Abstract
Background:
Educational interventions promoting the role of physical activity (PA) aim to address knowledge, poor exercise self-efficacy, and low outcome expectations, which are well-researched barriers to PA participation in healthy and in people with chronic conditions. However, little is known about the effectiveness of educational interventions in addressing these barriers in people with Parkinson’s (PwP).
Objective:
To examine the content of education interventions that promote PA behavior in PwP, and to assess their effectiveness on physical and psychosocial outcomes.
Methods:
An electronic search (12/2021) of MEDLINE, EMBASE, CINAHL, PubMed PsycINFO, the Web of Science and the Cochrane Library was conducted from 1990 to 2021. Education interventions, alone or combined with other strategies, promoting PA in PwP were included. Quality was assessed using the Johanna Briggs Institute and National Institute of Health quality assessment tools. A narrative synthesis was performed.
Results:
Six studies were identified. Five interventions were comprised of education and exercise sessions. Improvement in physical and psychosocial outcomes were suggested but delineating the exact impact of education was impeded due to lack of assessment.
Conclusion:
Few interventions exist that provide knowledge, and skills promoting PA participation, and fewer are addressed towards newly diagnosed PwP. There is lack of assessment over the effectiveness of education as a tool to facilitate PA participation in PwP. Lack of assessment poses the risk of potentially disregarding effective interventions or adopting ineffective approaches without the evidence. Education interventions can boost PA engagement by increasing factors such as exercise self-efficacy, but further interventions are required to assess this model of relationship.
INTRODUCTION
Parkinson’s disease (PD) is a chronic neurological condition clinically characterized by a wide range of progressive motor and non-motor symptoms [1, 2]. The occurrence and progression of those symptoms influence activities of daily living and consequently the person’s quality of life [3, 4]. Current management of PD centers on the provision of medication to control the impact of parkinsonian symptoms [5]. Although beneficial, the effects of medication are often time limited, with prolonged medication use associated with unwanted non-motor and motor side effects such as dyskinesias, which in themselves become debilitating [6]. The importance of non-pharmacological interventions such as exercise and physical activity (PA) are recognized for symptomatic relief in both motor and non-motor symptoms [7]. Specifically, studies discuss the beneficial role of PA and exercise for symptoms such as speech impairments, cognitive impairments, depression, postural instability and gait, which do not often respond to medication [8].
The role of PA and exercise in the management of the inherent functional decline associated with PD has been largely researched [9, 10]. Epidemiological studies have investigated the potential preventative role of exercise in lowering the risk of PD development [11]. Additionally, randomized controlled trials (RCT) have suggested a neuroprotective or neurorestorative function of high intensity exercise, which promotes neuronal health [12–17]. Specifically, systematic reviews and meta-analyses suggest that people with Parkinson’s (PwP) who exercise have better strength, flexibility and improve their cardiovascular fitness, as well as balance and quality of life [18, 19]. Hence, given the evidence on their physical, neurophysiological and psychosocial benefits PA and exercise are becoming accepted as fundamental components of the management of PD alongside the standard pharmacological and surgical management [7, 20].
As evidence over the significant and clinically meaningful benefits of exercise and PA for PwP accumulates [10, 17], PwP are in parallel asking for trusted, evidence-based education interventions around rehabilitation approaches [21] that will improve their ability to self-manage their condition more effectively, in order to live well and maintain a good quality of life [22]. Furthermore, guidelines and research advocate for physical health education interventions [23–25] that promote physical activity and exercise to help mitigate deconditioning and symptomatic decline accelerated by sedentary behavior associated in the PD population [26–29].
Education interventions have a prime role in addressing well-recognized barriers in PA participation [30–32]. Recent studies have suggested that educating PwP on the role of PA and exercise in PD progression can boost PA engagement by increasing enablers such as exercise self-efficacy [33] and removing barriers such as misinformation about exercise and exercise outcomes [32]. It is well established that high exercise self-efficacy is positively associated with exercise behavior both in healthy older adults [34] and in PwP [30, 31]. Thus, there is a strong rationale for educational interventions to address these barriers. However, despite the need and the recommendation for timely education interventions designed to help PwP manage their physical activity behavior [35, 36], there are yet no standardized guidelines on the information content, mode of delivery or duration of educational interventions.
Furthermore, little is known about the effective components that comprise an education intervention aimed at promoting the role of exercise and physical activity in PD education interventions. To answer these questions and fill the evidence gap, a systematic review of the literature on physical health education interventions for PwP was conducted.
With a growing population of people living with PD and the widely acknowledged health and well-being benefits of PA and exercise for PwP [19], the need for evidence-based recommendation on the content of education programs that promote PA and exercise is paramount in supporting both clinical practice and effective self-management for PwP.
This systematic review aims to: Examine the content, delivery method and duration of physical health education or information provision programs and their primary outcomes across the PD trajectory. Explore the effectiveness of physical health education or information provision programs. Identify barriers and facilitators in developing and implementing physical health educational programs across the across the PD trajectory.
MATERIALS AND METHODS
This systematic literature review was conducted in line with the Preferred Reporting Items for Systematic Reviews (PRISMA) reporting guidelines [37]. A protocol for this review (CRD42021241505) is registered on PROSPERO (https://www.crd.york.ac.uk/prospero/).
The review focuses on physical health education, defined for the purpose of this study as any education or information provision aimed to improve understanding of how and why exercise and PA might benefit PwP.
