Abstract
Patients with Parkinson’s disease (PD) carrying variants in the Glucocerebrosidase (GBA) gene (GBA-PD) suffer from orthostatic symptoms more frequently than idiopathic PD patients (IPD). Systematic measurements of the blood pressure have not yet been performed. In the present study, a prospective analysis of 33 GBA-PD and 313 IPD patients was carried out. Systolic blood pressure upon changing from the supine to the upright position dropped more strongly in GBA-PD compared to IPD patients. Diastolic blood pressure and heart rate did not differ between groups. This study provides further evidence for a pronounced involvement of the autonomic nervous system in GBA-PD.
INTRODUCTION
Variants in the Glucocerebrosidase (GBA) gene are the most common genetic risk factor for the development of Parkinson’s disease (PD). In a large international and comprehensive genetic screening study including 1,360 participants, 8.5% of the PD patients were found to carry heterozygous GBA variants, with the most frequent ones being c.1093G > A (p.Glu365Lys, also known as E326K) and c.1223C > T (p.Thr408Met, also known as T369M) [1]. These two variants were found to significantly increase the risk of PD (E326K: 1.97-fold; p < 0.00001 and T369M: 1.78-fold; p = 0.004) in non-Ashkenazi Jewish populations in a recent meta-analysis [2].
Orthostatic hypotension (OH) is defined as a sustained reduction of the systolic blood pressure of at least 20 mmHg or the diastolic blood pressure of 10 mmHg within 3 min of standing [3]. In PD, the prevalence of OH is 30–40%, depending on disease duration and disease severity [4, 5]. Patients with GBA-related PD (GBA-PD) report autonomic symptoms, such as OH, more frequently and to a greater extent than patients with idiopathic PD (IPD) [6]. Contrary to these reports, a large international study in 1,435 patients with PD and a recent meta-analysis did not find different frequencies of OH between GBA-PD and IPD patients; however, it remained uncertain how OH was assessed [7, 8].
OH is associated with cognitive decline [9] and the postural instability and gait difficulty phenotype (PIGD) [10, 11]. These clinical features are also more common in GBA-PD than in IPD [12].
OH in PD is predominantly a result of a nore-pinephrine deficiency due to cardiovascular sympathetic denervation [13]. A previous report found a stronger reduction of myocardial (123)I-metaiodo-benzylguanidine uptake on scintigraphy in GBA-PD compared to IPD [14], suggesting that sympathetic denervation is more pronounced in GBA-PD.
To address the conflicting reports of OH prevalence in GBA-PD compared to IPD, we conducted an analysis of OH in a previously described and meanwhile expanded PD cohort [15]. We aimed to assess the change in blood pressure upon moving from the supine into the upright position in GBA-PD compared to IPD and captured any orthostatic symptoms occurring during the change of position.
METHODS
Study design
We analyzed the baseline examination of the longitudinal population-based EPIPARK cohort. Details of the cohort structure were published earlier [15]. All participants were over 18 years of age, had a diagnosis of PD, and gave written informed consent. Patients carrying any rare variant in genes linked to parkinsonism (CHCHD2, DNAJC6, DNAJC13, LRRK2, GCH1, PARK7, PINK1, POLG, PRKN, SNCA, VPS35) were excluded from the analysis. The study was conducted in accordance with the Declaration of Helsinki and was approved by the local ethics committee.
Genetic analysis
Samples were tested for six different variants in the GBA gene including p.E365K, p.T408M, p.N409S, p.N431S, p.D448H, and p.V499L using the Global Screening Array with custom content (GSA, Illumina). In addition, the samples underwent GenePanel analysis covering all exons of GBA where no additional variants were found [1].
Clinical examination
The neurological examination was done by move-ment disorder specialists using the Unified Parkinson’s Disease Rating Scale (UPDRS) Part III protocol [16, 17]. Medical history, medication, and non-motor symptoms were recorded, and the Montreal Cognitive Assessment (MoCA) was performed. The PIGD phenotype score was calculated by adding up the following items from the UPDRS Part III: speech, leg agility, arising from chair, gait, freezing, and postural stability [18].
The levodopa equivalence daily dose (LEDD) was calculated according to Tomlinson et al. (2010) and updated according to Schade et al. (2020) [19, 20].
Blood pressure and heart rate assessment
Blood pressure and heart rate were assessed non-invasively using a manual monitor after study participants lay in the supine position for 2 min. The measurement was repeated 2 min after getting up to the upright position. We chose the 2-min time interval upon standing to be able to perform the blood pressure measurement in the third minute upon standing. The heart rate was assessed manually 1 min after lying down and 1 min after getting up, respectively. The pulse rate was measured 1 min upon standing to allow sufficient time to measure the blood pressure in the third minute.
