Abstract
Background
Apathy, defined as a quantitative reduction in goal-directed activity, is a non-motor manifestation that can be present in Parkinson's disease (PD). It seems to be a risk factor for conversion to dementia (PDD) in this population. Amyloid-β deposition also predicts progression to PDD.
Objective
We aimed to investigate whether PD patients with apathy showed higher amyloid burden than those without, as well as how these features may influence the rate of progression to dementia.
Methods
We conducted an observational cross-sectional and longitudinal study. Forty-eight PD patients were recruited, including 20 with apathy and 28 without it according to the Starkstein Apathy Scale. They underwent clinical and cognitive evaluations and [18F]-Flutemetamol PET. The neuropsychological assessment was repeated after 3 years. The predictive value of apathy and amyloid burden for conversion was assessed via logistic regression. Longitudinal trajectories across neuropsychological tests were modeled with linear mixed-effects.
Results
Patients with apathy showed worse performance on several cognitive domains. Using disease duration and global cognition Z-score as covariates, amyloid burden was higher in apathetic vs. non-apathetic patients, mainly in the frontal and temporal cortices. Non-apathetic patients did not have regions with higher amyloid burden in comparison with apathetic patients. After 3 years’ follow-up, the conversion rate to worse cognitive state was significantly higher in apathetic (47.4%) vs. non-apathetic (12.0%) patients (p < 0.05). Logistic regression showed that amyloid burden, but not apathy, predicted 3-year cognitive conversion (χ² = 9.95, p < 0.05).
Conclusions
Apathetic patients exhibit greater amyloid burden and higher cognitive deterioration over time than their non-apathetic counterparts.
Plain Language Summary
People with Parkinson's disease often show motivation loss (apathy) and experience cognitive difficulties. In this study, we aimed to investigate whether Parkinson's patients who show apathy also exhibit more amyloid protein accumulation in their brains than those without apathy, and to see how both apathy and brain amyloid levels affect the chance of developing dementia over three years. We followed 48 patients without dementia—20 with apathy and 28 without—measuring their apathy scores and their brain amyloid by PET scan. We tested their cognitive abilities via neurocognitive evaluations at baseline and again after three years. At baseline, patients with apathy showed worse performance on several cognitive domains and greater amyloid deposits in the frontal and temporal cortices. After three years, 47% of the apathetic group and 12% of the non-apathetic group had declined to mild cognitive impairment or dementia. However, when analyzing the relationship between apathy, amyloid deposition, and cognitive decline, only higher amyloid levels consistently predicted cognitive worsening. Apathy alone, did not add additional predictive power. These findings suggest that while apathy accompanies cognitive decline, it is the underlying amyloid accumulation that serves as the strongest early warning sign for dementia risk in Parkinson's disease. This study provides insights into potential pathological substrates and therapeutic implications and highlights the possibility of early interventions focusing on treatments to slow or prevent cognitive decline in this population.
Introduction
Parkinson's disease (PD) is the second most common neurodegenerative disease after Alzheimer's disease (AD). 1 It is typically characterized by degeneration of nigrostriatal dopaminergic neurons and misfolded α-synuclein (Lewy bodies) in surviving neurons. 2 In addition to the classic motor signs, non-motor manifestations in PD affect most patients. In this regard, cognitive decline and neuropsychiatric manifestations such as apathy are common. 3
According to the International Society for Central Nervous System Clinical Trials Methodology (ISCTM), apathy is defined as a quantitative reduction in goal-directed activity compared to the patient's previous level of functioning, affecting at least two of the three dimensions of apathy (behavior/cognition, emotion, and social interaction). 4 It has a reported prevalence of 40% among PD patients and is often associated with greater disease severity and lower cognitive scores,5,6 more frequently in executive functions, 7 as well as lower quality of life. 8
Furthermore, apathy occurs more frequently in PD patients with dementia (PDD) (54%) and seems to be a risk factor for conversion to dementia in this population.6,7,9,10 Additionally, the presence of dementia has been identified as a risk factor for subsequent development of apathy. 11
Apathy in PD has been largely related to dopaminergic denervation within the mesocorticolimbic pathway.12,13 In fact, some hypotheses suggest that motivational alterations in PD may represent a dopaminergic continuum, ranging from “hypodopaminergic” behaviors, such as apathy, anxiety, and depression, to “hyperdopaminergic” behaviors, like impulse control disorders. 14 However, non-dopaminergic systems such as serotoninergic, 15 noradrenergic,16,17 or cholinergic18,19 might also contribute.
Apathy is also common in AD, with a 5-year prevalence over 70% and significant prognostic value. 20 It is also associated with faster progression of cognitive, functional, and emotional impairment in these subjects.20,21
In AD the presence of apathy is negatively correlated with cerebrospinal fluid (CSF) amyloid-β (Aβ)42 concentrations 22 and has been associated with amyloid deposition, especially in the prefrontal cortex.23,24 Furthermore, the relationship between apathy and amyloid deposition has been described in other populations, such as late-life depression patients, where higher amyloid burden in the anterior cingulate cortex has been associated with increased apathy independent of depressive symptoms, 25 or preclinical populations, where frontal amyloid burden is a risk factor for the development of apathy. 26
AD-related pathology, including Aβ and tau deposition, as well as Lewy body pathology, independently predicts progression to PDD.26–28 Furthermore, AD pathology is the most common age-related pathology in individuals with PD and cognitive decline 26 and has been hypothesized to exacerbate Lewy body pathology in PD. 28 Additionally, amyloid burden in PD has been associated with a shorter interval to dementia.29,30
Apathy has been identified as a predictor of dementia in PD, and amyloid pathology has been linked to apathy in AD, suggesting that amyloidopathy may serve as a potential underlying mechanism for apathy in PD. This hypothesis may also explain the lack of therapeutic benefiter even worsening observed with dopaminergic or serotonergic drugs in some cases. 31
Moreover, recent findings suggest that apathy scores correlate with β-amyloidopathy in PD patients at risk for dementia, particularly in brain regions such as the inferior frontal cortex, cingulate cortex, and nucleus accumbens. 32
However, amyloid burden in apathetic PD subjects (with pathological cut-off scores) and its role in cognitive progression over time are not yet known. Thus, we aimed to investigate whether PD patients with apathy showed higher amyloid burden than those without apathy, as well as how these features may influence the rate of progression to dementia.