Search strategy and selection criteria
The electronic searches of MEDLINE, EMBASE, CINAHL, PubMed PsycINFO, the Web of Science, and the Cochrane Library covered publications from 1990 to December 2021. To ensure that the search captured all PD-specific education interventions, the search strategy was kept as broad as possible, and did not apply restrictions or used search terms such as exercise or physical activity, which might have resulted the focus to be on exercise interventions and potentially miss out on educational interventions. The search strategy included a combination of free-text words and MeSH terms such as “Parkinson* Disease” AND “patient education” OR “patient information” with boolean operators as specified by each electronic database settings (see the Supplementary Material for detailed MEDLINE search strategy). Additional searches were conducted in research reports of PD related non-for-profit organizations and charities, databases of doctoral dissertations and conference abstracts. If a relevant conference abstract was identified, searches were made to find the full paper; if no full paper was found, the authors of the abstract were contacted. The reference lists of included studies and identified reviews were screened for potential studies that were not picked up by the electronic searches.
All titles and abstracts were independently reviewed by at least two authors (LA, JJ, and JA). Studies were considered eligible if they met the following inclusion criteria: a) included education interventions that promoted the role of exercise and/or physical activity in the management of PD; b) reported on physical and/or psychosocial outcomes following the education intervention; c) were delivered to adults with PD in any setting, through variable modes of delivery and were singular or delivered on a weekly basis. Education interventions in which exercise and/or physical activity were mentioned but full content of education was not possible to be established, were excluded.
Study screening and data extraction
All retrieved studies were screened by one reviewer (LA) and checked independently by the two other reviewers (JJ and JA) using the predefined inclusion criteria and utilizing the Rayyan web application for systematic review management (http://www.rayann.qcri.org). All three reviewers confirmed the eligibility of the identified studies. Disagreement was resolved through consensus and by the opinion of a third reviewer (JJ or JA). Details of the flow of study selection throughout the process of assessment of eligibility criteria are presented in the PRISMA flow diagram (Fig. 1). Using a standardized form, three reviewers completed data extraction and recorded information on study year, author, duration of intervention, education content, sample size and results for each study. Relevant information and overview of the included studies are presented in Table 1. Attempts were made to contact authors of studies when data was missing, or data were ambiguously presented.
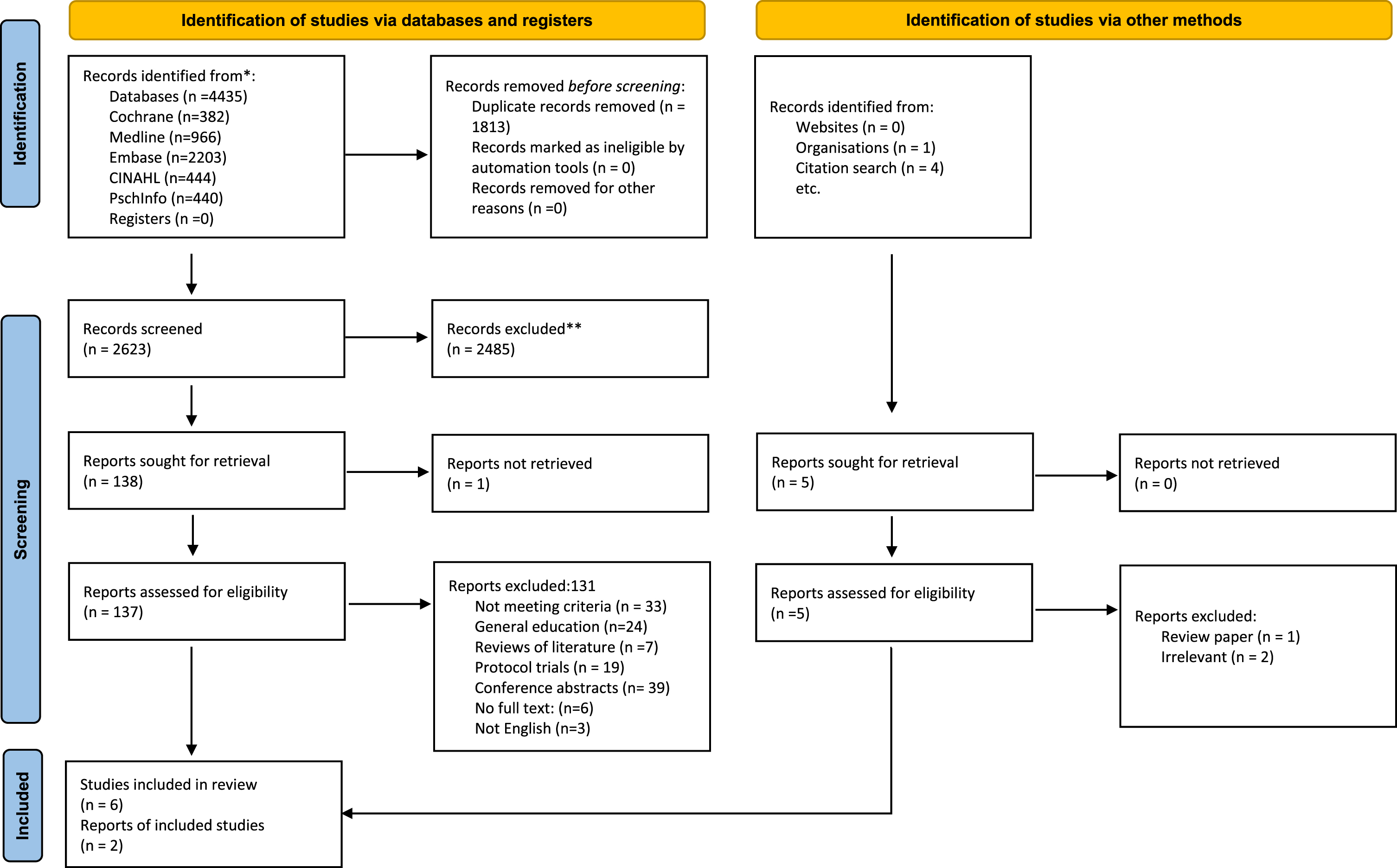
PRISMA flow diagram of included studies.
