Abstract
Background:
Apathy represents a core neuropsychiatric symptom in Parkinson’s disease (PD). As there is currently no established effective treatment for apathy in PD, further investigating the biological origin of this symptom is needed to design novel therapeutic strategies. Among the multiple neurotransmitter alterations that have been associated with apathy, the involvement of extra-striatal dopaminergic degeneration remains to be fully explored.
Objective:
To investigate whether apathy in PD reflects increased dopaminergic degeneration extending beyond striatal regions.
Methods:
In the de novo PD cohort of the Parkinson’s Progression Markers Initiative (PPMI), we performed whole-brain I123-Ioflupane Single Photon Emission Computed Tomography (DAT-SPECT) analyses to characterize cross-sectional and longitudinal differences in DAT uptake associated with the presence of apathy. We also assessed the relationship between apathy and cognition in this sample, as apathy has been suggested to herald cognitive decline.
Results:
Apathetic PD patients (N = 70) had similar sociodemographic, clinical, and biomarker profiles compared to the non-apathetic group (N = 333) at baseline. However, apathy was associated with an increased risk of developing cognitive impairment after a four-year follow-up period (p = 0.006). Compared to non-apathetic patients, apathetic patients showed a widespread reduction of extra-striatal DAT uptake at baseline as well as an increased longitudinal loss of DAT uptake (corrected p < 0.05).
Conclusions:
Isolated apathy in PD is associated with extra-striatal dopaminergic degeneration. As this abnormal dopamine depletion was in turn related to cognitive performance, this might explain, at least partially, the increased risk of apathetic PD patients to develop cognitive impairment or dementia.
INTRODUCTION
Apathy is probably the core neuropsychiatric sym-ptom of Parkinson’s disease (PD) [1]. It can be observed from early disease stages, showing a preva-lence of up to 40% in this population [2]. Characterized by reduced motivation, apathy has a direct impact on the behavioral, emotional, and socials domains of PD patients [3]. Moreover, apathy has been consistently associated with cognitive decline in PD [4], leading to a significant deterioration of quality of life [5]. As there is currently no established effective treatment available for apathy in PD [6], a better understanding of its pathological mechanisms is needed to design novel therapeutic strategies.
In PD, frontal-executive deficits due to the inherent nigrostriatal dopaminergic depletion are expected to contribute to the development of apathy. Yet, few studies have specifically assessed the pathological mechanisms underpinning apathy in PD and, interestingly, they suggest the involvement of multiple neurotransmitter deficiencies. While several studies have shown that apathy is associated to striatal dopaminergic depletion [7, 8], a serotoninergic—rather than dopaminergic—deficit was associated with apathy in drug-naïve PD patients [9].
Accurate in vivo studies of neurotransmitter alterations in PD are challenging due to the complex logistics of PET imaging and the possible interferences of anti-parkinsonian drugs. Thus, most studies in this field have suffered from important limitations in terms of sample size and the use of PD medication. These limitations may hinder the use of stringent statistical multiple-comparison testing in neuroimaging, and also preclude the investigation of the predictive power of imaging indicators in longitudinal cohorts.
Of note, apathy reflects an underlying structural brain damage that extends beyond fronto-subcortical regions [10–12]. Nonetheless, the biological origin of these extra-striatal damage remains poorly understood, which is in part related to the fact that some imaging studies restricted their analyses to the striatum. Apart from dopaminergic and serotoninergic alterations, cholinergic degeneration has also been suggested to play a key role in PD [13–15]. In particular, it has been suggested that posterior-cortical cholinergic depletion in PD is strongly related to more aggressive forms of mild cognitive impairment (PD-MCI), ultimately leading to dementia (PDD) [16, 17]. Similarly, prefrontal cholinergic denervation through the anterior cingulate cortex could play a role in apathy in PD and other neurodegenerative diseases [18–21].
Disentangling whether the cortical damage associated with apathy in PD is driven by dopaminergic degeneration or rather by other pathological processes is important for the design of optimized treatments. Recently, our group showed that the extra-striatal signal of I123-ioflupane SPECT images provides valuable information about dopamine transporter (DAT) density in the cerebral cortex [22]. Specifically, cortical DAT uptake was not only altered in PD, but also progressively decreasing along the course of the disease. Furthermore, cortical DAT uptake alterations contributed to cognitive dysfunction independently of the inherent striatal dopamine depletion occurring in PD.
