Abstract
Background:
Cognitive impairment is frequent in Parkinson’s disease (PD) and several neurotransmitter changes have been reported since the time of diagnosis, although seldom investigated altogether in the same patient cohort.
Objective:
Our aim was to evaluate the association between neurotransmitter impairment, brain metabolism, and cognition in a cohort of de novo, drug-naïve PD patients.
Methods:
We retrospectively selected 95 consecutive drug-naïve PD patients (mean age 71.89±7.53) undergoing at the time of diagnosis a brain [18F]FDG-PET as a marker of brain glucose metabolism and proxy measure of neurodegeneration, [123I]FP-CIT-SPECT as a marker and dopaminergic deafferentation in the striatum and frontal cortex, as well as a marker of serotonergic deafferentation in the thalamus, and quantitative electroencephalography (qEEG) as an indirect measure of cholinergic deafferentation. Patients also underwent a complete neuropsychological battery.
Results:
Positive correlations were observed between (i) executive functions and left cerebellar cortex metabolism, (ii) prefrontal dopaminergic tone and working memory (r = 0.304, p = 0.003), (iii) qEEG slowing in the posterior leads and both memory (r = 0.299, p = 0.004) and visuo-spatial functions (r = 0.357, p < 0.001).
INTRODUCTION
Cognitive impairment is a frequent finding in subjects with Parkinson’s disease (PD) and represents a key determinant of quality of life [1–3] in this population.
The main pathological hallmark in the early stage of the disease is the intracellular fibrillization of α-synuclein, along with Lewy body brainstem pathology [4–6]. Cognitive deficits in PD seem to be due to the uneven degeneration of several neurotransmitter systems, reflecting the complexity of the disease evolution [6]. Drug-naïve PD patients represent a suitable model to study the relationships among monoaminergic degeneration, regional neurodegeneration, and cognitive functions, given the presence of monoaminergic system deficits early on in the disease course [7], the relatively modest presence of cortical pathology [8] at this stage of the disease, and the lack of confounding factors from dopaminergic medications use.
In the last decade, several studies have been focused on the relationship between cognition and monoaminergic systems degeneration [9], and found that impaired nigrostriatal dopaminergic innervation in the caudate, anterior putamen, and ventral striatum was directly associated with attention/working memory, executive, and visuospatial functions [10–13]. Beside dopaminergic degeneration, also reduction of raphe serotonin receptors density and cortical cholinergic receptors, have been correlated both with non-motor symptoms [14] and the severity of cognitive impairment in a cohort of drug-naïve, de novo PD [6, 15]. Also, quantitative EEG (qEEG) studies have consistently shown the slowing down of background rhythm early on in the course of Lewy body diseases, thought to reflect mainly cortical cholinergic deafferentation [16, 17].
Another modality, i.e., brain [18F]FDG PET, can track metabolic changes reflecting both neurodegeneration and synaptic dysfunction in vivo. [18F]FDG PET studies have indeed demonstrated that an abnormal glucose brain metabolism profile is present at early stage of PD, especially in the lentiform nucleus, the thalamus, as well as the lateral frontal, paracentral, inferior parietal, and parietooccipital areas, known as Parkinson’s disease-related pattern (PDRP) [18]. Similarly, Huang et al. (2007) [19] described the Parkinson’s disease cognitive pattern (PDCP), characterized by relative hypometabolism in frontal and parietal association areas, along with relatively preserved metabolic levels in the cerebellar vermis and the dentate nuclei, correlating with executive function tasks [19–21]. From these findings, new interests rose in correlating these patterns with clinical and imaging variables. For example, it has been seen that, using [123I]FP-CIT SPECT and [18F]FDG PET, a reduced striatal DAT binding along with an increase in the expression of the PDRP is present at early stage of PD [22], as well as a reduced metabolism in the default mode network associated with an increased expression in the PDCP [23].
Based on these findings, and considering that few studies have combined information deriving from a multimodal approach, we combined [123I]FP-CIT SPECT (used in the thalamus and the striatum as a marker of serotonergic and dopaminergic degeneration, respectively; as well as a marker of extrastriatal prefrontal cortical dopaminergic expression) [24–26], [18F]FDG PET (used as a marker of neurodegeneration) [27], and qEEG (used as an indirect marker of cholinergic deafferentation) [28–30], in order to assess the potential impact of dopaminergic and serotonergic deafferentation, as well as of neurodegeneration and cholinergic tone, on the main cognitive domains in drug-naïve, de novo PD patients [31].
MATERIALS AND METHODS
Patients
In a 3-year period, we examined 95 consecutive, drug-naïve, de novo PD patients (age: 71.8±7.5 years; range: 50–84; 35 females; education: 10.9±4.0 years) who underwent neuropsychological assessment [32], brain [18F]FDG PET, [123I]FP-CIT SPECT, and qEEG recording at the time of the diagnosis. The inclusion criteria were a diagnosis of PD [33], confirmed by evidence of dopaminergic deficit on [123I]FP-CIT SPECT and by at least two years of follow-up. The main exclusion criteria were use of dopaminergic, cholinergic, or serotonergic medications, as well as of other medication known to affect brain function, such as benzodiazepines, the presence of neuropsychiatric or medical comorbidities, presence of dementia, and lack of informed consent. Table 1 shows the demographic, clinical, and imaging characteristics of the PD group in more detail. Nevertheless, all study procedures were performed before the beginning of any dopaminergic, cholinergic, and/or serotonergic medications. Structural imaging was available for all patients to help in the diagnostic process; however, MRI data were not used in the present study, as all images were acquired using different scanners and protocols.