Education intervention characteristics of included studies
Risk of bias and quality assessment
The included studies presented a variety of study designs and therefore to appropriately assess each study design, we chose the Johanna Briggs Institute Tools [38] for the randomized controlled trials (RCT), which are considered appropriate for study designs looking at feasibility, appropriateness, meaningfulness, and effectiveness of healthcare interventions. For non-randomized studies without a control group and before-after (Pre-Post) studies, the National Institute of Health quality assessment tool for before and after study was used (https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools). Three review authors (LA, JA, JJ) independently assessed and scored the selected studies; disagreement or uncertainty were resolved by discussion.
Data synthesis
A narrative synthesis of the results was performed, as due to heterogeneity in the interventions and outcome measures used, performing meta-analyses was not appropriate.
RESULTS
A total of 2,623 were identified by the initial search (Fig. 1). Following title and abstract screening, 2,485 were excluded resulting in 137 studies which underwent full text review. From those, 131 studies were excluded for the following reasons: conference abstracts (
Study characteristics
Of the 6 included studies, two were RCTs and four were pre- and post-test studies. The intervention in all studies comprised of a structured exercise program component and an education session component, apart from one study [33] which did not include an exercise session. The education sessions were provided in a group setting apart from two studies, which delivered the education intervention on a one-to-one basis [33, 39]. There was great variability in the outcome measures used to assess the efficacy of the intervention both for the physical and psychosocial outcomes (Table 2). Despite study interventions delivering exercise classes and education sessions, the efficacy of interventions was assessed as a whole, without taking into consideration the role of each individual component. Only one study [42] used different education content between the intervention and control group, indicating the potential impact that education might have, without further assessment or discussion over this distinction. Furthermore, studies failed to define the objectives of the education intervention and rationalize the use of education as one of the strategies used to impact physical or psychosocial outcomes. As such, all but one [33] of the included studies did not objectively measure the efficacy of the education provided.
Outcome measures used in the included studies
 Outcome measures used by at least two studies
Outcome measures used by at least two studies
Participants and settings
This review included 898 PwP who had been living with PD on average for more than 6 years (Fig. 2). Two studies included participants with a Hoehn & Yahr stage (H&Y) score ≤3 [39, 40], while three studies aimed their intervention specifically towards people in the early stage of Parkinson’s H&Y score ≤2 [33, 42]. One study included participants at H&Y stage ≤4 [43]. van Nimwegen et al. [39] specifically designed their intervention for sedentary populations, while Ridgel et al. [40] focused on PwP with depression. Three studies used hospital-based settings (outpatients) [39, 43] and three were based in the community [33, 41]. Two studies were carried out in USA [33, 40], one in Australia [42], one in Sweden [43], one in Canada [41], and one in the Netherlands [39].
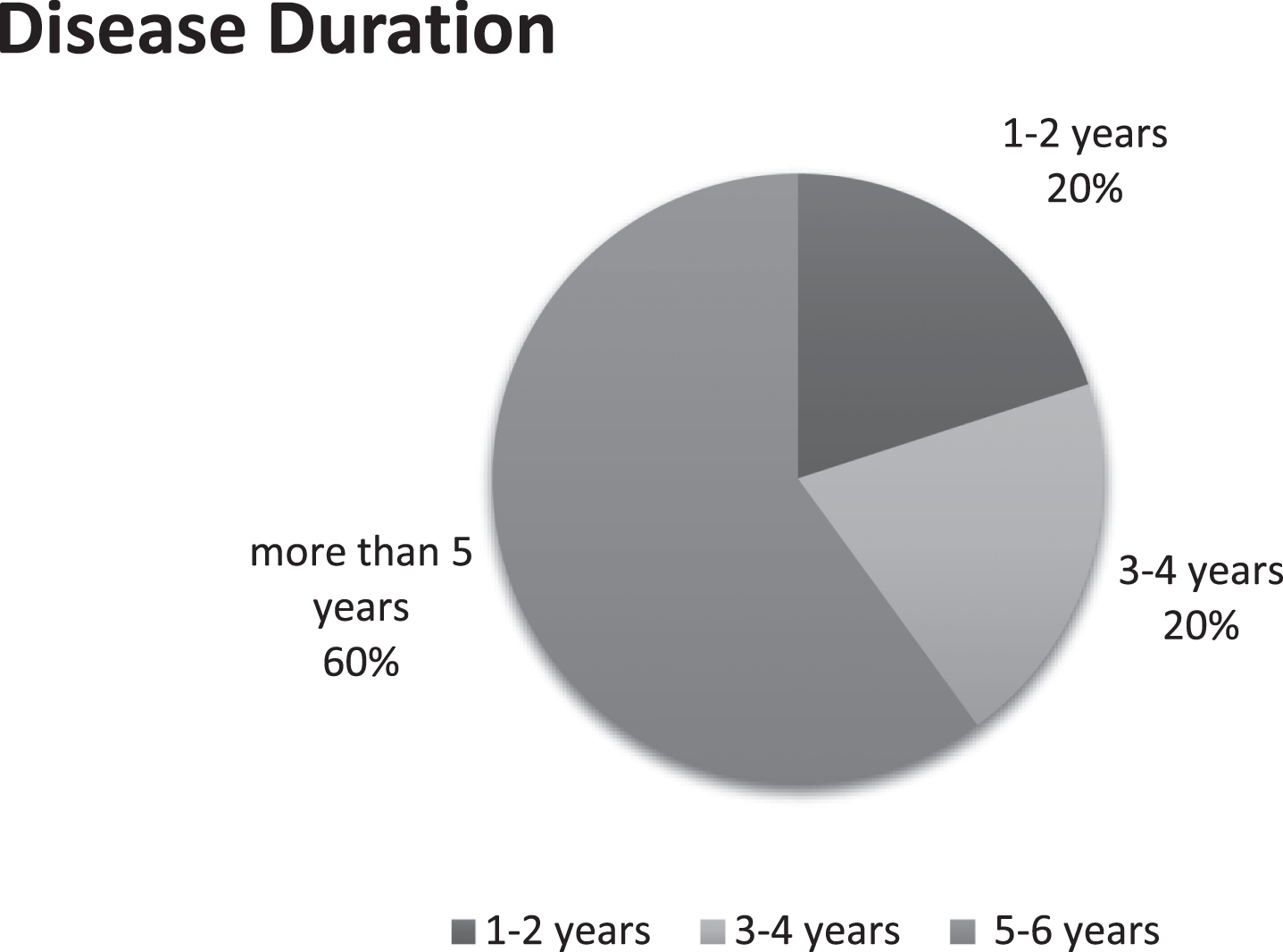
Average disease duration in years for participants in the included studies.
