Abstract
Background:
Patients with Parkinson’s disease (PD) and rest tremor may also have tremor during posture holding, with tremor being transiently suppressed during the transition between resting and posture holding. Other PD patients show no tremor suppression between resting and posture holding. The mechanisms responsible for tremor suppression in PD are unknown. Understanding the mechanisms of tremor suppression would expand our knowledge of tremor pathophysiology in PD.
Objective:
To investigate whether tremor suppression reflects the activity of the primary motor cortex (M1) and assess whether tremor features are different in patients with and without tremor suppression.
Methods:
We compared corticomuscular coherence (CMC) at tremor frequency and transcranial magnetic stimulation tremor resetting between 10 PD patients with tremor suppression and 10 patients without suppression. We also compared tremor spectral features between the two groups.
Results:
Patients with tremor suppression had higher CMC at tremor frequency during both rest tremor and postural tremor, and a higher postural tremor resetting index and stability when compared with patients without tremor suppression. Rest tremor frequency was similar between the two groups, but postural tremor frequency was lower in patients with tremor suppression as compared to patients without.
Conclusion:
M1 plays a major role in tremor suppression in PD, and the mechanisms of postural tremor may differ between patients with and without tremor suppression.
INTRODUCTION
Rest tremor is the most common type of tremor in Parkinson’s disease (PD) [1], but many patients also show postural tremor [2]. In PD patients with rest and postural tremor, rest tremor is typically suppressed by voluntary movements [3] and reemerges after a delay while holding a posture. This type of tremor is referred to as ‘re-emergent tremor’ (RET) [4–8]. However, in some patients, rest tremor is not suppressed while reaching a posture and thus postural tremor appears with no delay (patients without tremor suppression) [9, 10]. One previous study suggested that postural tremor may have a different pathophysiology in patients with tremor suppression as compared to patients without [10].
The mechanisms responsible for tremor suppression when holding a posture in PD are unknown. Tremor suppression may be due to proprioceptive inputs associated with postural changes [3], which may disrupt tremor activity in the cerebello-thalamic circuit via the spinocerebellar pathway. Alternatively, tremor suppression may be due to the encoding of voluntary motor commands that may disengage M1 from the tremor-generating network [4]. This hypothesis is supported by the observation that tremor can be voluntarily suppressed for several seconds in patients with PD [11].
In the present study, we tested the hypothesis that tremor suppression is strictly dependent on the critical involvement of M1 activity in PD tremor pathophysiology [8]. To do so, we compared M1 involvement in PD patients with and without tremor suppression by measuring the functional coupling between M1 activity and tremor activity, as measured by corticomuscular coherence (CMC) [12]. M1 involvement was also tested by measuring TMS-induced tremor resetting [13, 14]. If tremor suppression reflects greater M1 involvement in tremor pathophysiology, we would expect greater CMC and more resetting in PD patients with tremor suppression as compared to patients without tremor suppression. In order to confirm whether RET and non-suppressible postural tremor are two distinct tremor phenotypes, we also compared tremor frequency and power between the two groups of PD patients.
MATERIALS AND METHODS
We enrolled 20 patients with a diagnosis of PD according to standard clinical criteria [15, 16]. Patient clinical characteristics are shown in Table 1.
Clinical characteristics in patients with tremor suppression (TS+) and patients without tremor suppression (TS–)
H&Y, Hoehn & Yahr; 1Sum of tremor scores in MDS-UPDRS part III sub-items 3.17 and 3.18 of the most tremulous upper limb. 2Tremor score in MDS-UPDRS part III sub-item 3.15 of the more tremulous upper limb. Data are expressed as mean (SD).
All PD patients enrolled in the study had rest and postural tremor. Patients had to show postural tremor severity with a minimum score of 2 as measured by the Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS), part III, item 3.15. Similarly, patients had to show rest tremor with a minimum combined score of 4 for rest tremor amplitude (item 3.17) and constancy (item 3.18). We excluded patients with head tremor or other neurological or psychiatric conditions. Patients were asked to discontinue their antiparkinsonian medications for at least 12 hours prior to the experiment. Participants provided written informed consent prior to participating in the study. Study procedures were approved by the institutional review board and were in accordance with the Declaration of Helsinki.