Methods
Subjects
We conducted an observational, cross-sectional, and longitudinal study. Forty-eight PD patients were recruited at HM CINAC, Hospital Universitario HM Puerta del Sur, between 2017 and 2021. Patients were diagnosed with PD according to the United Kingdom Parkinson's Disease Society Brain Bank Criteria, 33 with a disease duration of less than 10 years and an age of onset >50 years. They were excluded if they presented with severe cerebrovascular disease, metabolic disease, active oncological processes, severe depression according to the Geriatric Depression Scale (GDS-30 > 20), 34 alterations on magnetic resonance imaging indicating other causes of cognitive impairment, or previous neurosurgical interventions. Subjects with dementia at baseline (PDD) were excluded according to the Movement Disorder Society (MDS) criteria. 35 The study was approved by the HM Puerta del Sur Ethics Committee (16.09.0942E1-GH), and all subjects provided informed consent prior to participation.
Clinical and neuropsychiatric assessment
The following clinical and demographic data were collected for each patient: age, gender, disease duration, educational level, disease severity (measured using the MDS-Unified Parkinson's Disease Rating Scale, MDS-UPDRS, part III [motor score], and Hoehn and Yahr stage), levodopa equivalent daily dose (LEDD), depression scores (using the 30-item Geriatric Depression Scale), and apathy scores. All assessments were conducted at baseline during the on-medication state.
Apathy was evaluated at baseline using the Starkstein Apathy Scale, 36 a shortened 14-item version scored on a 4-point Likert scale, derived from the preliminary Apathy Evaluation Scale (AES).37,38 This scale, specifically designed to assess apathy in patients with Parkinson's Disease (PD), evaluates impairments in the behavioral, affective, and cognitive components of motivation. It has shown reasonably good psychometric properties, 39 with a cut-off score of 14 that effectively classifies patients as apathetic or non-apathetic. 40
Neuropsychological evaluation
Patients underwent neuropsychological assessment at baseline and at 3-year follow up to evaluate the longitudinal progression of cognitive status. The neuropsychological battery included tests in the following domains: attention (Digit Span Forward, Trail Making Test A [TMT-A]); executive function (Stroop Test inhibition time, Trail Making Test B [TMT-B]); language (Boston Naming Test and semantic fluency); memory (Consortium to Establish a Registry for Alzheimer's Disease [CERAD] delayed recall for verbal memory, and the Wechsler Memory Scale for visual memory); and visuospatial function (Judgment of Line Orientation [JLO] and Silhouettes from the Visual Object and Space Perception Battery [VOSP] for the visuospatial domain). Although the MDS Task Force 41 initially classified semantic fluency as an executive function, later recommendation and validation studies have suggested that animal naming should be represented within the language domain, 42 but also based on expert consensus, semantic fluency is often the best available measure of language. 43 For each participant and cognitive test, we calculated a z-score based on the Neuronorma Project normative dataset44–48 or, in the case of CERAD, from a normative dataset from healthy subjects. 49 An overall global cognitive z-score was then computed by averaging all test scores, allowing it to be used as a moderating variable.
A diagnosis of mild cognitive impairment (MCI) was based on the MDS Level II criteria, 41 which indicate MCI when performance on at least two tests within one or more cognitive domains falls 1.5 standard deviations below expected levels, calculated using z-scores. PDD patients were diagnosed according to the MDS criteria, 35 which require deficits in at least two of the four core cognitive domains (attention, memory, executive, and visuo-spatial functions), demonstrated through clinical and cognitive assessment, and sufficiently severe to interfere with normal functioning. Patients who did not meet the diagnostic criteria for PD-MCI or PDD were classified as cognitively normal (PD-CN).
According to these criteria, patients were classified at baseline as being cognitively normal (PD-CN), having mild cognitive impairment (PD-MCI), or being demented (PDD). Patients underwent a 3-year follow-up evaluation to assess the progression of their initial cognitive status. They were classified as cognitively stable or converters (from PD-CN to PD-MCI/PDD or from PD-MCI to PDD). The Pfeffer Functional Activities Questionnaire 50 was administered to assess functional decline in these patients.
Neuroimaging assessment
At baseline, all subjects were scanned on a hybrid 3T mMR-Biograph system (Siemens Healthcare, Erlangen, Germany) using [18F]-Flutemetamol. After intravenous injection of 185 MBq of the radiotracer, there was a 90-min uptake period to estimate amyloid deposition. Images were reconstructed with an ordered subset expectation-maximization algorithm, incorporating 4-mm full-width half-maximum 3D Gaussian kernel smoothing. Attenuation correction was based on 4-compartment MR-derived maps generated from a dual-echo Dixon sequence (repetition time: 3.6 ms; echo time: 1.23–2.46 ms). The reconstructed images had a matrix size of 128 × 128 × 127 and a voxel size of 2.8 × 2.8 × 2.03 mm³. Simultaneously with each PET acquisition, a 3D T1-weighted magnetization-prepared rapid acquisition gradient echo (MPRAGE) sequence was obtained.
The SPM12 software package (The Wellcome Centre for Human Neuroimaging, UCL Queen Square Institute of Neurology, https://www.fil.ion.ucl.ac.uk/spm/software/spm12/) running in MATLAB R2021b was used. PET images were processed with the following steps: (1) nonlinear spatial normalization of T1-weighted volumes to Montreal Neurological Institute (MNI) reference space and segmentation into gray matter (GM), white matter, and CSF; (2) partial volume effect (PVE) correction, applied to match the spatial resolution of the PET data to account for its confounding effects; and (3) spatial normalization to reference space using nonlinear transformations, followed by isotropic spatial smoothing with an 8-mm Gaussian kernel. The cerebellar cortex was used as reference region to calculate voxelwise standardized uptake of the brain regions. The global amyloid load was also quantified, as standardized uptake value ratio (SUVR).