Description of interventions
All studies but one [33] provided both structured education and exercise as part of their intervention (Table 1). Two studies [33, 39] utilized goal setting, motivational interviewing & coaching in conjunction with education as part of their intervention and these were delivered on a one-to-one basis. Gruber et al. [41] also incorporated coaching and goal setting in their intervention which was delivered in a group format. One study [42, 44] in addition to exercise and education also provided a 20-min session for lunch and social discussion but its role in the intervention was not further assessed.
Control groups
Only two studies had a control group. Ridgel et al. [40] provided the same intervention to the control group but without a structured format. Instead, control group participants were advised to complete self-directed learning at their own time and exercise 3 times a week. van Nimwegen and colleagues [39] offered individual physiotherapy sessions to both intervention and control groups, however the control group did not receive coaching, motivation, and goal setting interventions to improve physical activity levels. The education in the control group also differed and lacked information on importance of exercise, but focused instead on safe movements in PD.
Risk of bias in the included studies
Selection bias was high in one of the RCT which did not specify if allocation of participants to groups was concealed [40]. Both RCTs had a high performance bias with difficulty demonstrating blinding of those delivering the intervention, partially due to the nature of the physical intervention [39] or self-management education [40]. The pre/post design interventions had high selection and detection bias. They all failed to demonstrate that the person conducting the assessment was blind to the participants group allocation. Moreover, with the exclusion of one study [42], pre and post studies had a small sample size to provide confidence in findings (Tables 3 and 4).
Risk of bias for pre/post intervention with no control group, NIH tool
Risk of bias for RCT, Joanna Briggs Institute Tool [38]
Effects of study interventions
Here we present the effect of the intervention (education and exercise) on the primary and secondary outcome measures used in each study (Table 1). Of the six included studies only two reported clear primary outcomes [39, 40]. Ridgel et al. had two primary outcome measures which included the number of exercise sessions attended by participants and the levels of physical activity utilizing the self-reported International Physical Activity Questionnaire (IPAQ). This study found no difference between the two groups in the number of exercise sessions in the gym (self-directed vs. structured group exercise session) but found the PA levels to be significantly lower in the control group at week 24 (
Studies demonstrated functional benefits as a result of their interventions including improvement in the physical functioning test maintained at 12-months post intervention [42], improvement in motor scores as measured by the UPDRS part 3, maintained up to 17 weeks post intervention [43], and improvement in functional reach [41]. A small effect in self-efficacy and controlled motivation and a medium effect in PA levels was detected in the study by Long [33], although this was a feasibility study and had a small sample size (