Demographic, clinical and imaging characteristics of Parkinson’s disease group. Values are shown as mean±standard deviation (SD)
M, male; F, female; MMSE, Mini-Mental State Examination; MDS-UPDRS-III, Movement Disorders Unified Parkinson’s Disease Rating Scale, motor section; MCI, mild cognitive impairment.
At the time of examinations, subjects gave their written consent for use of their anonymized data to research purposes, according to the protocol approved by our institutional review board. The study was conducted in respect of the Helsinki declaration.
Neuropsychological evaluations and cognitive domain quantification
All subjects underwent a complete neuropsychological evaluation, as previously described [32]. Based on the criteria for the evaluation of mild cognitive impairment in PD [31], we grouped tests into five domains (i.e., Executive Functions, Attention and Working Memory, Memory, Language, and Visuo-Spatial Functions), to compute a single score for each of these domains, based on previously published data [31]. To this aim, we used a principal component analysis with fixed factors for each domain, using the raw scores of individual tests to represent each of the five domains. The first principal component for each domain was retained for further analyses. The neuropsychological tests included in each cognitive domain are reported in Supplementary Table 1.
[18F]FDG PET
FDG PET images were acquired as a marker of regional neurodegeneration.
Brain [18F]FDG PET scan were acquired according to the guidelines of the European Association of Nuclear Medicine32, using a SIEMENS Biograph 16 PET/CT hybrid system with a total axial field of view of 15 cm and no interplane gap space. Attenuation correction was based on CT. Data were reconstructed through an ordered subset-expectation maximization (OSEM) algorithm, 16 subset and 6 iterations, with a reconstructed voxel size of 1.33×1.33×2.00 mm. All [18F]FDG PET images were acquired in static mode and then subjected to affine and nonlinear spatial normalization into Talairach and Tournoux space using SPM12 (Wellcome Department of Cognitive Neurology, London, UK). All the default choices of SPM were followed except for spatial normalization. For this study, the H215O SPM-default template was replaced by an optimized brain [18F]FDG PET template as described by Della Rosa and colleagues [36]. The spatially normalized set of images was then smoothed with a 10-mm isotropic Gaussian filter to blur individual variations in gyral anatomy and to increase the signal-to-noise ratio. In order to exclude white matter voxels and to limit the amount of voxels from hypometabolic regions from the reference value of the global mean, we fixed a ‘grey matter threshold’ of 1.0 instead of the default value of 0.8 [37].
[123I]FP-CIT SPECT
[123I]FP-CIT SPECT images were used to study nigro-striatal and raphe thalamic degeneration as well as prefrontal monoaminergic (i.e., dopaminergic and serotonergic) [26] innervation.
[123I]FP-CIT SPECT data was acquired by means of a 2-headed Millennium VG camera (G.E. Healthcare), as previously described [35].
The reconstructed [123I]FP-CIT SPECT images were processed using the BasGan software version 2 based on a high definition, 3D striatal template, derived from Talairach’s atlas [38]. Partial volume effect (PVE) correction is included in the process of uptake computation of caudate, putamen, and the occipital region background. The partial volume effect correction performed by the method consists of an activity assignment in a Talairach-Tornoux atlas-based 3-compartment model of basal ganglia. Background uptake was subtracted by putamen and caudate uptake as follows (caudate or putamen uptake –background uptake)/background uptake, to generate specific to non-displaceable binding ratio (SBR) values. Partial volume correction, a feature included in the BasGan pipeline, allows to reduce the impact of the limited SPECT spatial resolution of the assessment of midline structures. A single [123I]FP-CIT SBR value was computed for the caudate as well as for the putamen (representing the mean between the left and right nuclei SBR). In order to study SERT binding and prefrontal dopaminergic innervation, a customized brain [123I]FP-CIT SPECT template was built from 20 brain MRI and SPECT scans of healthy subjects as previously described [25], using SPM12. The SPECT images of the enrolled patients were then normalized to the MNI space in SPM12 and smoothed (3D gaussian filter with 10 mm full-width at half-maximum) [26]. In order to study cortical dopaminergic activity, we created region of interest (ROI) masks for the prefrontal cortex, consisting of Brodmann Areas 10, 11, 46, and 47 in both hemispheres, and for the bilateral thalami, using the WFU Pickatlas toolbox (https://fmri.wfubmc.edu/software/pickatlas, Wake Forest University Health Sciences Medical Center Blvd. Winston-Salem, NC), then we evaluated [123I]FP-CIT SPECT SBR in these ROIs. Prefrontal cortex SBR was computed as follows: (ROI –background uptake)/background uptake. All data represent a mean value across the two hemispheres.