Rest tremor was assessed with the patient’s forearm resting on a pillow and the hand hanging free. After the appearance of stable rest tremor (at least 10 seconds), patients were asked to extend their wrist and hold the posture. Patients were then divided into groups of patients with tremor suppression (i.e., RET) or without. Tremor suppression was evaluated clinically by visual inspection during consecutive resting, wrist extension, and 2 minutes of posture holding [3, 6]. We recruited 10 patients with tremor suppression and 10 patients without tremor suppression.
Electromyographic (EMG) activity from the extensor carpi radialis muscle (ECR) of the more tremulous arm was recorded with surface electrodes, amplified×1000, sampled at 5 kHz, and bandpass filtered between 5 and 2000 Hz (1401 Plus, CED). EEG was recorded from 32 channels mounted according to the 10–20 system, using POz as an online reference and FPz as a ground. EEG signal was sampled at 10 kHz and bandpass filtered between DC and 2500 Hz (NeurOne, Bittium). Impedance for all EEG electrodes was kept below 5 kΩ. EMG and EEG were recorded during three 1–minute blocks of rest tremor and postural tremor. Rest and postural tremor were measured when tremor had been present for at least 10 seconds. Following the same methods previously described [8], we measured rest and postural tremor peak frequency and tremor power from EMG recordings, and we computed CMC at rest and postural tremor peak frequency between EMG and EEG recordings. All participants were also asked to repeat ten trials of tremor suppression assessment consisting of consecutive resting, wrist extension, and 15 seconds of posture holding during EMG recording. These trials of tremor suppression assessment were used to confirm the group assignment for each patient. We measured the EMG power at tremor peak frequency [8] separately in the 2 seconds before and after the movement onset, then we computed the mean values over the ten trials, and we computed the percentage of reduction in tremor amplitude with the following formula: [1 –(mean tremor power after / mean tremor power before)]*100.
A Magstim® BiStim2 (Magstim Company, UK) and a 70-mm figure-of-eight coil were used for the TMS-induced tremor resetting experiment. TMS was delivered with anterior-posterior direction over M1 on the hotspot evoking the largest motor-evoked potential (MEP) in the contralateral ECR muscle on the tremor side. Coil positioning was monitored throughout the experiment with neuronavigation (Brainsight, Rogue Research) with an optical tracking system (Polaris Vicra, Northern Digital). We used the intensity that evoked a MEP of 1-mV amplitude at rest. Tremor resetting was assessed separately during rest and posture holding by five blocks of single-pulse TMS over the M1 contralateral to the tremor side, with each block consisting of 8 trials and an intertrial interval randomly varying between 7.5 and 12.5 seconds. Tremor resetting was assessed by calculating the average resetting index (RI) of the 5 tremor bursts following TMS and the resetting stability (RS) as a ratio of the RI between the first and fifth tremor bursts [8, 17].
For both CMC and tremor resetting experiments, blocks were separated by 30 seconds of rest to avoid muscle fatigue during posture holding and possible habituation during rest tremor. Data are expressed as mean±standard deviation (SD). Tremor resetting was considered significant when RI values were statistically different from zero as tested by a 1-sample t-test. We compared rest and postural tremor frequency, power, CMC, RI, and RS between patients with and without movement-related tremor suppression by means of unpaired t-test. A p value < 0.05 indicated statistical significance.
RESULTS
Clinical characteristics were similar between PD patients with and without movement-related tremor suppression (Table 1). All patients with clinically defined tremor suppression showed a clear reduction in tremor amplitude (EMG power at tremor peak frequency) in the 2 seconds following the movement onset compared to the 2 seconds before (81.73±6.59%), whereas no clear reduction was observed in patients without tremor suppression (1.64±2.68%).
