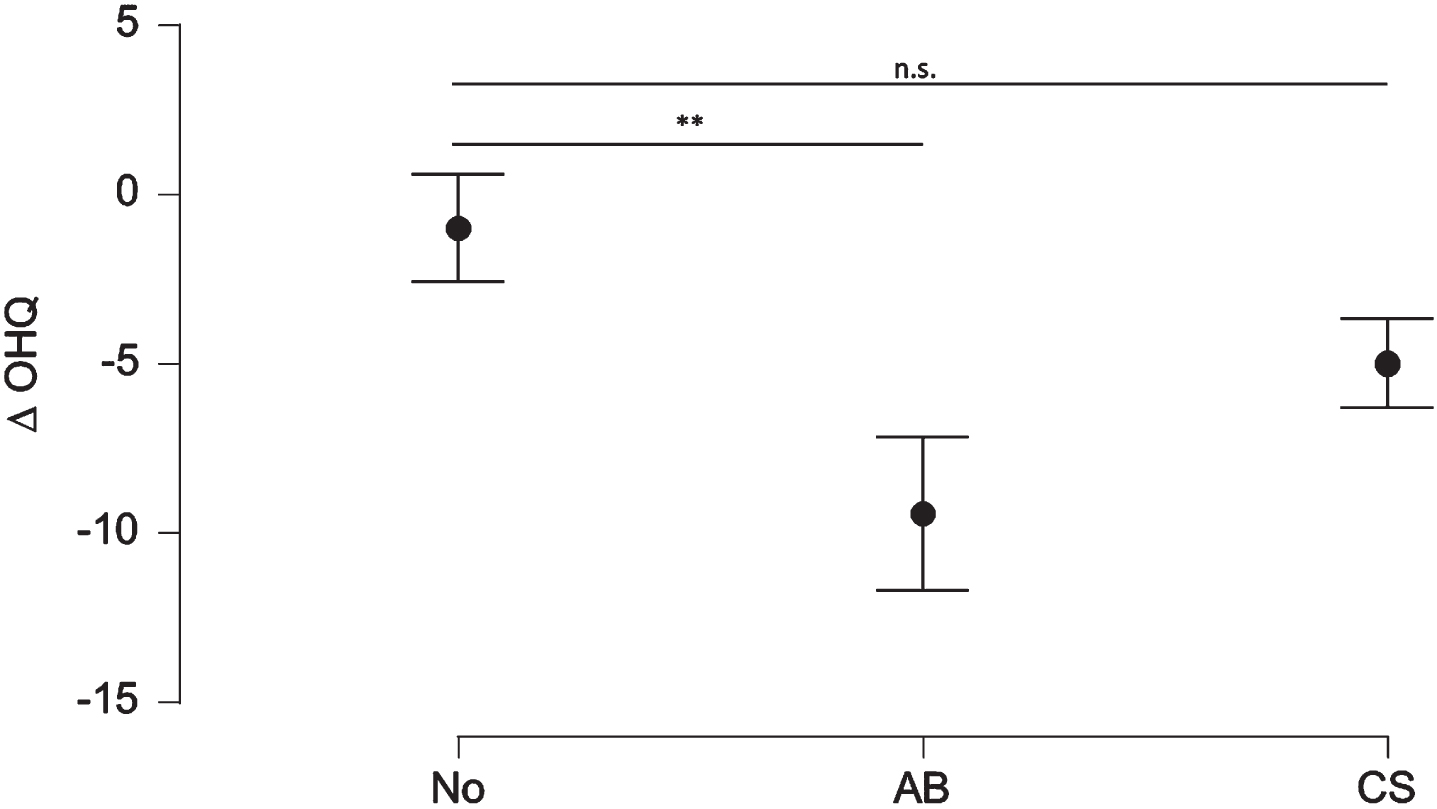
Symptoms associated with orthostatic hypotension (OH) as reported in the OH Questionnaire (OHQ) are reduced by treatment with abdominal binders (AB) (–8.4; SE: 2.5; pbonf < 0.01) but not compression stockings (CS) (–4.0; SE: 2.5; pbonf = 0.4). **p < 0.01. Data given as mean±SE.