We hypothesize that apathy in PD reflects a rather widespread pattern of dopaminergic depletion across cortical and subcortical brain regions. To test this hypothesis, we characterize the underlying whole-brain topography of dopaminergic degeneration associated with the presence of apathy symptoms in PD. Using the Parkinson’s Progression Markers Initiative (PPMI) database [23], we studied both cross-sectional and longitudinal whole-brain SPECT-DAT differences in apathetic PD patients with respect to non-apathetic. We further explored the clinical translation of the observed imaging alterations. Importantly, given that the PPMI is a large and biomarker-rich project, we were able to discard the presence of non-dopaminergic neurodegenerative processes that could potentially act as confounding factors in this study.
MATERIALS AND METHODS
Sample and assessments
In the PPMI database we identified de novo PD patients with available I123-ioflupane SPECT images in NIfTI format from https://ida.loni.usc.edu. The de novo PD PPMI cohort includes recently-diagnosed patients (< 2 years) who were drug naïve at the baseline visit and did not belong to the genetic PPMI cohorts.
123I-Ioflupane SPECT acquisition was performed as described in the PPMI Imaging Manual: The target dose was 185 MBq or 5.0 mCi of DaTSCANtrademark, and subjects were pretreated with saturated iodine solution or perchlorate prior to tracer injection. Subjects were imaged 4±0.5 h following the injection.
Inclusion criteria for this study were the availability of SPECT-DAT imaging at baseline and at least the following clinical data: the part I and III of the Movements Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS), global cognitive evaluation assessed by the Montreal Cognitive Assessment (MoCA) and depression evaluation by the Geriatric Depression Scale (GDS-15). The availability of these data was required as depression and cognition are the main potential confounding factors when targeting the study of isolated apathy [24, 25].
De novo PD patients were classified as apathetic or non-apathetic based on the item 5 (apathy item) of the MDS-UPDRS-I. Patients scoring 0 in this item were considered as non-apathetics, and those scoring ≥1 as apathetic.
For these participants, we also considered the following clinical and biomarker data at baseline: anxiety scores from the State-Trait Anxiety Inventory (STAI), cognitive indicators including Symbol Digit Modality Test (SDMT), Letter-Number Sequencing (LNS), Benton Judgment of Line Orientation (BJLO), Hopkins Verbal Learning Test (HVLT), and Semantic fluency; cerebrospinal fluid (CSF) biomarkers including α-Synuclein, Aβ1–42, tau/ptau, and Apolipoprotein E (APOE) status. This set of biomarker variables allowed us to discard the presence of non-dopaminergic signs of neurodegeneration in the apathy group that could act as confounding factors. We also computed total L-dopa equivalent daily doses (LEDD) for each PD patient at the 4-year follow-up visit and considered the proportion of patients taking dopamine agonist in that visit.
The site investigator’s clinical diagnosis of PD-MCI/PDD at the four-year follow-up was used to explore the role of apathy at predicting the development of clinically-relevant cognitive decline in PD. This cognitive categorization represents a sound procedure where each site investigator was provided an instruction sheet that outlined how to assess cognitive decline, functional impairment, and general interpretation of cognitive tests to achieve this type of diagnosis [26].
Finally, healthy controls (HC) with available SPECT-DAT imaging at baseline were also identified in the PPMI database to assess the specificity of the potential apathy-related dopaminergic deficits in the PD group.
Further details concerning the set of considered scales, biomarker procedures, imaging acquisition and clinical cognitive characterization are available at http://www.ppmi-info.org/.
Neuroimaging methods
To be consistent with our previous work [22], we applied the same neuroimaging procedures. Briefly, using the Statistical Parametric Mapping (SPM12) software package, SPECT images were intensity-scaled by cerebellar uptake, spatially normalized to stereotactic MNI space, and smoothed using a Gaussian kernel of 8×8×8 mm full-width at half maximum.
Statistical analyses
To examine between-group differences in terms of sociodemographic, clinical, and biomarker data, we used t-test analyses for continuous variables and χ2 for categorical variables.
Regarding neuroimaging data, whole-brain voxelwise between-group comparisons were performed to compare PD-apathetic with PD-nonapathetic patients, both in terms of baseline DAT uptake and in terms of changes in DAT uptake over one year.
Age, sex, education, disease duration, total UPDRS-III, total GDS-15, and total MoCA scores at baseline were used as covariates of no interest. Additionally, if any of the considered clinical or bio-marker variables showed significant differences between apathy groups, we repeated the imaging analyses including them as covariates.