The frequency of rest tremor was similar between patients with and without tremor suppression (4.3±0.4 vs. 4.6±0.9 Hz, t = –0.95, p = 0.356) and we observed a non-significant trend towards higher rest tremor power in patients with tremor suppression as compared to patients without (2.3±0.6 vs. 1.6±0.9, t = 2.02, p = 0.059). Postural tremor frequency was lower in patients with tremor suppression compared to patients without (4.7±0.4 vs. 5.4±0.6 Hz, t = –2.97, p = 0.008), whereas the two groups showed similar postural tremor power (2.3±0.9 vs. 2.0±0.5, t = 1.03, p = 0.317) (Fig. 1A).
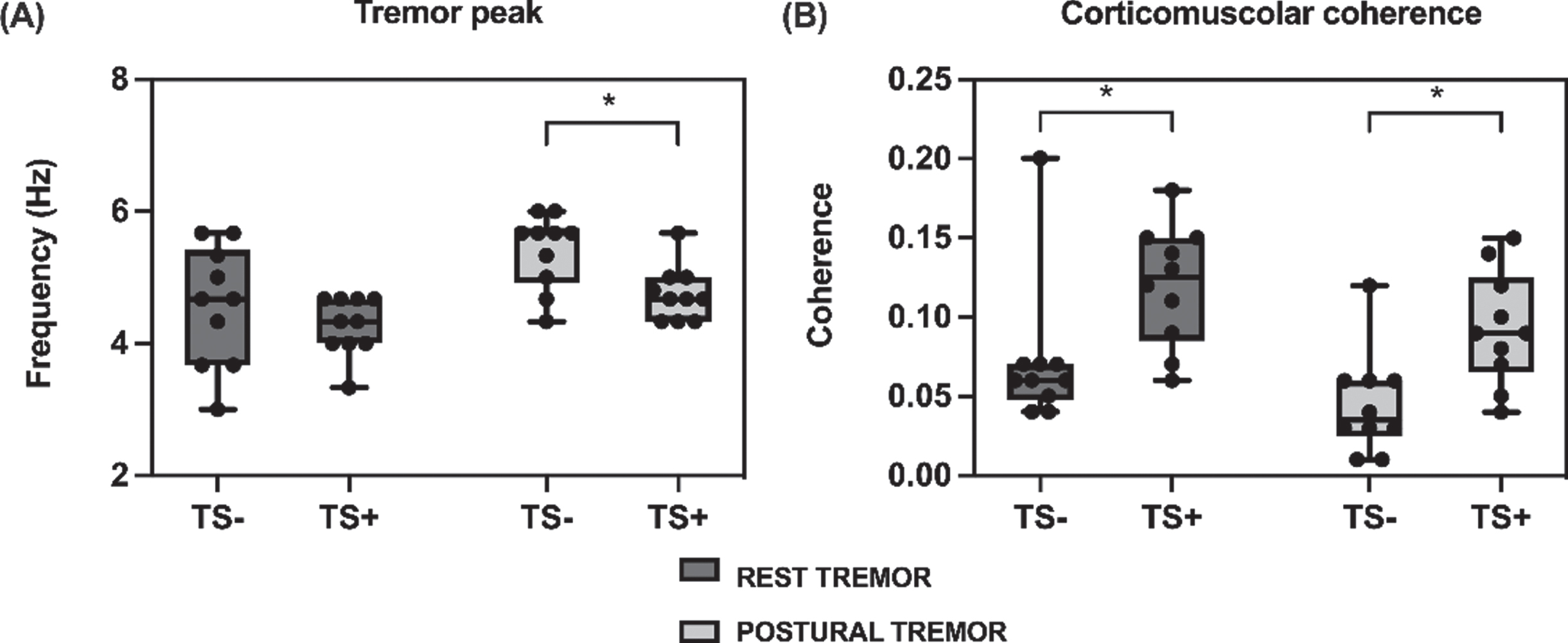
Tremor spectral features and corticomuscolar coherence. Comparison between patients with tremor suppression (TS+) and patients without tremor suppression (TS–). Black dots indicate individual data points, boxes indicate interquartile range, whiskers indicate minimum and maximum values, horizontal lines indicate median values. *p values < 0.05. A) Peak frequency of rest and postural tremor. B) Corticomuscolar coherence at tremor frequency over primary motor cortex contralateral to tremor side (C3, C4 channels) during rest and postural tremor.