Global amyloid load was computed as a volume-weighted average uptake across 116 cortical and subcortical regions using the automated anatomical labeling (AAL) atlas, 51 standardized against the whole cerebellar cortex as the reference regions, generating standardized uptake value ratios (SUVR). White matter and CSF probability maps from SPM segmentation were employed as masks to mitigate extra-cortical spill-in effects.
Statistical analysis
All statistical analyses were conducted using STATA software, version 18 (StataCorp, College Station, TX) and the SPM12 software package. Demographic, clinical, and neuropsychological variables, assessed at both baseline and follow-up, were compared between apathetic and non-apathetic patients using a two-sample t-test with a significance threshold of p < 0.05. When normality or homoscedasticity assumptions were not met, the Mann-Whitney U test and Welch t-tests were applied, respectively. Multiple comparisons were adjusted using the False Discovery Rate (FDR) via the Benjamini–Hochberg procedure. This method was chosen because it effectively controls the rate of false positives while preserving statistical power, which is particularly important given the correlated nature of the neuropsychological measures in our study. The same analyses were repeated to compare patients with mild to moderate depression to those without depression.
Whole-brain voxel-wise two-sample t-test was performed to compare amyloid burden between groups with and without apathy for the identification of regional differences. Mean global cognition z-score and disease duration were included as covariates. Significance was set at p < 0.005 uncorrected for multiple comparisons at voxel level, with a cluster size of k > 4888 mm3 and p < 0.05 family wise error (FWE) corrected at cluster level.52,53
GDS scores were significantly higher among participants exhibiting apathy, evidencing between-group differences in depressive symptomatology. However, since there is a shared apathy–depression variance in this scale and both scales correlated significantly, GDS scores were deliberately excluded from the regression models, to avoid masking the unique contribution of apathy—an approach further justified by factor-analytic work demonstrating a distinct apathy subscale within the GDS. Moreover, patients with baseline severe depression were removed a priori, and we subsequently compared clinical, cognitive, and 18F-Flutemetamol PET burden between individuals with mild to moderate versus absent depression. This strategy accords with standard psychometric guidance, which cautions against adjusting for variables that share substantial variance with the primary predictor, as such adjustment can distort interpretation of the predictor–outcome relationship.54,55
To determine the predictive value of apathy scores and amyloid burden for cognitive decline over three years, we fitted a series of logistic regression models, each controlling for disease duration. The first model included only apathy status; the second replaced apathy with global amyloid burden; and the third included both predictors. Model fit was compared via likelihood-ratio tests and pseudo-R², and discrimination was assessed by the area under the ROC curve.
Finally, longitudinal trajectories across neuropsychological tests were examined using a linear mixed-effects model. Fixed effects comprised apathy status, time, test scores, and their interactions; a random intercept for each participant accounted for within-subject variance. Significant interactions were followed up with Holm-corrected post-hoc simple-contrast comparisons.
Results
Clinical and neuropsychological data
A total of 48 PD patients (34 men and 14 women) with a mean disease duration of 5.50 ± 2.10 years were included in the study. According to the SAS score, 20 patients exhibited apathy, with a mean age of 68.05 ± 4.65 years, while 28 patients did not present apathy, with a mean age of 67.39 ± 5.20 years. When comparing both groups, there were no significant differences in clinical or demographic characteristics (Table 1), except for disease duration, which was longer in the apathetic group (mean duration of 6.40 ± 1.80 years vs. 4.86 ± 2.05 years in the non-apathetic group, p < 0.05). Additionally, patients with apathy showed worse performance on several cognitive tests, such as Digit Span Forward and Backward and TMT-A (attention), Stroop test inhibition time and TMT-B (executive function), and CERAD (memory) at baseline (Table 2).
Demographic variables at baseline.
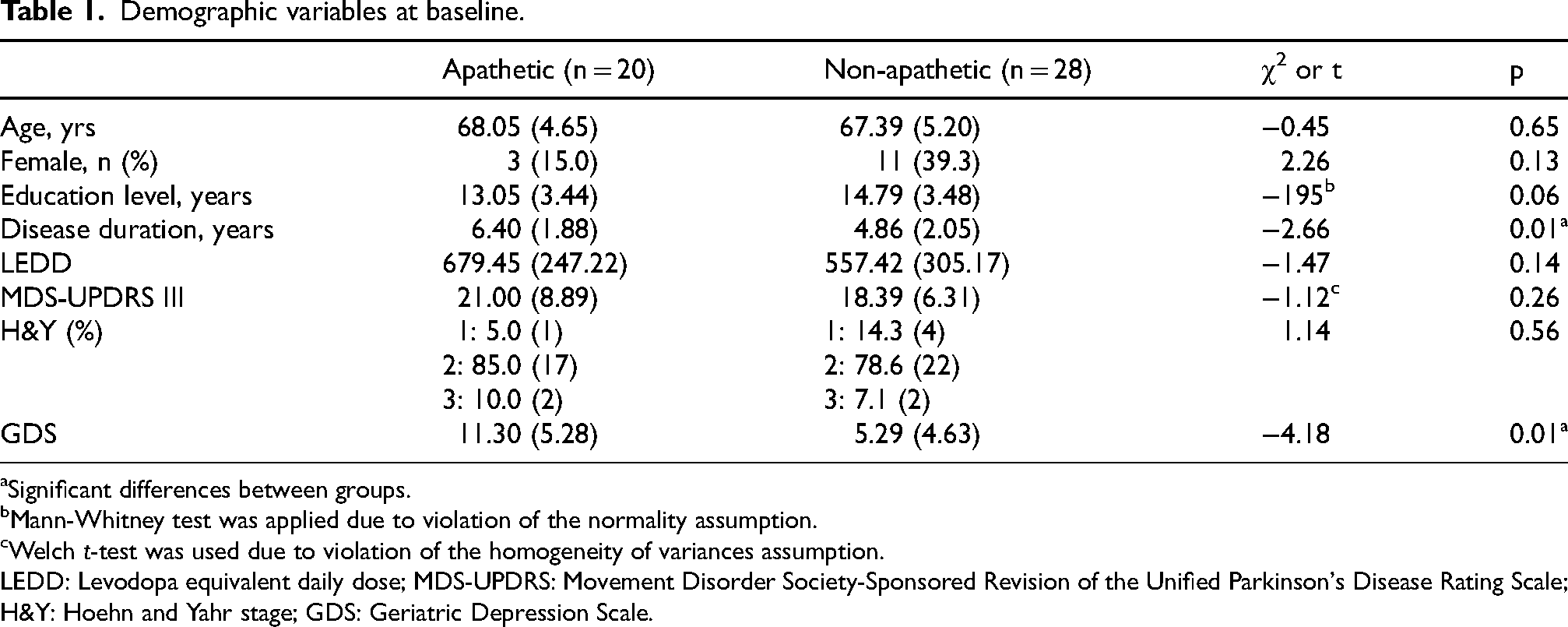
Significant differences between groups.