Quantitative EEG recording and posterior Alpha/Theta power ratio
qEEG was used as an indirect approach to study the cholinergic system integrity [28, 29].
EEG recordings were performed in the late morning to minimize drowsiness and obtained from patients seated in a comfortable chair with eyes closed. EEG electrodes were placed using standard 10–20 EEG electrode positions (Fp1, Fpz, Fp2, F3, Fz, F4, F7, F8, C3, Cz, C4, P3, Pz, P4, O1, Oz, O2, T3, T4). Electrode impedances were closely monitored and kept below 5 kOhm. Data were acquired using LTM system (EBNeuro, Florence, IT) at a sampling rate of 512 Hz and a bandpass of 0.3–70 Hz. To monitor eye movements, the horizontal electro-oculogram was simultaneously recorded with the same recording parameters of EEG. An EEG technician was present during the entire recording session to keep constant the level of vigilance and to monitor signal quality. Standard longitudinal bipolar montage was used for analysis. EEG data were manually analyzed off-line to reject artifacts. One minute of artifact-free EEG data was used for further analysis. Segments were two seconds continuous with no overlap. The Fast Fourier Transform (FFT, Welch method), applied to 4-s segments (Tukey window) with 2-s overlaps, was used to compute EEG spectral power for each derivation. The EEG spectrum was then divided into the following frequency bands: delta (2.25–4 Hz), theta (4.25–8 Hz), alpha (8.25–12 Hz), sigma (12.25–16 Hz), and beta (16.25–32 Hz) and relative power was computed for each band as normalized to the total EEG power.
Based on mean EEG spectra, the ratio between alpha (8.25–12 Hz) and theta (4.25–8 Hz) band power (alpha/theta ratio) was computed in the posterior (P3/O1, P4/O2, T5/O1, T6/O2) cortical region as indirect proxies of the cholinergic tone [28, 29]. Considering the right-skewed distribution of the alpha/theta ratio, the logarithmic (log-) transformation was applied in order to approximate a symmetric distribution of data.
We choose to explore the cholinergic tone expressed in the posterior leads since, in PD cohorts, deficits in cholinergic system exhibited mainly in the posterior cortices has been shown to be present at all stages of the disease, including non-demented subjects [39], and to consequently correlate with slowing at qEEG in the posterior leads [30].
Statistical analysis
Statistics were performed using IBM SPSS Statistics 25 (Armonk, NY, USA).
Partial correlations, correcting for age and MDS-UPRS-III score, were used to correlate each cognitive domain score with both the [123I]FP-CIT SPECT SBR in the basal ganglia and the prefrontal mask and qEEG metrics. Statistical threshold was set at 0.05 and p-values, both uncorrected and corrected using a false discovery rate (FDR) approach, were reported.
As for [18F]FDG PET, using SPM12 (https://www.fil.ion.ucl.ac.uk/spm/), a whole-brain voxel-wise correlation between whole-brain scaled FDG counts and each cognitive domain score was performed using a voxel height threshold p < 0.001 (uncorrected) and a minimum cluster size of 100 voxels for significant clusters. Analyses were corrected for age and MDS-UPDRS-III score, as a marker of disease severity. At cluster level, the accepted threshold of statistical significance was p < 0.05, family-wise error corrected for multiple comparisons.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
RESULTS
Executive functions
We found a positive correlation between executive functions and brain metabolic levels in the left cerebellar hemisphere, extending in the vermis. Resulting anatomic areas are shown in detail in Table 2 and Fig. 1.