To be consistent with our previous work [22], only clusters surviving voxelwise p < 0.01 and family-wise error (FWE) correction for multiple comparisons (cluster-wise p < 0.05 using Random Field Theory as implemented in SPM) were considered significant.
We also investigated the clinical implications of the observed imaging alterations using a set of exploratory analyses. For that, we first quantified average DAT uptake at the identified brain clusters showing significant between-group differences. Then, using Pearson’s correlation coefficients, we studied the association of these tracer uptakes with motor, cognitive and depression scales in the PD group, for which a p < 0.05 was considered significant. We also computed ROC curves to assess the predictive power of these imaging indicators at predicting cognitive impairment or dementia over a four-year follow-up period. Finally, we aimed to confirm that DAT uptakes in these regions were indeed altered in PD patients with respect to healthy controls with similar sociodemographics, for which t-tests at p < 0.05 were considered significant.
RESULTS
Table 1 summarizes the sociodemographic, clinical, and biomarker data of PD participants. APOE data was missing for 9% of participants, CSF tau/ptau/α-syn for 4%, CSF Aβ1–42 for 5%, and cognitive tests (except MoCA) and STAI for 0.9%.
Sample characteristics at baseline
Values are expressed as mean±standard deviation or percentage. MDS-UPDRS-III, total motor score for the Unified Parkinson’s disease rating scale; GDS, Geriatric Depression Scale; MoCA, Montreal Cognitive Assessment (MoCA); HVLT, Hopkins Verbal Learning Test; LNS, Letter-Number Sequencing; SDMT, Symbol Digit Modality Test; BJLO, Benton Judgment of Line Orientation. *p≤0.05.
At baseline, apathetic PD patients showed a similar clinical and sociodemographic profile compared to the non-apathetic group, with the only significant difference being that non-apathetic patients showed a very mild increase in total STAI scores. Up to 80% of these participants had available a clinical cognitive categorization at four-year follow-up. In these patients, apathy at baseline was significantly associated with the development of PD-MCI/PDD at follow-up: 19% of PD-MCI/PDD in the non-apathetic group versus 37% among PD-apathetic patients (p = 0.006 χ2).
Figure 1 shows the cross-sectional and longitudinal whole-brain differences in DAT uptake between PD-apathetic and non-apathetic patients, controlling for age, sex, education, disease duration, UPDRS-III, MoCA, and GDS-15 scores at baseline. Longitudinal SPECT-DAT imaging was available for 81% of PD participants (271 apathetic versus 58 non-apathetic).
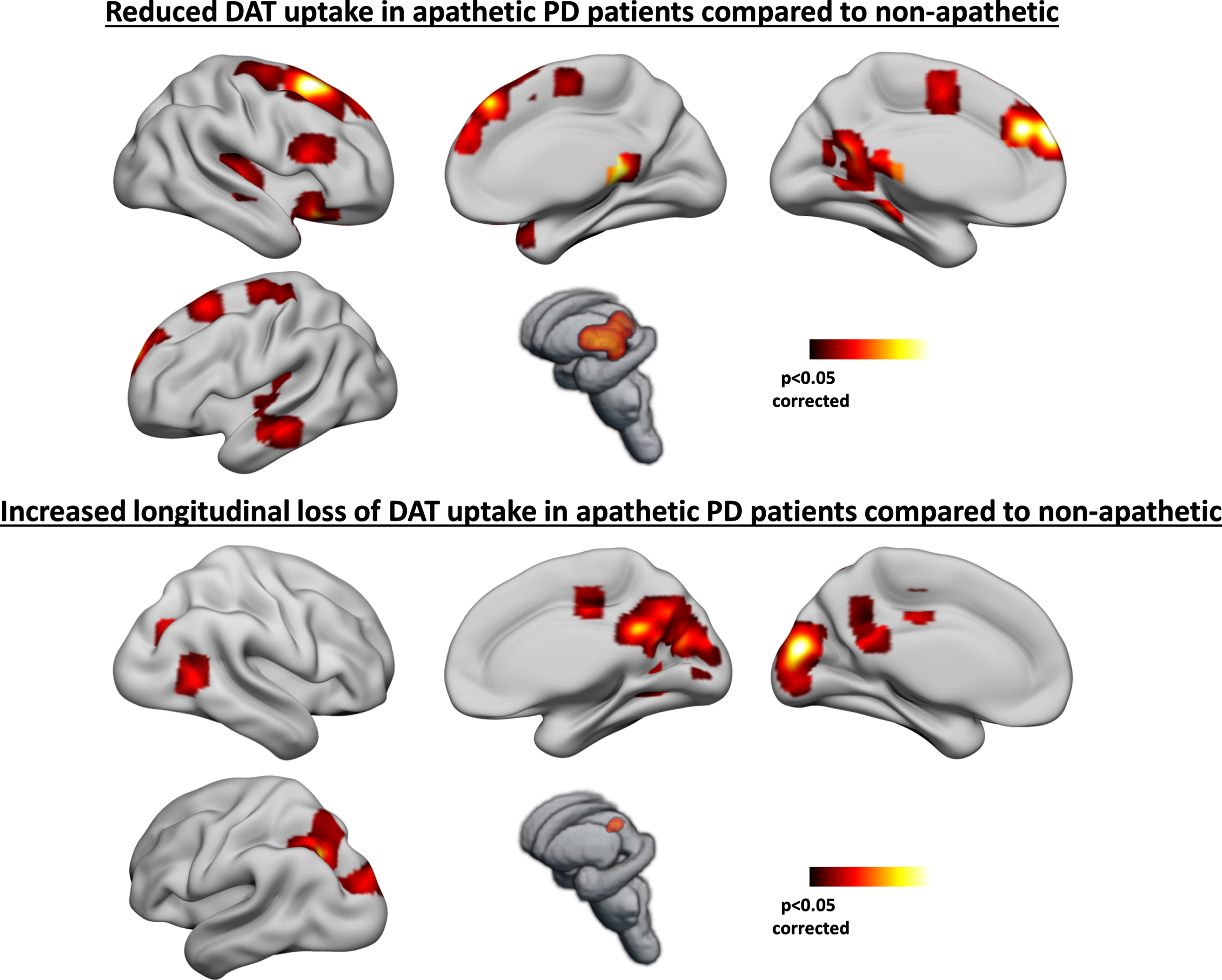