Mann-Whitney test was applied due to violation of the normality assumption.
Welch t-test was used due to violation of the homogeneity of variances assumption.
LEDD: Levodopa equivalent daily dose; MDS-UPDRS: Movement Disorder Society-Sponsored Revision of the Unified Parkinson's Disease Rating Scale; H&Y: Hoehn and Yahr stage; GDS: Geriatric Depression Scale.
Neuropsychological test scores assessed from baseline to 3-year evaluation.
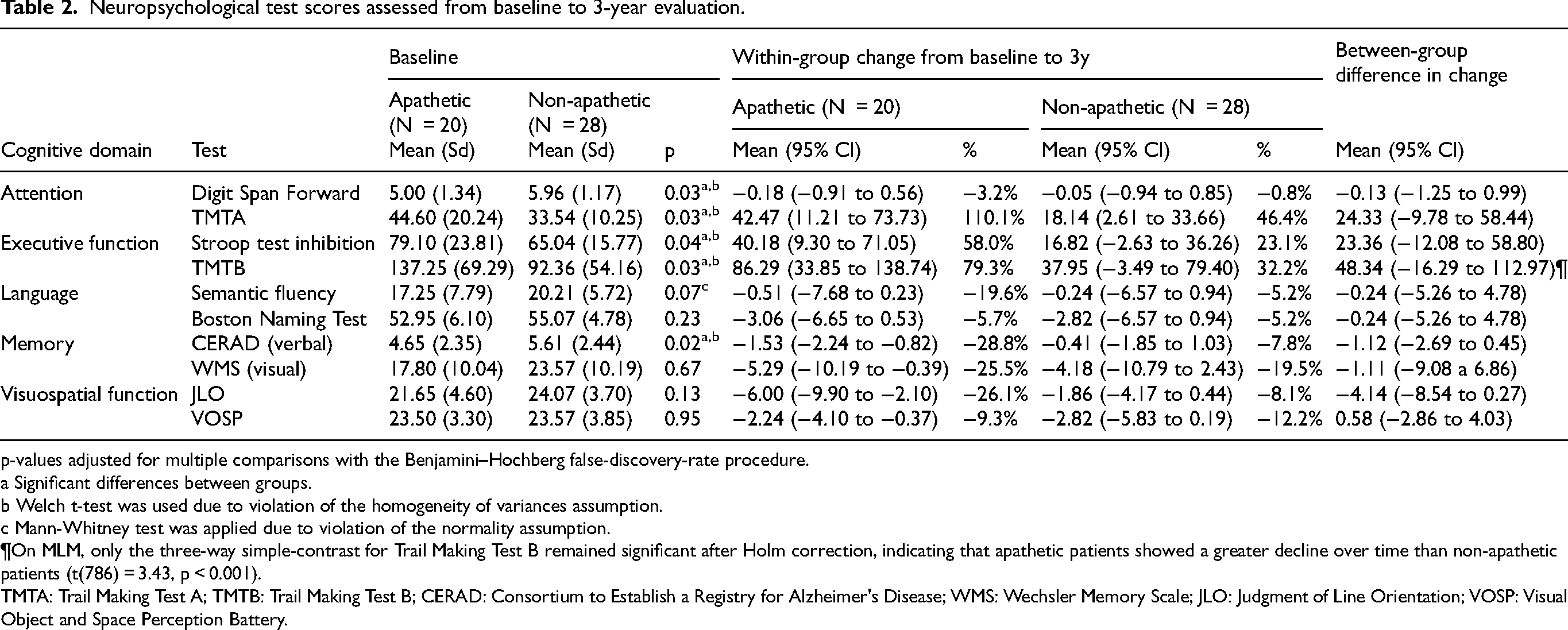
p-values adjusted for multiple comparisons with the Benjamini–Hochberg false-discovery-rate procedure.
Significant differences between groups.
Welch t-test was used due to violation of the homogeneity of variances assumption.
Mann-Whitney test was applied due to violation of the normality assumption.
¶On MLM, only the three-way simple-contrast for Trail Making Test B remained significant after Holm correction, indicating that apathetic patients showed a greater decline over time than non-apathetic patients (t(786) = 3.43, p < 0.001).
TMTA: Trail Making Test A; TMTB: Trail Making Test B; CERAD: Consortium to Establish a Registry for Alzheimer's Disease; WMS: Wechsler Memory Scale; JLO: Judgment of Line Orientation; VOSP: Visual Object and Space Perception Battery.
At baseline, 6 of 28 patients without apathy, and 10 of 20 patients with apathy fulfilled PD-MCI criteria. After 3 years of follow-up, 9 out of 19 patients with apathy (47.4%) progressed to a worse cognitive state (3 to PD-MCI and 6 to PDD), whereas only 3 of 25 non-apathetic patients (12.0%) converted (to PD-MCI) (χ² = 6.81, p < 0.05), resulting in 9 of the 25 non-apathetic patients meeting the criteria for PD-MCI. Four patients (1 with apathy and 3 without apathy) were lost to follow-up.