Anatomic areas resulting from the voxel-wise analysis between Executive Functions and frontal [18F]FDG values. Analyses were corrected for age and MDS-UPDRS-III score
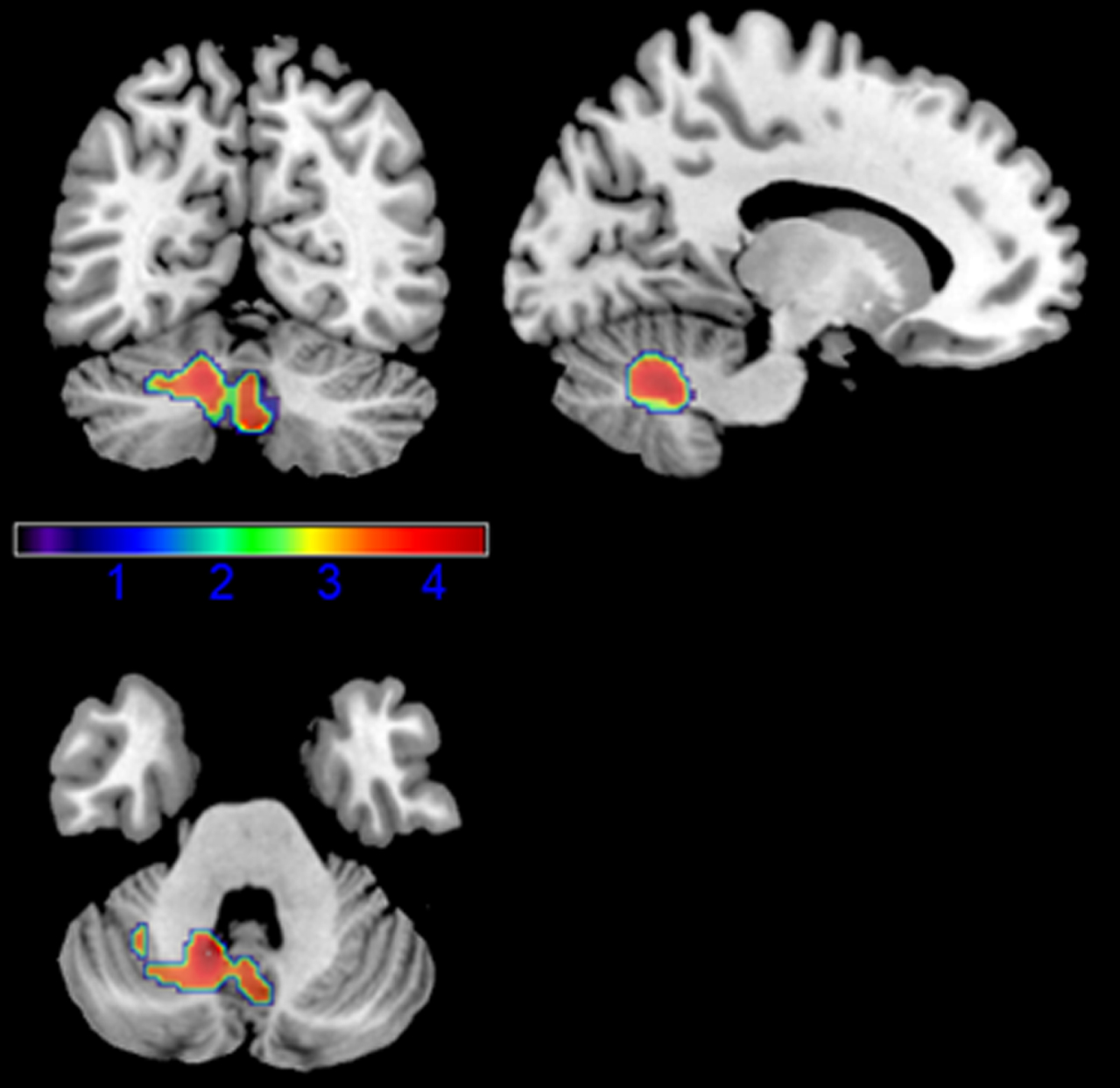
Results of the voxel-wise correlation analysis between executive functions and brain metabolism; nuisances: age and MDS-UPDRS-III score.
Attention and working memory
We found a positive correlation between attention and working memory and prefrontal [123I]FP-CIT SBR values (p = 0.003). Results are shown in Table 3 and Fig. 2.
Correlation between neuropsychological domain composite scores, striatal and thalamic [123I]FP-CIT SBR values, frontal [123I]FP-CIT