Description of educational interventions
Content
The topics covered in the physical health education intervention varied across studies (Fig. 3) but shared common themes such as the importance of exercise and PA [33, 42], goal setting [33, 41], overcoming barriers to exercise [33, 39], sleep and nutrition [40, 42], and social support [39–41, 43] (Fig. 3). Despite three papers specifically targeting their intervention to PwP in the early stage [33, 42], the education information provided in their intervention did not differ significantly from the content of the other studies included in this review, which were not stage specific. Interestingly, only one out of three studies aimed at newly diagnosed PwP included topics pertaining the importance of being an informed consumer and importance of communication with healthcare professionals [41] despite evidence emphasizing the importance of these topics in facilitating better self-management in PD [22, 23]. None of the included studies involved PwP in the development stage of their educational intervention.
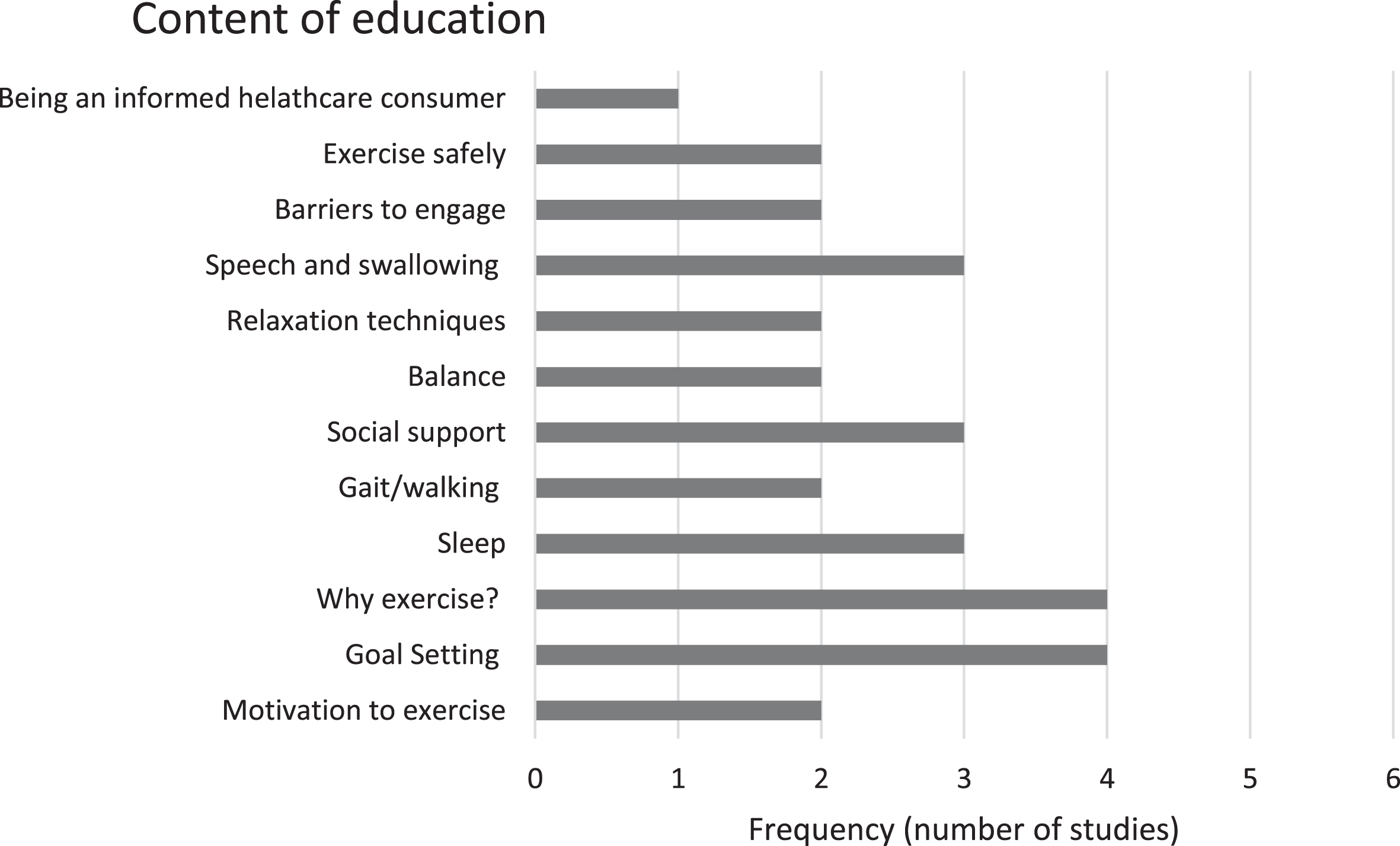
Content of education in the included studies.
Intervention development and theoretical framework
All but two studies [33, 40] developed their own educational content. In one study a coaching program designed to increase PA levels in people with Huntington’s disease was adapted for people with early-stage PD. This intervention was based on theoretical frameworks of behavioral change [33] (Fig. 4). The study by van Nimwegen et al. [39] was also based on similar theories and models of behavioral change. The Chronic Disease Self-Management Program first described by Lorig et al. and used with people with chronic conditions [45] was adapted by one study [40] for PwP who have depression. Another study [43] based their intervention on the Structure of Connection Model [46], which “focuses on interaction between a person and the environment which is an interaction among four domains of psychology, biology, sociology and physiology.” In one other study, principles of self-management such as goal setting and active problem solving were integrated in their intervention [41]. Whereas one intervention [42] was based on clinical practice with no mention of the underpinning theoretical basis.

Theoretical frameworks utilized within the included studies.
Delivery
Most of the education interventions were facilitated by a physiotherapist or nurse. Only one study utilized a specialist PD physiotherapist to deliver the intervention [39]. Two studies included a person with PD as a co-facilitator [40, 41] and one study used a multidisciplinary approach for their intervention delivery (Fig. 5). The main mode of delivery for the intervention was in-person sessions except for one study [33] which provided the option for virtual sessions and included telephone calls to follow up with and to motivate participants. All studies except one [41] provided a handout or workbook as part of the educational intervention.
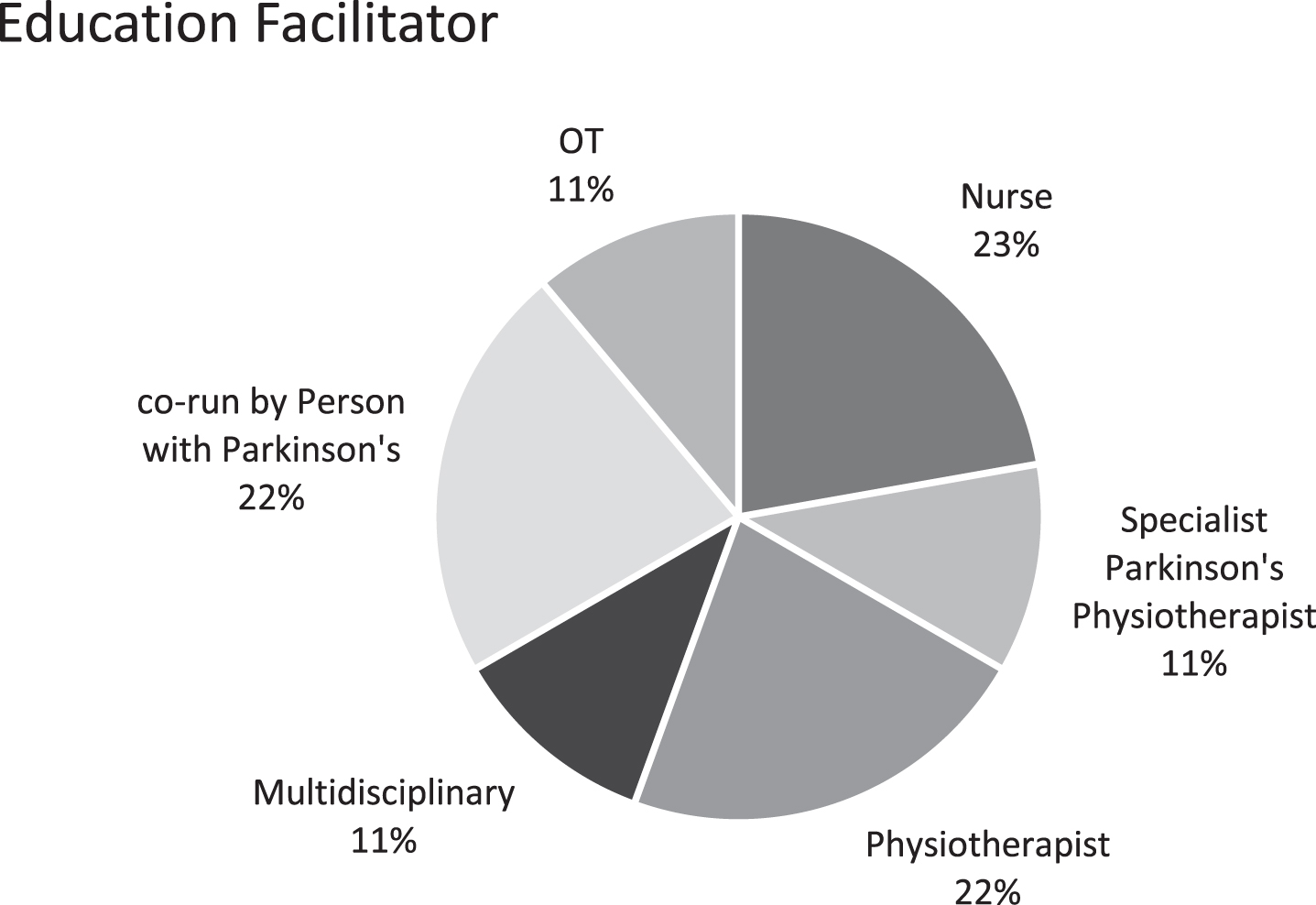
Education intervention facilitators in the included studies.