Regarding treatment, all patients were on dopaminergic therapy, with a mean LEDD of 679.45 ± 247.22 mg in the apathetic group and 557.42 ± 305.17 mg in the non-apathetic group (n.s.). In the apathetic group, ten patients (50%) were treated with dopamine agonists, whereas twenty patients (71%) in the non-apathetic group received dopamine agonists. No patient was receiving cholinesterase inhibitors.
Fifteen patients showed mild to moderate depression, and thirty-three did not report depressive symptoms. Disease duration was longer in patients with mild to moderate depression (mean duration: 6.67 ± 1.45 years vs. 4.97 ± 2.16 years in the non-depressed group, p < 0.05). They also showed lower educational level, higher LEDD, and worse cognitive performance in several neuropsychological tests: TMT-A (attention), Stroop test inhibition time, TMT B (executive function), Wechsler Memory Scale (memory), and Visual Object and Space Perception Battery (visuospatial function) (Supplemental material 1).
Regional differences in 18F-flutemetamol PET
Using disease duration and global cognition Z-score as covariates, amyloid burden was higher in apathetic vs. non-apathetic patients mainly in the frontal cortex (bilateral orbitofrontal cortex [OFC], rectus gyri) and to a lesser extent in the temporal cortex (right fusiform gyrus and middle and inferior temporal), the right insula and the left caudate (Figure 1 and Table 3). Non-apathetic patients did not show regions with higher amyloid burden in comparison with apathetic patients.
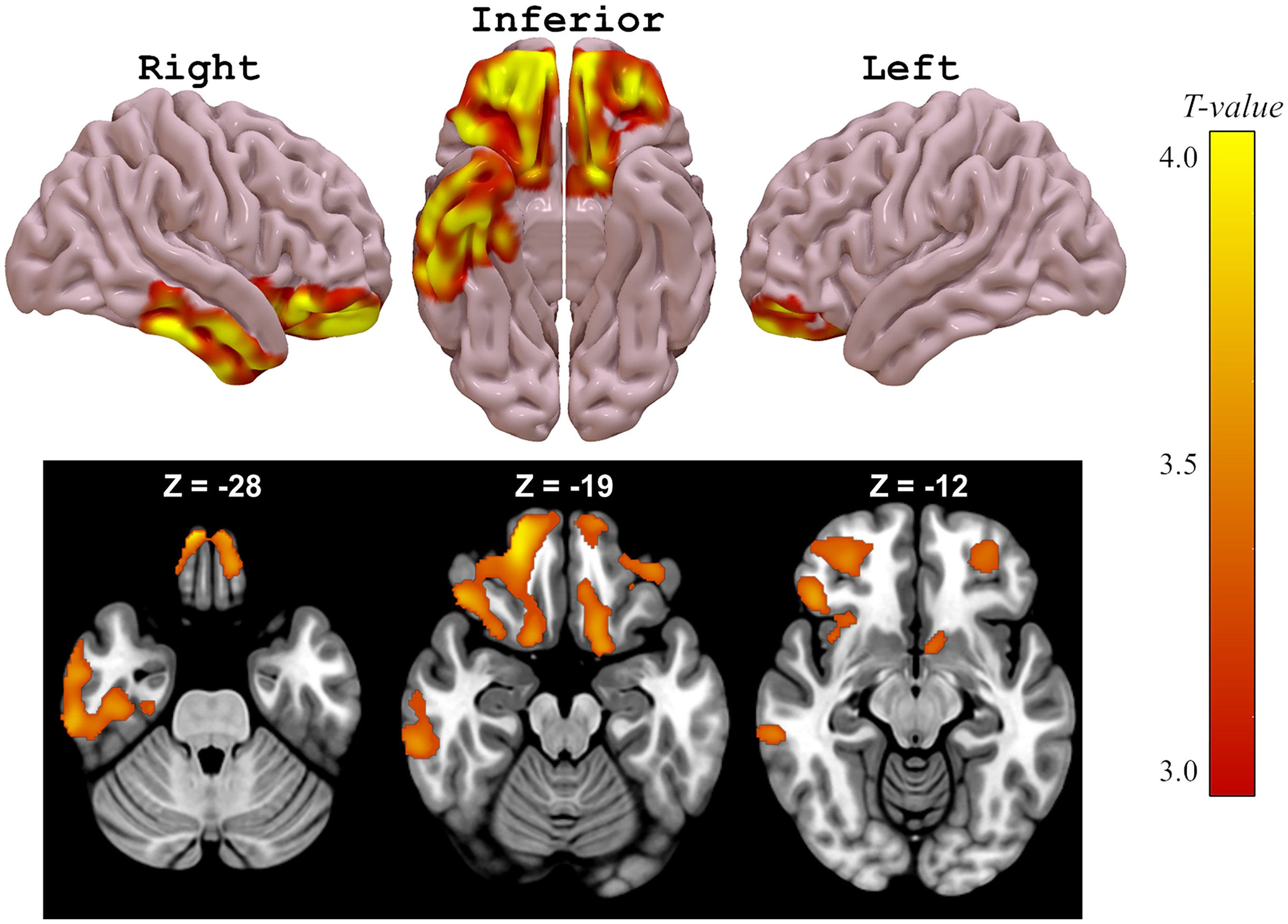
Cortical and subcortical increase uptake in [18F]-flutemetamol (voxelwise differences) between apathetic and non-apathetic patients. Data are thresholded at uncorrected p < 0.005, family-wise error corrected at cluster level p < 0.05.
Brain regions showing differences in amyloid load between apathetic and non-apathetic PD patients groups.

MNI coordinates (mm) [x y z = right hemisphere; -x y z = left hemisphere].
Z values at the peak of maximum significance applying a voxel-level threshold of p < 0.005 and an FWE–corrected cluster-size threshold of p < 0.05.
*Regions according to: AAL atlas; BA: Brodmann area.
For the depression related neuroimaging study, we used disease duration and global cognition Z-score as covariates. No amyloid differences between either group were found (mild to moderate depression vs. depression absence).