SBR values and qEEG Alpha/Theta ratio. Analyses were corrected for age and MDS-UPDRS-III score
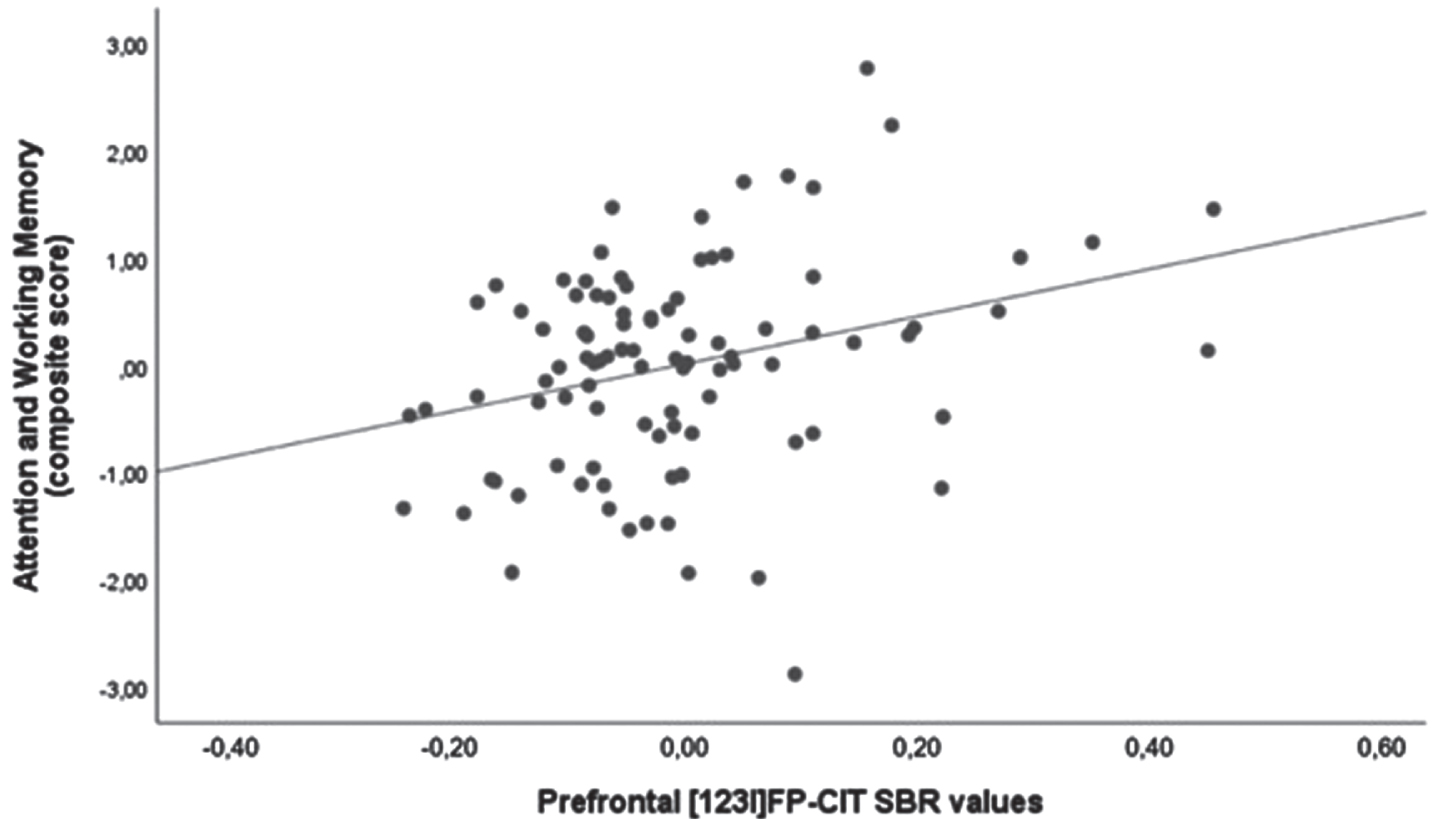
Scatter plot of the correlation between attention and working memory domain and prefrontal [123I]FP-CIT SBR values. Analysis was corrected for age and MDS-UPDRS-III score.
Memory
We found a positive correlation between memory domain and qEEG alpha/theta ratio (p = 0.004). Results are shown in Table 3 and Fig. 3.
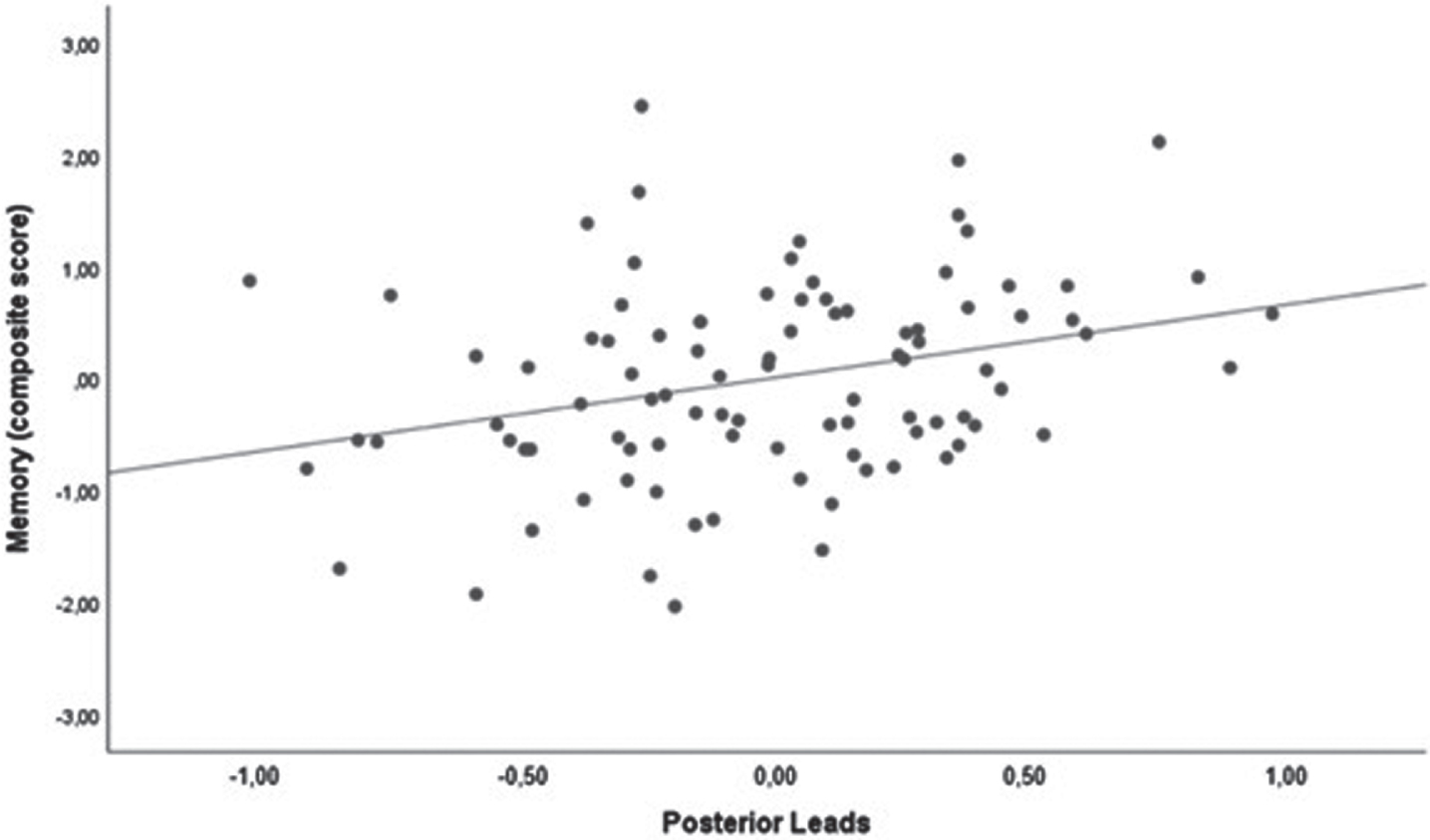
Scatter plot of the correlation between memory domain and the qEEG alpha/theta ratio in the posterior leads. Analysis was corrected for age and MDS-UPDRS-III score.