CMC values at tremor frequency over M1 contralateral to the tremor side (C3/C4 channel) were significantly higher in patients with tremor suppression compared to patients without during rest (0.12±0.04 vs. 0.07±0.04, t = 2.66, p = 0.016) and postural tremor (0.09±0.04 vs. 0.05±0.03, t = 2.93, p = 0.009) (Fig. 1B).
The TMS intensity used for tremor resetting was statistically similar between patients with tremor suppression (65±10%) and without (67±8%). We found no significant differences in rest tremor RI and RS values between patients with and without tremor suppression (RI: 2.1±0.6 vs. 1.8±0.5, t = 1.59, p = 0.12; RS: 0.99±0.3 vs. 0.9±0.4, t = 0.66, p = 0.516), whereas patients with tremor suppression showed significantly higher postural tremor RI (2.0±0.3 vs. 1.4±0.7, t = 2.56, p = 0.020) and RS values (1.0±0.2 vs. 0.7±0.3, t = 3.20, p = 0.005) compared to patients without tremor suppression (Fig. 2).
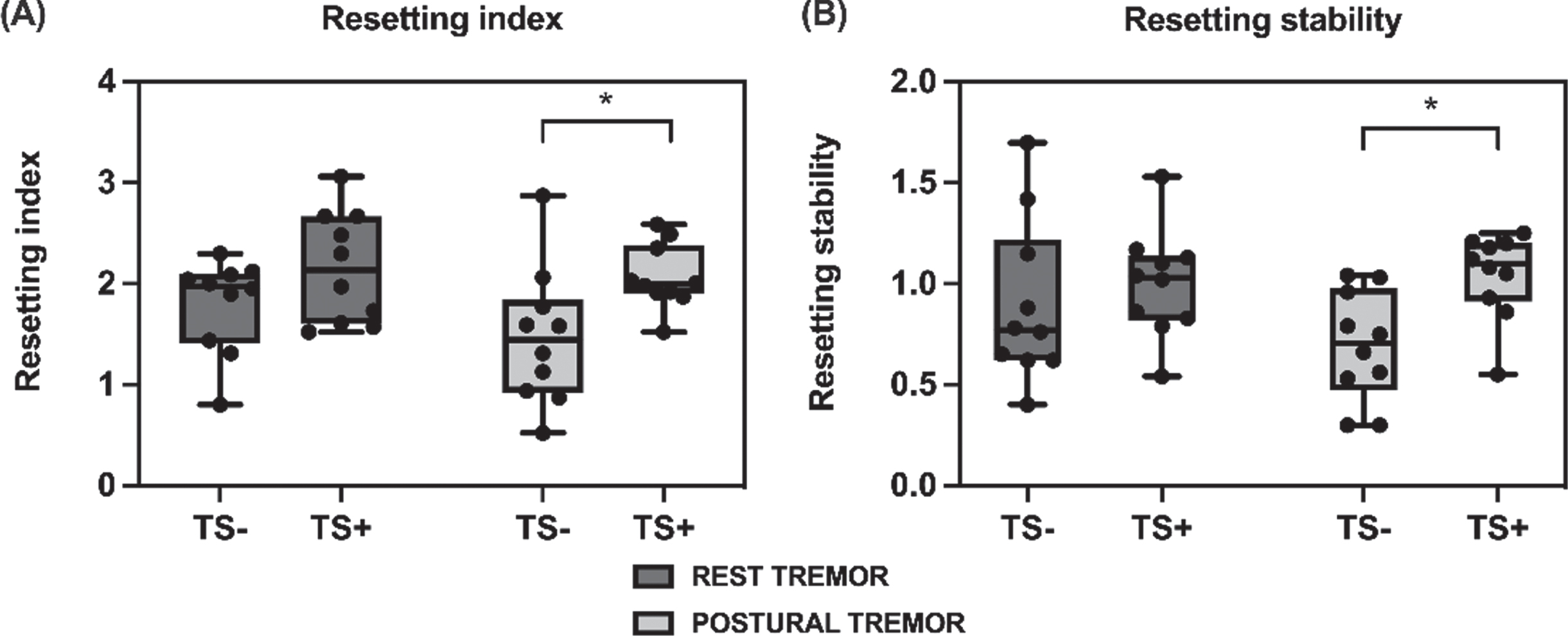
Tremor resetting induced by transcranial magnetic stimulation over the primary motor cortex. Comparison between patients with tremor suppression (TS+) and patients without tremor suppression (TS–). Black dots indicate individual data points, boxes indicate interquartile range, whiskers indicate minimum and maximum values, horizontal lines indicate median values. *p values < 0.05. A) Tremor resetting index of rest and postural tremor. B) Tremor resetting stability of rest and postural tremor, calculated as ratio of the resetting index between the first and fifth tremor burst.