Logistic regression models predicting cognitive conversion
We fit three logistic regression models, each adjusted for disease duration, to examine the predictive value of apathy and amyloid burden for cognitive conversion over three years (Table 4). In this case, we did not include global cognition Z-score as a covariate due to circularity concerns. In Model 1 (apathy + disease duration), the overall fit was not significant (χ²(2) = 4.36, p = 0.113; McFadden's R² = 0.0845), and discrimination was poor (AUC = 0.677). Replacing apathy with amyloid burden in Model 2 yielded a significant improvement over the null (χ²(2) = 9.95, p = 0.007; R² = 0.193) and substantially better discrimination (AUC = 0.826), with amyloid burden emerging as a significant predictor. Model 3, which included both apathy and amyloid burden alongside duration, also attained a significant omnibus test (χ²(3) = 10.4, p = 0.016; R² = 0.201; AUC = 0.830), but a likelihood-ratio comparison against Model 2 showed no further gain from adding apathy (Δχ²(1) = 1.24, p = 0.265). Thus, amyloid burden drove the improvement in predictive accuracy, whereas apathy did not contribute incremental value beyond amyloid burden.
Logistic regression model fit and predictive performance for cognitive conversion at 3-year follow-up (N = 44).

p < 0.05. Δχ² tests compare each model to the one immediately above. AIC: Akaike's Information Criterion; R² McFadden: McFadden's pseudo-R²; χ² & p: omnibus test vs. prior (?).
Longitudinal effects of apathy on neuropsychological performance across domains
A linear mixed-effects model was conducted to examine the effects of apathy, time, and their interactions with performance on multiple neuropsychological tests. The model showed good fit (conditional R² = 0.65) (Table 5). Main effects were significant for time (F = 20.50, p < 0.05) and neuropsychological test type (F = 161.49, p < 0.05), whereas apathy alone was not significant (F = 0.94, p = 0.338). Notably, the Apathy × Time interaction was significant (F = 4.14, p < 0.05), as was the Apathy × Time × Test interaction (F = 1.94, p < 0.05), indicating that the effect of apathy on cognitive change over time varied across neuropsychological domains (Table 6). Post-hoc simple-contrast analyses (Holm-corrected) revealed a significant three-way interaction for TMT-B, where patients with apathy exhibited a greater decline at follow-up compared to those without apathy. No significant interactions were observed for other cognitive tests (Table 2).
Linear mixed-effects model results on neurocognitive performance and apathy over time. Model fit.
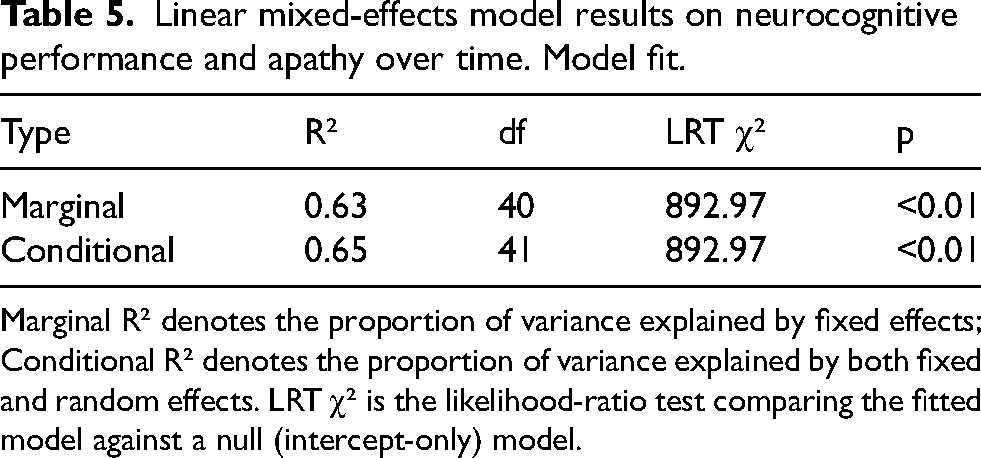
Marginal R² denotes the proportion of variance explained by fixed effects; Conditional R² denotes the proportion of variance explained by both fixed and random effects. LRT χ² is the likelihood-ratio test comparing the fitted model against a null (intercept-only) model.
Linear mixed-effects model results on neurocognitive performance and apathy over time. Fixed–effects omnibus tests.
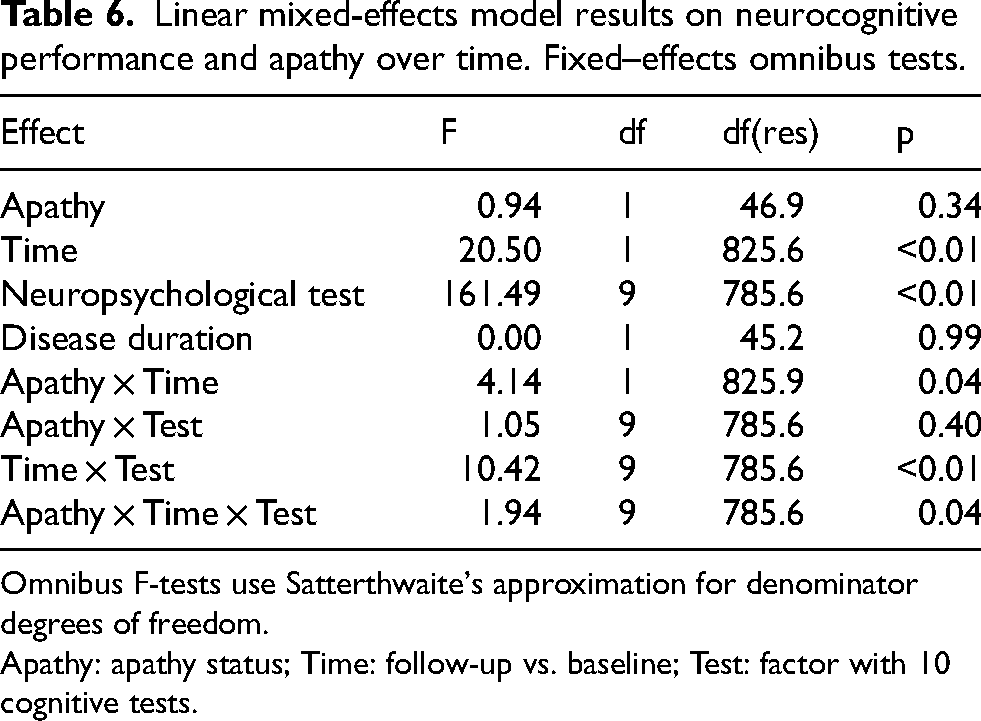
Omnibus F-tests use Satterthwaite's approximation for denominator degrees of freedom.