Language
We found a positive correlation between language and qEEG alpha/theta ratio, not surviving correction for multiple comparisons. Results are shown in Table 3.
Visuo-spatial functions
We found a positive correlation between visuo-spatial functions and qEEG alpha/theta ratio (p < 0.001). Results are shown in Table 3 and Fig. 4.
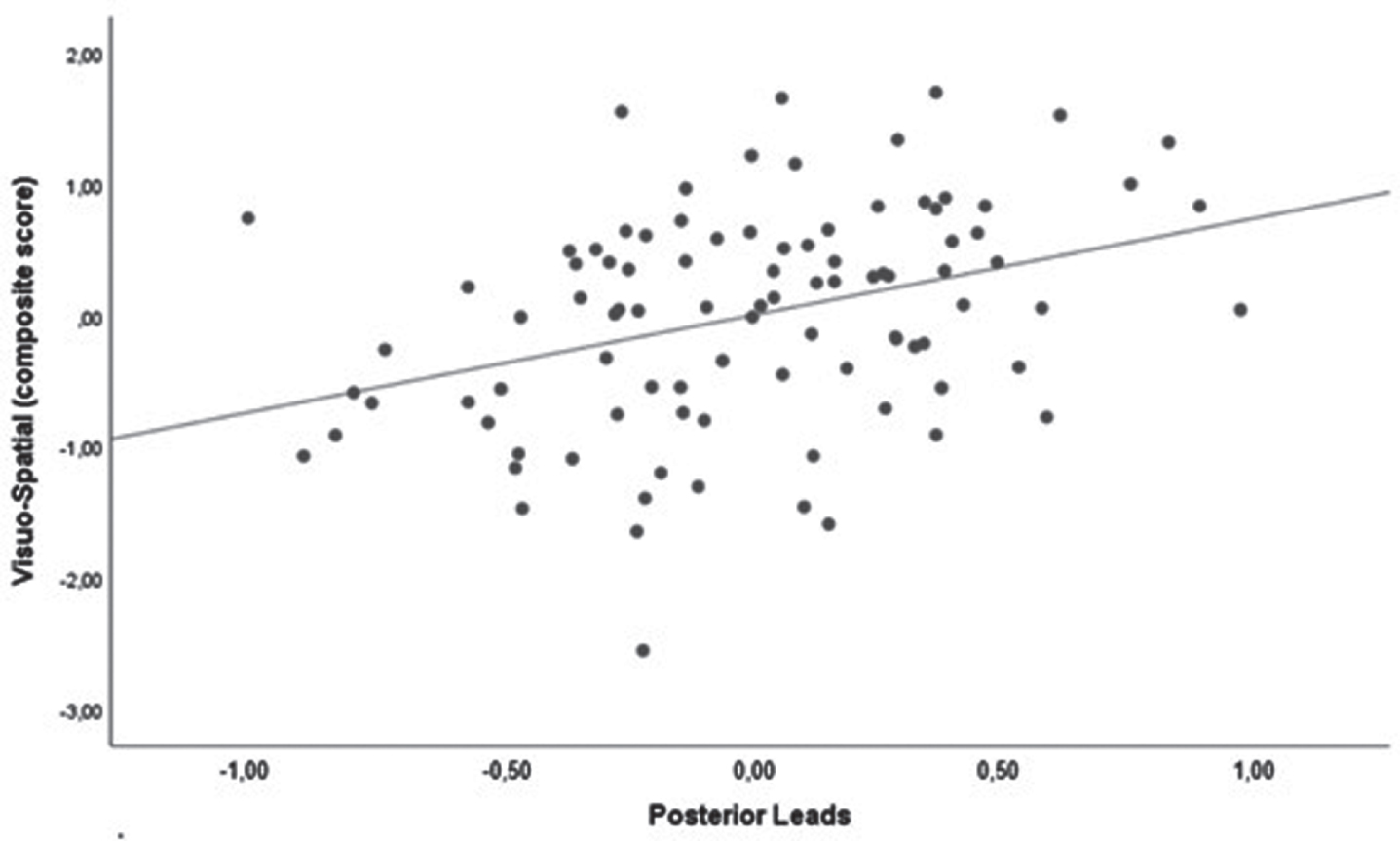
Scatter plot of the correlation between visuo-spatial domain and the qEEG alpha/theta ratio in the posterior leads. Analysis was corrected for age and MDS-UPDRS-III score.