Statistical analysis
Statistical analyses using SPSS27 (IBM SPSS Statistics for Macintosh, Armonk, NY: IBM Corp) comprised chi-square tests for categorical, Mann-Whitney U for ordinal, and t-tests or ANOVA for continuous variables. The comparison of blood pressure in the supine and upright position was done using repeated-measures and mixed-model ANOVA. Correlations were calculated using Pearson’s correlations. A significance threshold of < 0.05 was applied.
RESULTS
Clinical, demographic, and genetic characteristics
Out of 346 included PD patients with blood pressure and heart rate measurements, 33 (9.5%) carried a heterozygous variant in the GBA gene: 13 patients were heterozygous and one homozygous for p.E365K, 17 were heterozygous, and one homozygous for p.T408M (both variants are not associated with Gaucher disease), and one heterozygous for p.N409S.
GBA-PD patients did not significantly differ from IPD patients in age, sex, age at onset, disease duration, UPDRS Part III, MoCA, LEDD, presence of orthostatic symptoms, and Hoehn and Yahr (H&Y) scale. Clinical and demographic data are displayed in Table 1.
Clinical and demographic characteristics of the study participants
N, number; y, years; UPDRS, Unified Parkinson’s Disease Rating Scale; H&Y, Hoehn and Yahr score; MoCA, Montreal Cognitive Assessment; LEDD, levodopa equivalence daily dose; DA, dopamine agonists; RR, blood pressure; HR, heart rate; SD, standard deviation; IQR, interquartile range.
Comparison of blood pressure and heart rate between GBA-PD and IPD
The mixed-models ANOVA with the within-subject factor ‘body position’ and the between-subject factor ‘genotype’ revealed a main effect of ‘body position’ on systolic blood pressure (F (1, 332) = 30.77, p < 0.001) and an interaction of ‘body position’ and ‘genotype’ (F (1, 332) = 4.007, p = 0.046) with a more severe drop of systolic blood pressure in GBA-PD patients. The diastolic blood pressure did not change in PD patients depending on ‘body position’ (F (1, 332) = 0.276, p = 0.599, Fig. 1B), and there was no interaction between ‘body position’ and ‘genotype’ (F (1, 332) = 0.105, p = 0.746). There was a main effect of ‘body position’ on heart rate with higher rates in the upright position (F (1, 329) = 35.133, p < 0.001), but no interaction with the ‘genotype’ (F (1, 329) = 1.388, p = 0.240) (Fig. 1).
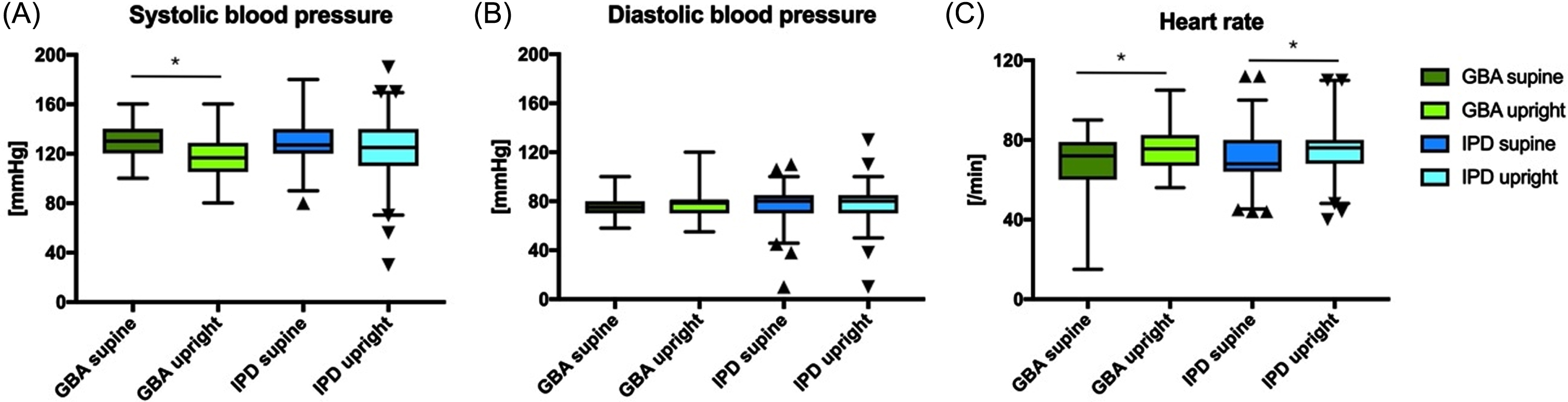
Comparison of systolic (A) and diastolic (B) blood pressure and heart rate (C) between GBA-PD and IPD. Data are shown as box plots. The whiskers indicate the 1st to 99th percentile. *p < 0.05.