Apathy: apathy status; Time: follow-up vs. baseline; Test: factor with 10 cognitive tests.
Discussion
Apathy is a prevalent emotional state in the PD population. It is often linked to reduced quality of life and increased caregiver burden.56,57 It is frequently associated with cognitive decline, which leads to higher levels of disability.
In this study, we compared amyloid burden, cognitive performance at baseline, and cognitive progression in PD patients without dementia, categorized based on the presence or absence of apathy.
Apathetic patients exhibited a greater amyloid burden, predominantly in the frontal cortex and, to a lesser extent, in the temporal cortex. No brain regions showed higher amyloid deposition in non-apathetic patients. Additionally, PD patients with apathy demonstrated poorer performance on several neuropsychological tests and exhibited a higher conversion rate to worse cognitive states after 3 years of follow-up compared to their non-apathetic counterparts. Nonetheless, logistic regression showed that only amyloid burden significantly predicted conversion, whereas apathy did not. Our mixed-effects model demonstrated that impairment was most pronounced on executive functions compared with other cognitive domains.
Our findings align with previous studies that have reported more significant cognitive deficits in apathetic PD patients compared to non-apathetic ones. These deficits span global cognition, executive function, delayed verbal memory, and semantic verbal fluency.7,58 In addition, these findings indicate that the presence of apathy is a predictor of cognitive deterioration. 7 We found progression to PDD was nearly four times higher in apathetic patients. Our findings suggest that apathy may serve as a marked of underlying Alzheimer–type pathology in PD patients. They link studies that have reported increased brain amyloid deposition in apathetic PD patients32,59 with those that have demonstrated accelerated cognitive decline and higher conversion rates in amyloid-positive PD cohorts,60,61 most notably driving steeper executive-function decline. 29 These findings are consistent with our analysis, which showed that, although the apathetic group exhibited a pronounced cognitive decline over time, only performance on the TMT-B (executive function) remained significant in the post-hoc simple-contrast analyses. This is likely a consequence of the effect size and the statistical power of our study.
The neurobiology of apathy in PD likely involves multiple mechanisms. Dopaminergic denervation within the mesocorticolimbic circuit plays a pivotal role in its pathogenesis.12,62 Serotoninergic degeneration, as measured by [11C] DASB PET, has also been implicated, even in the early stages of PD. 15 Notably, lower serotonin levels in the neocortex and striatum have been inversely correlated with amyloid pathology, suggesting that serotoninergic dysfunction might modulate Aβ production. 63
Amyloid deposition in PD has been linked to synergistic interactions with Lewy body pathology, which accelerate cognitive decline and progression to dementia.30,64 Mixed AD pathology (amyloid and tau) is common in Lewy body diseases like PD and has been shown to independently predict progression to dementia.65,66 Part of this AD pathology seems to be related to apathy in non-demented cases. Our findings indicate that apathetic PD patients exhibit a selective increase in Aβ deposition within the OFC, gyrus rectus, and insula, with less prominent involvement of temporal regions. The only previous study that evaluated the amyloid-apathy correlations in PD also reported significantly greater Aβ burden in OFC and gyrus rectus, and to a lesser extent in insular cortices and lateral temporal regions, even after controlling for disease duration and global cognition. 32
The OFC and adjacent ventral prefrontal cortex are critical for encoding reward value and motivational salience.67–69 Given the OFC key role in encoding reward value and motivational salience, amyloid-driven dysfunction here likely disrupts reward valuation and motivational processes mediated by this region. 70 Moreover, dopaminergic denervation of the caudate nucleus in PD has shown a correlation with cognitive slowing and reduced initiative, 71 and greater atrophy of the dorsolateral head of the left caudate has been described in apathetic PD patients. 72 This potentially impairs the associative and limbic striatal loops that connect the caudate and ventral striatum to prefrontal regions and thus undermines the neural substrates of motivated action. 73 Indeed, voxel-based morphometry and SPECT studies have likewise reported bilateral OFC, anterior cingulate cortex, and left caudate nucleus atrophy or hypometabolism in apathetic patients with AD or frontotemporal dementia.23,24,74
The insula, a region traditionally involved in motivational processes, 75 exhibits a bilateral reduction in gray matter density in apathetic PD patients.76,77 This structure also participates in a thalamocortical network that may serve a compensatory role during reward feedback, 78 and its dopaminergic denervation has been linked to the development of apathy in animal models of PD. 79
In contrast, studies of structural alterations in temporal regions and the fusiform gyrus among apathetic PD patients have yielded inconsistent results.76,77,80,81 Nevertheless, functional impairment within these areas have been reported, 82 and cortical thinning in the temporal lobe and fusiform gyrus appears to progress with disease severity, potentially underpinning our observed clinical findings. 83
Taken together, converging evidence from neuroimaging and neuropathology implicates dysfunction of fronto-subcortical reward circuits. This is potentially driven by regional amyloid pathology as a core substrate of apathy in PD and related neurodegenerative disorders, supporting the link between apathy, cognition, and amyloid in PD.
In our study, we did not observe any significant difference in amyloid burden within the cingulate cortex or prefrontal regions when we compared apathetic PD patients with their non-apathetic counterparts. This finding contrasts with earlier studies that had consistently identified structural or functional abnormalities in prefrontal cortical circuits and anterior cingulate regions in apathetic PD patients.76,82,84 However, prior studies that demonstrated brain amyloid-apathy links often examined patients at later stages or “at risk for dementia”, 32 who tend to harbor more Alzheimer-type pathology. Our PD sample may have been at an earlier disease stage (or had a severe degree of cognitive impairment), potentially with lower overall global amyloid burden. Furthermore, apathy in PD is likely multifactorial. The structural atrophy or hypometabolism reported in cingulate and prefrontal cortices of apathetic PD patients do not necessarily stem from amyloid deposition.