DISCUSSION
In this study, we tried to assess the impact of cortical and deep grey matter monoaminergic tones, as well as of regional neurodegeneration, on the main cognitive domains in drug-naïve, de novo PD patients.
Overall, we found that executive functions directly correlated with metabolic levels in the left cerebellum, while attention and working memory directly correlated with prefrontal dopaminergic tone, and memory and visuo-spatial functions were related to the alpha/theta ration in the posterior leads at qEEG, reflecting cholinergic tone.
Executive functions
We showed a positive correlation between executive functions and cerebellar glucose metabolism. Over the years there has been an increasing interest in the association between cerebellum and cognition. Grafman et al. (1992) [40] found that deficit in higher-order cognitive functions, such as executive functions, were associated with cerebellum, basal ganglia, and frontal regions damage. Different theories have been proposed to describe the role played by the cerebellum in cognitive processes. Firstly, the cerebellum is included in a vast network spanning also the basal ganglia and the prefrontal cortex, structures that are known to play a key role in executive functions [41]. Schmahmann & Sherman (1998) [42] observed impaired executive function and deficient planning and abstract reasoning in patients with focal cerebellar injury. Other studies, using different imaging techniques such as resting state functional connectivity MRI and diffusion MRI, found that the dorsal and ventral attention, frontoparietal and both default mode and salience networks map onto focal areas within the posterior lobe of the cerebellum [43, 44]. In PD, a role for abnormal functional connectivity between the cerebellum and the cerebral cortex has been proposed as a potential substrate for cognitive deficits, also possibly mediated by changes in cholinergic activity [45].
Our findings expand on the observations found within the PDCP [19, 46], and link cerebellar focal metabolic changes with executive functions.
Attention and working memory
Attention and working memory were directly correlated with prefrontal dopaminergic tone. It has been previously shown that dopamine is thought to play a significant role in attentional process both in healthy subjects and in neuropsychiatric disorders [47, 48].
In rats, lesions of the meso-cortical dopaminergic bundles are associated with difficulties in task-switching and in the space exploration, in line with the proposed role of prefrontal dopamine in attentional processes [49, 50].
In healthy controls, prefrontal dopamine levels, as assessed by PET with 6-[18F]fluoro-L-DOPA (FDOPA), have been shown to correlate with executive functions independently from regional grey matter volumes [47]. Also, in subjects with psychiatric conditions, such as attention deficit-hyperactivity disorder and schizophrenia, prefrontal dopamine levels have been found to directly correlate with cognitive performance [47, 48]. In PD, prefrontal dopamine presents a complex relationship with cognition. In fact, both high and low levels of regional prefrontal dopamine were associated with reduced reaction times, in line with the known U-inverted shape of the dopamine - cognitive performance association [51, 52]. Our results broaden previous literature exploring the selective role of cortical dopaminergic tone in attentional processes in de novo, drug-naïve PD subjects.
Memory, visuo-spatial functions
We found that cholinergic tone, indirectly expressed by the alpha/theta ratio in the posterior cortical regions, positively correlated with memory and visuospatial functions. The alpha/theta power ratio mirrors the reduction of alpha frequency power and the increase of theta power during wake state at rest [16] and reliably, though indirectly, reflects the cholinergic-correlated background slowing in the brain; in particular, it has been associated with cognitive impairment in PD [16], as well as with response to acetylcholinesterase inhibitors [53].
Among diffuse projection systems, the impact of acetylcholine on cognition is relatively well characterized. A reduction in cholinergic cortical innervation is thought to play a key role in cognitive performance in subjects with Alzheimer’s disease and dementia with Lewy bodies [54] in which cholinergic drugs are currently used to improve cognition.
Cholinergic system deficits in posterior cortices in PD has been found in all disease stages, including non-demented subjects [39], and to correlate with posterior slowing at qEEG [30].