Brain regions showing lower DAT uptake (top) and increased longitudinal loss of DAT uptake (bottom) in apathetic PD patients compared to non-apathetic. Age, sex, education, disease duration, UPDRS-III, MoCA, and GDS-15 scores at baseline were included as covariates of no interest. Only clusters surviving multiple comparison correction were considered significant.
At baseline, apathetic PD patients showed reduced DAT uptake in the following regions: midbrain, thalami, medial frontal gyri, posterior-cingulate cortex (PCC), precuneus, right orbitofrontal cortex (OFC), right insula, left supplementary motor area (SMA), superior frontal gyri, and left middle temporal gyrus. Apathetic PD patients also showed increased longitudinal loss of DAT uptake over one year in the following regions: PCC/precuneus, cuneus, cingulum middle, left inferior parietal, occipital cortex and right calcarine sulcus. Similar results were obtained when including anxiety scores as a covariate in the voxelwise statistical models.
The following significant correlations between DAT uptake in these regions and clinical indicators were found. Total UPDRS-III scores inversely correlated with average DAT uptakes in all of these regions (p < 0.05) except for the PCC/precuneus and occipital clusters. No significant correlations between DAT uptake in these clusters and total GDS-15 or STAI scores were found. Regarding cognitive indicators, total MoCA scores correlated with DAT uptake in the PCC and middle cingulum clusters (p = 0.027 and p = 0.010, respectively), SDMT scores correlated with midbrain, OFC and PCC uptakes (p = 0.006, p = 0.027, p = 0.041, respectively), LNS scores correlated with midbrain and middle cingulum uptakes (p = 0.032 and p = 0.039, respectively), HVLT scores correlated with DAT uptakes in PCC/Precuneus, inferior parietal, calcarine, cingulum middle, OFC and midbrain (p < 0.001, p = 0.001, p = 0.019, p = 0.044,p=0.003, p = 0.002, respectively), BJLO scores correlated with inferior parietal DAT uptake (p = 0.019), and Semantic Fluency scores correlated with DAT uptake in all these regions of interest regions (p < 0.05) except for the superior frontal and calcarine clusters.
Moreover, DAT uptake in the midbrain, OFC, right insula and middle cingulum clusters significantly predicted the development of clinically-significant cognitive impairment or dementia over a four-year follow-up period: AUC = 0.66 (p < 0.001), AUC = 0.64 (p < 0.001), AUC = 0.65 (p < 0.001), and AUC = 0.60 (p = 0.010), respectively. Lastly, in a group of 192 healthy controls with similar sociodemographics (age 60.9±11.3 years, 34% female, education 16.0±3 years), we confirmed that DAT uptake in all of the brain regions involved in this study was significantly lower in the PD group compared to HC (p < 0.001), except for the left inferior parietal cluster (p = 0.73).