In addition, some of the differences in findings might be attributed to distinct patient populations since Zhou et al. 32 included individuals with more advanced disease and additional risk factors for dementia like older age, subjective cognitive complaints, and mild cognitive impairment, the latter associated with amyloid pathology. 84 The scales for apathy were different, but both the SAS 38 and Apathy Evaluation Scale 85 display clinical validity for assessment of apathy in PD patients. 86
Our findings highlight apathy as a potential clinical marker for increased amyloid burden and faster cognitive decline in PD. This underscores the importance of distinguishing apathy from depression, as apathy can occur independently of a depressed mood. Apathy is not so much a negative mood, but rather a reduced ability to initiate voluntary, goal-directed actions. 4 On the other hand, depression includes feelings of pessimism, hopelessness, anhedonia, self-criticism, and even thoughts of death. 87 The differentiation between both concepts has great clinical significance. We excluded patients with severe depression and performed separate analyses comparing amyloid burden in patients with mild to moderate versus absent depression. No differences were found, which supports the notion that amyloid deposition in apathetic patients is unrelated to depressive symptoms, which is consistent with imaging studies in the general population. 88
Treatment for apathy in PD is challenging. While dopamine agonists and cholinesterase inhibitors have shown potential benefits, 89 sometimes they may be due to treatment of the associated depression or cognitive impairment 90 and some patients are refractory. In addition, some selective serotonin reuptake inhibitors typically produce emotional blunting and increased apathy.31,91,92 Moreover, dopamine agonists can worsen cognition in PD patients with cognitive decline, where apathy is more prevalent. 35 On the other hand, non-pharmacological interventions offer an essential strategy for managing apathy in PD. Thus, the combination of Behavioral Activation Therapy, physical exercise, Cognitive-Behavioral Therapy and caregiver support tailored to the individual needs of each patient can significantly improve motivation, participation, and quality of life.93,94
Given the association between amyloid pathology and apathy, anti-amyloid therapies might offer a novel approach. These treatments could potentially improve both cognitive function and decrease apathy, especially in patients who are unresponsive to conventional dopaminergic or antidepressant treatments.
A limitation of our investigation was the lack of multiple comparisons correction of the neuroimaging study at a voxel level. Nevertheless, a cluster level analysis, a widely used and recommended approach in the literature, 95 was performed that increased the sensitivity to detect broader patterns while controlling for Type I error. 52 In addition, semantic fluency has been considered to be a measure of executive fluency 41 ; however, more recent studies focused on the application of the PD-MCI MDS Criteria Task Force recommended placing it within language,42,43 and category fluency has been applied this way in numerous research studies on PD-MCI (including PD-MCI MDS Criteria Task Force members45–48). Finally, although apathetic patients exhibited significantly higher GDS scores, we did not include GDS as a covariate to preserve apathy's unique effect; nevertheless, we acknowledge that some of the observed group differences may instead reflect depressive symptoms.
The main strengths of the study were the large number of subjects evaluated, its longitudinal nature, the comprehensive neuropsychological assessment, and the application of a validated scale for excluding or detecting apathy in PD patients. 52
Conclusion
This study expands current knowledge regarding the relationship between apathy, amyloid burden, and cognitive decline in PD. By identifying apathy as an amyloid- and cognition-related emotional state with specific regional deposition patterns, we provide insights into its potential pathological substrates and therapeutic implications. Future research should focus on validating these findings and exploring the relationship between apathy, amyloid burden, and cognition, thus leading to targeted interventions that address neuropathological aspects of apathy in PD.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251370969 - Supplemental material for Apathy linked to higher amyloid burden and cognitive deterioration in Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251370969 for Apathy linked to higher amyloid burden and cognitive deterioration in Parkinson's disease by Carmen Gasca-Salas, Roberto Fernández-Fernández, Rafael Rodríguez-Rojas, Beatriz Fernández-Rodríguez, Lina García-Cañamaque, Guillermo Lahera and Clara Trompeta in Journal of Parkinson's Disease
Footnotes
Acknowledgements
The authors would like to thank and acknowledge to all patients for their time and help, as well as to Dr José A. Obeso for supporting the project, and to Dr Lawrence Philips for the English revision.
ORCID iDs
Ethical considerations
The study was approved by the HM Puerta del Sur Ethics Committee (ref. 16.09.0942E1-GH) and was conducted in accordance with the Declaration of Helsinki and local regulations governing human research.
Consent to participate
All patients provided written informed consent before enrollment in the study.
Consent for publication
All patients gave additional consent for the publication of anonymized data arising from this research.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: PET-magnetic resonance studies were funded by the collaboration agreement between General Electric, HM Hospitales 1989 S.A., and Fundación de Investigación HM Hospitales.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.G.S has received lecture honoraria from Insightec, Società Italiana Parkinson e Disordini del Movimento and Asociación Madrileña de Neurología, and reimbursement of travel expenses to attend scientific conferences from Boston Scientific and Esteve. R.F-F. declares no financial or competing interests in relation to this work. R.R-R. has received speaker honoraria from Insightec and Zambon. B.F-R. declares no financial or competing interests in relation to this work. L. G-C. declares no financial or competing interests in relation to this work. G.L. has been a consultant for, spoken in activities of, or received grants from: Instituto de Salud Carlos III, Fondo de Investigación Sanitaria (FIS), Centro de Investigación Biomédica en Red de Salud Mental (CIBERSAM), Lundbeck, Otsuka, Angelini, Adamed, Boehringer and ROVI. C.T. declares no financial or competing interests in relation to this work.
Data availability statement
The data that support the findings of this study are available from the corresponding author, [CGS], upon reasonable request. The data are not publicly available, they contain information that could compromise the privacy of research participants.
Supplemental material
Supplemental material for this article is available online.
Previous presentation
Partial results from this study were included as an e-Poster abstract submitted to the European Congress of Psychiatry, which was held in April 2025.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