The SCOPA-AUT showed an improvement by AB therapy (F(1.6,24.5)=4.2, p = 0.033, ω2 = 0.08; post hoc testing: reduction of 3 points, SE±1.2, pbonf = 0.046, Table 2). The reduction of the SCOPA-AUT by compression therapy approached significance (–3, SE±1.2, pbonf = 0.059). Both AB and CS therapies reduced the non-motor symptoms assessed via the NMSS (F(2,30)=9.5, p = 0.0064, ω2 = 0.1, Table 2). Post hoc testing showed significant differences between both, no therapy and AB (mean difference = 10.2, SE±2.6, pbonf = 0.0037) and between no therapy and CS (mean difference = 9.2, SE±2.6, pbonf = 0.0013). No significant differences were observed comparing AB and CS treatment, neither for the SCOPA-AUT nor for the NMSS. The evaluation of the other secondary endpoints (PDQ-39, MDS-UPDRS, part I-IV, ADL/iADL, and STS duration) showed no significant differences between the three conditions (no therapy, AB and CS; Table 2, Supplementary Table 3).
The evaluation of the STS duration showed no significant differences between no therapy, therapy with abdominal binder and therapy with compression stockings (detailed results are provided in Supplementary Table 3).
Real life movement intensity, turning duration, and real-life turning frequency were not significantly different between the three conditions (for details, see Supplementary Table 3). Depressive symptoms assessed with the BDI-II were significantly reduced by AB but not with CS therapy (F(1.3, 18.9)=4.6, p = 0.037, ω2 = 0.049; post hoc testing against T1: mean difference 2.5, SE±0.8, p = 0.019). No significant differences could be observed in the CGI and PGI. Details are reported in Table 2.
Classification of evidence
Although this study could not provide the anticipated Class I evidence that the physical activity surrogate marker “sit-to-stand frequency in usual environment” shows treatment effects in AB or CS therapies, the significant results of the secondary endpoints OHQ, SCOPA-AUT and mean MAP in the AB treatment suggest a superiority of AB over CS.
DISCUSSION
Treating OH with AB and CS was not associated with increase in continuously assessed, objective real-life surrogate markers of physical activity, and these parameters did not differ between the two interventions. More specifically, the primary outcome measure of this single blinded cross-over trial, real-life STS frequency, was not significantly changed by the interventions. The results of the secondary and exploratory physical activity endpoints argue against a relevant change in quality of STS (and turning) movements, and the amount of overall physical activity.
The following reasons may explain, at least partly, this “negative” result. The participants included in this trial were already at a relatively high physical activity level at inclusion, which may make it difficult to further increase this parameter. Already under control conditions, our patient population performed about twice as many STS/h as the study participants of an earlier, in some parts comparable study [30]. Interestingly, the population of the latter was even younger, less severely affected and had a shorter duration of illness, than our cohort, which suggests that no reliable predictions about daily activity and mobility can be made from a person’s demographic and gross clinical data. Similar results were obtained in a review comparing mobility parameters under supervised with mobility parameters under unsupervised conditions [31]. Moreover, an increase in physical activity also requires a change in habits. Although the home measurements were taken over 1 week, a longer observation period (e.g., 1–2 months) could possibly show an effect. Moreover, as expected in a cohort with advanced PD patients, also these participants suffered from multiple PD-associated symptoms beyond OH (i.e., akinesia, rigidity, and postural instability) that might have prevented them from increasing their physical activity significantly due to an (efficient) OH treatment. Therefore, future trials in this field should also assess the participants’ satisfaction with their existing mobility and their general desire to improve mobility (if they are satisfied with their current mobility, it will not improve as a result of an improvement in a symptom), as well as the effective restriction of mobility due to the symptom to be treated. A recent systematic review presents a conceptual framework for walking experience from the perspective of patients [32]. It can be assumed that the similar applies to STS.
As already mentioned, this is possibly the first trial worldwide that has used a specific digital endpoint of mobility as the primary endpoint for the evaluation of a (potentially) mobility-influencing symptom and can therefore provide preliminary but important insights for the design of such future trials in the field.
Concerning clinical and PRO-related endpoints, our analysis showed favorable effects of AB, compared to CS therapy. Especially the OHQ, a questionnaire focusing on orthostatic symptoms (OHSA) and associated activities (OHDAS), showed a significant and, based on Frith and Newton [33] a clinically relevant reduction of symptoms by AB but not by CS. A positive effect of AB on OH-associated symptoms has already been reported by use of the OHQ in a 4-week open-label follow up study [15]. While in an older study with 14 patients superiority of AB over CS stockings for BPD was demonstrated, this was evident in our study only for subjectively reported symptoms [17].