No significant differences were found in LEDD at the 4-year follow-up visit between apathetic and non-apathetic patients (648±472 versus 655±543, p = 0.93), and the proportion of patients taking dopamine agonists was not significantly different either (33% 43%, p = 0.13). The 4-year LEDD did not differ significantly either between those patients showing cognitive impairment at follow-up and those showing normal cognition (709±527 versys 640±535, p = 0.33). However, there was a significantly lower proportion of cognitively-impaired patients at follow-up taking dopaminergic agonists (30%) compared to the cognitively-unimpaired (45%), p = 0.03.
DISCUSSION
We found that apathy in de novo and drug-naïve PD patients reflects extra-striatal dopaminergic depletion. The dopaminergic integrity in the set of compromised cortico-subcortical regions in apathetic patients correlated, in turn, with cognitive performance. Importantly, the observed dopaminergic deficiency in apathetic PD patients was associated with an increased risk of developing PD-MCI or PDD after a four-year follow-up period.
Apathetic de novo PD patients had a very similar clinical and sociodemographic profile compared to non-apathetic. This reinforces that isolated apathy is associated with early cognitive decline in PD, independently of the basal cognitive or depressive symptoms. After confirming the clinical motivation of this study, we investigated the dopaminergic contribution of apathy in PD.
Apathy was associated with reduced cross-sectional DAT uptake in frontal, temporo-parieto-occipital, midbrain, and thalamic regions. Observing brain alterations extending beyond fronto-striatal regions was expected, given the current understanding of apathy in PD as a complex multidimensional syndrome that probably reflects widespread brain alterations [10, 27]. The added value of the present study lies in observing temporo-parieto-occipital dopaminergic alterations associated with apathy in PD. This suggest a role of the extra-striatal dopaminergic system in this context, which could coexist with the involvement of other neurotransmitter systems such as the cholinergic system [27]. Moreover, apathy was also associated with an increased longitudinal loss of extra-striatal DAT uptake, suggesting that apathy reflects the presence of a more severe form of active dopaminergic degeneration.
Interestingly, in those brain regions showing apathy-related reduced DAT uptake, the following cognitive-imaging associations were observed: DAT uptake in frontal regions correlated with frontal-dominant cognitive tests such as the SDMT or LNS; posterior-cortical-dominant tests such as the HVLT and BJLO correlated with DAT uptake mainly in parieto-occipital regions; and semantic fluency, known to involve fronto-temporo-parietal preservation, correlated with DAT uptake in both frontal and posterior-cortical regions. Moreover, in line with previous works [22], global cognition assessed by total MoCA scores correlated with PCC/Precuneus integrity. This region, where dopamine receptors have been identified [28], represents the key node of the default mode network, which is known to play a key role in cognition [29]. Importantly, depression scores appeared to be unrelated to the observed DAT alterations, reinforcing that apathy—but not depression—may be reflecting increased dopaminergic degeneration in PD.
However, in contrast to other works [7], we did not find striatal DAT uptake alterations associated with apathy in this sample. This may be due to differences in sample characteristics or to the use of distinct image analysis pipelines (e.g., voxelwise whole-brain vs striatal-restricted ROI approach, the choice of reference region, the inclusion of motor/cognitive/depression scores as covariates, etc.). Alternatively, it could be argued that, at least in very early disease stages, apathy would not be a mere direct consequence of striatal dopamine depletion. If that was the case, virtually all PD patients should show at least mild apathy symptoms when compared to healthy controls, given the inherent striatal dopaminergic damage occurring in this disorder. Similarly, apathy symptoms would improve after dopaminergic treatment in parallel to motor symptoms, which is not the case for most apathetic patients [6]. Therefore, in fact, it is plausible that the involvement of additional extra-striatal dopaminergic degeneration is needed to develop apathy symptoms in PD.
Those patients showing apathetic symptoms at the baseline visit did not show significant PD medication differences at the 4-year follow-up visit. Interestingly, however, whereas total LEDD was not significantly different between cognitively-impaired and cognitively-unimpaired patients at follow-up, there was a lower proportion of patients taking dopamine agonists in the former group. This finding could be related to the fact that dopamine agonist may have been used more cautiously in patients showing signs of cognitive dysfunction or minor hallucinations [30]. Alternatively, this finding could be attributed to a suggested protective effect of dopamine agonists on cognitive functions [31, 32]. Further clinical research is needed to appropriately address this potential association.