Duration and frequency
The duration of the education intervention varied hugely within the six studies; in one study the intervention was completed in 5 weeks [43], whilst one study [39] delivered the education intervention in 35 one-to one sessions per year over a 2-year period. The frequency also ranged from weekly to twice a week [41–43]. The length of the education session was 1 h in 4 out of 6 studies; in one study it was 30 min [39] per session, and in one other it was not specified [33]. The follow up time varied from 8 weeks to 104 weeks (Fig. 6).
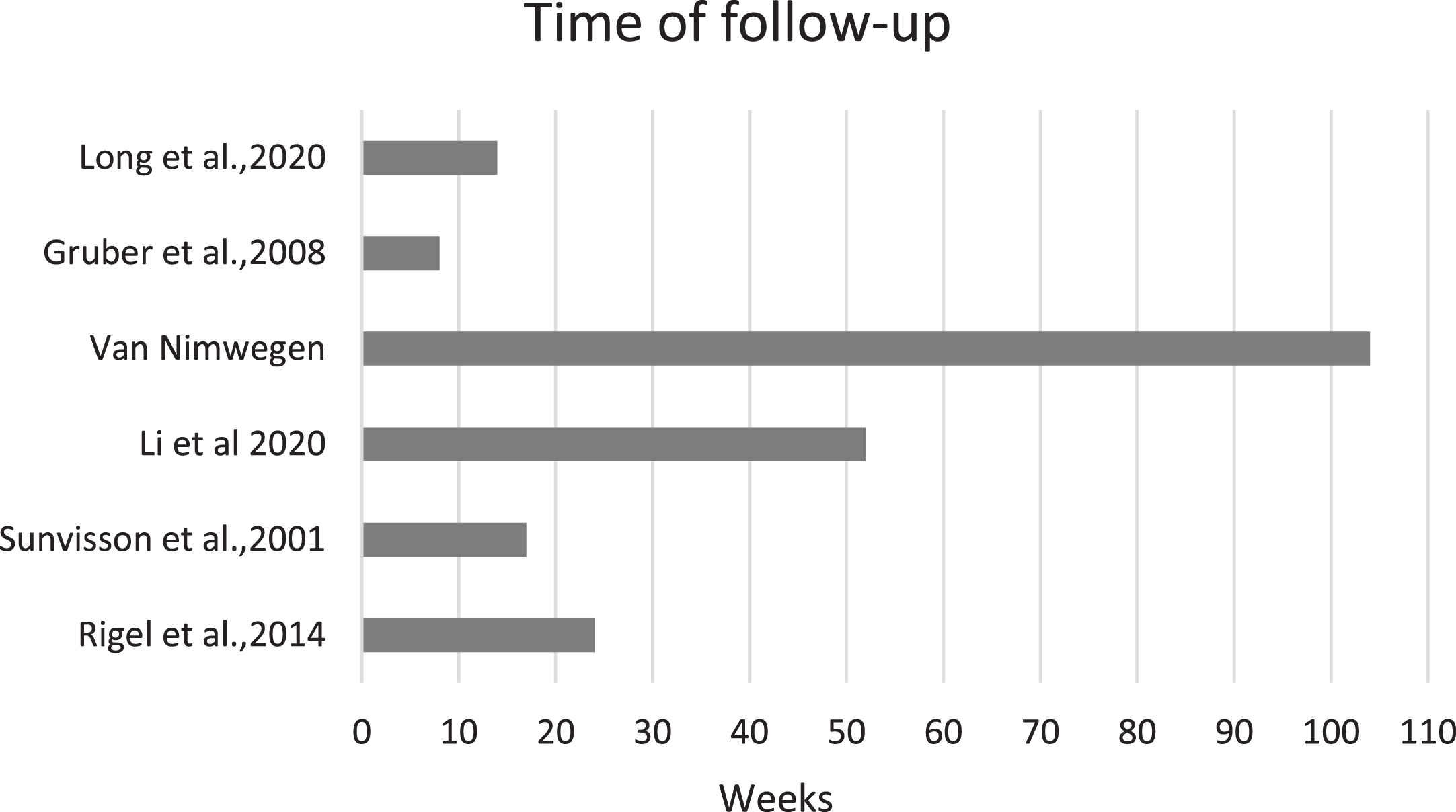
Time of follow up in the included studies.
DISCUSSION
This review focused on interventions that aimed to promote PA in PwP and to specifically examine the effectiveness of physical health education on physical and psychosocial outcomes.