DISCUSSION
In the present study, we investigated the role played by M1 in tremor suppression in PD. Patients with tremor suppression had higher CMC at tremor frequency during both rest tremor and postural tremor, and higher RI and RS values during postural tremor when compared with patients without tremor suppression. The two groups of patients had similar tremor RI and RS values during rest tremor. We also compared tremor features and found that rest tremor frequency was similar between the two groups, but postural tremor frequency was lower in patients with tremor suppression as compared to patients without. Overall, these findings suggest that M1 plays a major role in tremor suppression and that the mechanisms of postural tremor may differ between patients with and without tremor suppression.
Since the two PD groups we studied had similar disease duration, severity, and tremor clinical scores, the differences in postural tremor frequency, CMC, and resetting did not depend on these clinical features. We excluded patients with postural tremor alone without rest tremor because postural tremor alone is not considered for the diagnosis of PD [16]. Postural tremor resetting may depend, at least in part, on mechanical perturbation of the stretch reflex arc related to MEP regardless of cortical mechanisms [14, 18–20]. However, since stretch reflex arc-related resetting depends on tremor power [18], the observation of similar postural tremor power in patients with and without tremor suppression excluded possible confounding due to peripheral mechanisms.
The first finding of the present study is that patients with tremor suppression had greater CMC at rest and postural tremor frequency than those without tremor suppression. Previous studies demonstrated that both rest tremor and postural tremor showed significant coherence at tremor frequency between EMG and EEG electrodes over the contralateral sensorimotor cortex in patients with tremor suppression [8, 21–23]. Conversely, to date no studies have investigated tremor CMC in PD patients without tremor suppression. CMC measures the coupling between sensorimotor cortical activity and EMG bursts and has been used to indicate M1 involvement in driving tremor-related activity [12]. CMC, however, does not exclusively reflect M1 output to the muscle, but also reflects tremor-related sensory reafference to the cortex [24, 25]. Sensory reafference related to posture holding may influence CMC results. However, the observation that the two patient groups also showed different CMC during rest tremor makes it unlikely that CMC differences during postural tremor simply reflect changes in sensory input related to posture holding. We believe that the greater CMC reflects a higher correlation between M1 oscillatory activity and tremor in patients with tremor suppression compared to patients without. These results support our hypothesis that tremor suppression is related to M1-specific mechanisms.
The second novel finding is that RI values for postural tremor were significantly different from zero in both patients with tremor suppression and in patients without, suggesting that both tremors depend on M1 activity. Previous studies that showed a significant postural tremor RI in PD did not distinguish between patients with and without tremor suppression [13, 26]. However, the higher RI in patients with tremor suppression compared to patients without observed in the present study suggests that M1 activity is more involved in driving tremor in patients with tremor suppression. Since the tremor RI alone may result from M1 circuit perturbation or the perturbation of cortico-subcortical loops [13, 17], it does not clarify whether M1 is only an output station for tremor oscillations or whether it directly participates in generating oscillations [27]. A tremor RS close to 1 means a stable RI value across tremor bursts following TMS and reflects the ability to reset the entire central oscillator at a particular phase in its cycle [28]. Therefore, a transient resetting indicates a passive role of M1 as an output station, whereas a stable resetting suggests that M1 directly participates in generating oscillations as a node of the tremor network [13, 14]. The higher RS in patients with tremor suppression compared to patients without therefore indicates that M1 is more involved in the oscillating network responsible for RET. Overall, CMC and tremor resetting suggest that M1 is more directly involved in the pathophysiology of tremor in patients with tremor suppression.