Taken together, these cognitive-imaging associations suggest that the observed dopaminergic alterations in apathetic PD patients may play an important role in their future cognitive disturbances. In fact, DAT uptake in a subset of the altered brain regions predicted a PD-MCI/PDD diagnosis after a four-year follow-up period. From a biological perspective, the fact that apathetic PD patients in this sample did not show signs of increased non-dopaminergic neurodegeneration at baseline is of utmost importance. If we had observed differences in CSF biomarkers or APOE status in apathetic patients, these could have acted as critical confounding factors, as they have been associated with both apathy and cognitive decline in PD [26, 33].
Finally, the fact that posterior-cortical DAT uptake did not predict the longitudinal development of PD-MCI/PDD may be consistent with the dual syndrome hypothesis for cognitive decline in PD. This hypothesis distinguishes two pathological pathways jointly leading to cognitive disturbances in PD [16]: dopaminergic-mediated fronto-striatal alterations and visuospatial alterations due to posterior-cortical cholinergic degeneration. In the light of our results, it could be interpreted that it is the cholinergic -and not the dopaminergic- posterior-cortical component which drives PD-MCI/PDD onset. Overall, given the complex nature of cognitive deficits in PD, further research is needed to better delineate the contribution of each pathological pathway in this clinical context.
Strengths of this study include the use of a large and biomarker-rich sample of well-characterized drug-naïve PD patients with available baseline and longitudinal DAT-SPECT data. Limitations of this work include the use of low-resolution SPECT images to quantify dopaminergic depletion. Additionally, we could not control for the effects of non-selective 123I-Ioflupane binding to serotonin or noradrenaline transporters. Finally, the use of Part 1.5 of the MDS-UPDRS to identify apathetic patients is clearly suboptimal. Even though this instrument was confirmed to be an easy-to-assess and suitable instrument for detecting apathy syndrome in PD [34], the use of more precise and specific instruments for apathy assessment and further exploring the imaging and clinical characteristics of apathy subtypes [35] would have clearly allowed a better characterization of this core neuropsychiatric PD feature. Nonetheless, it is noteworthy that an item-based identification of apathy symptoms was able to identify a subgroup of recently-diagnosed and drug-naïve PD patients with increased risk of cognitive decline, who also show significant cross-sectional and longitudinal neuroimaging alterations.
To conclude, apathy in PD appears to be driven by the presence of extra-striatal dopaminergic degeneration. As this cortico-subcortical dopamine depletion was in turn related to cognitive deficits, this might explain, at least partially, the increased risk of apathetic PD patients to develop cognitive impairment and dementia.
Footnotes
ACKNOWLEDGMENTS
This work was partially supported by CERCA and CIBERNED funding, and grants from la Marató de TV3 (grants #2014/U/477 and #20142910) and Fondo de Investigaciones Sanitarias (FIS) from the Instituto de Salud Carlos III (ISCIII) and Fondo Europeo de Desarrollo Regional (FEDER) grants #PI15/00962 and #PI18/01717.
Data used in the preparation of this article were obtained from the Parkinson’s Progression Markers Initiative (PPMI) database (http://www.ppmi-info.org/data). For up-to-date information on the study, visit ![]() .
.
PPMI, a public-private partnership, is funded by the Michael J. Fox Foundation for Parkinson’s Research and funding partners, including Abbvie, Acurex, Allergan, Amathus, ASAP, Avid Radiopharmaceuticals, Bial Biotech, Biogen, BioLegend, Bristol-Myers Squibb, Calico, Celgene, Cerevel, Coave, Dacapo, Denali, Edmond J Safra, 4D pharma plc, GE Healthcare, Genentech, GlaxoSmithKline, Golub capital, Handl Therapeutics, insitro, janssen, Lilly, Lundbeck, Merck, Meso Scale Discovery, Neurocrine, Pfizer, Piramal, Prevail Therapeutics, Roche, Sanofi Genzyme, Servier, Takeda, Teva, UCB, Vanqua bio, Verily, Voyager and Yumanity.
CONFLICT OF INTEREST
None of the authors of this manuscript have any potential conflict of interest related to the content of this study.
AVAILABILITY OF DATA AND MATERIAL
CODE AVAILABILITY
The complete neuroimaging pipeline used in this work is available upon request.