The role of cholinergic system in memory is well known, as acetylcholine is thought to be relevant for the consolidation of memory traces, both in healthy controls and among different neurodegenerative conditions, including PD [55]. In particular, nicotinic alpha7 acetylcholine receptors (α7 nAChRs) are highly expressed in the hippocampus and are associated with cholinergic pathways. Their depletion accompanies cognitive decline, specifically memory impairment, observed in different neurodegenerative conditions [56]. In a cohort of 25 idiopathic PD patients, Lorenz et al. (2014) [57] found a significant correlation between nAChR density loss and the results of CERAD (the Consortium to Establish a Registry for Alzheimer’s Disease), suggesting that acetylcholinesterase inhibitors could be useful in the treatment of cognitive impairment in PD [58], since therapy with acetylcholinesterase inhibitors has been shown to improve memory performance and to modulate the memory network in PD patients [59]. On the other hand, also muscarinic receptors expression is correlated with cognitive deficits. Antagonists, such as scopolamine, impair the cognitive performance in animal models [60].
As for visuo-spatial functions, they have been associated with cholinergic deficits, assessed using transcranial magnetic stimulation with the short-latency afferent inhibition, showing that a reduced central cholinergic tone was associated with a worse performance at visuo-spatial tests [61]. Interestingly, in PD visual hallucinations are thought to be supported by visuo-spatial deficits [61] and, from a pharmacological point of view, respond to an increase of cholinergic tone instead than to antipsychotics [62].
Lack of correlation with deep grey matter metabolism and monoaminergic innervation
We did not find significant correlation between cognitive domains and both cortical and deep grey matter metabolism, nigro-striatal and raphe-thalamic pathway degeneration.
The lack of correlation between cognition, putamen metabolism, and nigro-putaminal integrity is not surprising, given both the role of this structure involved mainly in motor control, and its lack of association with cortical metabolism found in previous studies in PD [63].
We also failed to find a correlation between executive functions and fronto-parietal metabolism, included in the PDCP [19]. This lack of correlation might be explained when taking into consideration the characteristics of our cohort, composed by de novo PD patients, thus with fairly preserved brain metabolism and executive functions, compared to the more advanced disease stage of patients included in the PDCP studies.
Conversely, the lack of correlation between caudate measures and cognition was partly unexpended. However, it must be noted that the nigro-caudate pathway degenerates later on in the disease course than putamen [64], with half of de novo PD patients showing a normal dopaminergic innervation of the caudate [65], especially in absence of frank cognitive decline. Moreover, from imaging perspective, the correlation between nigro-caudate metrics and cognition in early PD is somewhat controversial, with some studies failing to show significant associations [66].
Regarding the lack of findings in the raphe-thalamic serotonergic pathway, it stems from the cognitive domains explored. Indeed, we have previously shown that alteration in this pathway are associated in PD with theory of mind and not with more conventional cognitive domains [35].
Strength and limitations
This study represents a proof-of-concept investigation of the different roles played by regional neurodegeneration and by cortical and deep grey matter monoaminergic tones on cognitive functions in de novo PD patients. To our knowledge this is the first study combining multi-tracer molecular imaging and qEEG to shed light on the contribution of multiple diffuse projection systems on each cognitive domain in PD.
De novo PD is an interesting model to investigate deficits in multiple diffuse projection systems because the degree of neurodegeneration is still moderate at this stage, while later on in the disease course, the severity of damage and the effects of drugs may hamper meaningful conclusions.
Regarding the assessment of cognition, we did not explore a single cognitive construct, such as general cognition, but we systematically explored each cognitive domain using an a priori model [31].
We did not use a specific serotonergic marker, but the [123I]FP-CIT SBR in the thalami, taking advantage of the [123I]FP-CIT affinity for SERT and also the lack of DAT in the thalami [26, 67]. Although this approach has been previously used in a number of studies [24, 25], it would be worthwhile to confirm our findings using a SERT specific PET tracer; however, it is not currently approved for clinical use nor is widely available. Moreover, the cholinergic marker here used (i.e., qEEG) is an indirect measure of cholinergic activity, thus caution is needed when interpreting qEEG results.
These limitations are however balanced by the large number of enrolled patients, the lack of confounding factors (e.g., drugs,) and the use of multimodal molecular imaging-neurophysiological approach, acquired for all patients in the same center.
CONCLUSION
We have shown the peculiar neurotransmitter and metabolic correlates of specific cognitive functions in drug naïve, de novo PD patients, using a multi-modal approach. Our results suggest possible novel treatment approaches for cognitive decline in de novo PD. Future longitudinal studies are warranted to explore changes in each specific cognitive domain.
Footnotes
ACKNOWLEDGMENTS
Matteo Pardini is partly supported by a University of Genoa “curiosity-driven” grant.
This work was developed within the framework of the DINOGMI Department of Excellence of MIUR 2018-2022 (legge 232 del 2016).
The study was partly supported by a grant from the Italian Ministry of Health to IRCCS Ospedale Policlinico San Martino (Fondi per la Ricerca Corrente 2019/2020) and by a University of Genoa Curiosity Grant to M.P.
CONFLICT OF INTEREST
The authors declare no Competing Non-Financial Interests but the following Competing Financial Interests: Matteo Pardini: fees from Novartis, Merck and Biogen. Dario Arnaldi: fees from Fidia, Bioproject and Jazz. Silvia Morbelli: speaker Honoraria from G.E. Healthcare. Flavio Nobili: fees from Roche, Bial, Biogen and G.E. Healthcare. All other authors report no conflicts of interest.