To further investigate whether tremor suppression is associated with different tremor types, we compared tremor spectral features between patients with tremor suppression and without. Patients with tremor suppression had lower postural tremor frequency than patients without tremor suppression, in line with previous studies [9, 10]. The lower postural tremor frequency in patients with tremor suppression than in patients without may be influenced by different proprioceptive inputs associated with posture holding in the two groups. However, a previous study in both types of tremor found no effect of weight loading on tremor frequency, thus making it unlikely that peripheral mechanisms account for the difference in frequency between patients with and without tremor suppression [10]. We conclude that differences in postural tremor frequency in patients with and without tremor suppression are due to differences in central oscillator activity, thus suggesting differences in postural tremor mechanisms in PD patients with and without tremor suppression. The presence of two distinct postural tremor types in PD patients with rest tremor is in line with previous observations [4, 10]. Postural tremor in patients with rest tremor suppression has been referred to as RET by several authors [4–8], whereas one previous study referred to postural tremor in patients without rest tremor suppression as ‘pure postural tremor’ [10].
Our study expands previous clinical observations showing that mechanisms related to M1 involvement in tremor pathophysiology may underlie the phenomenon of tremor suppression in PD. Rest tremor in PD results from oscillatory M1 activity generated by pathological interplay between the basal ganglia, particularly the globus pallidus internus (GPi) and cerebello-thalamo-cortical circuit [27]. The GPi triggers oscillatory activity in the cerebello-thalamo-cortical loop, which entrains M1 and in turn modulates tremor amplitude [27]. Similar to rest tremor, RET is also associated with oscillations in the cerebello-thalamo-cortical loop [21]. Since M1 plays a similar role in rest tremor and RET [8], transitory tremor suppression between resting and posture holding in patients with rest tremor and RET may be related to M1 mechanisms. When patients are asked to reach and hold a posture, the encoding of voluntary movement at the M1 level may temporarily disengage M1 from the cerebello-thalamo-cortical loop. Tremor suppression at the M1 level in patients with rest tremor and RET may occur via a competition between movement-encoding and tremor-generating activity. In patients with rest tremor and postural tremor without suppression, the encoding of a motor command would have less impact on tremor activity. Subcortical areas may also be involved differently in RET and non-suppressible postural tremor, which may contribute to tremor suppression. This hypothesis is supported by recent data showing that tremor suppression is shorter following M1 TMS than after the execution of a voluntary fast wrist extension [27]. Therefore, subcortical structures involved in both voluntary actions and tremor may contribute to tremor suppression. One possibility is that the GPi, which is thought to trigger tremor-related activity, may also actively interfere with tremor activity during movements due to its ability to inhibit unwanted movements [29, 30]. At the same time, the cerebellar-receiving motor thalamus may be responsible for tremor suppression since it plays an important role in the transition between motor planning and execution [31] and there is an overlap between planning- and tremor-related activation at the level of the motor thalamus [32]. In addition, intact cerebellar output seems necessary for movement-related rest tremor suppression in PD [33, 34]. We speculate that subcortical mechanisms may contribute to tremor suppression by modulating the ability of the GPi and cerebello-thalamic circuit to engage M1 activity in tremor oscillations.
In conclusion, our study provides new insight into the pathophysiological mechanisms responsible for tremor suppression in PD. We found evidence supporting a relatively larger role of M1 in tremor pathophysiology in PD patients showing transient suppression of tremor with voluntary movements. The present study also supports the hypothesis that different pathophysiological mechanisms underlie tremor in patients with rest tremor and RET and in patients with rest tremor and postural tremor without suppression. These results shed light on the link between voluntary movement and tremor in PD and may represent a basis for developing non-invasive neuromodulation paradigms aimed at treating rest tremor in PD.
CONFLICTS OF INTEREST
The authors have no conflict of interest to report.