More GBA-PD patients experienced a systolic blood pressure drop of≥20 mmHg (37.5% versus 12.9% in the IPD group; p < 0.001). Upon changing to the upright position, 9/31 (29%) of GBA-PD and 68/288 (23.6%) of IPD patients reported orthostatic symptoms during the test; however, this difference was not significant (p = 0.503). We did not find a significant difference in systolic difference (p = 0.794), diastolic difference (p = 0.315), or heart rate difference (p = 0.289) between the study participants who exhibited orthostatic symptoms and those who did not.
Furthermore, there was no correlation between LEDD and change in systolic blood pressure (Pearson’s r = 0.277; p = 0.132).
PIGD phenotype score
PIGD phenotype scores did not differ between groups (GBA-PD, mean (±SD): 8.56 (±4.46); IPD: 7.75 (±4.09); p = 0.371). The PIGD phenotype scores did not correlate with systolic (Pearson’s r = 0.02; p = 0.723) or diastolic (Pearson’s r = 0.05; p = 0.931) blood pressure drop in the group-spanning analysis (data not shown). The PIGD phenotype scores inversely correlated with the heart rate change (Pearson’s r = -0.125; p = 0.028).
DISCUSSION
The main finding of this study is that GBA-PD patients experience a more pronounced systolic blood pressure drop upon changing from the supine to the upright position than IPD patients. Moreover, a larger proportion of participants exhibited a drop in systolic blood pressure ≥ 20 mmHg in GBA-PD than in IPD.
Both groups matched well with regard to age, sex, age at onset, disease duration, and disease severity which allowed us to specifically assess the frequency of OH in GBA-PD compared to IPD. The intake of levodopa or dopamine agonists is known to exacerbate OH [21], but we observed no difference between groups in intake of these medications in our sample. Further, the PIGD phenotype score and intake of antihypertensive drugs were not different between both groups, indicating a distinct effect of the GBA genotype on the occurrence of OH.
A recent study including 80 patients with GBA-PD did not reveal significant differences between GBA-PD and IPD with regards to autonomic symptoms. However, the analysis was restricted to the early disease phase and was solely based on a questionnaire without measurements of blood pressure [22]. Our data suggest that these differences may become apparent only later in the disease course, as the mean disease duration in our study was ∼7 years compared to ∼3 years in the previous study. However, it would be important to conduct longitudinal studies to establish the first appearance of OH in GBA-PD versu s IPD. Further, the measurement of the blood pressure is more precise than a questionnaire. A recent survey of patients with PD and OH has found that they often hide or minimize OH symptoms and are reluctant to discuss symptoms with their healthcare providers [23], or are unable to recognize them [24]. While in some patients, the lack of reports of orthostatic symptoms may be due to chronic adaptive changes in cerebral blood flow regulation, in other patients, the inability to recognize symptoms might be attributable to the underlying neurodegenerative process [25]. Importantly, these patients are at a higher risk of falls [26]. Therefore, orthostatic blood pressure measurements are important to identify individuals with OH to prevent falls although the frequency of falls was not assessed in the present study.
GBA variants have been associated with the in-creased susceptibility to synucleinopathies other than PD, such as dementia with Lewy bodies and multiple system atrophy [27–30]. These synucleinopathies have been associated with increased autonomic symptoms and are in the beginning stages not always easily distinguishable from PD.
In summary, GBA-PD patients display OH more often than IPD patients. This finding has an important clinical implication as the broad consequences of potential falls and reduced quality of life easily outweigh the small effort of autonomic screening. Clinicians should therefore specifically test GBA-PD patients for autonomic dysfunction and symptoms attributable to OH to optimize their PD management.
Footnotes
ACKNOWLEDGMENTS
This research was partially supported by the German Research Foundation (DFG; FOR2488).
CONFLICT OF INTEREST
C.K. serves as a medical advisor on genetic testing reports for Centogene (excluding Parkinson’s disease) and is a member of the Scientific Advisory Board of Retromer Therapeutics.