It found a small number of interventions that aimed to alter PA behaviors in this population by providing strategies and education specific to exercise and PA, and identified a significant evidence gap with regard to the efficacy of education
Educational interventions are associated with changing attitudes, self-efficacy and beliefs which can impact positively health status [47–51]. More specifically, educational and psychosocial PA interventions in chronic conditions, including other neurodegenerative conditions, have been found to increase exercise outcome expectations and exercise self-efficacy [52–54]. Therefore, assessing changes in these behavioral and psychological factors following the educational intervention would have been appropriate to establish its effectiveness in the PD population. Education alone, if well-tailored, might drive participation in some PwP, including in the early stages of the disease trajectory, where PA and exercise have been shown to be beneficial. The evidence-base to inform practice is as of yet insufficient.
Three studies included outcome measures addressing factors related to behavior change. A feasibility study found a small effect in exercise self-efficacy and motivation post the education intervention [33]. Another study found significant improvement in the Chronic Disease Self-Management questionnaire [41] scores following education and exercise, whereas one study found no difference in the General self-efficacy scores following the intervention [55]. From these studies, it is difficult to draw any conclusion on the role of education on these behavioral factors. The studies did not consider the mechanism of action of the intervention, and how, or whether, individual components have additive or multiplicative effect on PA behaviors. Hence, delineating the impact of the education component from that of the exercise class was impeded.
Furthermore, each study employed different outcome measures to assess their intervention. There is a debate over the appropriative outcome measures to use when evaluating the impact of complex multi-component interventions [56], which could possibly be addressed when interventions are based on theoretical principles [57]. In the included studies, there was a variety of theoretical principles underpinning the interventions. Three out of six studies based their intervention in the Bandura’s self-efficacy theory and the transtheoretical model of behavior change. Self-efficacy, motivation and exercise outcome expectations have been strongly associated with PA behavior [30–32, 59], yet the effect of these mediating factors was not accounted for in the included studies.
Another significant strategy that drives motivation, as identified by PwP, is increased knowledge around exercise and its importance in the management of PD [60]. This is because PwP correlate knowledge with improved confidence and self-efficacy, as knowledge allows better preparation, leads to awareness and an increased sense of control [61, 62]. Indeed, gaining knowledge and expertise is the most prevalent reason for PwP attending a self-management program [61]. However, the relationship between education, self-efficacy and motivation is not fully explored in PD specific intervention.
Poorly explored is also the actual content of an education intervention. This review aimed to assess how and whether the content of education changed as the condition progressed. Although, three studies targeted their interventions to specific stages of PD [33, 42] or to specific group characteristics, i.e., PwP with depression [40] and sedentary PD population [39], there was no significant difference in the content of the education component across the different interventions. This highlights the disconnect between the request by PwP for education and information specific to their needs and stage [63] and what is delivered both in research and in practice [22, 64].
The content of education in the included studies although it varied, also shared some key topics such as the importance of exercise, goal setting and identifying barriers to participating in exercise. Studies that included these topics showed significant improvements in a range of outcomes following intervention and at follow up. Specifically, two studies [39, 42] included covered the importance of exercise, what is exercise and PA, personalizing exercises to participants’ characteristics. They found significant improvements in physical functioning tests such as 6 Minute Walk Test as well as PA levels as measured by digital activity monitors, with improvements being sustained over 1 and 2 years, respectively [39, 42]. One study also discussed the large effect of specific and tailored exercise education on participants’ exercise performance perception [33]. This is in line with previous literature which has indicated that specific skills, knowledge and targeted information around a person’s PD stage and needs, lead to improved sense of control and better self-management [60].
Self-management education programs aim to impart knowledge and skills that will enable and empower patients to become self-reliant in the day-to day management of their condition [65]. Hence, understanding the role of exercise in PD management, targeting exercise programs to PD-specific needs and knowing how to exercise and progress the exercises safely are identified by PwP as crucially important topics in interventions that aim to facilitate motivation and participation in PA [30, 66]. In contrast, interventions that promote physical activity without providing specific information on the role of the activity and its benefits have not been found effective, even when exercise programs were designed to be as individualized as possible [67], which underlines the role of education and knowledge in facilitating better physical self-management [68].
The effectiveness of health education interventions also depends on how well the person’s needs and preferences are addressed, in order to enable shared decision making and achieve person-centered care [69, 70]. However, none of the interventions in this review had involved a PwP in the development of the educational content. Co-designed methods in other conditions have been found effective and successful as they provide better awareness of concerns among stakeholders, increase the feeling of shared ownership of a product and lead to better health outcomes [71–73]. This stresses the need for interventions that are co-designed with PwP to address and represent their needs, perspectives and preferences around PA and exercise education.
Regarding the timing of such interventions, research recommends that education interventions aiming to increase physical activity and exercise levels need to be delivered as early as possible in the in Parkinson’s trajectory [35], given their potential to modify disease’s progression [74, 75]. Yet, participants in the included studies had an average disease duration of 6.6 years at the time of participation. Only one out of six studies was delivered to participants with a mean disease duration of 1.7 years. Results from this intervention suggested statistically improved self-efficacy levels in participating in exercise, managing stress, and communicating effectively with healthcare professionals [41]. These topics have been found to be directly linked with health related quality of life [76] and access to healthcare services around the time of diagnosis in PwP [77].
The duration of the education intervention in the identified studies varied from 5 weeks to 2 years, and the frequency varied from twice a week to weekly or monthly sessions, making it impossible to draw conclusion on what an effective duration and frequency might be. Appropriateness of the duration of an intervention might be also determined by the main objective of the study. For example, in a diabetes education intervention that aimed to promote knowledge, self-management and metabolic control, a longer intervention (> 24 weeks) produced larger effects for all 3 outcomes [78]. In behavior change interventions though, it has been suggested that more time has to pass before the psychological benefits of the intervention can be experienced [79]. Future studies should consider addressing the question of the intervention duration to PwP to understand their perspective from an acceptability point of view. From an effectiveness of intervention point of view, it is important that future studies follow up participants sufficiently for any long-term impact of the intervention to be observed. In this review, the follow-up period ranged from weeks to months, with only 2 studies following participants up to 1 year and 2 years respectively [39, 42].