In addition, autonomic dysfunction assessed with the SCOPA-AUT was significantly improved only in the AB intervention but failed to reach significance with CS therapy. Finally, the AB and CS both improved non-motor symptoms (NMSS) compared to no therapy. These results show 1) that the interventions investigated here have indeed an effect that most probably translates into real life but may be detectable/relevant only in the “non-physical activity” area, and 2) that AB therapy may achieve even more effect, compared to CS therapy. Future studies should investigate these aspects on a more granular level, and also test whether the combination of both therapies may lead to additional benefit for the patients.
This trial shows additional, to our best knowledge not yet published results: We observed that continuous compression therapy, somehow contrary to the above-mentioned positive effects patients reported, did not decrease BPD but even increased it. This was significant after one week of treatment with CS (CS+), but not with AB (AB+). This result also tends to speak for the superiority of AB therapy over CS therapy.
Most interestingly, mean BPD increased even further when compression therapy was stopped, even after a short washout phase of 15 min, and reached now significance in both treatment arms compared to baseline. It is conceivable that AB and CS treatment lead to an accelerated decompensation of the blood pressure-regulating system. To our knowledge, this paradox effect of permanent compression therapy has not been described before. All previous studies consistently report positive effects of compression therapy on BPD [5]. There are some possible explanations for this discrepancy. First, permanent compression therapy might reduce physiologic self-regulation of vaso-compression and sympathetic activation as venous pooling and return is taken over by compression of the abdominal splanchnic veins (AB) or femoral veins (CS). This functional loss might result in an even more pronounced BPD in extreme situations as in tilting table tests where additional compensatory mechanisms like activation of the muscle pump cannot be used. However, such situations do rarely occur in daily life. Second, there is evidence that BPD and clinical symptoms may be relatively independent from each other. A study reported that only 23 of 37 patients with a moderate BPD (>20/10 mmHg) had OH-associated symptoms but 14 had not [1], which was confirmed in another, more recent and independent study [34]. The reason(s) for the low association between OH and clinical symptoms are not completely understood, but effective compensation to the variable (peripheral) pressures on the cerebral blood flow level might play a role [35, 36]. Third, a compression therapy might affect the overall feeling of safety which could attenuate OH-associated symptoms reported by patients. Long-term studies with compression therapy are needed to examine a possible habituation effect (on subjective patient reported outcomes). Fourth, in previous studies effects of compression therapy were evaluated after wearing AB or CS just a few hours. Washout times between baseline and compression therapy differed between 20 min [14, 17], 1 h [13] and 1 day [15]. The key difference is the length of time wearing the compression therapy which was 7 days for each device in our study instead of just a few hours. All other factors are less likely to explain the differences. In particular, the applied compression pressures are almost identical to the previous studies [13–17].
Our study has limitations. First, the evaluation was performed in a small patient collective with some heterogeneity that cannot be avoided in such groups, e.g., with regard to medication. A larger case number of participants may have provided more insight, including results from correlation analyses that are not possible with such a small N. However, we consider this a thoroughly performed pilot trial for hypothesis generation, especially as it is, to our best knowledge, the first trial investigating a neurological cohort that used a digital real-life parameter as a primary endpoint. Second, our trial shows surprising results especially concerning BPD, which requires further investigation into the pathophysiology of OH treatment. In subsequent studies, possible discrepancies between patient self-assessment and objective measurement of physical activity should be considered.
Conclusion
Wearing of AB or CS does most probably not increase physical activity, as measured with mean sit-to-stand frequency during respective 7-day assessment phases, or PD motor symptoms. Patient-reported outcomes, such as orthostatic symptoms, autonomic symptoms, and other non-motor symptoms, were significantly improved by AB, and to a lesser extent by CS therapy. BPD in tilting table test was significantly increased after one week of CS and augmented on trend after one week of AB therapy. The results of this study support the superiority of AB therapy compared to CS in treatment of OH in PD patients on a symptomatic level, which however did not translate in a measure of daily mobility and activity.