Finally, regarding the mode of delivery of education intervention, all interventions were delivered face to face with one intervention including telephone calls in-between sessions to increase motivation [33]. It is worth mentioning that the healthcare and delivery of service interventions have been metamorphosed by the recent pandemic, driving a rapid expansion of telemedicine use [80]. Hence, many recent studies have utilized telehealth to deliver interventions and have showed that telehealth is a feasible and acceptable delivery mode to promote physical activity in PwP [67, 81]. This indicates the opportunity to be flexible in the delivery mode of future interventions, considering both the barriers (i.e., population with no access to internet) and the facilitators (i.e., younger population still in occupancy).
Despite the inability to determine the efficacy of education intervention in facilitating PA behavior, this systematic review has highlighted the lack of physical health education interventions promoting PA for PwP, especially within the first 2 years since diagnosis. Interestingly, this review identified 24 general self-management education interventions for PwP aiming to improve day to day management of PD and overall well-being. Topics on exercise and PA were mentioned in the intervention but specific information on content or duration was often missing. Qualitative systematic reviews on these self-management interventions have highlighted that PwP perceived the topics around exercise as one of the most important topics of the education content and have expressed their need for such content to be expanded [60, 63]. Hence it was surprising to identify only 6 studies focusing on exercise and PA education in PD.
Further studies are needed to explore the role of physical health education in facilitating healthy lifestyle in PwP. Interventions need to clearly present the rational and define the objectives for incorporating education as one of the behavior change strategies, choose outcome measures that mirror those objectives, and power the trials accordingly. This is essential to develop evidence-based recommendations on what constitute an effective physical health education program for PwP with regards to content, duration, frequency, and delivery.
CONCLUSION
In light of the building evidence on the role of PA and exercise in the management of PD, it is important to design interventions that translate those findings and implement them in practice as soon as diagnosis is received. Not only because of the well-researched benefits of PA, but also to provide evidence-led education interventions that improve the sense of control over the condition, keep hope and drive motivation in PwP. More theory-based interventions are needed with structured objectives and outcome measures that can accurately assess those objectives. The existing literature on the efficacy of education interventions in people with chronic disease indicates there are subgroups of responders and non-responders to education intervention [82]. There is a need to explore effectiveness of physical health education as a tool in facilitating physical activity behavior in PD. Lack of assessment poses the risk of potentially disregarding effective interventions or adopt ineffective approaches without the evidence. Furthermore, we need to work with PwP to design interventions that address their needs to ensure not only acceptability but also effectiveness of these interventions.
Strengths and limitations
This systematic review aimed to provide a comprehensive overview of the literature on physical health education interventions which aimed to promote PA and exercise behavior in the PD population. The search strategy used in this review was purposefully wide, including all education interventions at the screening phase, in an attempt not to miss out potentially relevant studies that might not have fully described their education intervention and focused on other elements of their intervention such as exercise. Data extraction was completed in a reliable method by three independent reviewers. The reviewers attempted to provide detailed information on the education content, frequency, and duration. When this information was not present, reviewers contacted the study authors to collect missing information. It is encouraged that future research should include the content of their education or information provision as this will improve the ability of replicability of potentially successful interventions.
A selection bias of studies written in the English language is a possible limitation of this systematic review, as it potentially excluded relevant studies not written in English. Finally, this review leaves the main objective of the study around effectiveness of education unanswered, which in turn highlights a substantial evidence gap, that future studies should address by utilizing education as a strategy to promote physical self-management in PD population.
Implications for practice
The results of this review provide further evidence that interventions that include information about PA and exercise classes for people with Parkinson’s can improve physical and psychosocial outcomes and may have a positive impact on improving exercise behaviors. The direct effect of education on knowledge, self-efficacy, health behavior and resource-use were not assessed in the included studies and therefore a clear conclusion cannot be drawn. Furthermore, the marked heterogeneity of the content of education intervention also impairs the ability to make recommendation on the specific content of physical health educational interventions. Yet, it was clear that interventions that included targeted information on the role and benefits of exercise in managing symptoms, overcoming obstacles, and setting meaningful goals translated in a significant improvement in the physical function outcomes in the short and the long-term. This review builds the case for a shift towards designing education interventions that are stage specific [33, 42] or addressed towards populations with similar activity levels [39] as this targeted approach might reflect better the needs and preferences of that population. Most interventions were delivered face to face, but given the recent pandemic, data supports that people have become more technology friendly and delivery of online interventions are feasible and acceptable.
A clear and transparent process of intervention evaluation should be adopted to allow for replicability of effective education interventions or provide indications for improvements or change in practice. Furthermore, there is a call for more robust studies to bridge both the identified gap in the literature and to address the expressed needs of PwP for tailored interventions to their condition. Input from PwP regarding their needs, preferences and priorities should be included in the design and development stage of an intervention [83], as they are the experts in their condition.
Footnotes
ACKNOWLEDGMENTS
The authors would like to thank the School of Clinical Medicine librarian at the University of Cambridge for their assistance with the search strategy. All authors have contributed to the work, agree with the presented findings. We would like to state that the work has not been published before nor is being considered for publication in another journal.
Ledia Alushi, University of Cambridge is supported by the National Institute for Health and Care Research (NIHR) Applied Research Collaboration East of England (NIHR ARC EoE) at Cambridge and Peterborough NHS Foundation Trust. The views expressed are those of the author[s] and not necessarily those of the NIHR or the Department of Health and Social Care.’
CONFLICT OF INTEREST
The authors have no conflicts of interest to declare.
